Venclexta in Acute Myelogenous Leukemia JULIA BERNAS Introduction

Venclexta in Acute Myelogenous Leukemia JULIA BERNAS

Introduction to MC is a 77 year old female with acute myelogenous leukemia (TP 53 mutation) PMH: Esophageal reflux disease Hypertension Hyperlipidemia Peripheral vascular disease Osteopenia Left breast invasive ductal carcinoma (SP lumpectomy and radiation to left breast) Intracranial hemorrhage status post gamma knife in 2012 Diverticulosis Dysfunctional uterine bleeding

December 2015 – March 2016 Treatment with Cytarabine and Mitoxantrone (with residual disease) After 2 cycles of Decitabine, was admitted for lower left lobe pneumonia and neutropenic fever Started on IV Ceftriaxone and PO Ciprofloxacin

April 2016 After 1 additional dose of Decitabine MC was admitted with: Prolonged weakness and fatigue C. diff positive Dehydration Atrial Fibrillation

May 2016 Last dose of Decitabine Admitted for worsening nausea and bloating and small bowel obstruction Volume overload Diuresed C. diff positive Discharged on oral vancomycin

Medications Acyclovir 400 mg – 1 tablet PO BID Amiodarone HCl 200 mg tablet – 1 tablet PO BID Vancomycin 125 mg PO q 12 h Neurontin Noxafil 300 mg PO daily Zofran prn Oxybutynin Paroxetine 10 mg tablet – 1 tablet PO every other day Prilosec 20 mg tablet Lasix 20 mg po daily
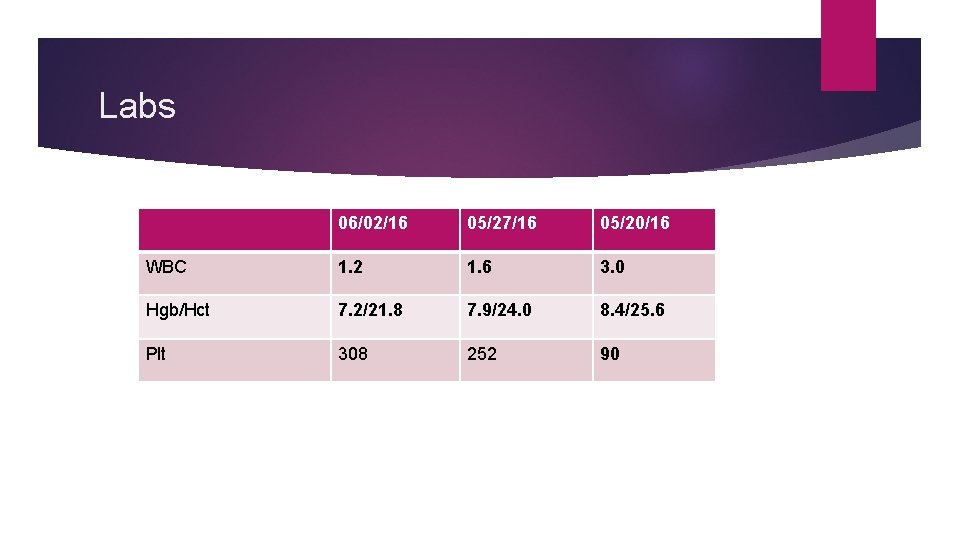
Labs 06/02/16 05/27/16 05/20/16 WBC 1. 2 1. 6 3. 0 Hgb/Hct 7. 2/21. 8 7. 9/24. 0 8. 4/25. 6 Plt 308 252 90

Venclexta

Objectives • Discuss trials that evaluated the effectiveness of Venclexta in AML • Evaluate the safety of Venclexta in AML

Venclexta BCL-2 inhibitor recently approved for treatment of CLL patients with a 17 p deletion who have received at least 1 prior therapy Formulations: 10 mg, 50 mg and 100 mg tablets Dosing (for CLL): Week 1: 20 mg once daily Week 2: 50 mg once daily Week 3: 100 mg once daily Week 4: 200 mg once daily Week 5 and thereafter: 400 mg once daily (continue until disease progression or unacceptable toxicity)

Venetoclax Monotherapy Phase 2 Study Phase 2, open-label, multicenter, dose escalation study to evaluate the safety and efficacy of Venclexta as: Monotherapy in patients with relapsed or refractory (R/R) AML Frontline therapy in patients unfit for intensive treatment 32 patients enrolled
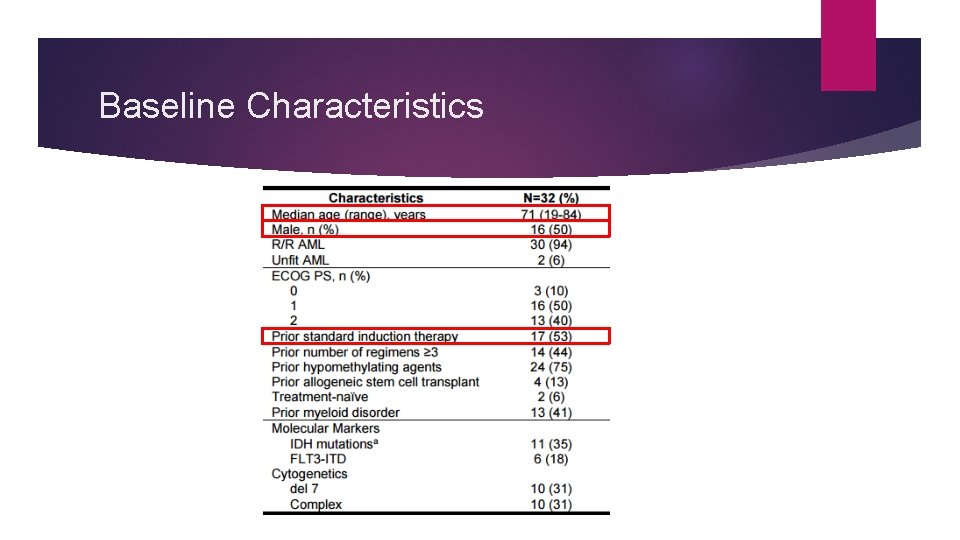
Baseline Characteristics

Endpoints Primary endpoint To evaluate the preliminary efficacy measured by overall response rate Secondary endpoints Evaluation of preliminary safety Assessment of pharmacokinetics
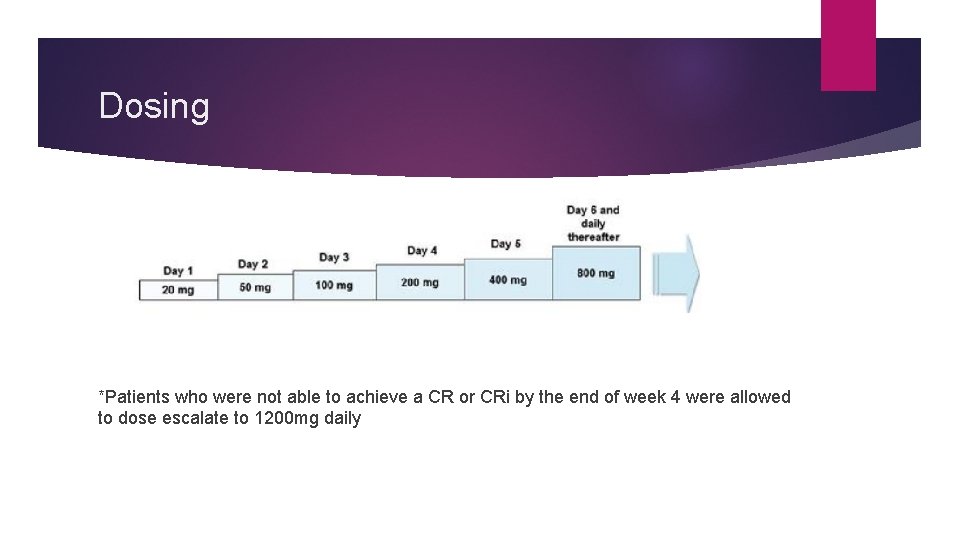
Dosing *Patients who were not able to achieve a CR or CRi by the end of week 4 were allowed to dose escalate to 1200 mg daily

Results 32 patients with R/R AML were enrolled As of the data cut-off date patients were on therapy for a median of 63 days (range 13 -246) 31 patients discontinued therapy due to: Progressive disease (28) Adverse events (1) – terminal ileitis Withdrawn consent (1) Proceed to transplant (1)

Overall Response Objective response (CR + CRi) – 6 patients CR – 2 patients CRi – 4 patients Anti-leukemic Activity – 6 patients Treatment Failure – 20 patients Preliminary analysis of 11/32 patients with IDH mutation found objective response in 4 (36%) including 2 (18%) with a CR and 2 (18%) with a CRi

Adverse Reactions Most common (>20%): Hypokalemia Nausea Hypokalemia Vomiting Cough Diarrhea Epistaxis Hypomagnesemia Hypocalcemia Pneumonia Dyspnea Fatigue Hypotension Headache Peripheral Edema Abdominal Pain Pyrexia Febrile Neutropenia

Venetoclax in Combination with Decitabine or 5 -Azacitidine Design: Phase 1 b Open-label Nonrandomized 2 -arm 2 stage study To evaluate Venetoclex in treatment naïve patients with AML > 65 yo not eligible for standard induction therapy due to comorbidity or other factors

Patient Assignment Arm A: Venetoclax + Decitabine Arm B: Venetoclax + 5 -Azacitidine 20 mg/m 2, Days 1 -5 for 28 -day cycles 75 mg/m 2, Days 1 -7 for 28 -day cycles Patients were hospitalized and received TLS prophylaxis 72 h prior to dosing of Venetoclax in cycle 1
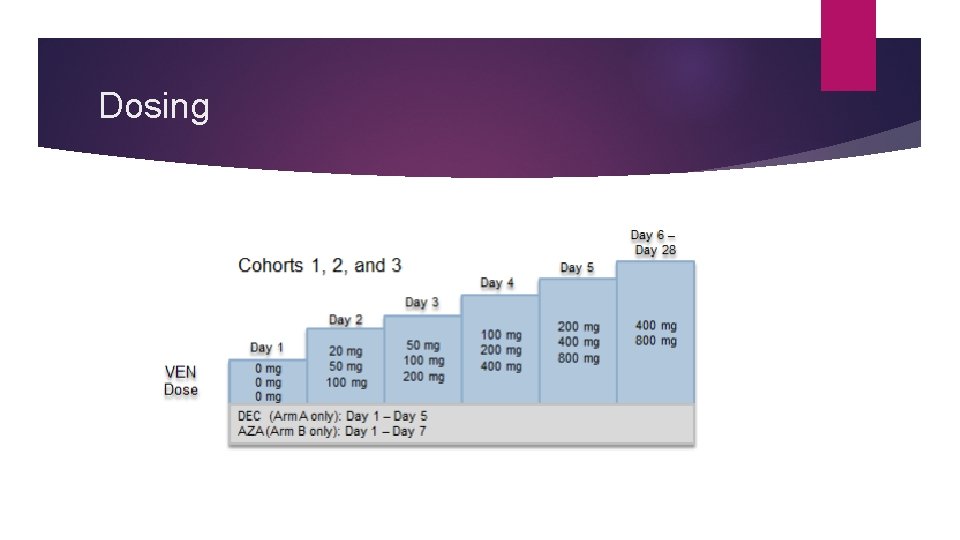
Dosing
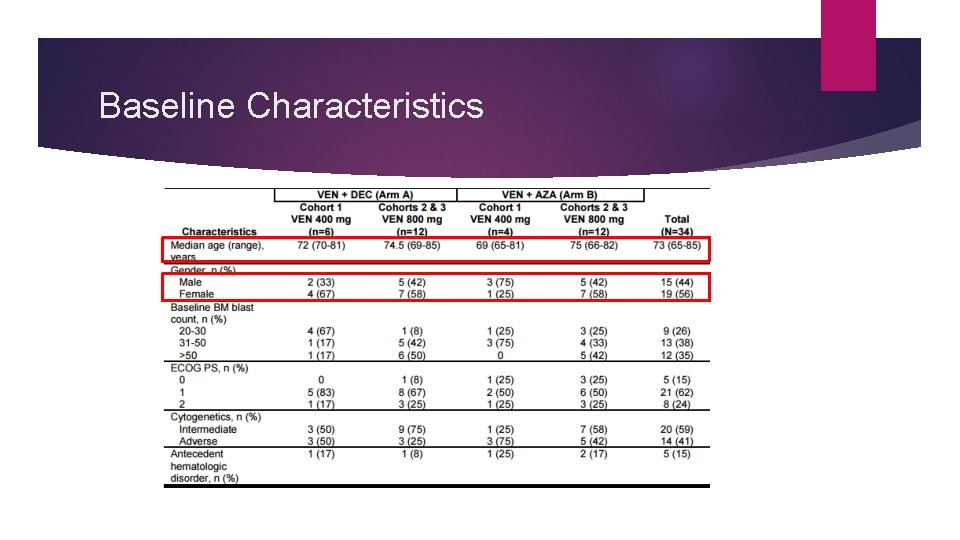
Baseline Characteristics

Endpoints Primary Endpoints Assessment of safety and PK of Venetoclax in combination with 5 -AZA or Decitabine Secondary Endpoints: Evaluation of Preliminary efficacy Changes in clinical laboratory test results Percent of patients who progress to stem cell transplant

Results September 19, 2015 - Analysis was conducted on 34 patients with AML Of the 34 patients 23 discontinued therapy due to: PD (6) Allogenic Stem Cell Transplant (6) Withdrawn Consent (6) Resistant Disease (2) AE not due to AML progression (3) 11 Remain 7 on Arm A and 4 on Arm B (Max tolerated dose was not reached on either arm)

Results Overall Response Rate for Patients Receiving 400 mg daily 50% (3/6) with Decitabine 83% (10/12) with Azacitadine 800 mg daily – 100% (4/4) in combination with Decitabine 75% (9/12) in combination with 5 -Azacitadine

Safety No dose-limiting toxicities or TLS reported Treatment was delayed/interrupted for 24 patients due to neutropenia and/or other AEs, and 13 patients delays occurred in Cycle 2 to allow for neutropenia recovery Adverse Effects (> 30% of patients) Nausea Diarrhea Febrile Neutropenia Peripheral Edema

What Happened to MC? Currently in a rehab facility Started on Venclexta on May 31 st 20 mg PO daily x 1 week 50 mg PO daily x 1 week 100 mg PO daily x 1 week 200 mg PO daily x 1 week We will be following up with MC at her appointment tomorrow

References A phase 2 study of ABT – 199 in subjects with acute myelogenous leukemia (AML), NLM Identifier: NCTNCT 01994837. Clinical. Trials. gov Web site. https: //clinicaltrials. gov/ct 2/show/NCT 01994837 Updated December 18, 2015. Accessed June 4, 2016. Phase 1 b acute myelogenous leukemia (AML) study with ABT-199 + Decitabine or Azacitidine (chemo combo). NLM identifier: NCT 02203773. Clinical. Trials. gov Web site. http: //clinicaltrials. gov/ct 2/show/NC 02203773. Updated February 15, 2016. Accessed June 4, 2016. Di. Nardo C, Pollyea D, Pratz K, et al. A phase 1 b study of venetoclax (ABT-199/GDC-0199) in combination with decitabine or azacitidine in treatment-naive patients with acute myelogenous leukemia who are ≥ 65 years and not eligible for standard induction therapy. Presented at: American Society of Hematology Annual Meeting; December 5 -8, 2015; Orlando, FL. Oral Presentation 327. Konopleva M, Pollyea DA, Potluri J, et al. A phase 2 study of venetoclax (ABT-199/GDC-0199) in patients with acute myelogenous leukemia (AML). Presented at: American Society of Hematology Annual Meeting; December 6 -9, 2014; San Francisco, CA

Questions?
- Slides: 28