Vascular Access and Vascular Closure Devices February 21

Vascular Access and Vascular Closure Devices February 21, 2010 Nelson Lim Bernardo, MD Washington Hospital Center

DISCLOSURES Nelson L. Bernardo, MD Honoraria – The Medicines Company, Cordis, a Johnson & Johnson company, Bristol-Myers Squibb/Sanofi Pharmaceuticals Partnership

Faculty Disclosure No conflict of interest related to this presentation
Vascular Access • Successful vascular access is paramount in the performance of catheterization – Diagnostic or Therapeutic/Interventional • Uncomplicated hemostasis of the vascular access site is key to the success of the invasive procedure • GOAL: “ 0” groin complication Ø 1 -stick during access Ø Hemostasis ASAP

Vascular Access for Dxtic/Txtic caths • Vascular access complications will happen Ø Diagnostic caths – 0. 8 - 1. 8% Ø Therapeutic/PCI – 1. 5 - 9% Anti-coagulation, anti-platelet Tx, bigger sheath size, delayed sheath removal, etc. • Complications lead to: Ø Increased length of stay Ø Morbidity/Mortality Ø Adds to health care cost
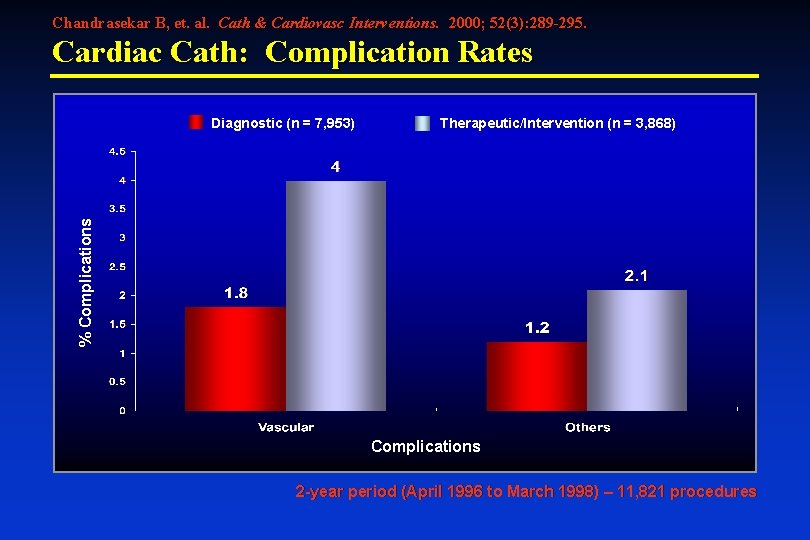
Chandrasekar B, et. al. Cath & Cardiovasc Interventions. 2000; 52(3): 289 -295. Cardiac Cath: Complication Rates Therapeutic/Intervention (n = 3, 868) % Complications Diagnostic (n = 7, 953) Complications 2 -year period (April 1996 to March 1998) – 11, 821 procedures

Vascular Access Site Management: Complications • Femoral artery complications: 2 -10% • Complications: Ø Groin Hematoma Ø Dissection (intimal) (1 -10%) Ø Vessel Laceration Ø Pseudoaneurysm Ø Neural injury (1 -6%) Ø AV fistula Ø Infection (<1%) Ø Venous thrombosis Ø Retroperitoneal bleed (+/- right heart cath) (0. 1 -0. 9%) Ø Sheath/catheter clot Ø Acute closure (<1%)

Vascular Access Site Complications • Factors increasing risk: Ø Advanced age - >70 years Ø Female Ø Percutaneous coronary intervention (PCI) Ø Chronic kidney disease (CKD) Ø + venous sheath • Operator driven/dependent
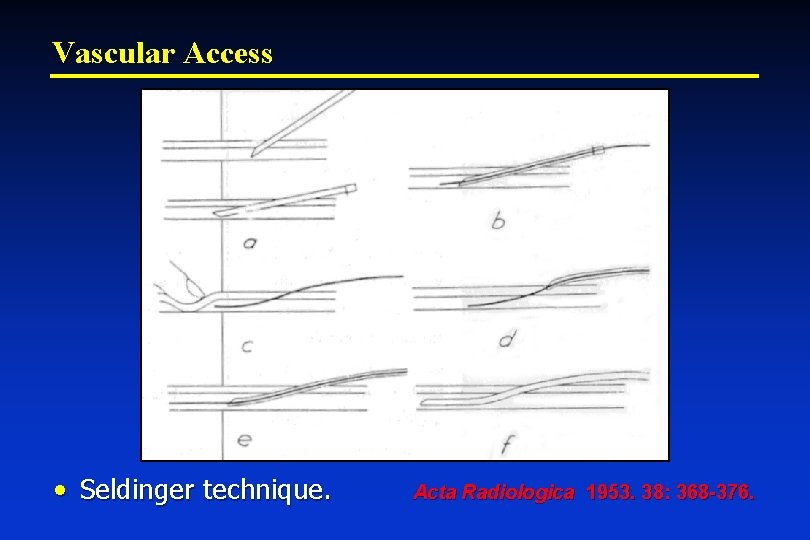
Vascular Access • Seldinger technique. Acta Radiologica 1953. 38: 368 -376.

Vascular Access: Simple rules • 1 -stick – anterior wall only with good blood return “NO-NO” – multiple sticks, ‘through & through’ else: Don’t rush – compress until hemostasis is achieved • Needle to access artery Ø Use of micropuncture 21 -gauge access needle Ø 21 -gauge ‘hole’ is more ‘forgiving’ if puncture fails Ø For ultrasound guidance – ‘sono’ tip

Vascular Access of Common Femoral Artery • U. S. – Use of the common femoral artery (CFA) as the primary access site is still the most common • Puncture site (of CFA) is key “Not too-high” (RPH) & “Not too-low” (PSA) Ø Bony landmark Ø Skin/inguinal crease Ø Pulsation Ø Guidance – doppler needle, ‘road-map’ Ø Ultrasound ‘direct’ visualization
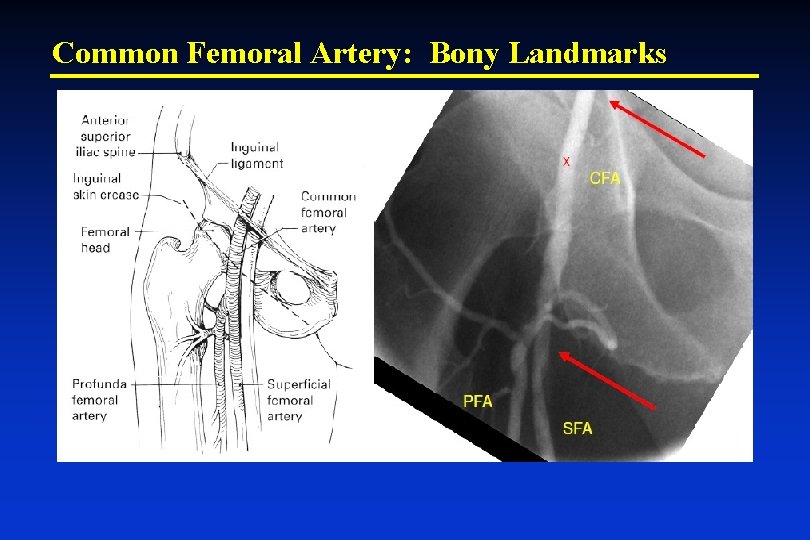
Common Femoral Artery: Bony Landmarks
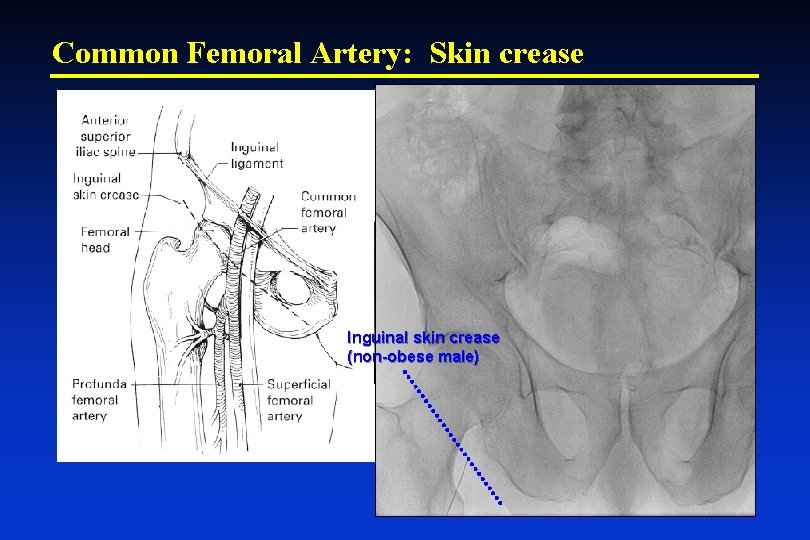
Common Femoral Artery: Skin crease Inguinal skin crease (non-obese male)
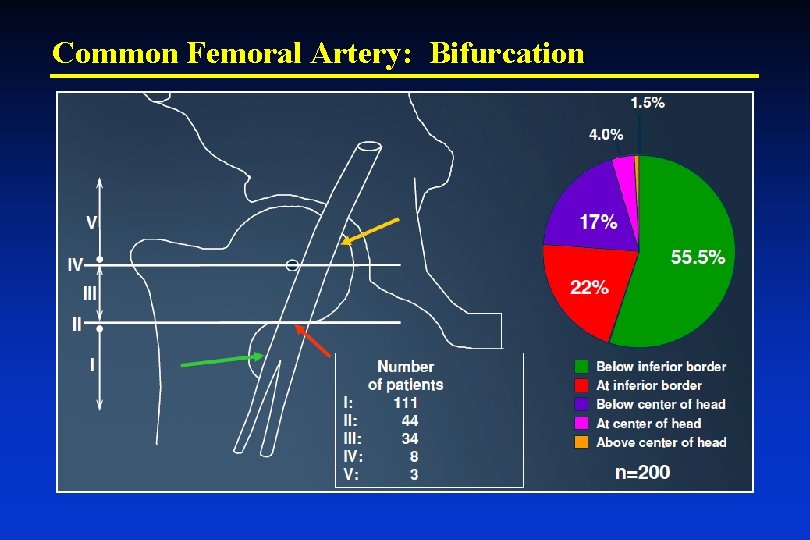
Common Femoral Artery: Bifurcation
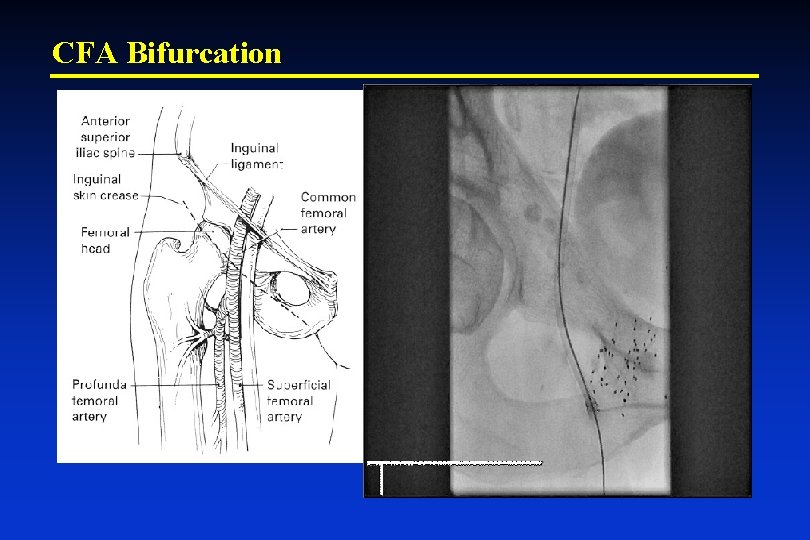
CFA Bifurcation
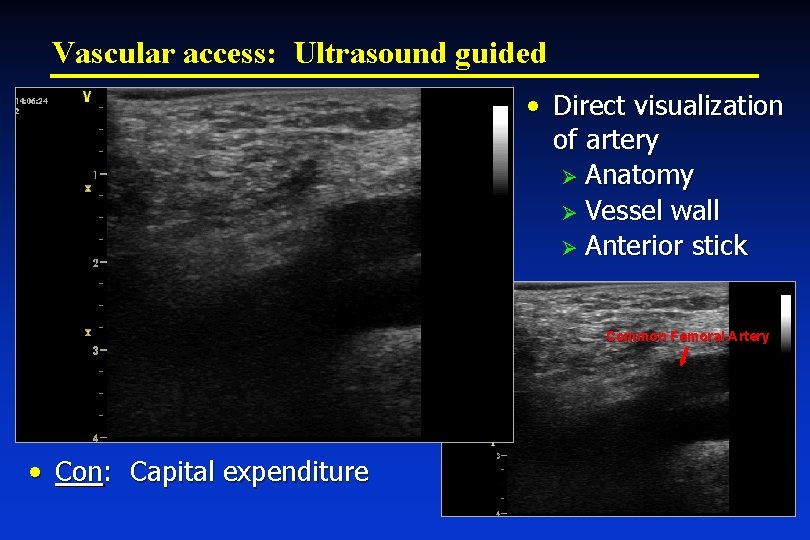
Vascular access: Ultrasound guided • Direct visualization of artery Ø Anatomy Ø Vessel wall Ø Anterior stick Common Femoral Artery • Con: Capital expenditure
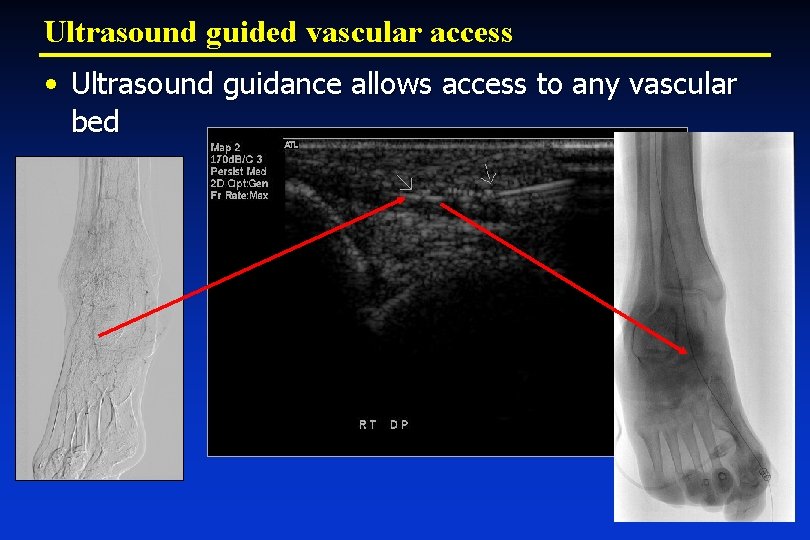
Ultrasound guided vascular access • Ultrasound guidance allows access to any vascular bed

Hemostasis of Vascular Access Site • Manual compression – ‘standard of care’ Ø With anticoagulation, i. e. , Heparin – need to check ACT before pulling Ø Delayed sheath removal ~ increase complication • Adjunctive mechanical compression devices Ø Pressure dressings – ? ? evidence base Ø Compression devices – concern about compression necrosis

Hemostasis: Vascular Closure Device (VCD) • Rationale for Use of VCD: Ø Earlier sheath removal – hemostasis Ø Patient comfort – earlier ambulation Ø Reduced Length of stay – healthcare cost Ø Patient preference • ? ? compelling evidence for lower complication rates. [Koreny 2004, Nikolsky 2004, Applegate 2006]

Arora N, et. al. Am Heart J. 2007; 153: 606 -611. Risk of Vascular Complication with VCD • Single-center. Jan. 1, 2002 to Dec. 31, 2005. Ø 12, 937 patients – 53% (n = 6, 913) underwent PCI Ø 77% (n = 9, 996) usage of vascular closure device (VCD) • Contraindication to VCD deployment: Ø Arteriotomy at or below CFA bifurcation Ø CFA with >50% stenosis (Ca++ or plaque) Ø CFA diameter <5 mm • VCD Usage: Ø Angioseal >> Perclose, Vasoseal, Duett
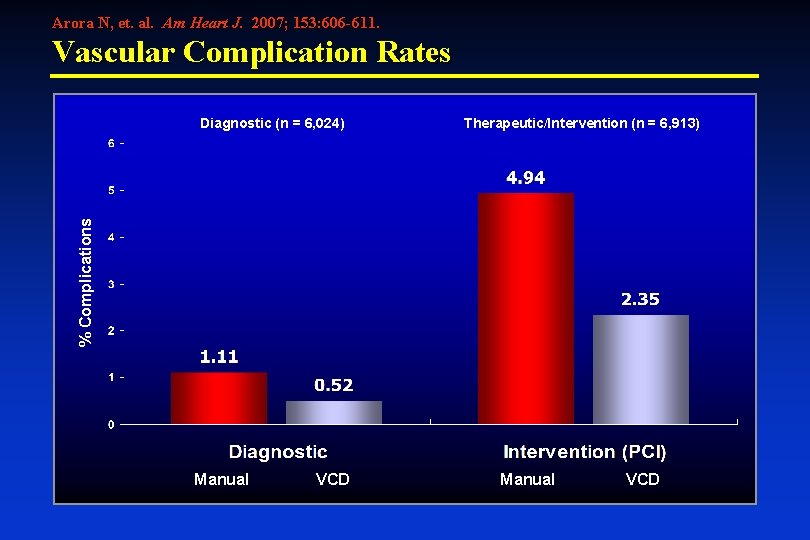
Arora N, et. al. Am Heart J. 2007; 153: 606 -611. Vascular Complication Rates Therapeutic/Intervention (n = 6, 913) % Complications Diagnostic (n = 6, 024) Manual VCD
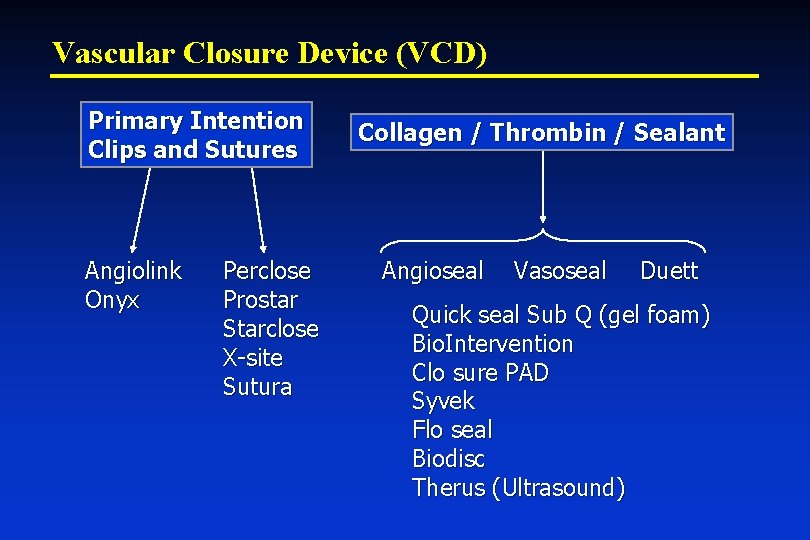
Vascular Closure Device (VCD) Primary Intention Clips and Sutures Angiolink Onyx Perclose Prostar Starclose X-site Sutura Collagen / Thrombin / Sealant Angioseal Vasoseal Duett Quick seal Sub Q (gel foam) Bio. Intervention Clo sure PAD Syvek Flo seal Biodisc Therus (Ultrasound)

Vascular Closure Device (VCD) • Device Types – NO “ideal” device • Device selection ~ Operator “comfort zone”

Vascular Closure Device (VCD) • Contraindications to Deployment Ø Multiple sticks, posterior sticks Ø ‘Low’ sticks Ø PAD - Calcified tortuous vessel Ø Arteriotomy through ‘plaque’ Ø ‘Gut feeling’ – Trust instinct, if there is any doubt, do not deploy Ø Inexperience/unfamiliarity with particular device
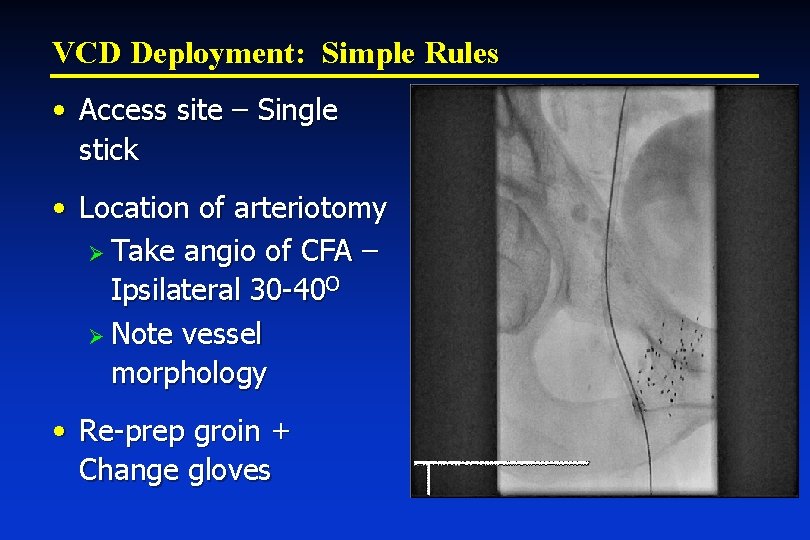
VCD Deployment: Simple Rules • Access site – Single stick • Location of arteriotomy Ø Take angio of CFA – Ipsilateral 30 -40 O Ø Note vessel morphology • Re-prep groin + Change gloves

Vascular Closure Device (VCD) • Use of vascular closure devices for hemostasis is not devoid of access site complications but is associated with device related/device deployment complications. • Complications: Ø Acute vessel closure Ø Subacute occlusion Ø Infection Ø Device malfunction
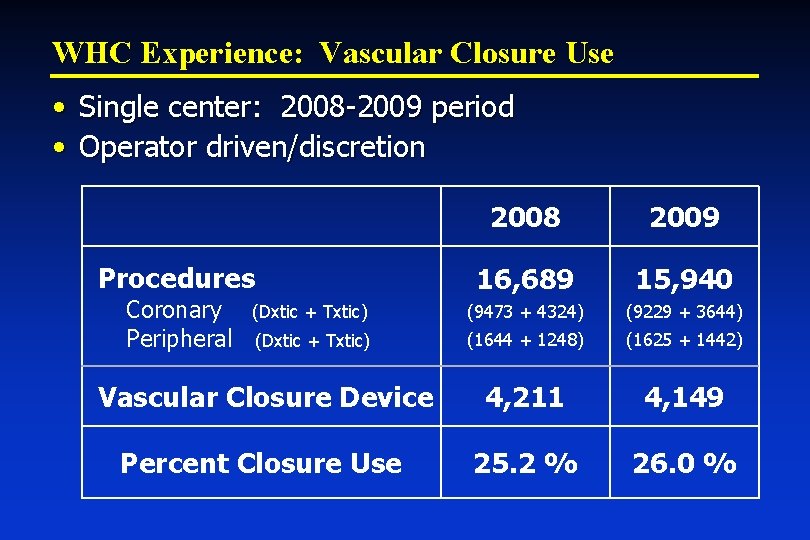
WHC Experience: Vascular Closure Use • Single center: 2008 -2009 period • Operator driven/discretion 2008 2009 16, 689 15, 940 (9473 + 4324) (9229 + 3644) (1644 + 1248) (1625 + 1442) Vascular Closure Device 4, 211 4, 149 Percent Closure Use 25. 2 % 26. 0 % Procedures Coronary (Dxtic + Txtic) Peripheral (Dxtic + Txtic)
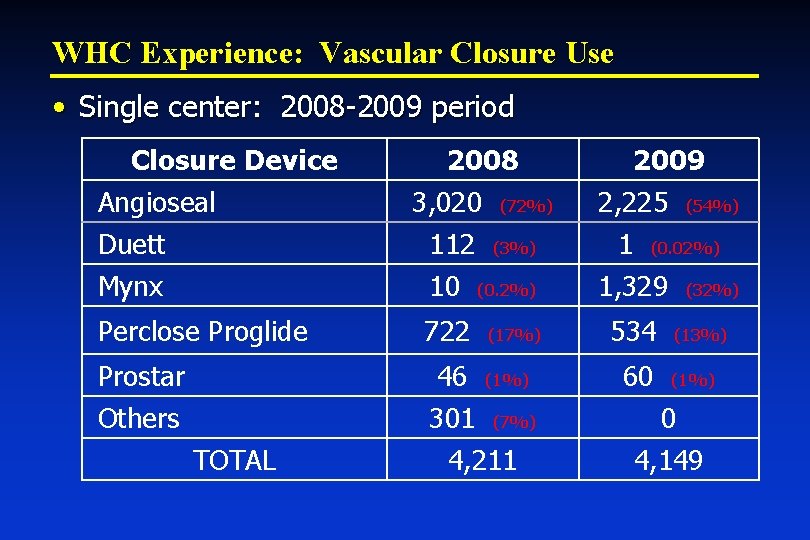
WHC Experience: Vascular Closure Use • Single center: 2008 -2009 period Closure Device Angioseal Duett 2008 3, 020 (72%) 112 (3%) 2009 2, 225 (54%) 1 (0. 02%) Mynx 10 (0. 2%) 1, 329 (32%) Perclose Proglide 722 (17%) 534 (13%) Prostar Others 46 (1%) 301 (7%) 4, 211 60 (1%) 0 4, 149 TOTAL

Hemostasis of Access Site • Earlier sheath removal, earlier hemostasis Better outcome. • Post-intervention: With anti-coagulation on board, VCD allows ‘immediate’ sheath removal. • However: If VCD is not an option. . . Heparin – Reverse with Protamine if OK Bivalirudin – Wait >2 hours
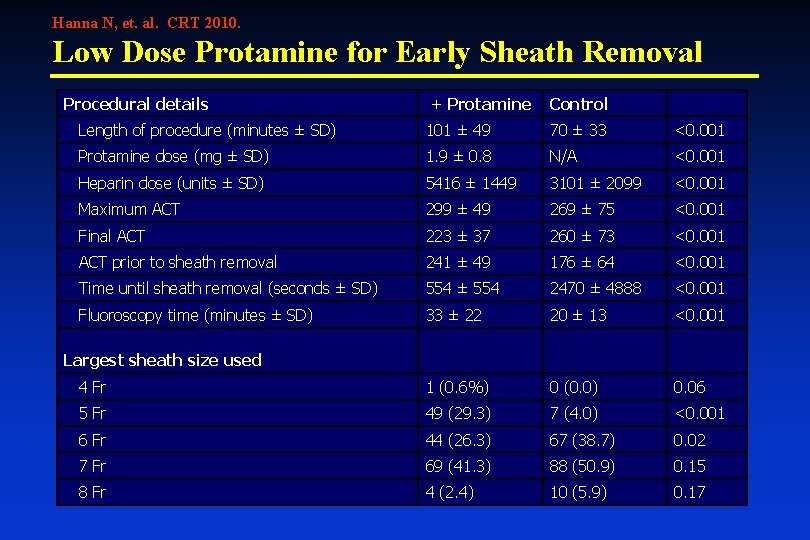
Hanna N, et. al. CRT 2010. Low Dose Protamine for Early Sheath Removal Procedural details + Protamine Control Length of procedure (minutes ± SD) 101 ± 49 70 ± 33 <0. 001 Protamine dose (mg ± SD) 1. 9 ± 0. 8 N/A <0. 001 Heparin dose (units ± SD) 5416 ± 1449 3101 ± 2099 <0. 001 Maximum ACT 299 ± 49 269 ± 75 <0. 001 Final ACT 223 ± 37 260 ± 73 <0. 001 ACT prior to sheath removal 241 ± 49 176 ± 64 <0. 001 Time until sheath removal (seconds ± SD) 554 ± 554 2470 ± 4888 <0. 001 Fluoroscopy time (minutes ± SD) 33 ± 22 20 ± 13 <0. 001 4 Fr 1 (0. 6%) 0 (0. 0) 0. 06 5 Fr 49 (29. 3) 7 (4. 0) <0. 001 6 Fr 44 (26. 3) 67 (38. 7) 0. 02 7 Fr 69 (41. 3) 88 (50. 9) 0. 15 8 Fr 4 (2. 4) 10 (5. 9) 0. 17 Largest sheath size used
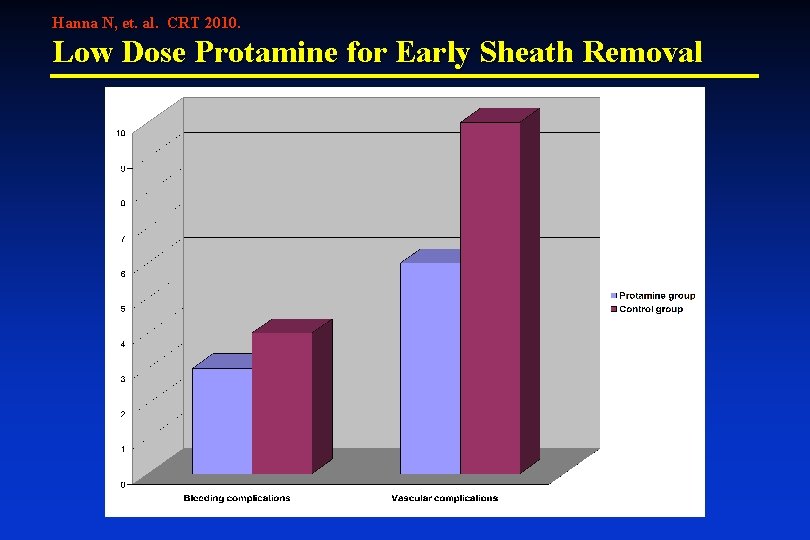
Hanna N, et. al. CRT 2010. Low Dose Protamine for Early Sheath Removal

Percutaneous EVAR of AAA (with no cutdown) • Subgroup of AAA patients who are ‘high-risk’, even to undergo surgical cutdown for vascular access. • Reports of successful ‘purely’ percutaneous implantation of stent-grafts with the use of vascular closure device for hemostasis, i. e. Perclose Proglide or Prostar. • Challenge: Achieve hemostasis in a 24 French access site (8 -mm ‘hole’).

WHC Experience: Percutaneous EVAR • Single center experience, consecutive EVAR patients. February 2008 to August 2009 – 30 patients • Demographics: Age: 77 ± 8 years (56 - 95 y. o. ) Sex: Male = 22 (73%) Female = 8 (27%) Race: Caucasian = 21 (70%) African American = 9 (30%) • Co-morbidities: CAD = 21 (70%) DM = 8 (27%) HTN = 27 (90%) COPD (O 2) = 4 (13%) Lipids = 29 (97%)

WHC Experience: Percutaneous EVAR • 2 patients excluded: Ø Patient #2 – needed right iliac bypass conduit; contralateral limb closed with Prostar. Ø Patient #16 – “calcified” femoral artery, elective cutdown; contralateral limb closed with Prostar. • Technique: Ø Access obtained via both common femoral arteries using the Micropuncture technique. Ø Pre-deployment of Prostar closure device. • Local anesthesia +/- conscious sedation – patient typically awake. • Successful hemostasis obtained = 28/28.

WHC Experience: Percutaneous EVAR • ‘Purely’ percutaneous implantation of EVAR is feasible and safe – even with the current “large” 24 French device systems. • This allows the ‘option’ of treating ‘high-risk’ patients with AAA who are not good surgical candidate. • Allows the implantation of stent-graft under local anesthesia short hospital stay, (-) post-anesthesia care • Proper training and appropriate case selection are critical to optimize outcomes & minimize complications.
WHC Experience: Percutaneous EVAR Only drawback is. . . “no bragging rights of having had an aneurysm repair” CB 6/2009
Conclusions: • Successful vascular access with uncomplicated hemostasis of the vascular access site is key to the success of any invasive procedure. • Adjunctive use of ‘new’ devices and imaging modalities improves success in vascular access and outcome of hemostasis management. • Proper training and appropriate case selection are critical to optimize outcomes in device usage & minimize complications.

Thank you. Have a Good Day! On the road to Mount Everest ‘Touching the Bells’
- Slides: 38