Venous Thromboembolism VTE Recent Advances in Reducing the

Venous Thromboembolism (VTE) Recent Advances in Reducing the Disease Burden Gary E. Raskob, Ph. D. Dean, College of Public Health Regents Professor, Epidemiology and Medicine University of Oklahoma Health Sciences Center CDC Division of Blood Disorders, Webinar November 6, 2014
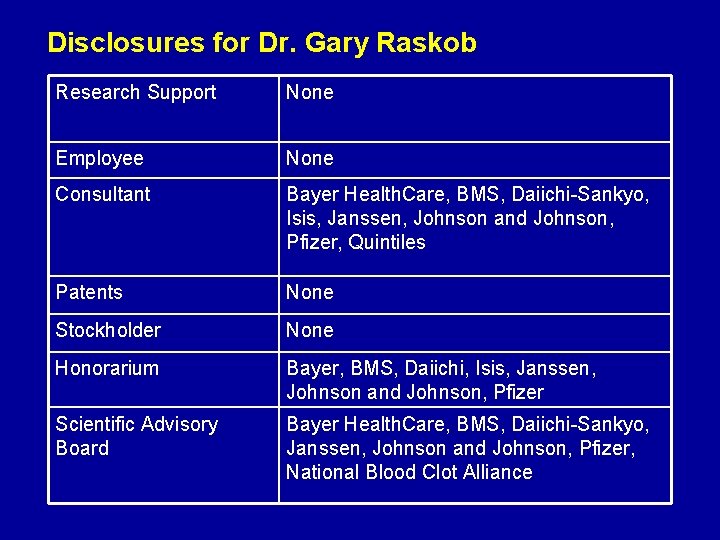
Disclosures for Dr. Gary Raskob Research Support None Employee None Consultant Bayer Health. Care, BMS, Daiichi-Sankyo, Isis, Janssen, Johnson and Johnson, Pfizer, Quintiles Patents None Stockholder None Honorarium Bayer, BMS, Daiichi, Isis, Janssen, Johnson and Johnson, Pfizer Scientific Advisory Board Bayer Health. Care, BMS, Daiichi-Sankyo, Janssen, Johnson and Johnson, Pfizer, National Blood Clot Alliance

Objectives ® Describe the burden of disease from VTE ® Describe the clinical features of VTE ® Describe the difference between provoked and unprovoked VTE ® Describe the risk factors for VTE and the steps people can take to reduce their risk of VTE ® Describe the evidence-based recommendations for treatment of VTE ® Describe how recent advances in oral anticoagulant therapy may help reduce the burden of VTE ® Describe the evidence - based strategies for the prevention of new cases of VTE

Disease Burden of VTE ® 1 to 3 episodes per 1, 000 population ® 2 to 7 per 1, 000 population age > 70 yrs ® 547, 596 hospitalizations with VTE in US 2007 - 2009 ® Estimated 900, 000 cases per year in US ® 100, 000 to 300, 000 VTE-related deaths in US each year ® 684, 000 DVT, 434, 000 PE, and 543, 000 VTE-related deaths in European Union 2004 (pop 454. 4 million) ® VTE a leading cause of hospital - associated premature death and disability (DALY) world wide Heit J, Cohen A, Anderson F. Estimated annual number of incident and recurrent, fatal and non-fatal venous thromboembolism (VTE) events in the US. Blood 2005; 106: 267 a Yusuf H et al. MMWR 2012; 61: 22: 401 – 404 ISTH Steering Committee for World Thrombosis Day. Thrombosis: a major contributor to global disease burden. J Thromb Haemost 2014; 12: doi: 10. 111/jth. 12698
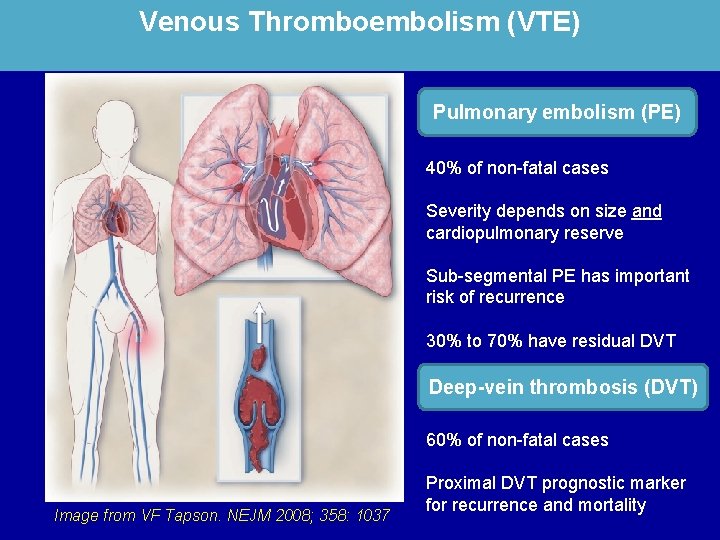
Venous Thromboembolism (VTE) Pulmonary embolism (PE) 40% of non-fatal cases Severity depends on size and cardiopulmonary reserve Sub-segmental PE has important risk of recurrence 30% to 70% have residual DVT Deep-vein thrombosis (DVT) 60% of non-fatal cases Image from VF Tapson. NEJM 2008; 358: 1037 Proximal DVT prognostic marker for recurrence and mortality
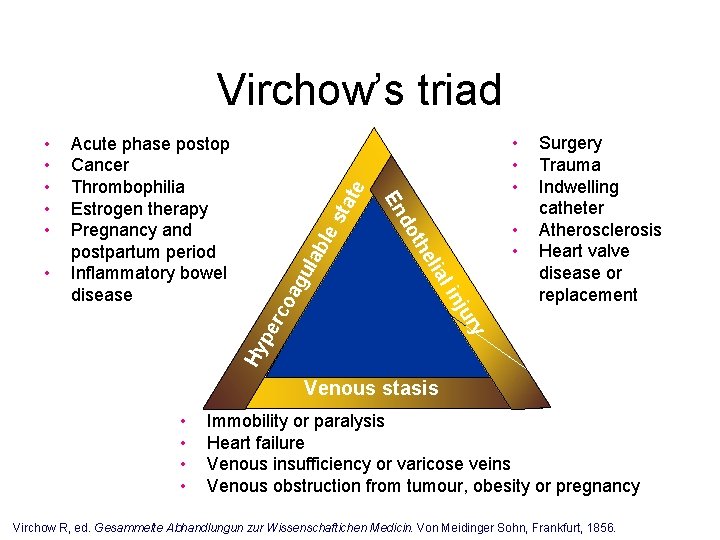
sta erc Hy p Surgery Trauma Indwelling catheter Atherosclerosis Heart valve disease or replacement y jur l in lia ula ble • • he oa g • • • ot • Acute phase postop Cancer Thrombophilia Estrogen therapy Pregnancy and postpartum period Inflammatory bowel disease d En • • • te Virchow’s triad Venous stasis • • Immobility or paralysis Heart failure Venous insufficiency or varicose veins Venous obstruction from tumour, obesity or pregnancy Virchow R, ed. Gesammelte Abhandlungun zur Wissenschaftichen Medicin. Von Meidinger Sohn, Frankfurt, 1856.
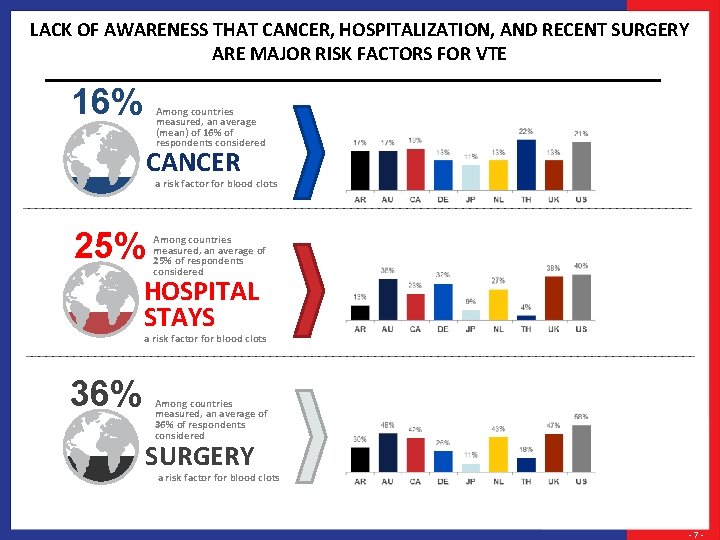
LACK OF AWARENESS THAT CANCER, HOSPITALIZATION, AND RECENT SURGERY ARE MAJOR RISK FACTORS FOR VTE 16% Among countries measured, an average (mean) of 16% of respondents considered CANCER a risk factor for blood clots 25% Among countries measured, an average of 25% of respondents considered HOSPITAL STAYS a risk factor for blood clots 36% Among countries measured, an average of 36% of respondents considered SURGERY a risk factor for blood clots -7 -

Clinical Presentations of VTE ® Provoked (70% of all patients) · · Associated with known risk factors Hospital, surgery, cancer, medical illness Risk factors may be continuing (cancer, APLA) If risk factor reversible (transient), 2% per year recurrence after 3 months of anticoagulant therapy ® Unprovoked (30% of all patients) · Absence of identifiable risk factor · Also called “idiopathic” · 7% to 11% per year recurrence for DVT or PE if anticoagulant therapy stopped after 3, 6, 12 or 24 months Kearon C, Akl E. Blood 2014; 123 (12) 1794 -1801. Boutitie F et al. BMJ 2011, May 24; 342: d 3036

Goals of Treatment for VTE ® Prevent death from pulmonary embolism ® Prevent symptomatic recurrent VTE 25% risk of symptomatic recurrent VTE during 3 if inadequate therapy ® Prevent and/or reduce morbidity from post-thrombotic syndrome (PTS) 25% at 2 yr chronic pulmonary hypertension 4% at 2 yr ® Minimize the risk of bleeding and other side effects

Evidence- based Guidelines for Treatment of VTE ® AHA Guidelines PE, iliofemoral DVT, CTEPH Jaff M et al Circulation 2011; 123: 1788 – 1830 ® American College of Chest Physicians (ACCP) 9 th ed 2012 Kearon et al CHEST 2012; 141: (2) Suppl: e 419 s-e 494 s ® International Union of Angiology - Consensus Statement Nicolaides AN et al Int Angiology 2013; 32: 111260 ® Treatment of Venous Thromboembolism Wells P et al JAMA 2014; 311: 717 -728 ® European Society of Cardiology 2014 ESC Guideline on the diagnosis and management of acute pulmonary embolism European Heart Journal 2014; doi 10. 1093/eurheart/ehu 283

Treatment of VTE 9 th ACCP Guideline Recommendations ® Anticoagulant therapy over other approaches for most acute DVT or PE (2 C) parenteral therapy using LMWH or fondaparinux (1 B) long-term therapy for at least 3 months (1 B) evaluate risk-benefit of extended therapy ® Catheter - Directed Thrombolytic (CDT) therapy for DVT anticoagulant therapy alone over CDT most patients (2 C) selected patients with DVT may benefit ® Anticoagulant therapy over no anticoagulation for extensive superficial vein thrombosis (2 B) (fondaparinux over LMWH, 2 C) ® Thrombolytic therapy for PE acute PE + hypotension (2 C) acute PE, high risk of hypotension, low risk of bleeding (2 C) intracranial bleeding in 2 to 3% in contemporary studies ® Inferior vena cava filter anticoagulants contraindicated (1 B) Kearon et al CHEST 2012; 141: (2) Suppl: e 419 s - e 494 s

Treatment of Venous Thromboembolism Heparin LMWH Fondaparinux Thrombolysis Thrombus Removal IVC filter Initial treatment Vitamin K Antagonists LMWH Long term-treatment Vitamin K Antagonists Extended treatment 5 to 10 days 3 to 6 months > 6 months
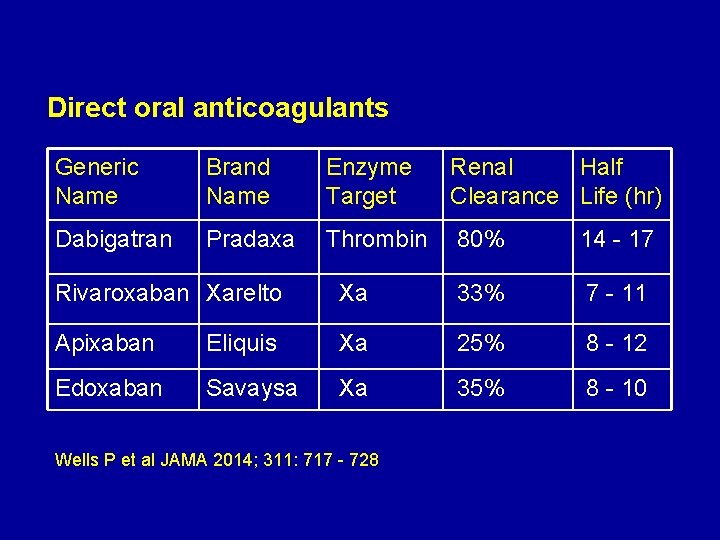
Direct oral anticoagulants Generic Name Brand Name Enzyme Target Renal Half Clearance Life (hr) Dabigatran Pradaxa Thrombin 80% 14 - 17 Rivaroxaban Xarelto Xa 33% 7 - 11 Apixaban Eliquis Xa 25% 8 - 12 Edoxaban Savaysa Xa 35% 8 - 10 Wells P et al JAMA 2014; 311: 717 - 728

Direct oral anticoagulants for VTE FDA approved indications and regimens ® Rivaroxaban (November 2012) Treatment of Deep Vein Thrombosis (DVT) Treatment of Pulmonary Embolism (PE) Reduction in the risk of recurrence of DVT and PE 15 mg orally twice daily with food for first 21 days (initial treatment), then 20 mg orally once daily with food ® Dabigatran (April 2014) Treatment of Deep Venous Thrombosis and Pulmonary Embolism Reduction in the risk of recurrence of DVT and PE 150 mg orally twice daily after 5 to 10 days of parenteral anticoagulation ® Apixaban (August 2014) Treatment of Deep Vein Thrombosis Treatment of Pulmonary Embolism 10 mg orally twice daily for 7 days, then 5 mg orally twice daily Reduction in the risk of recurrence of DVT and PE 2. 5 mg orally twice daily
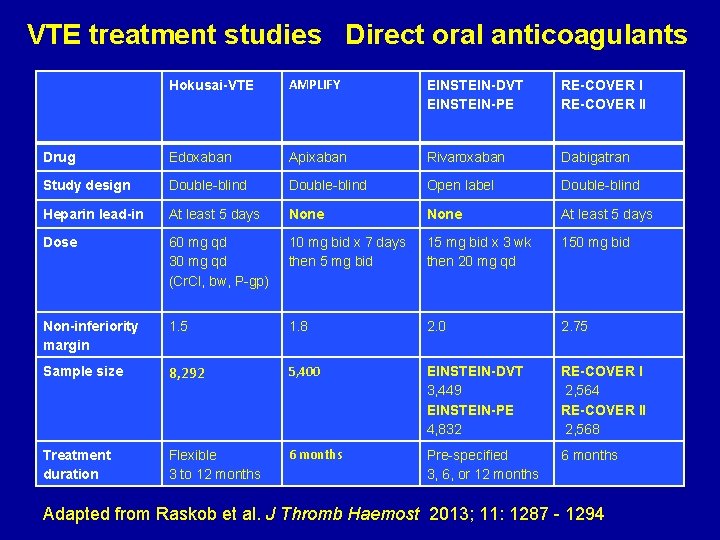
VTE treatment studies Direct oral anticoagulants Hokusai-VTE AMPLIFY EINSTEIN-DVT EINSTEIN-PE RE-COVER II Drug Edoxaban Apixaban Rivaroxaban Dabigatran Study design Double-blind Open label Double-blind Heparin lead-in At least 5 days None At least 5 days Dose 60 mg qd 10 mg bid x 7 days 30 mg qd then 5 mg bid (Cr. Cl, bw, P-gp) 15 mg bid x 3 wk then 20 mg qd 150 mg bid Non-inferiority margin 1. 5 1. 8 2. 0 2. 75 Sample size 8, 292 5, 400 EINSTEIN-DVT 3, 449 EINSTEIN-PE 4, 832 RE-COVER I 2, 564 RE-COVER II 2, 568 Treatment duration Flexible 3 to 12 months 6 months Pre-specified 3, 6, or 12 months 6 months Adapted from Raskob et al. J Thromb Haemost 2013; 11: 1287 - 1294

Clinical Trials of Rivaroxaban for VTE Open-label, non-inferiority 3, 6, or 12 months N=3, 449 R Rivaroxaban 15 mg bid Day 21 Rivaroxaban 20 mg od Double-blind, superiority Enoxaparin bid for at least 5 days, plus 6 or 12 months VKA target INR 2. 5 (INR range 2– 3) Rivaroxaban 20 mg od N=1, 197 Open-label, non-inferiority 3, 6, or 12 months N~4, 500 R Rivaroxaban 15 mg bid Day 21 R Placebo Rivaroxaban 20 mg od Enoxaparin bid for at least 5 days, plus Outside of the EINSTEIN programme Patients with confirmed symptomatic DVT or PE completing 6 or 12 months of VKA target INR 2. 5 (INR range 2– 3) N Engl J Med 2010; 363: 2499 -510 N Engl J Med 2012; 366: 1287 -1297 16
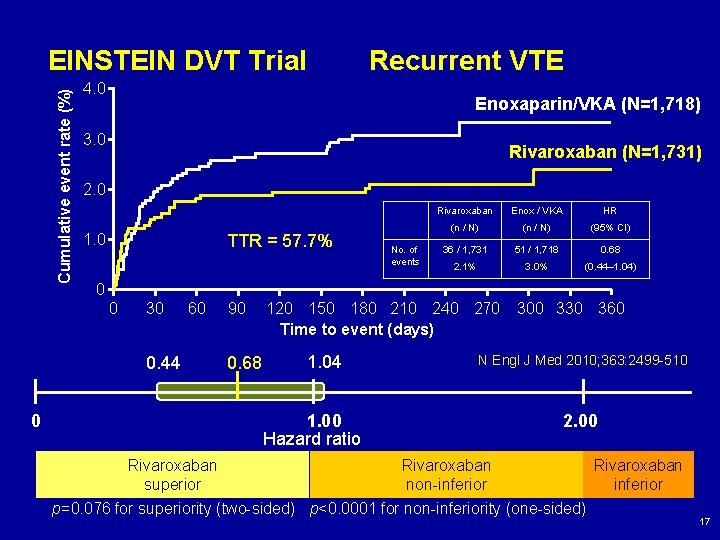
Cumulative event rate (%) EINSTEIN DVT Trial 4. 0 Enoxaparin/VKA (N=1, 718) 3. 0 Rivaroxaban (N=1, 731) 2. 0 1. 0 TTR = 57. 7% No. of events Rivaroxaban Enox / VKA HR (n / N) (95% CI) 36 / 1, 731 51 / 1, 718 0. 68 2. 1% 3. 0% (0. 44– 1. 04) 0 0 30 0. 44 0 Recurrent VTE 60 90 0. 68 120 150 180 210 240 270 Time to event (days) 1. 04 1. 00 Hazard ratio 300 330 360 N Engl J Med 2010; 363: 2499 -510 2. 00 Rivaroxaban superior non-inferior p=0. 076 for superiority (two-sided) p<0. 0001 for non-inferiority (one-sided) 17
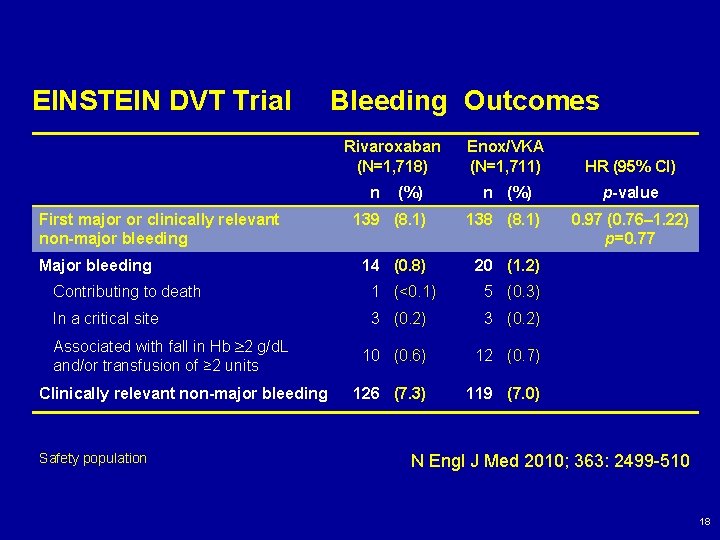
EINSTEIN DVT Trial Bleeding Outcomes Rivaroxaban (N=1, 718) Enox/VKA (N=1, 711) HR (95% CI) n (%) p-value 139 (8. 1) 138 (8. 1) 0. 97 (0. 76– 1. 22) p=0. 77 14 (0. 8) 20 (1. 2) n First major or clinically relevant non-major bleeding Major bleeding (%) Contributing to death 1 (<0. 1) 5 (0. 3) In a critical site 3 (0. 2) 10 (0. 6) 12 (0. 7) 126 (7. 3) 119 (7. 0) Associated with fall in Hb 2 g/d. L and/or transfusion of ≥ 2 units Clinically relevant non-major bleeding Safety population N Engl J Med 2010; 363: 2499 -510 18

EINSTEIN PE Trial Recurrent VTE Rivaroxaban (N=2419) First symptomatic recurrent VTE Recurrent DVT + PE Non-fatal PE Fatal PE/unexplained death where PE cannot be ruled out HR 0. 75 0 Enoxaparin/VKA (N=2413) n (%) 50 (2. 1) 44 (1. 8) 18 0 22 (0. 7) (0. 9) 17 2 19 (0. 7) (<0. 1) (0. 8) 10 (0. 4) 6 (0. 2) 1. 12 1. 68 1. 00 Rivaroxaban superior p=0. 57 for superiority (two-sided) 2. 00 Rivaroxaban non-inferior Rivaroxaban inferior P=0. 0026 for non-inferiority (one-sided) Absolute risk difference 0. 24% , 95% CI - 0. 5 to 1. 02 N Engl J Med 2012; 366: 1287 -1297
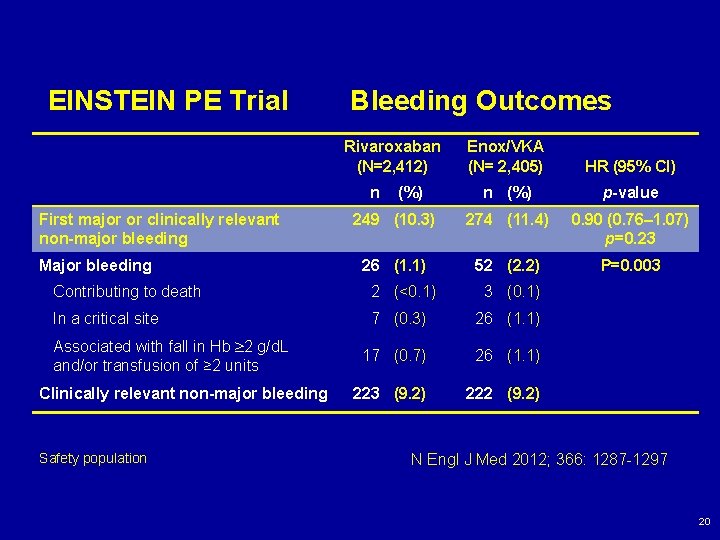
EINSTEIN PE Trial Bleeding Outcomes Rivaroxaban (N=2, 412) Enox/VKA (N= 2, 405) HR (95% CI) n (%) p-value 249 (10. 3) 274 (11. 4) 0. 90 (0. 76– 1. 07) p=0. 23 26 (1. 1) 52 (2. 2) P=0. 003 n First major or clinically relevant non-major bleeding Major bleeding (%) Contributing to death 2 (<0. 1) 3 (0. 1) In a critical site 7 (0. 3) 26 (1. 1) 17 (0. 7) 26 (1. 1) 223 (9. 2) 222 (9. 2) Associated with fall in Hb 2 g/d. L and/or transfusion of ≥ 2 units Clinically relevant non-major bleeding Safety population N Engl J Med 2012; 366: 1287 -1297 20
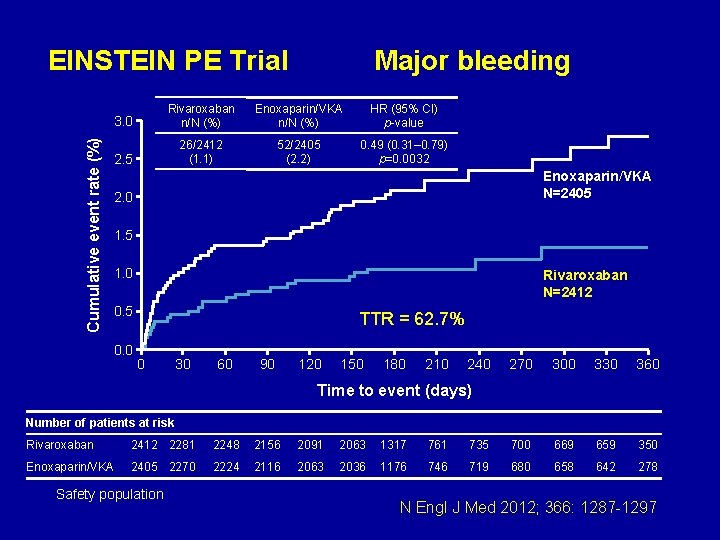
Cumulative event rate (%) EINSTEIN PE Trial Major bleeding 3. 0 Rivaroxaban n/N (%) Enoxaparin/VKA n/N (%) HR (95% CI) p-value 2. 5 26/2412 (1. 1) 52/2405 (2. 2) 0. 49 (0. 31– 0. 79) p=0. 0032 Enoxaparin/VKA N=2405 2. 0 1. 5 1. 0 Rivaroxaban N=2412 0. 5 0. 0 TTR = 62. 7% 0 30 60 90 120 150 180 210 240 270 300 330 360 Time to event (days) Number of patients at risk Rivaroxaban 2412 2281 2248 2156 2091 2063 1317 761 735 700 669 659 350 Enoxaparin/VKA 2405 2270 2224 2116 2063 2036 1176 746 719 680 658 642 278 Safety population N Engl J Med 2012; 366: 1287 -1297
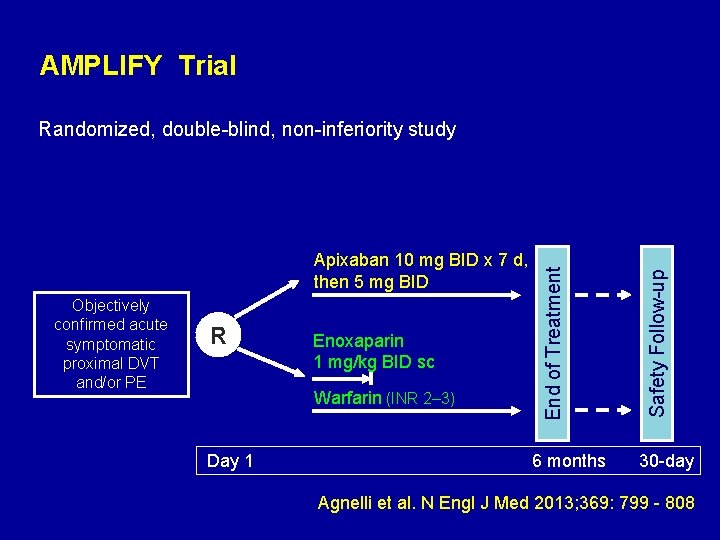
AMPLIFY Trial Objectively confirmed acute symptomatic proximal DVT and/or PE R Enoxaparin 1 mg/kg BID sc Warfarin (INR 2– 3) Day 1 6 months Safety Follow-up Apixaban 10 mg BID x 7 d, then 5 mg BID End of Treatment Randomized, double-blind, non-inferiority study 30 -day Agnelli et al. N Engl J Med 2013; 369: 799 - 808
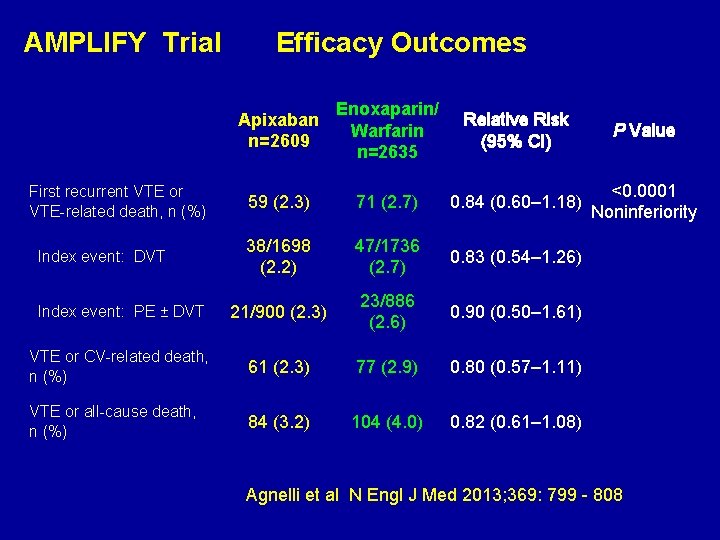
AMPLIFY Trial Efficacy Outcomes Apixaban n=2609 Enoxaparin/ Warfarin n=2635 Relative Risk (95% CI) P Value 59 (2. 3) 71 (2. 7) 0. 84 (0. 60– 1. 18) <0. 0001 Noninferiority 38/1698 (2. 2) 47/1736 (2. 7) 0. 83 (0. 54– 1. 26) Index event: PE ± DVT 21/900 (2. 3) 23/886 (2. 6) 0. 90 (0. 50– 1. 61) VTE or CV-related death, n (%) 61 (2. 3) 77 (2. 9) 0. 80 (0. 57– 1. 11) VTE or all-cause death, n (%) 84 (3. 2) 104 (4. 0) 0. 82 (0. 61– 1. 08) First recurrent VTE or VTE-related death, n (%) Index event: DVT Agnelli et al N Engl J Med 2013; 369: 799 - 808
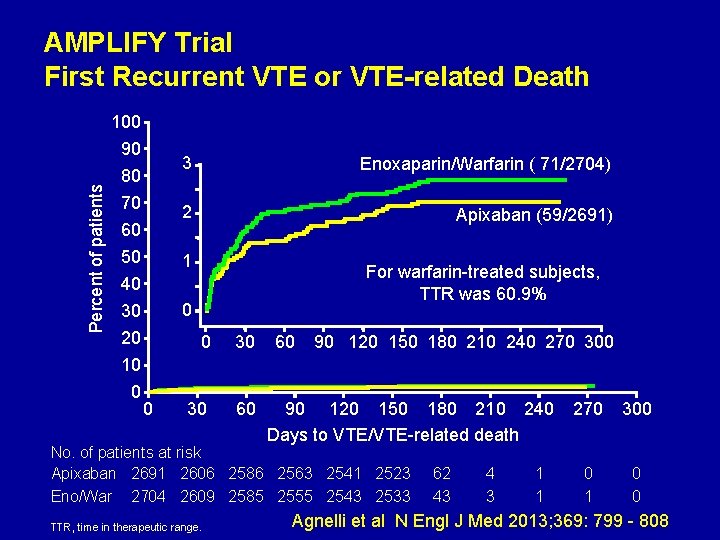
AMPLIFY Trial First Recurrent VTE or VTE-related Death Percent of patients 100 90 80 70 60 50 Enoxaparin/Warfarin ( 71/2704) 2 Apixaban (59/2691) 1 40 30 20 10 0 3 For warfarin-treated subjects, TTR was 60. 9% 0 0 0 30 30 60 60 90 120 150 180 210 240 270 300 90 120 150 180 210 240 Days to VTE/VTE-related death No. of patients at risk Apixaban 2691 2606 2586 2563 2541 2523 Eno/War 2704 2609 2585 2555 2543 2533 TTR, time in therapeutic range. 62 43 4 3 1 1 270 300 0 1 0 0 Agnelli et al N Engl J Med 2013; 369: 799 - 808
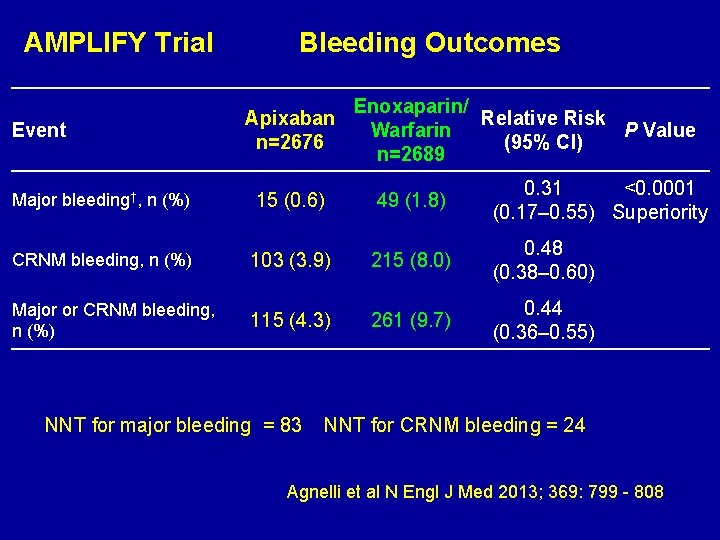
AMPLIFY Trial Event Bleeding Outcomes Apixaban n=2676 Enoxaparin/ Relative Risk Warfarin P Value (95% CI) n=2689 Major bleeding†, n (%) 15 (0. 6) 49 (1. 8) 0. 31 <0. 0001 (0. 17– 0. 55) Superiority CRNM bleeding, n (%) 103 (3. 9) 215 (8. 0) 0. 48 (0. 38– 0. 60) Major or CRNM bleeding, n (%) 115 (4. 3) 261 (9. 7) 0. 44 (0. 36– 0. 55) NNT for major bleeding = 83 NNT for CRNM bleeding = 24 Agnelli et al N Engl J Med 2013; 369: 799 - 808
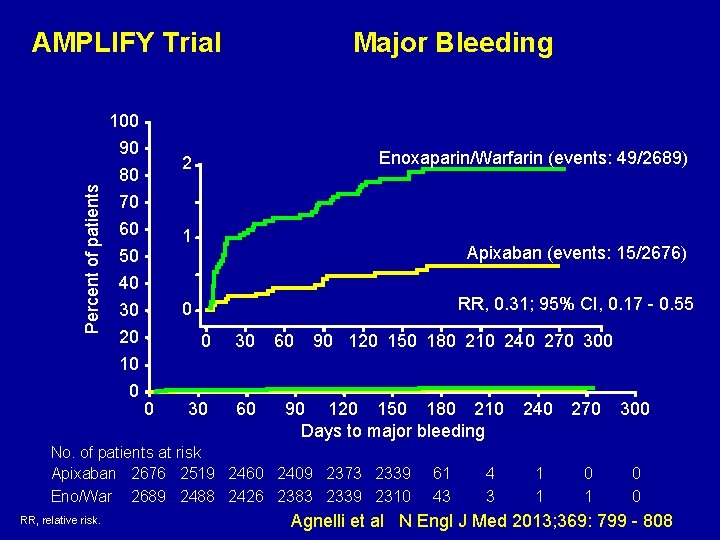
Percent of patients AMPLIFY Trial 100 90 80 Enoxaparin/Warfarin (events: 49/2689) 2 70 60 50 40 30 20 10 0 Major Bleeding 1 Apixaban (events: 15/2676) RR, 0. 31; 95% CI, 0. 17 - 0. 55 0 0 0 30 30 60 60 90 120 150 180 210 240 270 300 90 120 150 180 210 Days to major bleeding No. of patients at risk Apixaban 2676 2519 2460 2409 2373 2339 Eno/War 2689 2488 2426 2383 2339 2310 RR, relative risk. 61 43 4 3 240 270 300 1 1 0 0 Agnelli et al N Engl J Med 2013; 369: 799 - 808
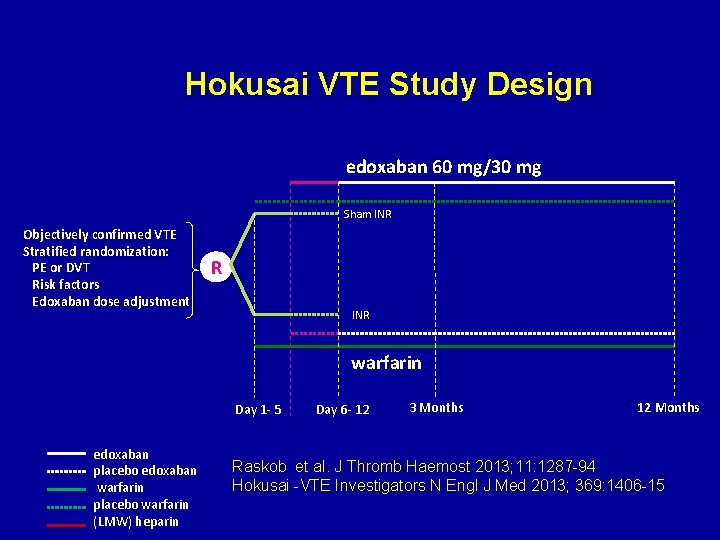
Hokusai VTE Study Design edoxaban 60 mg/30 mg Sham INR Objectively confirmed VTE Stratified randomization: PE or DVT Risk factors Edoxaban dose adjustment R INR warfarin Day 1 - 5 edoxaban placebo edoxaban warfarin placebo warfarin (LMW) heparin Day 6 - 12 3 Months 12 Months Raskob et al. J Thromb Haemost 2013; 11: 1287 -94 Hokusai -VTE Investigators N Engl J Med 2013; 369: 1406 -15
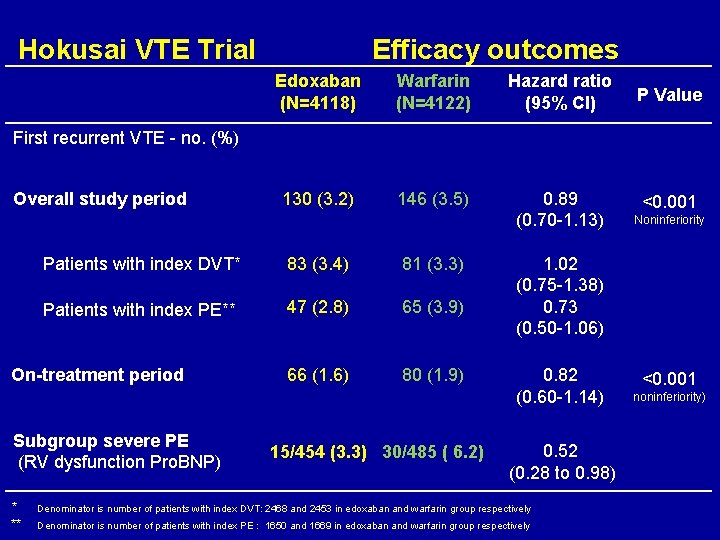
Hokusai VTE Trial Efficacy outcomes Edoxaban (N=4118) Warfarin (N=4122) Overall study period 130 (3. 2) 146 (3. 5) 0. 89 <0. 001 Noninferiority (0. 70 -1. 13) Patients with index DVT* 83 (3. 4) 81 (3. 3) Patients with index PE** 47 (2. 8) 65 (3. 9) 1. 02 (0. 75 -1. 38) 0. 73 (0. 50 -1. 06) On-treatment period 66 (1. 6) 80 (1. 9) Hazard ratio (95% CI) P Value First recurrent VTE - no. (%) Subgroup severe PE 15/454 (3. 3) 30/485 ( 6. 2) (RV dysfunction Pro. BNP) 0. 82 <0. 001 noninferiority) (0. 60 -1. 14) 0. 52 (0. 28 to 0. 98) * Denominator is number of patients with index DVT: 2468 and 2453 in edoxaban and warfarin group respectively ** Denominator is number of patients with index PE : 1650 and 1669 in edoxaban and warfarin group respectively
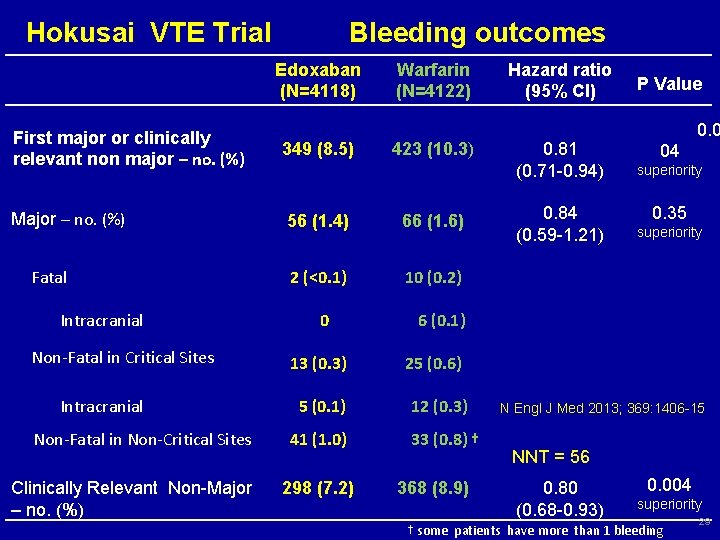
Hokusai VTE Trial Bleeding outcomes Edoxaban (N=4118) Warfarin (N=4122) First major or clinically relevant non major – no. (%) 349 (8. 5) Major – no. (%) 56 (1. 4) 66 (1. 6) 2 (<0. 1) 10 (0. 2) Fatal Intracranial Non-Fatal in Critical Sites Intracranial Non-Fatal in Non-Critical Sites Clinically Relevant Non-Major – no. (%) 0 13 (0. 3) 5 (0. 1) 41 (1. 0) 298 (7. 2) 423 (10. 3) Hazard ratio (95% CI) 0. 81 (0. 71 -0. 94) P Value 0. 0 04 superiority 0. 84 0. 35 superiority (0. 59 -1. 21) 6 (0. 1) 25 (0. 6) 12 (0. 3) 33 (0. 8) † 368 (8. 9) N Engl J Med 2013; 369: 1406 -15 NNT = 56 0. 80 (0. 68 -0. 93) 0. 004 superiority † some patients have more than 1 bleeding 29
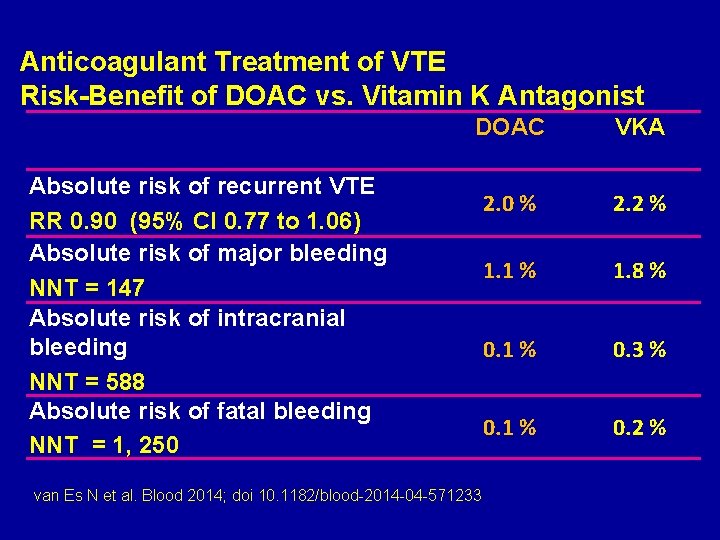
Anticoagulant Treatment of VTE Risk-Benefit of DOAC vs. Vitamin K Antagonist DOAC VKA 2. 0 % 2. 2 % 1. 1 % 1. 8 % 0. 1 % 0. 3 % 0. 1 % 0. 2 % Absolute risk of recurrent VTE RR 0. 90 (95% CI 0. 77 to 1. 06) Absolute risk of major bleeding NNT = 147 Absolute risk of intracranial bleeding NNT = 588 Absolute risk of fatal bleeding NNT = 1, 250 van Es N et al. Blood 2014; doi 10. 1182/blood-2014 -04 -571233

Acute PE without shock or hypotension ESC 2014 Guideline Recommendations ® Anticoagulant is recommended with objective to prevent both early death and recurrent symptomatic or fatal VTE parenteral anticoagulation LMWH or fondaparinux (I A) in parallel, VKA target INR 2. 5 (range 2. 0 - 3. 0) (I B) ® Alternative to combined parenteral anticoagulation with VKA rivaroxaban 15 mg bid x 3 weeks, followed by 20 mg daily (I B) apixaban 10. 0 mg bid x 7 days, followed by 5. 0 mg bid (I B) ® Alternative to VKA treatment , following parenteral anticoagulation dabigatran 150 mg bid (110 mg age > 80 yr, or verapamil) (I B) edoxaban (subject to regulatory review) (I B) European Heart Journal 2014 doi: 10. 1093/eurheartj/ehu 283

Duration of Anticoagulant Therapy 9 th ACCP Recommendations First episode VTE Provoked (surgery or reversible risk factor) 3 months over longer therapy (1 B) Unprovoked at least 3 months (1 B), evaluate for extended therapy (low bleeding risk, extended therapy (2 B), high bleeding risk 3 months (1 B) Cancer LMWH over VKA (2 B), extended therapy (1 B or 2 B) Second episode VTE, unprovoked extended therapy (1 B or 2 B) Kearon et al CHEST 2012; 141: (2) Suppl: e 419 s - e 494 s
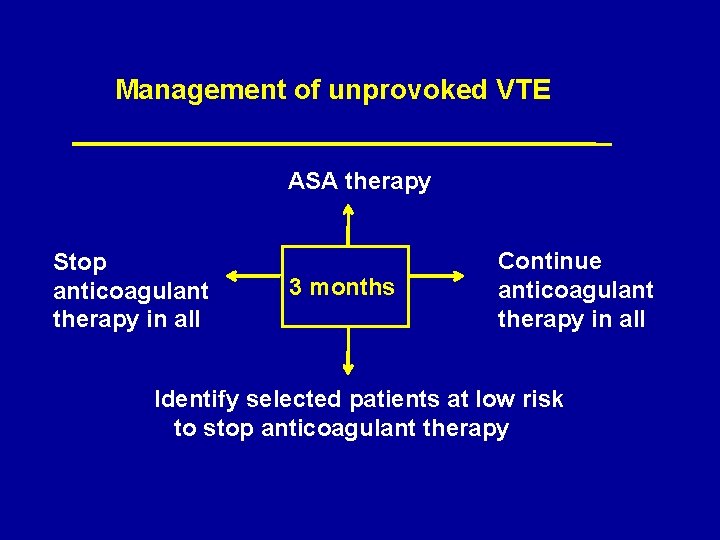
Management of unprovoked VTE ASA therapy Stop anticoagulant therapy in all 3 months Continue anticoagulant therapy in all Identify selected patients at low risk to stop anticoagulant therapy

Duration of Anticoagulant Therapy Factors Influencing Decision ®Risk of recurrent VTE ®Risk of bleeding ®Patient preference
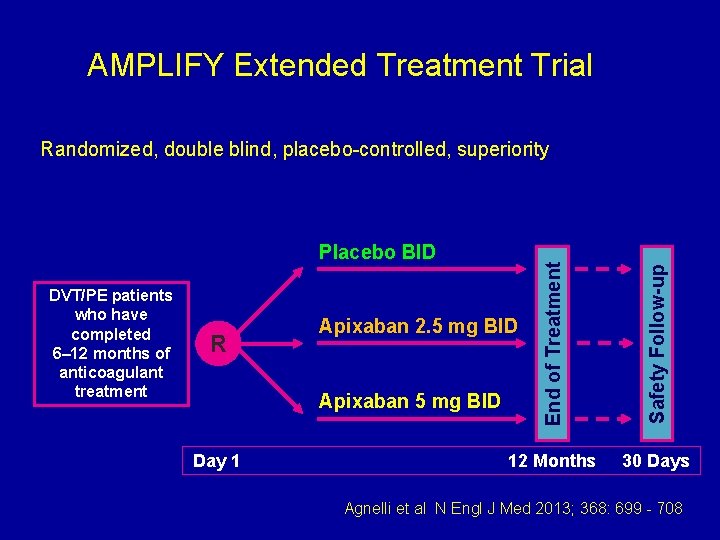
AMPLIFY Extended Treatment Trial Randomized, double blind, placebo-controlled, superiority R 12 Months 30 Days Apixaban 2. 5 mg BID Apixaban 5 mg BID Day 1 Safety Follow-up DVT/PE patients who have completed 6– 12 months of anticoagulant treatment End of Treatment Placebo BID Agnelli et al N Engl J Med 2013; 368: 699 - 708
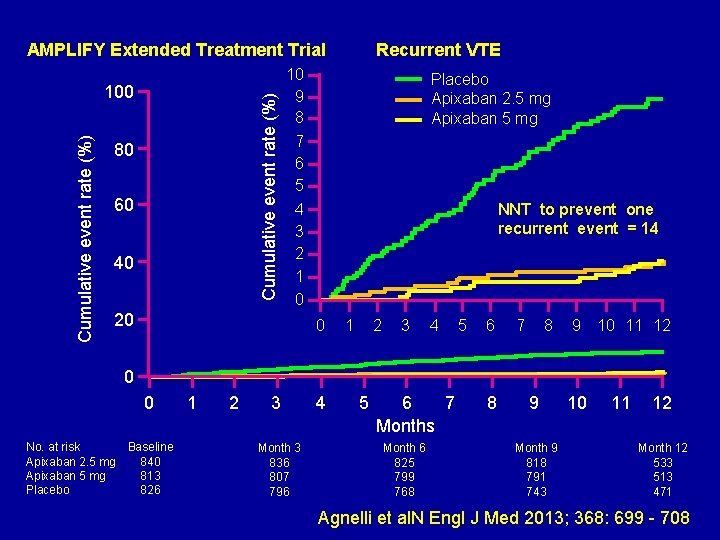
AMPLIFY Extended Treatment Trial Cumulative event rate (%) 100 80 60 40 Recurrent VTE 10 9 8 7 6 5 4 3 2 1 0 20 Placebo Apixaban 2. 5 mg Apixaban 5 mg NNT to prevent one recurrent event = 14 0 1 2 3 4 5 6 7 8 9 10 11 12 0 0 No. at risk Baseline Apixaban 2. 5 mg 840 Apixaban 5 mg 813 Placebo 826 1 2 3 Month 3 836 807 796 4 5 6 7 Months Month 6 825 799 768 8 9 Month 9 818 791 743 10 11 12 Month 12 533 513 471 Agnelli et al. N Engl J Med 2013; 368: 699 - 708
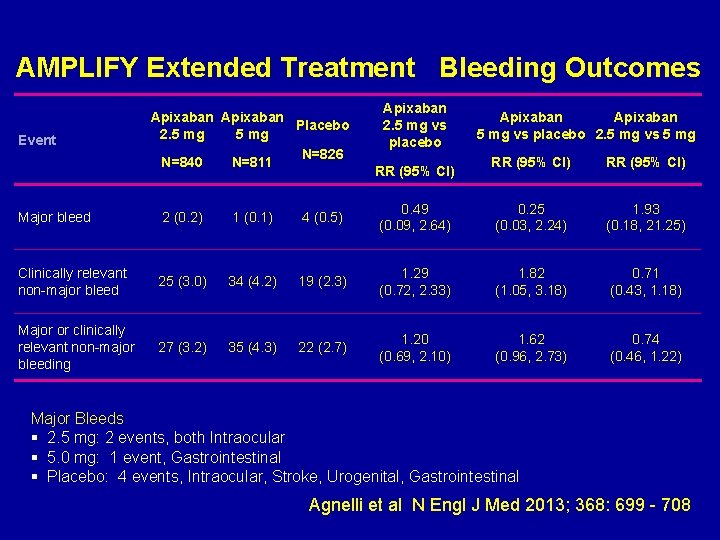
AMPLIFY Extended Treatment Bleeding Outcomes Event Apixaban Placebo 2. 5 mg N=826 N=840 N=811 Apixaban 2. 5 mg vs placebo RR (95% CI) Apixaban 5 mg vs placebo 2. 5 mg vs 5 mg RR (95% CI) Major bleed 2 (0. 2) 1 (0. 1) 4 (0. 5) 0. 49 (0. 09, 2. 64) 0. 25 (0. 03, 2. 24) 1. 93 (0. 18, 21. 25) Clinically relevant non-major bleed 25 (3. 0) 34 (4. 2) 19 (2. 3) 1. 29 (0. 72, 2. 33) 1. 82 (1. 05, 3. 18) 0. 71 (0. 43, 1. 18) Major or clinically relevant non-major bleeding 27 (3. 2) 35 (4. 3) 22 (2. 7) 1. 20 (0. 69, 2. 10) 1. 62 (0. 96, 2. 73) 0. 74 (0. 46, 1. 22) Major Bleeds § 2. 5 mg: 2 events, both Intraocular § 5. 0 mg: 1 event, Gastrointestinal § Placebo: 4 events, Intraocular, Stroke, Urogenital, Gastrointestinal Agnelli et al N Engl J Med 2013; 368: 699 - 708
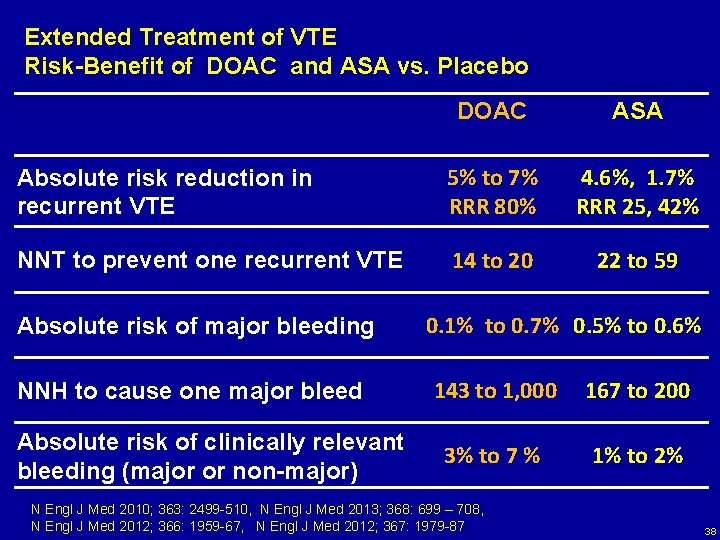
Extended Treatment of VTE Risk-Benefit of DOAC and ASA vs. Placebo DOAC ASA Absolute risk reduction in recurrent VTE 5% to 7% RRR 80% 4. 6%, 1. 7% RRR 25, 42% NNT to prevent one recurrent VTE 14 to 20 22 to 59 Absolute risk of major bleeding NNH to cause one major bleed Absolute risk of clinically relevant bleeding (major or non-major) 0. 1% to 0. 7% 0. 5% to 0. 6% 143 to 1, 000 167 to 200 3% to 7 % 1% to 2% N Engl J Med 2010; 363: 2499 -510, N Engl J Med 2013; 368: 699 – 708, N Engl J Med 2012; 366: 1959 -67, N Engl J Med 2012; 367: 1979 -87 38

Treatment of Venous Thromboembolism Heparin LMWH Fondaparinux Thrombolysis Thrombus Removal IVC filter Rivaroxaban Apixaban Initial (acute) treatment Vitamin K Antagonists LMWH Oral Xa. I or dabigatran Vitamin K Antagonists ASA 100 mg Oral Xa. I or dabigatran Long term-treatment Extended treatment 5 to 10 days 3 to 6 months > 6 months

Hospital Associated VTE ® Age, hospital, surgery, prior VTE, cancer are the major risk factors ® 60% of incident VTE associated with recent hospitalization ® Risk period often extends beyond hospital stay ® Hospital is opportune access point to implement prevention Heit JA. The epidemiology of Venous Thromboembolism in the Community. Arteriosclerosis, Thrombosis, and Vascular Biology 2008; 28: 370 -372
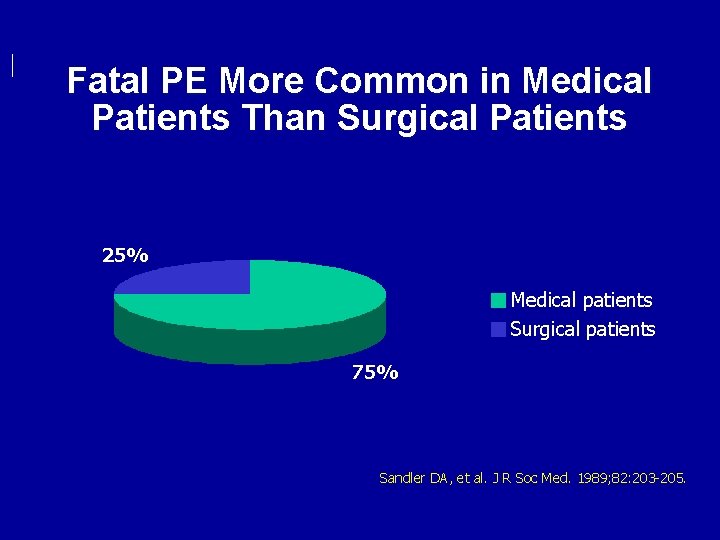
Fatal PE More Common in Medical Patients Than Surgical Patients 25% Medical patients Surgical patients 75% Sandler DA, et al. J R Soc Med. 1989; 82: 203 -205.

Hospitalized Medical Patients ® ACCP Evidence-based Practice Guidelines 2008 LMWH, Unfractionated Heparin, or Fondaparinux (Grade 1 A) Mechanical methods if above contraindicated Patients with heart failure, sever respiratory disease, or confined to bed with 1 or more risk factors (cancer, previous VTE, sepsis, acute neurologic disease, IBD) Duration not specified, 4 to 14 days in clinical trials ® ACP Clinical Practice Guideline 2011 Risk assessment for VTE and bleeding, heparin or related drug unless bleeding risk outweighs benefit Geerts et al. Prevention of venous thromboembolism. American College of Chest Physicians Evidence Based Practice Guidelines (8 th edition). CHEST 2008; 133: 381 -453. Qaseem A et al. Venous thromboembolism prophylaxis in hospitalizedpatients: A Clinical Practice Guideline from the American College of Physicians. Annals of Internal Medicine 2011; 155: 625 -632.

American College of Chest Physicians Guidelines: 9 th Edition • For the Non-surgical and Non-orthopedic surgical chapters, a primary shift is towards an individualized approach of risk assessing the patients’ bleeding risk factors as well as their VTE risk factors for the appropriate thromboprophylactic strategy Kahn et al. CHEST 2012; 141: (2 Suppl): e 195 S-226 S

Clinical Trials of Extended Duration (28 - 35 days) vs 10 days of Prophylaxis in Medical Patients Absolute Risk Differences Study Major VTE Major Bleeding EXCLAIM (n= 5, 963) - 1. 5 % + 0. 5 % enoxaparin 40 mg od NNT 67 NNH 200 MAGELLAN (n= 8, 101) - 1. 3 % + 0. 7 % rivavoxaban 10 mg od NNT 77 NNH 143 ADOPT (n= 6, 528) - 0. 39 % (NS) + 0. 3 % apixaban 2. 5 mg bid NNT ? NNH 333 Hull R et al Ann Intern Med 2010; 153: 8 -18. Cohen et al. N Engl J Med 2013; 368: 513 - 523 Goldhaber et al. N Engl J Med 2011; 365: 2167 -2177

9 th ACCP Recommendations 2. 8. For acutely ill hospitalized medical patients who receive an initial course of thromboprohylaxis, we suggest against extending the duration of thromboprophylaxis beyond the period of patient immobilization or acute hospital stay (Grade 2 B) Kahn et al. CHEST 2012; 141; e 195 S-e 226 S
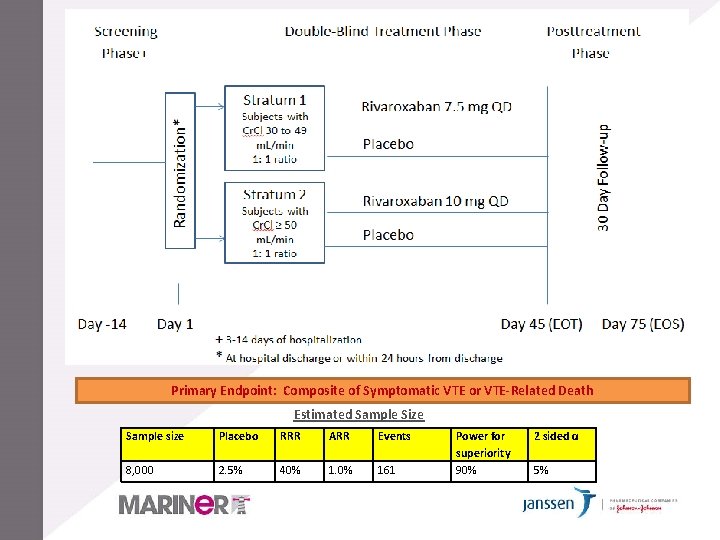
Primary Endpoint: Composite of Symptomatic VTE or VTE-Related Death Estimated Sample Size Sample size Placebo RRR ARR Events 8, 000 2. 5% 40% 161 Power for superiority 90% 2 sided α 5%

Reducing the Disease Burden of VTE ® VTE is a “winnable battle” ® Improve utilization of effective prevention ® If you are hospitalized or having surgery, ask about your VTE risk and prevention ® New oral anticoagulants improve safety and enhance secondary prevention of recurrent VTE ® Improve public awareness of VTE, risk factors and prevention ® Continued research

We’re starting a global movement. Help us stop blood clots and save lives. Join us and 100+ organizations from around the world. Learn how at World. Thrombosis. Day. org World. Thrombosis. Day @Thrombosis. Day #Join. WTDay #WTDay 14 - 48 -
- Slides: 48