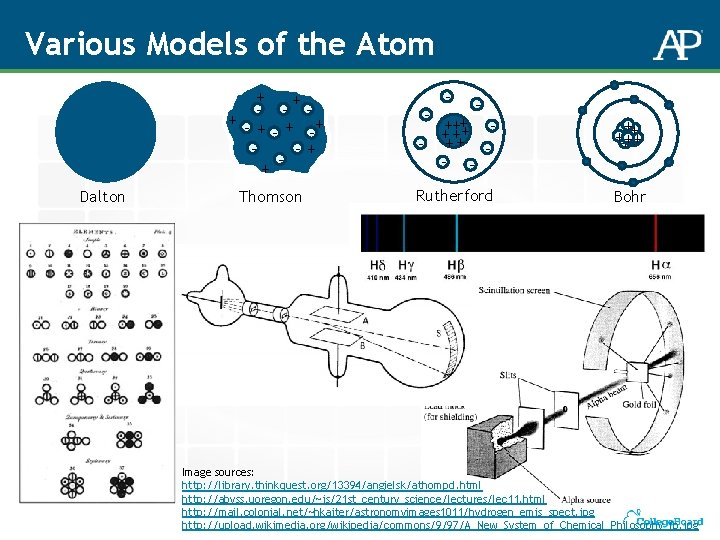
Various Models of the Atom Dalton Thomson Rutherford



![Periodic Table [Enter Presentation Title in Header and Footer] 6 Periodic Table [Enter Presentation Title in Header and Footer] 6](https://slidetodoc.com/presentation_image_h/b668189a26d8998094a39c5da78bf94b/image-6.jpg)










![But not all elements ‘follow the rules’ 1 s [Ar]4 s 13 d 5 But not all elements ‘follow the rules’ 1 s [Ar]4 s 13 d 5](https://slidetodoc.com/presentation_image_h/b668189a26d8998094a39c5da78bf94b/image-28.jpg)















- Slides: 68

Various Models of the Atom + + - - + + -+ -+ + Dalton Thomson - - - ++ + ++++ - - + +++++++ Rutherford Bohr Image sources: http: //library. thinkquest. org/13394/angielsk/athompd. html http: //abyss. uoregon. edu/~js/21 st_century_science/lectures/lec 11. html http: //mail. colonial. net/~hkaiter/astronomyimages 1011/hydrogen_emis_spect. jpg http: //upload. wikimedia. org/wikipedia/commons/9/97/A_New_System_of_Chemical_Philosophy_fp. jpg

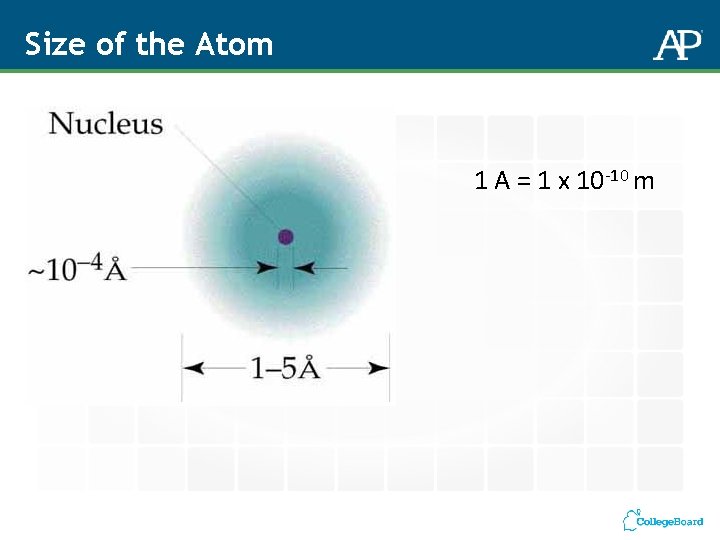
Size of the Atom 1 A = 1 x 10 -10 m

Atomic Numbers, Mass Numbers, Isotopes

Atomic Weights 1 amu = 1. 66054 x 10 -24 g 1 g = 6. 02214 x 1023 amu Based on the measured and calculated mass of 1 Hydrogen atom Derived from the mass of 1 Hydrogen atom – how many it would take to add up to 1 gram

Atomic Weights and Isotopes are just versions of atoms with the same number of protons – some heavier than others, but they are the same element. Atomic Weight = Σ[(isotope mass) x (fractional isotope abundance)]
![Periodic Table Enter Presentation Title in Header and Footer 6 Periodic Table [Enter Presentation Title in Header and Footer] 6](https://slidetodoc.com/presentation_image_h/b668189a26d8998094a39c5da78bf94b/image-6.jpg)
Periodic Table [Enter Presentation Title in Header and Footer] 6

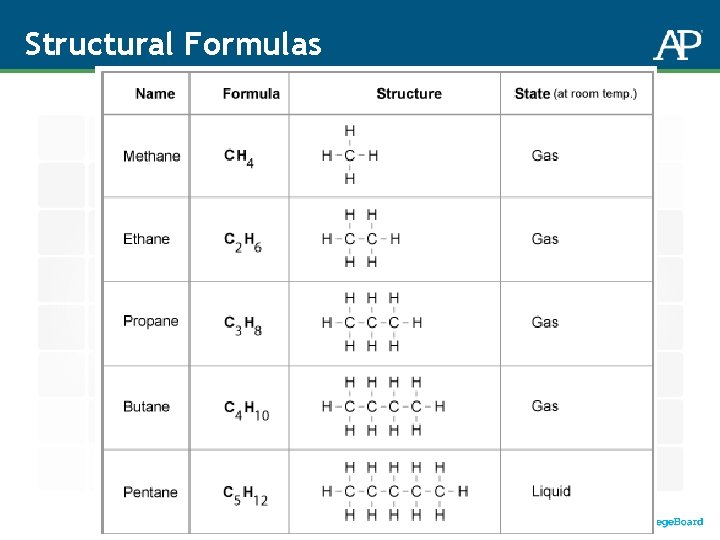
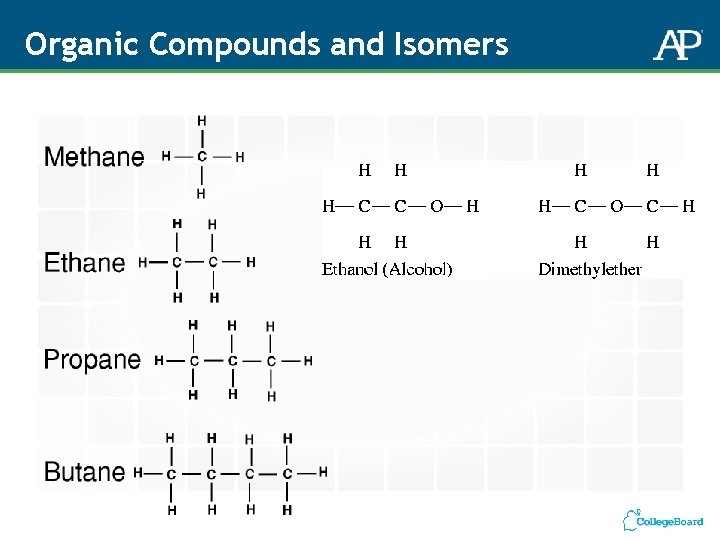
Molecules and Molecular Compounds Chemical Formulas: CO 2 H 2 O C 3 H 8 Subscripts tell you how many atoms of each element are in the molecule. No subscript = 1 Molecular Compounds: Contain more than one type of atom – most contain non-metals Molecular Formulas: Show the actual numbers of atoms Example: Ethane = C 2 H 6 Empirical Formulas: Show the lowest whole number ratio of atoms – the relative amounts Example: Ethane = CH 3

Structural Formulas

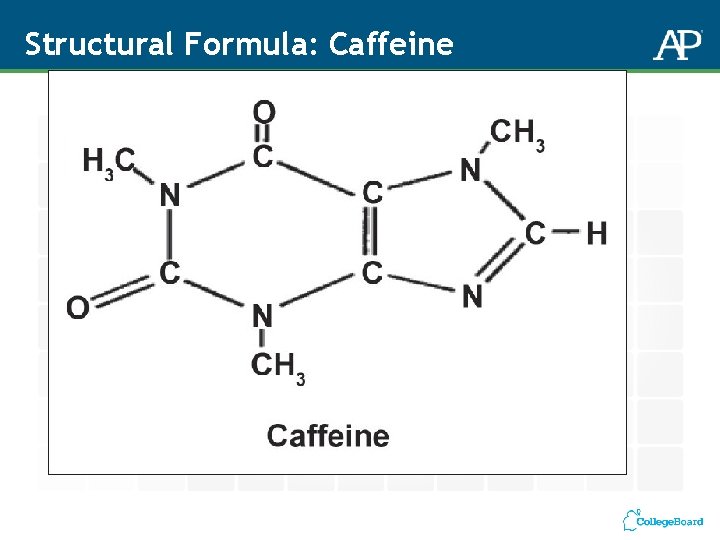
Structural Formula: Caffeine

Space Filling Model: Crystal Meth

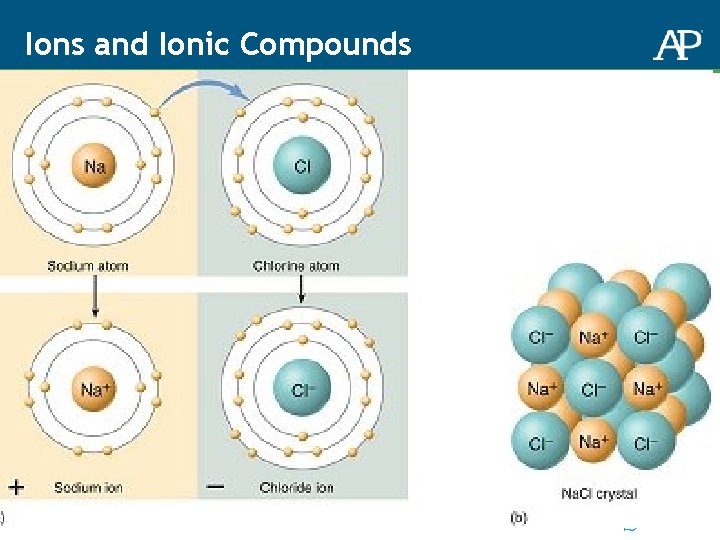
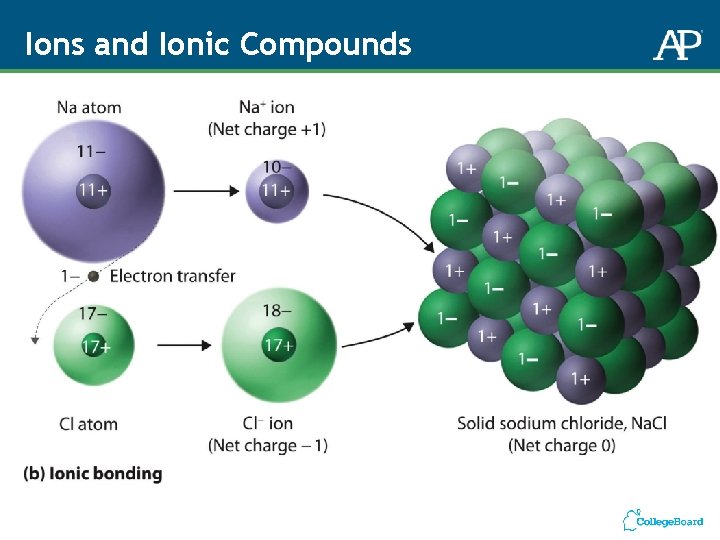
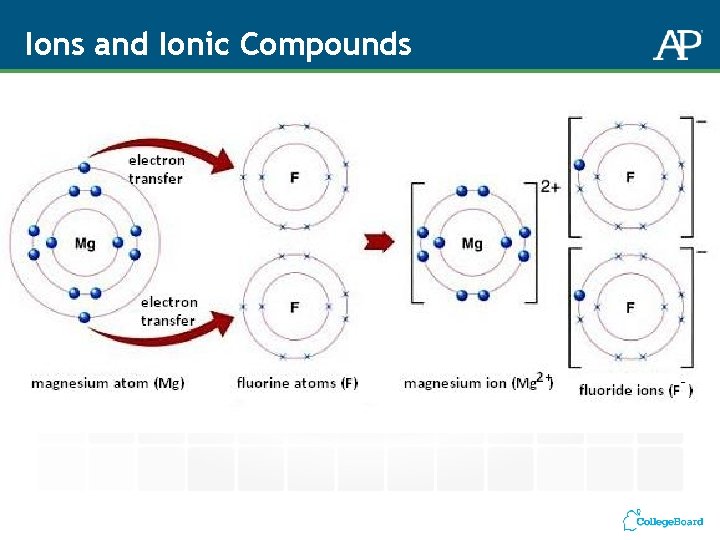
Ions and Ionic Compounds

Ions and Ionic Compounds

Ions and Ionic Compounds

Common Ion Names Memorize!

Naming Ionic and Molecular Compounds Ionic? Metal and Non-metal 1. Cation first: If it is type II, use the correct name 2. Anion second 3. Replace end of Anion with “ide” 4. If Anion is polyatomic, use its name Molecular? Non -metals only 1. Least electronegative atom goes first 2. Change the ending to ide 3. Use prefixes (see page 68)

Organic Compounds and Isomers

HOMEWORK TONIGHT Watch video on electron configurations at bozemanscience. com http: //www. bozemanscience. com/apchem-005 -electron-configuration/

How do we know? hν - hν - +

Tuesday 9. 23. 14 Can you complete these sentences? The Bohr Model of the atom shows us… However, it doesn’t… Be ready to share your thoughts

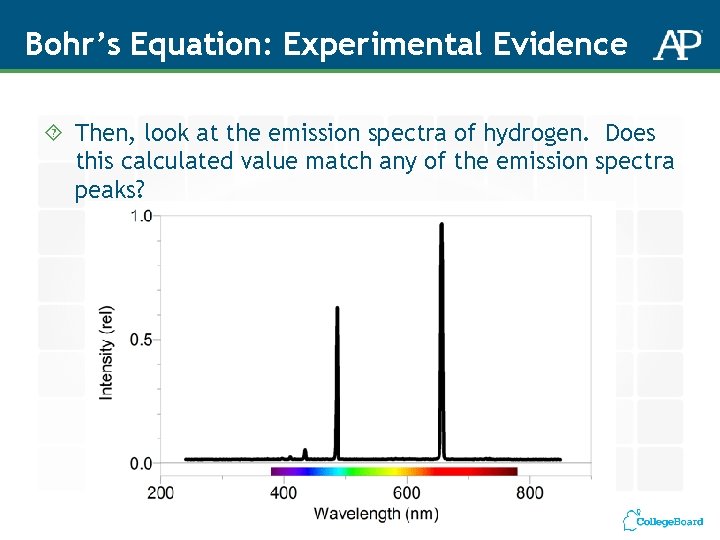
Bohr’s Equation: Experimental Evidence 1. First by yourself, then with your team, use the Rydberg equation to calculate the Energy of an electron moving from n = 3 to n = 1 OR from n = 2 to n =1. Just choose one. 2. Then, using the two equations below, calculate the wavelength of of this electron. You have to be able to manipulate these equations – what do they have in common? Equation 1 E = hv Where h is Planck’s constant = 6. 626 x 10 -34 J and where v represents frequency of UV light Equation 2 c = λv Where again v is frequency of UV light, and λ represents the wavelength, and c is the speed of light at 3. 00 x 108 m/s Hint: first solve for v, frequency of UV light

Bohr’s Equation: Experimental Evidence Then, look at the emission spectra of hydrogen. Does this calculated value match any of the emission spectra peaks?

Warm Up: 7 mins, You might present! 1. First by yourself, then with your team, use the Rydberg equation to calculate the ΔE of an electron moving from n = 4 to n= 3 2. Then, using the two equations below, calculate the wavelength of of this electron. Show your work using dimensional analysis. The Equation 1 E = hv Where h is Planck’s constant = 6. 626 x 10 -34 J s and where v represents frequency of UV light Equation 2 c = λv Where again v is frequency of UV light, and λ represents the wavelength, and c is the speed of light at 3. 00 x 108 m/s Hint: first solve for v, frequency of UV light

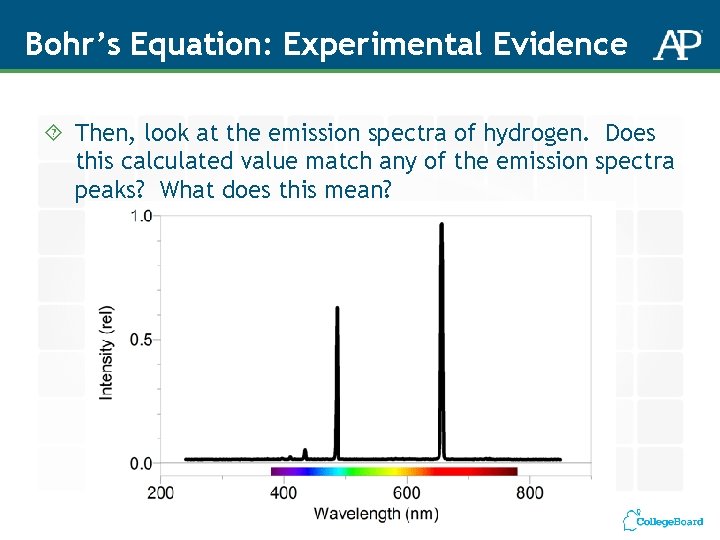
Bohr’s Equation: Experimental Evidence Then, look at the emission spectra of hydrogen. Does this calculated value match any of the emission spectra peaks? What does this mean?

Plan for This Week and Next - Homework: Watch Video and Do “From Shells to subshells” Assignment - http: //www. bozemanscience. com/ap-chem 004 -coulombs-law

Tuesday 9. 23. 14 Can you complete these sentences? The Bohr Model of the atom shows us… However, it doesn’t… Be ready to share your thoughts

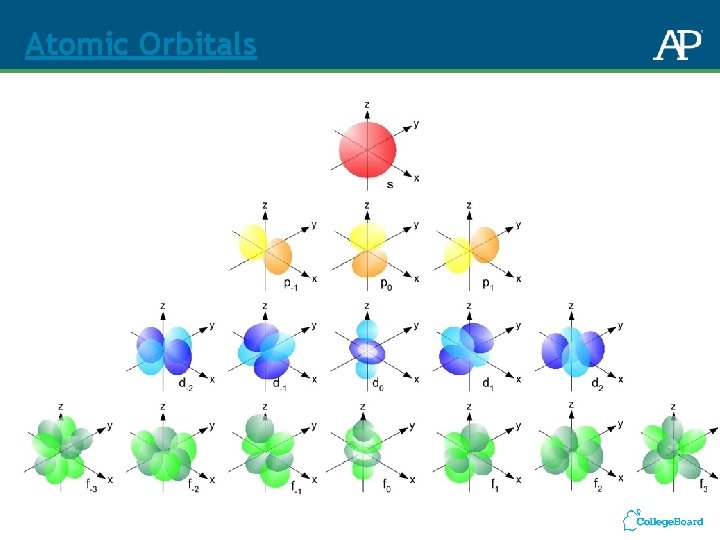
Atomic Orbitals

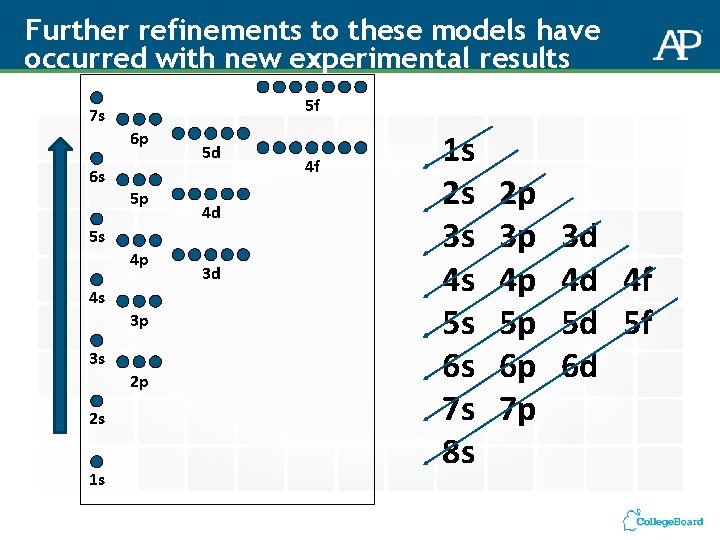
Further refinements to these models have occurred with new experimental results 5 f 7 s 6 p 6 s 5 p 5 d 4 d 5 s 4 p 4 s 3 p 3 s 2 p 2 s 1 s 3 d 4 f 1 s 2 s 3 s 4 s 5 s 6 s 7 s 8 s 2 p 3 p 4 p 5 p 6 p 7 p 3 d 4 d 4 f 5 d 5 f 6 d
![But not all elements follow the rules 1 s Ar4 s 13 d 5 But not all elements ‘follow the rules’ 1 s [Ar]4 s 13 d 5](https://slidetodoc.com/presentation_image_h/b668189a26d8998094a39c5da78bf94b/image-28.jpg)
But not all elements ‘follow the rules’ 1 s [Ar]4 s 13 d 5 1 s [Ar]4 s 13 d 10 2 s 2 p 3 s 3 p 4 s 3 d 4 p 5 s 4 d 5 p 6 s 5 d 6 p 7 s 6 d 7 p 4 f 5 f

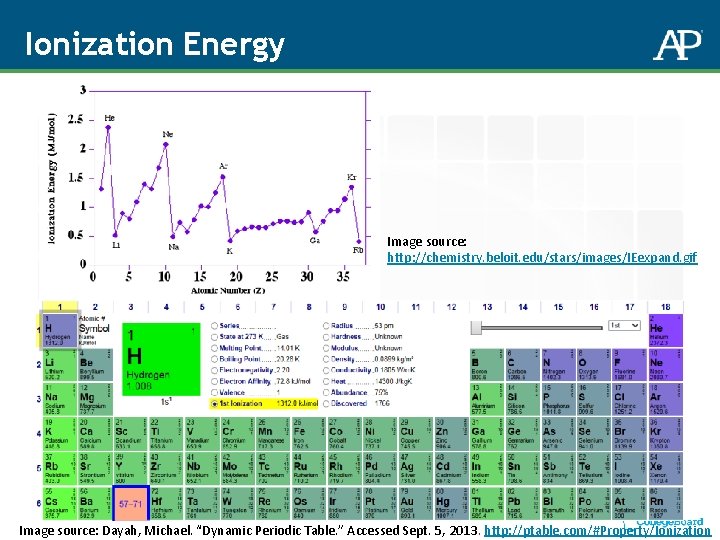
Ionization Energy Image source: http: //chemistry. beloit. edu/stars/images/IEexpand. gif Image source: Dayah, Michael. “Dynamic Periodic Table. ” Accessed Sept. 5, 2013. http: //ptable. com/#Property/Ionization

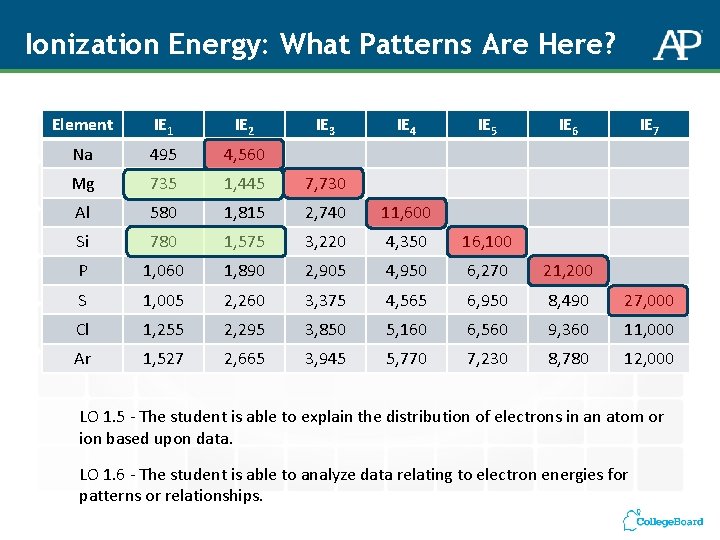
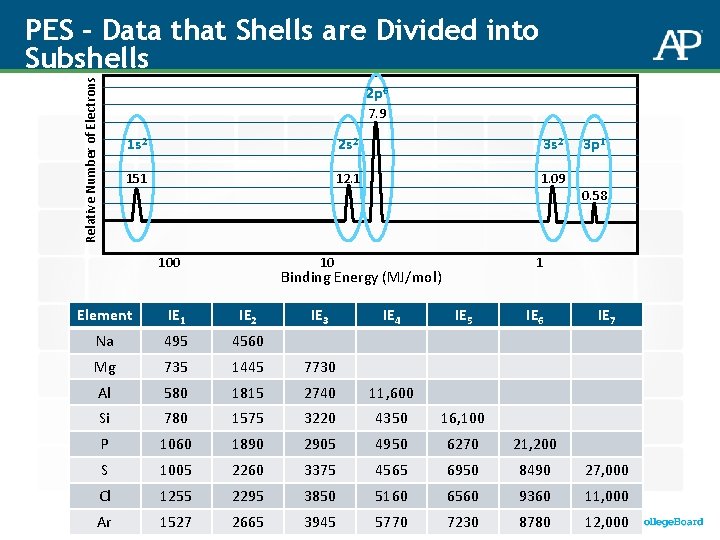
Ionization Energy: What Patterns Are Here? Element IE 1 IE 2 IE 3 IE 4 IE 5 IE 6 IE 7 Na 495 4, 560 Mg 735 1, 445 7, 730 Al 580 1, 815 2, 740 11, 600 Si 780 1, 575 3, 220 4, 350 16, 100 P 1, 060 1, 890 2, 905 4, 950 6, 270 21, 200 S 1, 005 2, 260 3, 375 4, 565 6, 950 8, 490 27, 000 Cl 1, 255 2, 295 3, 850 5, 160 6, 560 9, 360 11, 000 Ar 1, 527 2, 665 3, 945 5, 770 7, 230 8, 780 12, 000 LO 1. 5 - The student is able to explain the distribution of electrons in an atom or ion based upon data. LO 1. 6 - The student is able to analyze data relating to electron energies for patterns or relationships.

How do we probe further into the atom? Radiation Type ν E Aspects Probed Microwaves 109 – 1011 Hz 10 -7 – 10 -4 MJ/mol Molecular rotations Infrared (IR) 1011 – 1014 Hz 10 -4 – 10 -1 MJ/mol Molecular vibrations Visible (ROYGBV) 4 x 1014 – 7. 5 x 1014 Hz 0. 2 - 0. 3 MJ/mol Ultraviolet (UV) 1014 – 1016 Hz X-ray 1016 – 1019 Hz hν - - - 11+ - 102 – 105 MJ/mol - - hν 0. 3 – 100 MJ/mol - Valence electron transitions in atoms and molecules Core electron transitions in atoms

Removing Core Electrons - - - 11+ hν - E = 103. 3 MJ/mol E = 1. 033 x 108 J/mol - - v = E/h = (1. 033 x 108 J/mol)/(6. 626 x 10 -34 J s) v = (1. 559 x 1041 mol-1 s-1)x( 1 mol /6. 022 x 1023 e vmin = 2. 59 x 1017 Hz Radiation Type X-ray ν 1016 – 1019 Hz E 102 – 105 MJ/mol Aspects Probed Core electron transitions in atoms

Removing Core Electrons hν - - 11+ hν - - - E 1 st = 103. 3 MJ/mol E 2 nd = 3 -6 MJ/mol Any frequency of light that is sufficient to remove electrons from the 1 st shell can remove electrons from any of the other shells.

PES Instrument Image Source: SPECS Gmb. H, http: //www. specs. de/cms/front_content. php? idart=267

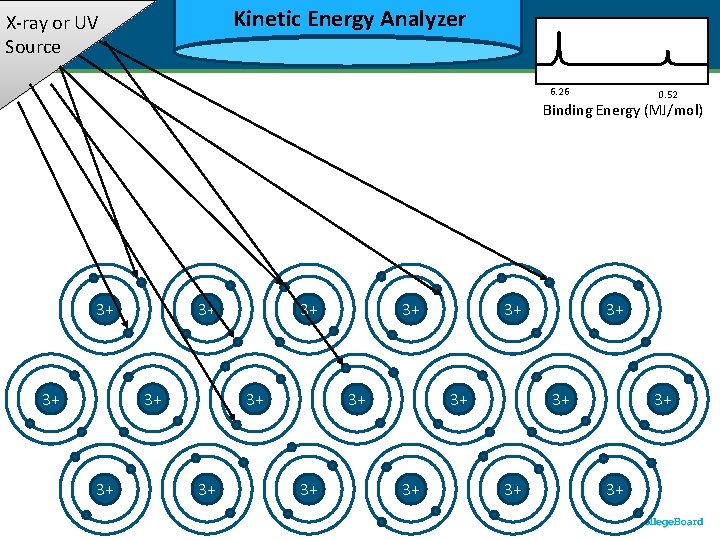
Kinetic Energy Analyzer Kinetic X-ray or UV Source 6. 26 0. 52 Binding Energy (MJ/mol) 3+ 3+ 3+ 3+ 3+

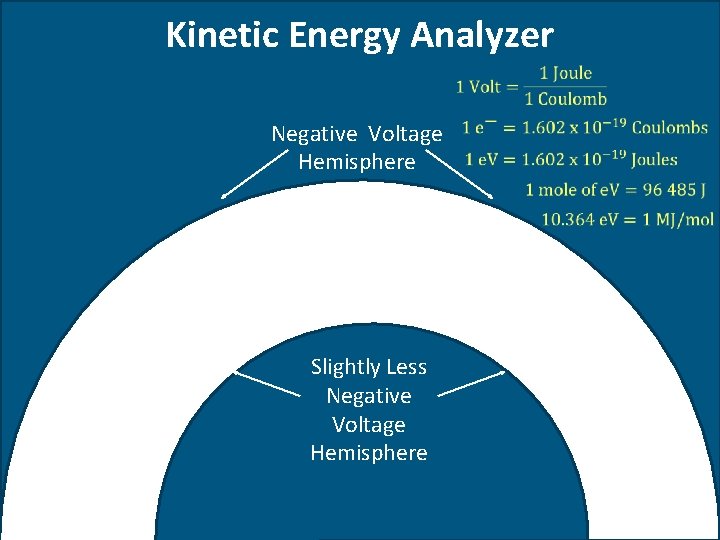
Kinetic Energy Analyzer Negative Voltage Hemisphere Slightly Less Negative Voltage Hemisphere

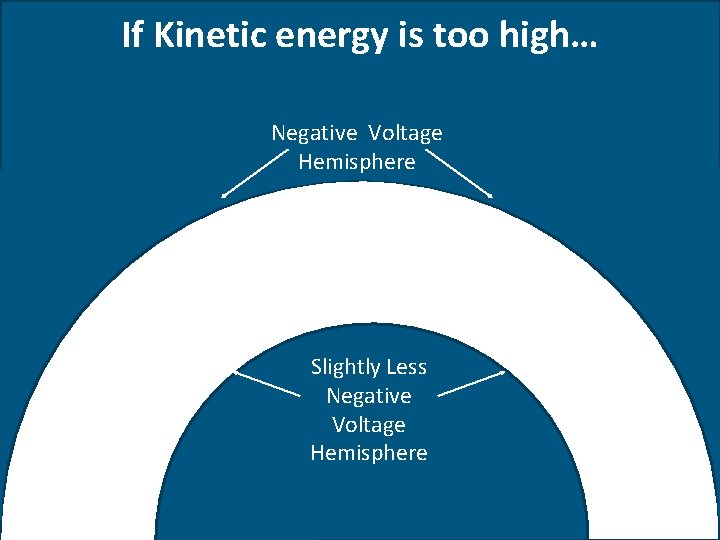
If Kinetic energy is too high… Negative Voltage Hemisphere Slightly Less Positive Negative Voltage Hemisphere

If voltage is too high… Negative Voltage Hemisphere Slightly Less Positive Negative Voltage Hemisphere

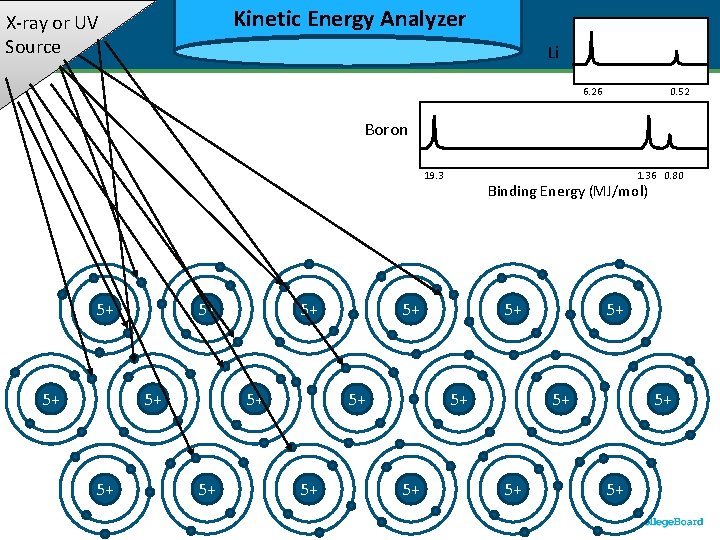
Kinetic Energy Analyzer Kinetic X-ray or UV Source Li 6. 26 Binding Energy (MJ/mol) Boron 1. 36 0. 80 19. 3 5+ 5+ 3+ 3+ 5+ 5+ 3+ 3+ 5+ Binding Energy (MJ/mol) 5+ 3+ 3+ 5+ 0. 52 5+ 3+ 3+ 5+ 5+ 3+

Monday 9. 29. 14 What does PES data show that emission spectra do not?

Plan for the Week Chapter 2 Problem Set: Q & A The Evolving Atom: Experimental Evidence • Lecture: Bohr’s Equation and the Hydrogen Atom Lecture: Aufbau Diagrams and Ionization Energy Activity: Shells Class Activity Lecture: PES = Evidence for Subshells (Orbitals) Activity: Shells to Subshells Activity Discussion: Practice Problems Lab: Paramagnetism and Electron Configurations (More Evidence)

Analyzing Data from PES Experiments

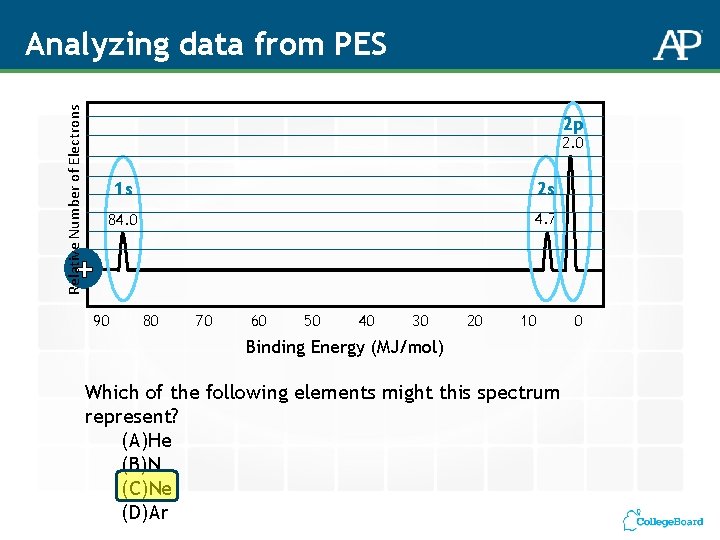
Relative Number of Electrons Analyzing data from PES 2 p 2. 0 1 s 2 s 84. 0 4. 7 + 90 80 70 60 50 40 30 20 10 Binding Energy (MJ/mol) Which of the following elements might this spectrum represent? (A)He (B)N (C)Ne (D)Ar 0

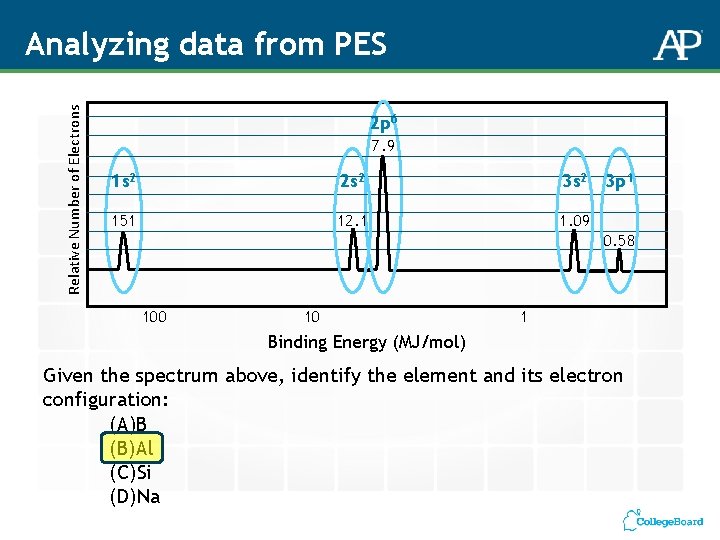
Relative Number of Electrons Analyzing data from PES 2 p 6 7. 9 1 s 2 2 s 2 3 s 2 151 12. 1 1. 09 3 p 1 0. 58 100 10 1 Binding Energy (MJ/mol) Given the spectrum above, identify the element and its electron configuration: (A)B (B)Al (C)Si (D)Na

Real Spectrum

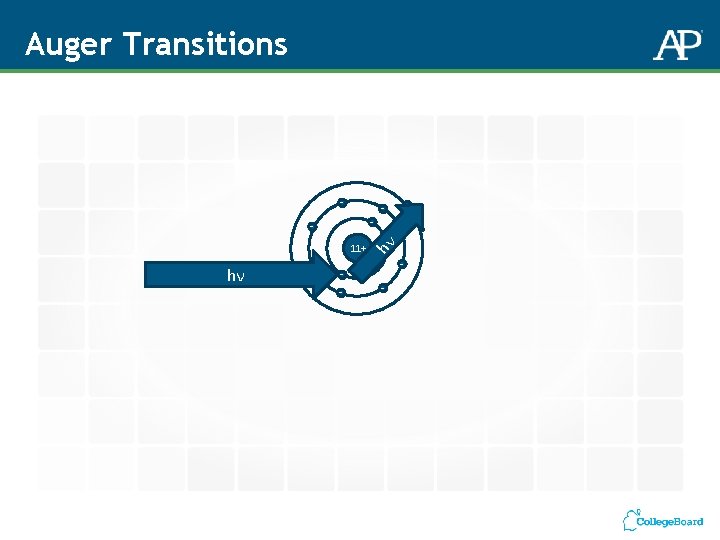
Auger Transitions 11+ hν - - -

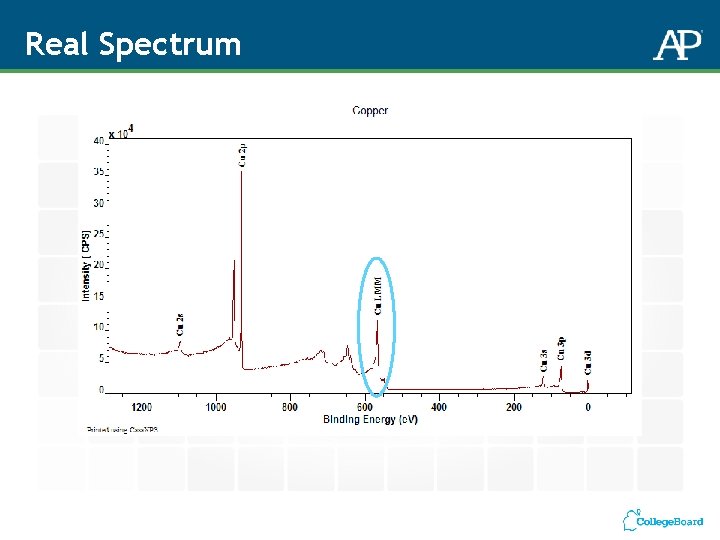
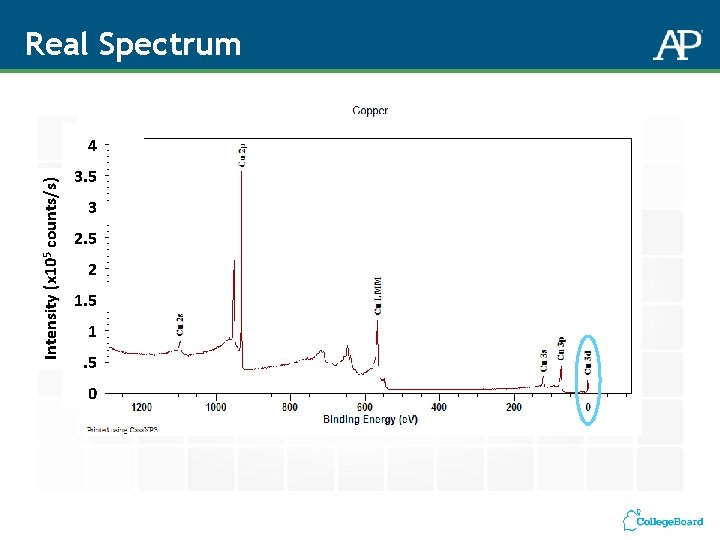
Real Spectrum Intensity (x 105 counts/s) 4 3. 5 3 2. 5 2 1. 5 0

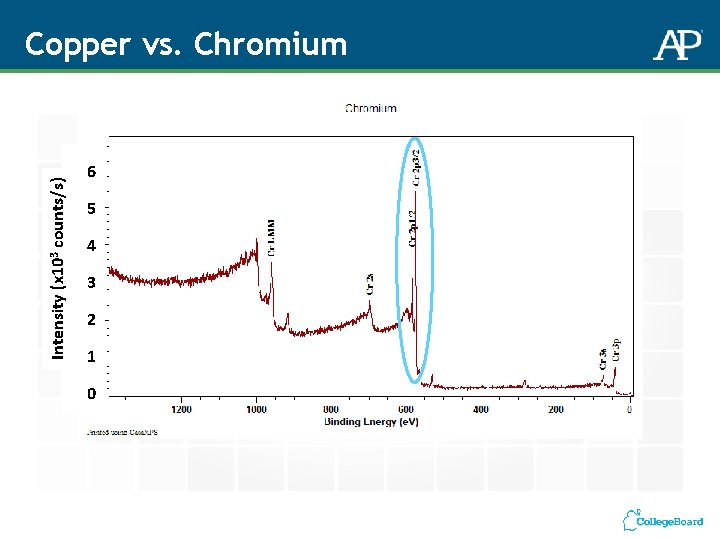
Intensity (x 103 counts/s) Copper vs. Chromium 6 5 4 3 2 1 0

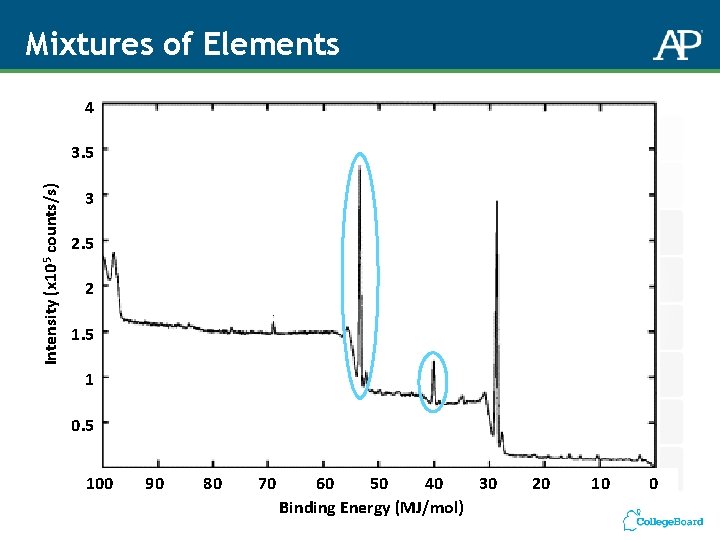
Mixtures of Elements 4 Intensity (x 105 counts/s) 3. 5 3 2. 5 2 1. 5 1 0. 5 100 90 80 70 60 50 40 30 Binding Energy (MJ/mol) 20 10 0

PES Sample Questions

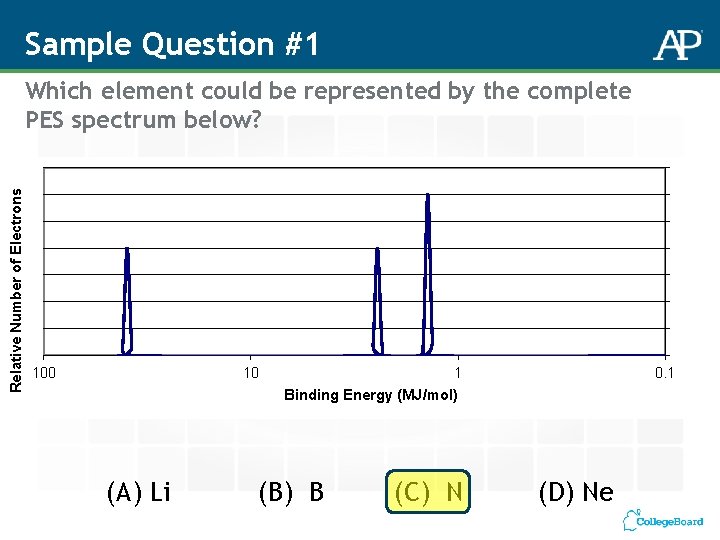
Sample Question #1 Relative Number of Electrons Which element could be represented by the complete PES spectrum below? 100 10 (A) Li 1 Binding Energy (MJ/mol) (B) B (C) N 0. 1 (D) Ne

Sample Question #2 Intensity Which of the following best explains the relative positioning and intensity of the 2 s peaks in the following spectra? Li 12 10 8 6 4 Binding Energy (MJ/mol) 2 0 Intensity 14 Be 14 (A) (B) (C) (D) 12 10 8 6 4 Binding Energy (MJ/mol) 2 0 Be has a greater nuclear charge than Li and more electrons in the 2 s orbital Be electrons experience greater electron-electron repulsions than Li electrons Li has a greater pull from the nucleus on the 2 s electrons, so they are harder to remove Li has greater electron shielding by the 1 s orbital, so the 2 s electrons are easier to remove

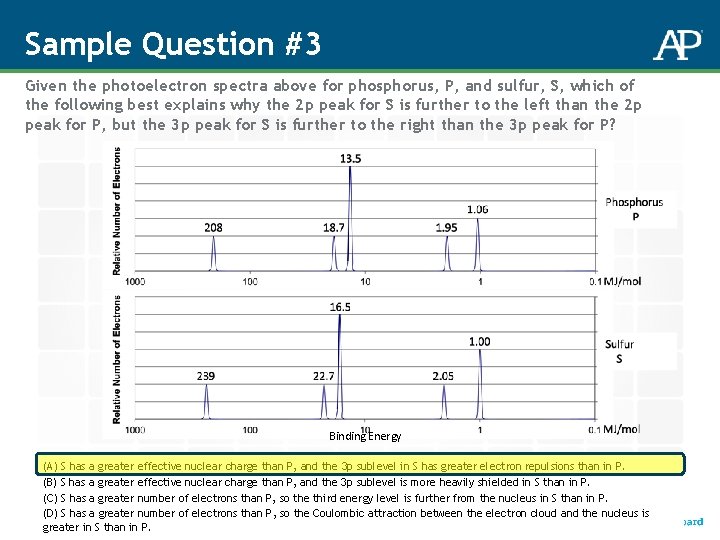
Sample Question #3 Given the photoelectron spectra above for phosphorus, P, and sulfur, S, which of the following best explains why the 2 p peak for S is further to the left than the 2 p peak for P, but the 3 p peak for S is further to the right than the 3 p peak for P? Binding Energy (A) S has a greater effective nuclear charge than P, and the 3 p sublevel in S has greater electron repulsions than in P. (B) S has a greater effective nuclear charge than P, and the 3 p sublevel is more heavily shielded in S than in P. (C) S has a greater number of electrons than P, so the third energy level is further from the nucleus in S than in P. (D) S has a greater number of electrons than P, so the Coulombic attraction between the electron cloud and the nucleus is greater in S than in P.

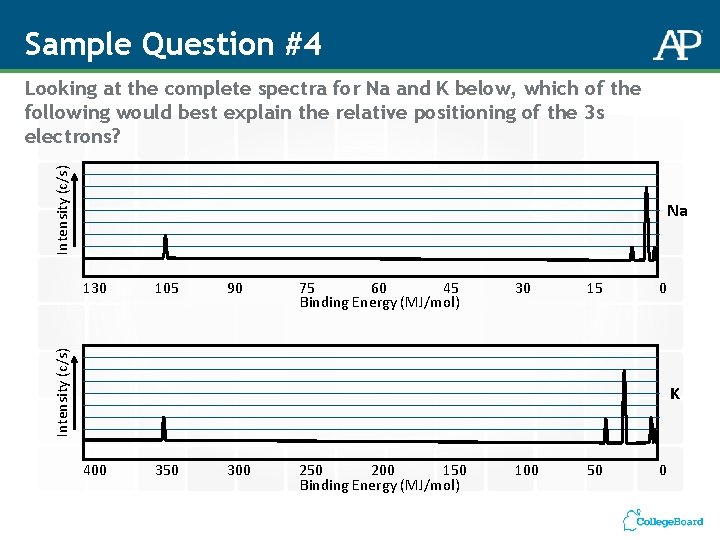
Sample Question #4 Intensity (c/s) Looking at the complete spectra for Na and K below, which of the following would best explain the relative positioning of the 3 s electrons? Na 105 90 75 60 45 Binding Energy (MJ/mol) 30 15 0 Intensity (c/s) 130 K 400 350 300 250 200 150 Binding Energy (MJ/mol) 100 50 0

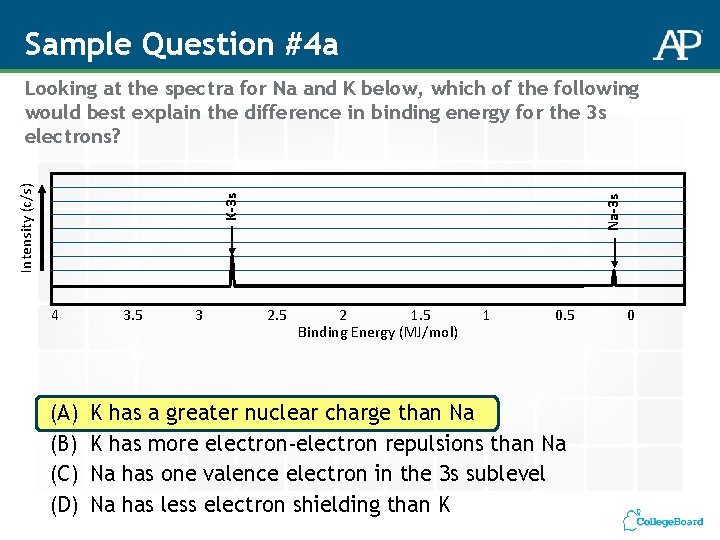
Sample Question #4 a 4 (A) (B) (C) (D) 3. 5 3 Na-3 s K-3 s Intensity (c/s) Looking at the spectra for Na and K below, which of the following would best explain the difference in binding energy for the 3 s electrons? 2. 5 2 1. 5 Binding Energy (MJ/mol) 1 0. 5 K has a greater nuclear charge than Na K has more electron-electron repulsions than Na Na has one valence electron in the 3 s sublevel Na has less electron shielding than K 0

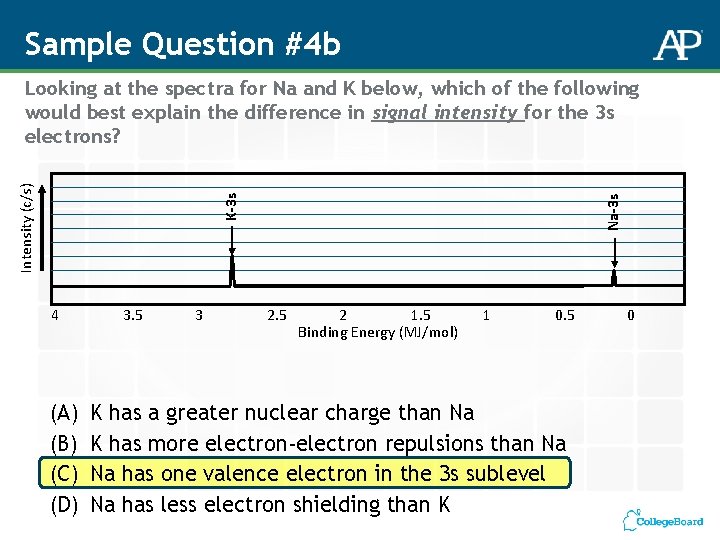
Sample Question #4 b 4 (A) (B) (C) (D) 3. 5 3 Na-3 s K-3 s Intensity (c/s) Looking at the spectra for Na and K below, which of the following would best explain the difference in signal intensity for the 3 s electrons? 2. 5 2 1. 5 Binding Energy (MJ/mol) 1 0. 5 K has a greater nuclear charge than Na K has more electron-electron repulsions than Na Na has one valence electron in the 3 s sublevel Na has less electron shielding than K 0

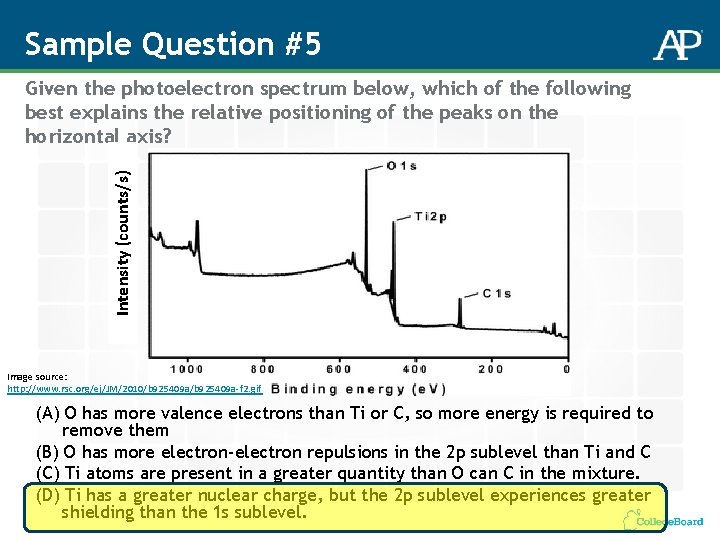
Sample Question #5 Intensity (counts/s) Given the photoelectron spectrum below, which of the following best explains the relative positioning of the peaks on the horizontal axis? Image source: http: //www. rsc. org/ej/JM/2010/b 925409 a-f 2. gif (A) O has more valence electrons than Ti or C, so more energy is required to remove them (B) O has more electron-electron repulsions in the 2 p sublevel than Ti and C (C) Ti atoms are present in a greater quantity than O can C in the mixture. (D) Ti has a greater nuclear charge, but the 2 p sublevel experiences greater shielding than the 1 s sublevel.

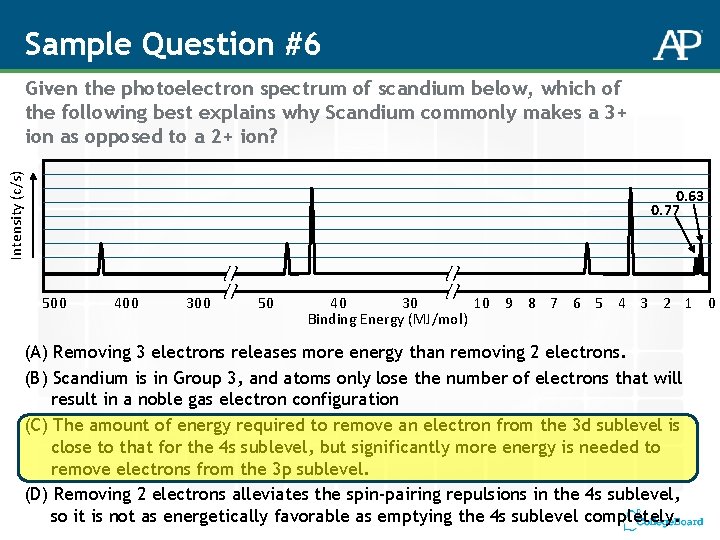
Sample Question #6 Intensity (c/s) Given the photoelectron spectrum of scandium below, which of the following best explains why Scandium commonly makes a 3+ ion as opposed to a 2+ ion? 0. 63 0. 77 500 400 300 50 40 30 10 9 8 7 6 5 4 3 2 1 0 Binding Energy (MJ/mol) (A) Removing 3 electrons releases more energy than removing 2 electrons. (B) Scandium is in Group 3, and atoms only lose the number of electrons that will result in a noble gas electron configuration (C) The amount of energy required to remove an electron from the 3 d sublevel is close to that for the 4 s sublevel, but significantly more energy is needed to remove electrons from the 3 p sublevel. (D) Removing 2 electrons alleviates the spin-pairing repulsions in the 4 s sublevel, so it is not as energetically favorable as emptying the 4 s sublevel completely.

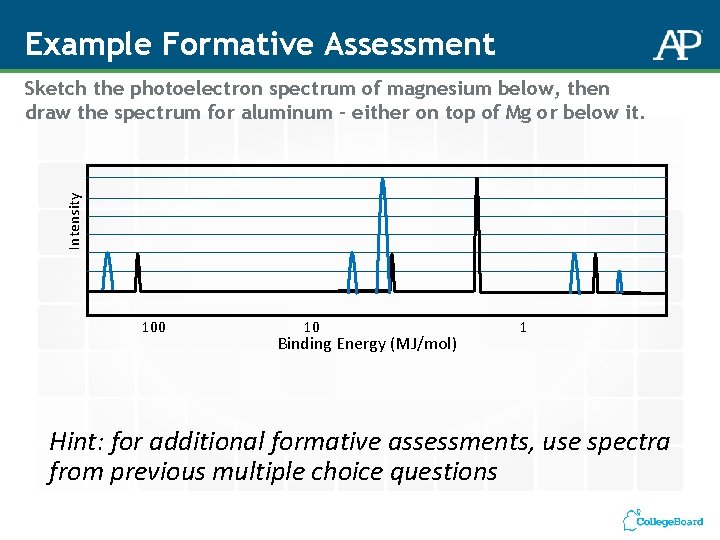
Example Formative Assessment Intensity Sketch the photoelectron spectrum of magnesium below, then draw the spectrum for aluminum – either on top of Mg or below it. 100 10 Binding Energy (MJ/mol) 1 Hint: for additional formative assessments, use spectra from previous multiple choice questions

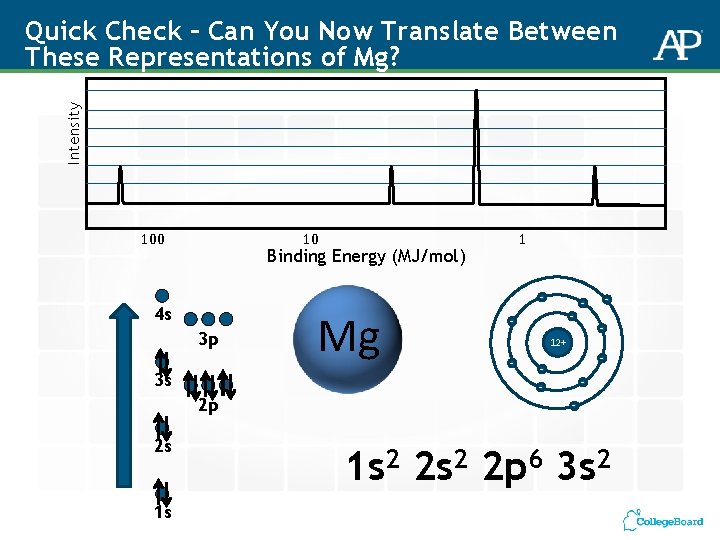
Intensity Quick Check – Can You Now Translate Between These Representations of Mg? 100 10 Binding Energy (MJ/mol) - 4 s 3 p 3 s 2 p 2 s 1 s 1 Mg - - - 12+ - - - 1 s 2 2 p 6 3 s 2

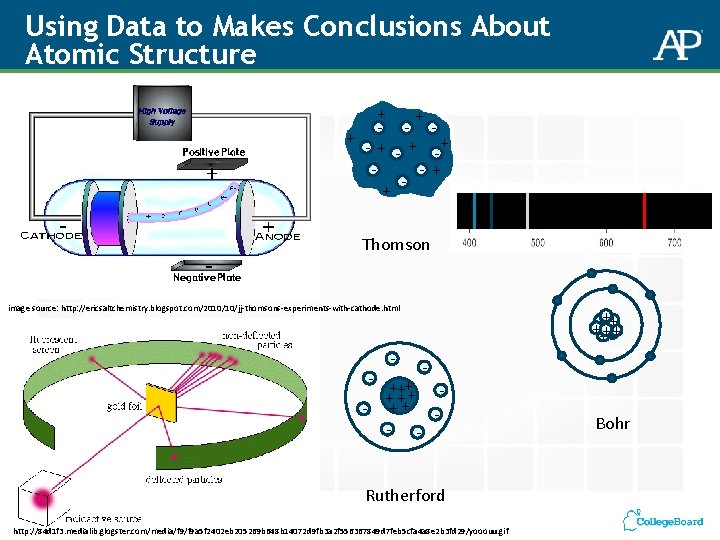
Using Data to Makes Conclusions About Atomic Structure + + -+ - + + Thomson image source: http: //ericsaltchemistry. blogspot. com/2010/10/jj-thomsons-experiments-with-cathode. html - - + +++++++ - +++ + +++ - Rutherford http: //84 d 1 f 3. medialib. glogster. com/media/f 9 a 5 f 2402 eb 205269 b 648 b 14072 d 9 fb 3 a 2 f 556367849 d 7 feb 5 cfa 4 a 8 e 2 b 3 fd 29/yooouu. gif Bohr

Relative Number of Electrons PES – Data that Shells are Divided into Subshells 2 p 6 7. 9 1 s 2 2 s 2 3 s 2 151 12. 1 1. 09 100 10 IE 4 0. 58 1 Binding Energy (MJ/mol) IE 3 3 p 1 Element IE 1 IE 2 IE 5 IE 6 IE 7 Na 495 4560 Mg 735 1445 7730 Al 580 1815 2740 11, 600 Si 780 1575 3220 4350 16, 100 P 1060 1890 2905 4950 6270 21, 200 S 1005 2260 3375 4565 6950 8490 27, 000 Cl 1255 2295 3850 5160 6560 9360 11, 000 Ar 1527 2665 3945 5770 7230 8780 12, 000

Plan for Today Questions on Packet? Lab Notebooks? Some Fun With Stoichiometry What’s a Mole? Video(s) HW [Enter Presentation Title in Header and Footer] 63

Mole Challenge If you lined up a mole of your shoes end to end, toe to heel, what distance would this cover? What distance is this similar to? How many semi-trucks would it take to haul a mole of Voodoo Doughnut boxes? (Hint: you have to figure out the volume of a doughnut box, and the volume of a semitruck container = 75. 3 m 3) If you lined the containers up end to end, what distance would it cover? Hint: Each container is 12. 192 m long) [Enter Presentation Title in Header and Footer] 64

Applicable Science Practices From the AP Chemistry Curriculum Framework: SP 3. 2 • The student can refine scientific questions SP 3. 3 • The student can evaluate scientific questions SP 6. 3 • The student can articulate the reasons that scientific explanations are refined or replaced.

Applicable Learning Objectives From the AP Chemistry Curriculum Framework: 1. 5 – The student is able to explain the distribution of electrons in an atom or ion based upon data. 1. 6 – The student is able to analyze data relating to electron energies for patterns and relationships. 1. 7 – The student is able to describe the electronic structure of the atom, using PES data, ionization energy data, and/or Coulomb’s law to construct explanations of how the energies of electrons within shells in atoms vary. 1. 8 – The student is able to explain the distribution of electrons using Coulomb’s law to analyze measured energies. 1. 12 – The student is able to explain why a given set of data suggests, or does not suggest, the need to refine the atomic model from a classical shell model with the quantum mechanical model. 1. 13 – Given information about a particular model of the atom, the student is able to determine if the model is consistent with specified evidence. 1. 14 – The student can justify the selection of a particular type of spectroscopy to measure properties associated with vibrational or electronic motions of molecules.

Supporting Resources (cont. ) Arizona simulated photoelectron spectra http: //www. chem. arizona. edu/chemt/Flash/photoelectron. html Guided inquiry activities on PES • John Gelder (Oklahoma State University) • Moog and Farrell, Chemistry: A Guided Inquiry • POGIL Books on PES technical specs • Van der Heide, Paul. X-Ray Photoelectron Spectroscopy: An Introduction to Principles and Practices. New Jersey: John Wiley & Sons, Inc, 2012. • Ellis, Andrew M. , Miklos Feher, and Timothy Wright. Electronic and Photoelectron Spectroscopy: Fundamentals and Case Studies. New York: Cambridge University Press, 2005.

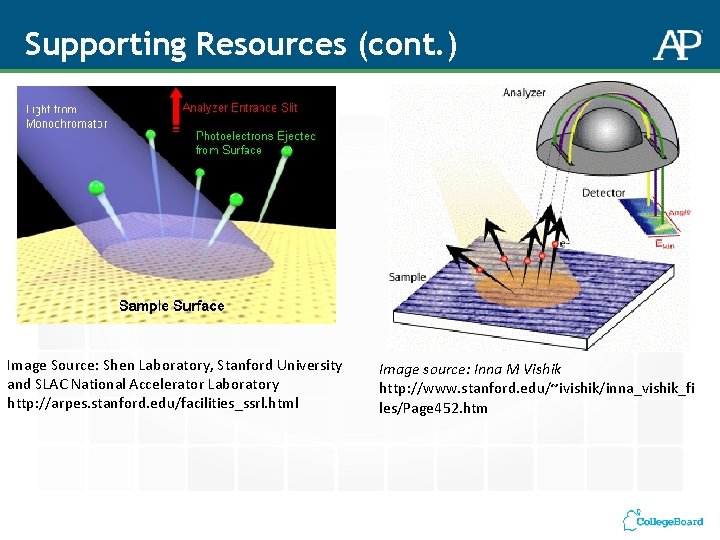
Supporting Resources (cont. ) Image Source: Shen Laboratory, Stanford University and SLAC National Accelerator Laboratory http: //arpes. stanford. edu/facilities_ssrl. html Image source: Inna M Vishik http: //www. stanford. edu/~ivishik/inna_vishik_fi les/Page 452. htm