VAPORLIQUID EQUILIBRIUMIntroduction ERT 206 Thermodynamics Miss Anis Atikah

VAPOR/LIQUID EQUILIBRIUMIntroduction ERT 206: Thermodynamics Miss Anis Atikah Ahmad Email: anis atikah@unimap. edu. my

OUTLINE 1. 2. 3. 4. 5. The Nature of Equilibrium Duhem’s Theorem Simple Models for VLE by Modified Raoult’s Law VLE from K-value Correlations

1. The Nature of Equilibrium • Equilibrium is a static condition in which no changes occur in the macroscopic properties of a system with time. – Eg: An isolated system consisting of liquid & vapor phase reaches a final state wherein no tendency exists for change to occur within the system. The temperature, pressure and phase compositions reach final values which thereafter remain fixed.

• At microscopic level, conditions are not static. – Molecules with high velocities near the interface overcome surface forces and pass into the other phase. – But the average rate of passage of molecules is the same in both directions & no net interphase transfer of material occurs.

Measures of Composition 1. Mass fraction: the ratio of the mass of a particular chemical species in a mixture or solution to the total mass of mixture or solution. 2. Mole fraction: the ratio of the number of moles of a particular chemical species in a mixture or solution to the number of moles of mixture or solution.

Measures of Composition 3. Molar concentration: the ratio of the mole fraction of a particular chemical species in a mixture or solution to the molar volume of mixture or solution. Molar flow rate Volumetric flow rate 4. Molar mass of mixture/solution: mole-fractionweighted sum of the molar masses of all species present.

2. Duhem’s Theorem Similar to phase rule, but it considers extensive state. • Duhem’s Theorem: for any closed system formed initially from given masses of prescribed chemical species, the equilibrium state is completely determined when any two independent variables are fixed. – Applies to closed systems at equilibrium – The extensive state and intensive state of system are fixed No of variables No of equations

3. SIMPLE MODELS FOR VAPOR/LIQUID EQUILIBRIUM • Vapor/liquid equilibrium (VLE): the state of coexistence of liquid and vapor phase. • VLE Model: to calculate temperatures, pressures and compositions of phases in equilibrium. • The two simplest models are: – Raoult’s law – Henry’s law

3. SIMPLE MODELS FOR VAPOR/LIQUID EQUILIBRIUM 3. 1 Raoult’s Law • Assumptions: – The vapor phase is an ideal gas (low to moderate pressure) – The liquid phase is an ideal solution (the system are chemically similar) *Chemically similar: the molecular species are not too different in size and are of the same chemical nature. eg: n-hexane/n-heptane, ethanol/propanol, benzene/toluene Liquid phase mole fraction Vapor pressure of pure species i at system temperature
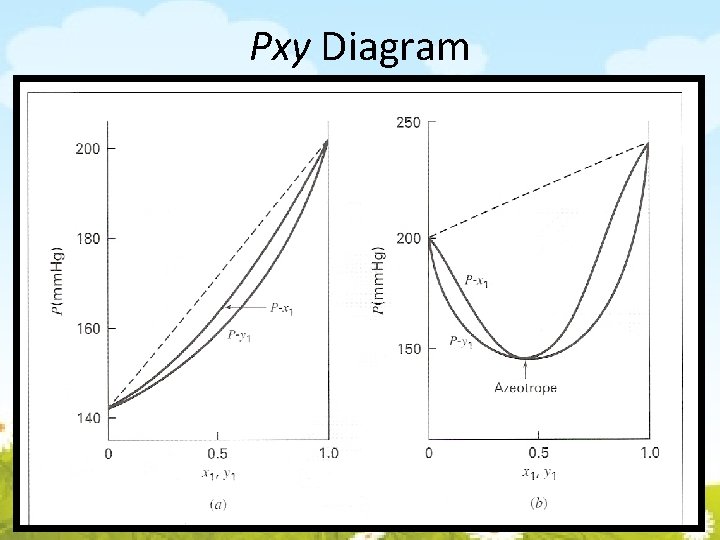
Pxy Diagram

3. 2 Dewpoint & Bubblepoint Calculations with Raoult’s Law 4 Calculations • • BUBL P : Calculate {yi} and P, given {xi} and T DEW P : Calculate {xi} and P, given {yi} and T BUBL T : Calculate {yi} and T, given {xi} and P DEW T : Calculate {xi} and T, given {yi} and P If the vapor-phase composition is unknown, may be assumed; thus For bubble point calculation

3. 2 Dewpoint & Bubblepoint Calculations with Raoult’s Law If the liquid-phase composition is unknown, may be assumed; thus For dew point calculation

3. 2. 1 BUBL P CALCULATION (Calculate {yi} and P, given {xi} and T) Find P 1 sat & P 2 sat using Antoine equation Find P Calculate yi

Example 1 Binary system acetronitrile (1)/ nitromethane (2) conforms closely to Raoult’s law. Vapor pressure for the pure species are given by the following Antoine equations: Prepare a graph showing P vs. X 1 and P vs. Y 1 for a temperature of 75°C.

3. 2. 1 BUBL P CALCULATION (Calculate {yi} and P, given {xi} and T) Find P 1 sat & P 2 sat using Antoine equation At 75°C, by Antoine Equations, Find P Calculate yi

3. 2. 1 BUBL P CALCULATION (Calculate {yi} and P, given {xi} and T) Find P 1 sat & P 2 sat using Antoine equation Find P Taking at any value of x 1, say x 1=0. 6, Calculate yi

3. 2. 1 BUBL P CALCULATION (Calculate {yi} and P, given {xi} and T) Find P 1 sat & P 2 sat using Antoine equation Find P Calculate yi At 75°C, a liquid mixture of 60 mol-% acetonitrile and 40 mol-% nitromethane is in equilibrium with a vapor containing 74. 83 mol-% acetonitrile at a pressure of 66. 72 k. Pa
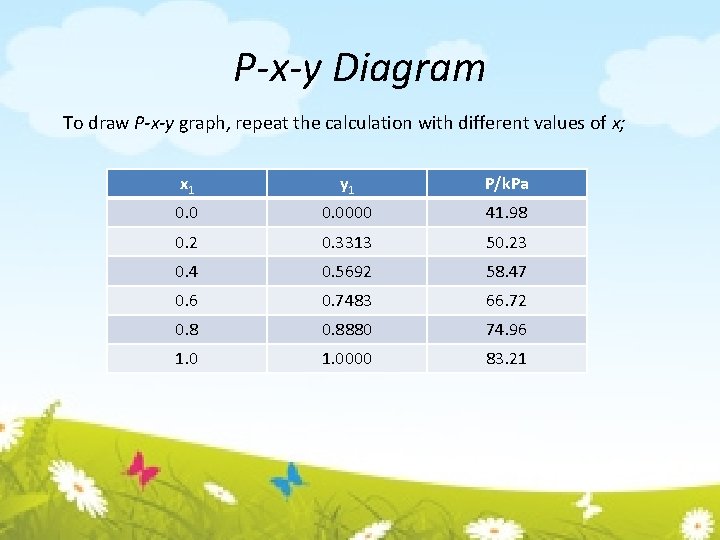
P-x-y Diagram To draw P-x-y graph, repeat the calculation with different values of x; x 1 y 1 P/k. Pa 0. 0000 41. 98 0. 2 0. 3313 50. 23 0. 4 0. 5692 58. 47 0. 6 0. 7483 66. 72 0. 8880 74. 96 1. 0000 83. 21
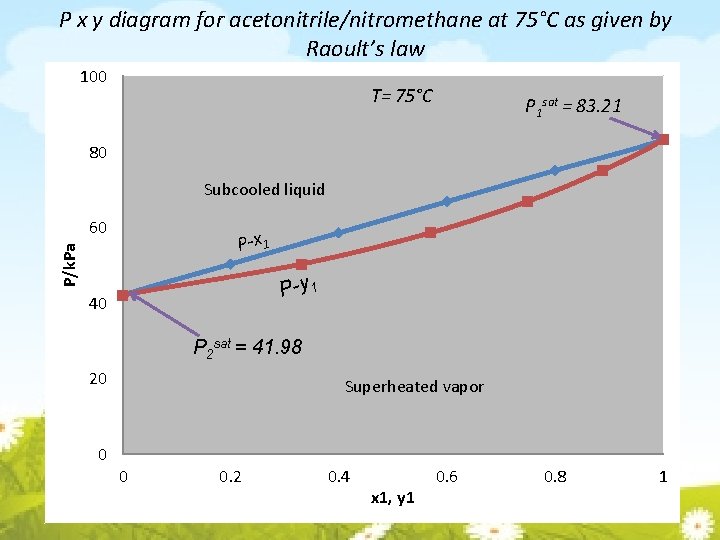
P x y diagram for acetonitrile/nitromethane at 75°C as given by Raoult’s law 100 T= 75°C P 1 sat = 83. 21 80 Subcooled liquid 60 P/k. Pa P-x 1 P-y 1 40 P 2 sat = 41. 98 20 0 Superheated vapor 0 0. 2 0. 4 x 1, y 1 0. 6 0. 8 1
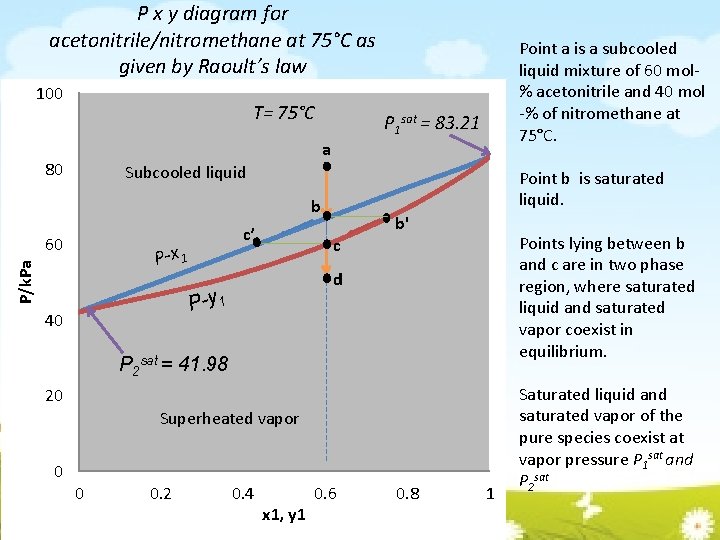
P x y diagram for acetonitrile/nitromethane at 75°C as given by Raoult’s law 100 T= 75°C Point a is a subcooled liquid mixture of 60 mol% acetonitrile and 40 mol -% of nitromethane at 75°C. P 1 sat = 83. 21 a 80 Subcooled liquid Point b is saturated liquid. b 60 c’ P/k. Pa P-x 1 Points lying between b and c are in two phase region, where saturated liquid and saturated vapor coexist in equilibrium. c d P-y 1 40 b' P 2 sat = 41. 98 20 Superheated vapor 0 0 0. 2 0. 4 x 1, y 1 0. 6 0. 8 1 Saturated liquid and saturated vapor of the pure species coexist at vapor pressure P 1 sat and P 2 sat
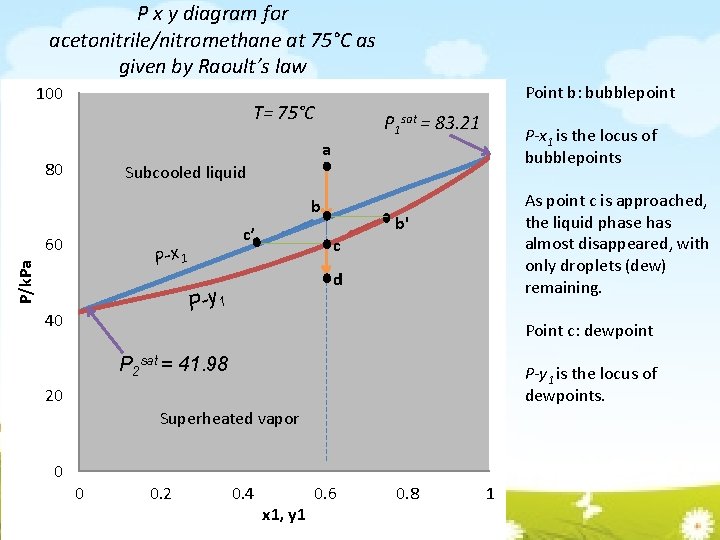
P x y diagram for acetonitrile/nitromethane at 75°C as given by Raoult’s law 100 Point b: bubblepoint T= 75°C P 1 sat = 83. 21 P-x 1 is the locus of bubblepoints a 80 Subcooled liquid b 60 c’ P/k. Pa P-x 1 b' c d P-y 1 40 As point c is approached, the liquid phase has almost disappeared, with only droplets (dew) remaining. Point c: dewpoint P 2 sat = 41. 98 P-y 1 is the locus of dewpoints. 20 Superheated vapor 0 0 0. 2 0. 4 x 1, y 1 0. 6 0. 8 1
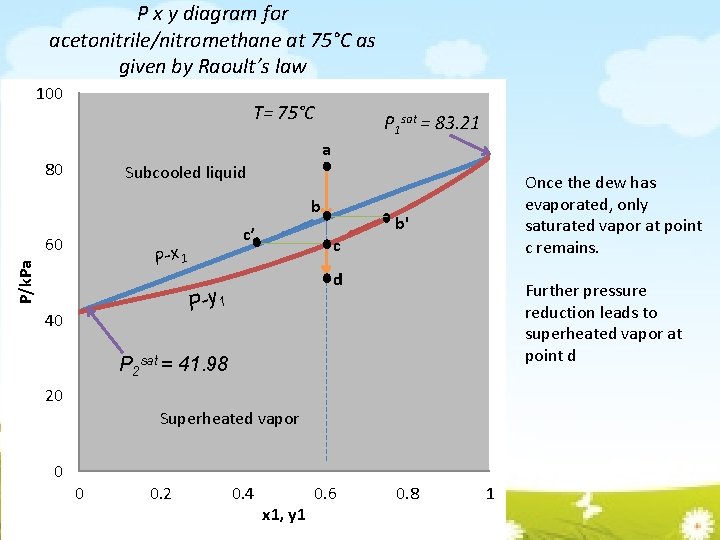
P x y diagram for acetonitrile/nitromethane at 75°C as given by Raoult’s law 100 T= 75°C P 1 sat = 83. 21 a 80 Subcooled liquid b 60 c’ P/k. Pa P-x 1 b' c d P-y 1 40 Once the dew has evaporated, only saturated vapor at point c remains. Further pressure reduction leads to superheated vapor at point d P 2 sat = 41. 98 20 Superheated vapor 0 0 0. 2 0. 4 x 1, y 1 0. 6 0. 8 1
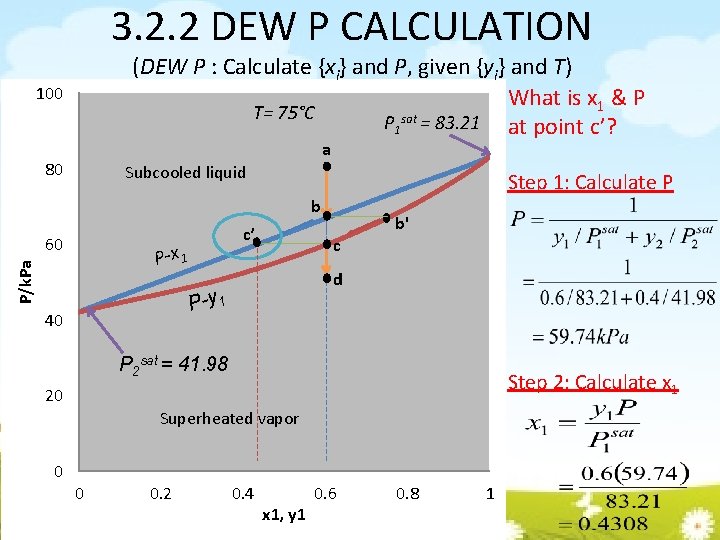
3. 2. 2 DEW P CALCULATION (DEW P : Calculate {xi} and P, given {yi} and T) What is x 1 & P T= 75°C P 1 sat = 83. 21 at point c’? 100 a 80 Subcooled liquid Step 1: Calculate P b 60 c’ P/k. Pa P-x 1 c d P-y 1 40 b' P 2 sat = 41. 98 Step 2: Calculate x 1 20 Superheated vapor 0 0 0. 2 0. 4 x 1, y 1 0. 6 0. 8 1

3. 2. 2 DEW P CALCULATION (DEW P : Calculate {xi} and P, given {yi} and T) Find P from Raoult’s Law assuming Calculate xi
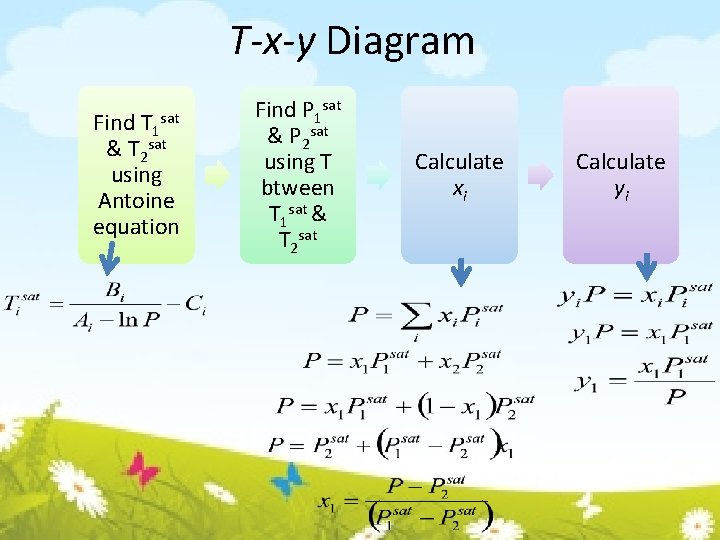
T-x-y Diagram Find T 1 & T 2 sat using Antoine equation sat Find P 1 sat & P 2 sat using T btween T 1 sat & T 2 sat Calculate xi Calculate yi

Example 2 Binary system acetronitrile (1)/ nitromethane (2) conforms closely to Raoult’s law. Vapor pressure for the pure species are given by the following Antoine equations: Prepare a graph showing T vs. X 1 and T vs. Y 1 for a pressure of of 70 k. Pa.
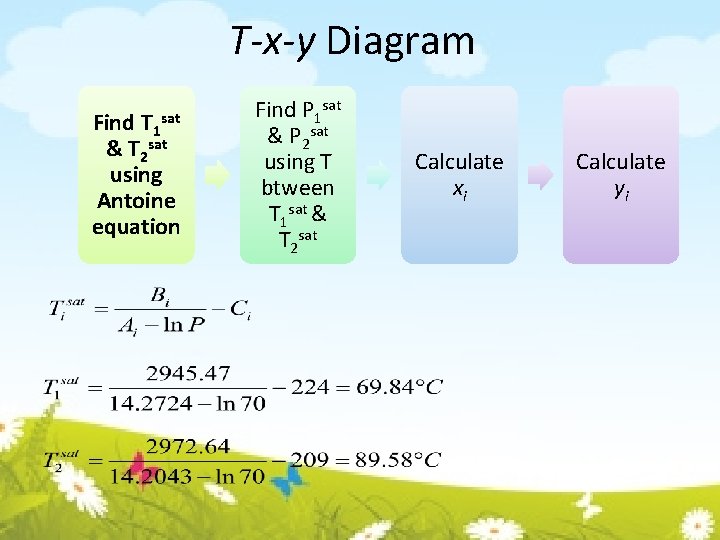
T-x-y Diagram Find T 1 & T 2 sat using Antoine equation sat Find P 1 sat & P 2 sat using T btween T 1 sat & T 2 sat Calculate xi Calculate yi
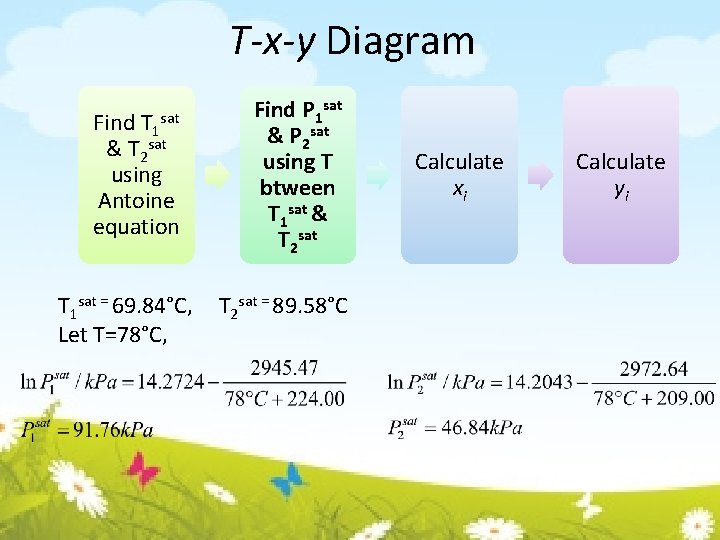
T-x-y Diagram Find T 1 & T 2 sat using Antoine equation sat T 1 sat = 69. 84°C, Let T=78°C, Find P 1 sat & P 2 sat using T btween T 1 sat & T 2 sat = 89. 58°C Calculate xi Calculate yi
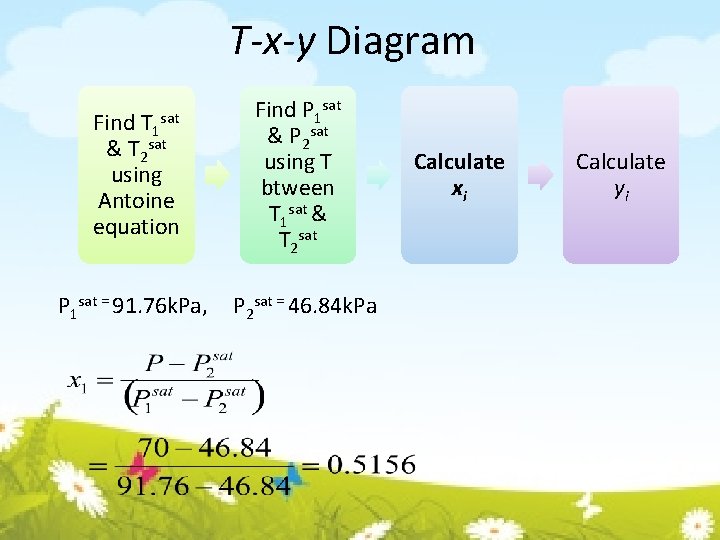
T-x-y Diagram Find T 1 & T 2 sat using Antoine equation Find P 1 sat & P 2 sat using T btween T 1 sat & T 2 sat P 1 sat = 91. 76 k. Pa, P 2 sat = 46. 84 k. Pa sat Calculate xi Calculate yi
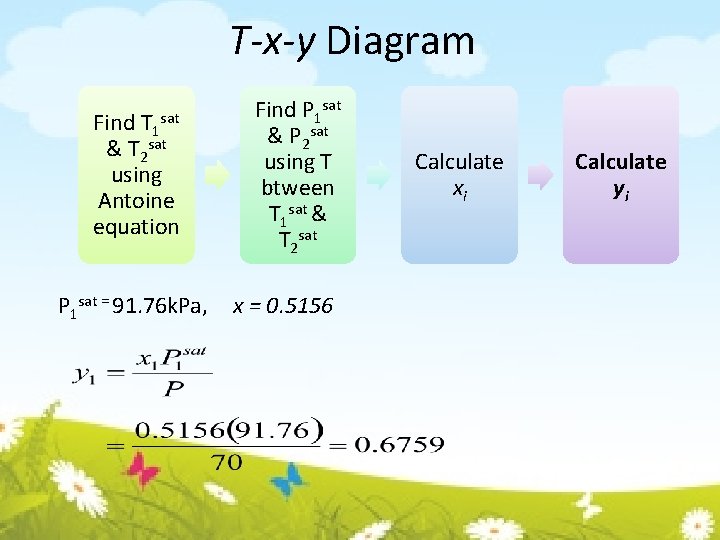
T-x-y Diagram Find T 1 & T 2 sat using Antoine equation sat P 1 sat = 91. 76 k. Pa, Find P 1 sat & P 2 sat using T btween T 1 sat & T 2 sat x = 0. 5156 Calculate xi Calculate yi
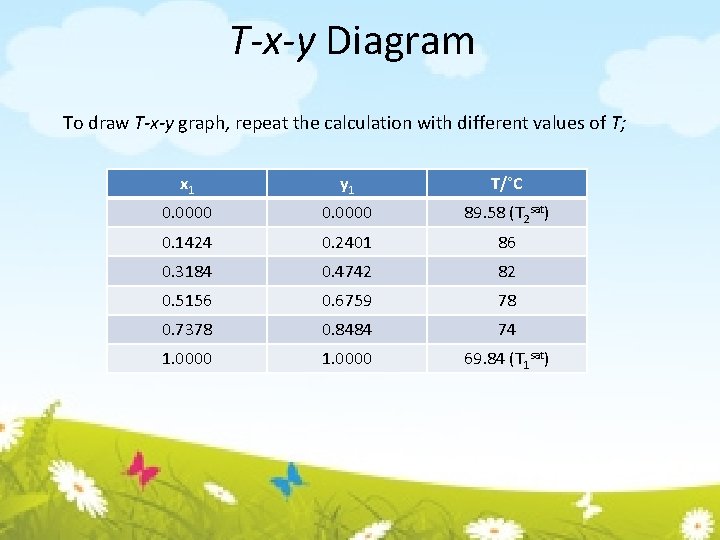
T-x-y Diagram To draw T-x-y graph, repeat the calculation with different values of T; x 1 y 1 T/°C 0. 0000 89. 58 (T 2 sat) 0. 1424 0. 2401 86 0. 3184 0. 4742 82 0. 5156 0. 6759 78 0. 7378 0. 8484 74 1. 0000 69. 84 (T 1 sat)
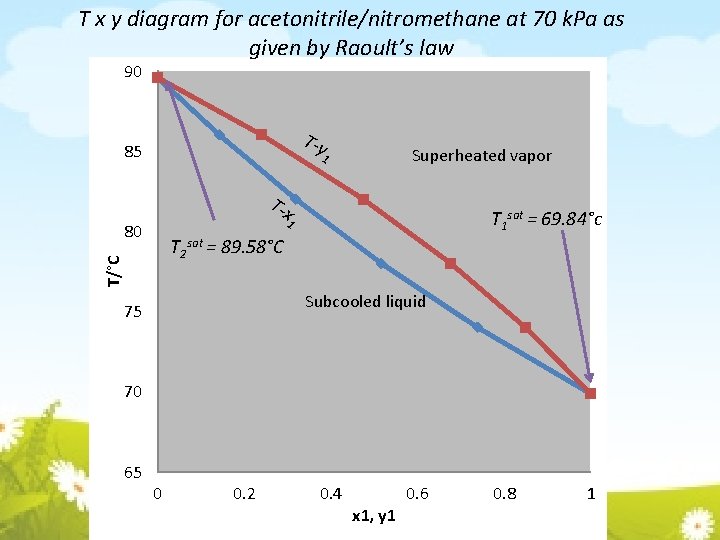
T x y diagram for acetonitrile/nitromethane at 70 k. Pa as given by Raoult’s law 90 T-y 85 Superheated vapor 1 T-x T 1 sat = 69. 84°c 1 80 T/°C T 2 sat = 89. 58°C Subcooled liquid 75 70 65 0 0. 2 0. 4 x 1, y 1 0. 6 0. 8 1
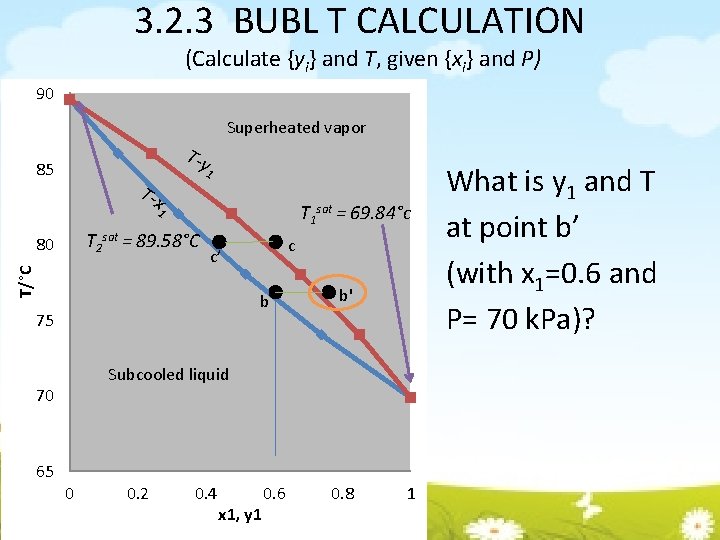
3. 2. 3 BUBL T CALCULATION (Calculate {yi} and T, given {xi} and P) 90 Superheated vapor T-y 85 1 T-x T 1 sat = 69. 84°c 1 T 2 sat = 89. 58°C T/°C 80 b 75 b' Subcooled liquid 70 65 c c’ 0 0. 2 0. 4 x 1, y 1 0. 6 0. 8 1 What is y 1 and T at point b’ (with x 1=0. 6 and P= 70 k. Pa)?

3. 2. 3 BUBL T CALCULATION (Calculate {yi} and T, given {xi} and P) Start with α=1, find P 2 sat Find T using Antoine eq & substitute P 2 sat obtained in step 1 Find new α by substituting T Repeat step 1 by using new α until similar value of α is obtained Find P 1 sat & find y 1 using Raoult’s law The substraction of ln P 1 sat & P 2 sat from Antoine Equation

3. 2. 3 BUBL T CALCULATION (Calculate {yi} and T, given {xi} and P) Start with α=1, find P 2 sat Iteration 1 Iteration 2 Find T using Antoine eq & substitute P 2 sat obtained in step 1 Iteration 3 Iteration 4 Find new α by substituting T Repeat step 1 by using new α until similar value of α is obtained Find P 1 sat & find y 1 using Raoult’s law
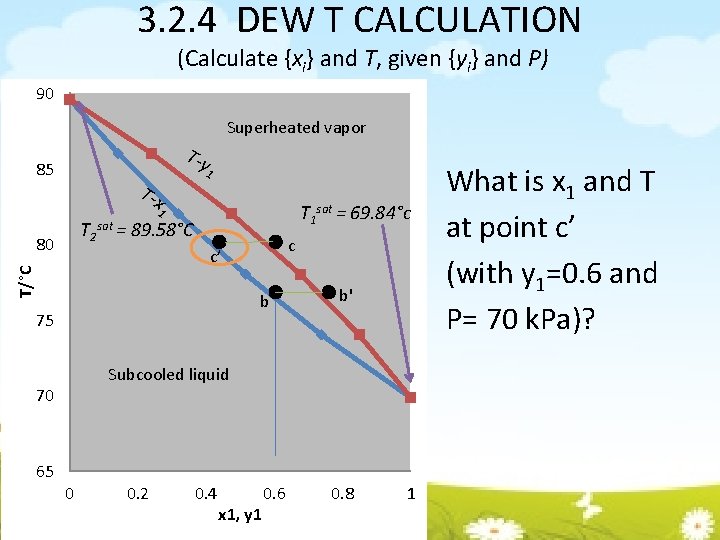
3. 2. 4 DEW T CALCULATION (Calculate {xi} and T, given {yi} and P) 90 Superheated vapor T-y 85 T-x 1 T 1 sat = 69. 84°c 1 T 2 sat = 89. 58°C 80 T/°C b 75 b' Subcooled liquid 70 65 c c’ 0 0. 2 0. 4 x 1, y 1 0. 6 0. 8 1 What is x 1 and T at point c’ (with y 1=0. 6 and P= 70 k. Pa)?

3. 2. 4 DEW T CALCULATION (Calculate {xi} and T, given {yi} and P) Start with α=1, find P 1 sat Find T using Antoine eq & substitute P 1 sat obtained in step 1 Find new α by substituting T Repeat step 1 by using new α until similar value of α is obtained Find x 1

3. 3 Henry’s Law • Used for a species whose critical temperature is less than the temperature of application, in which Raoult’s Law could not be applied (since Raoult’s Law requires a value of Pisat). Where Hi is Henry’s constant and obtained from experiment.

4. VLE by Modified Raoult’s Law • Used when the liquid phase is not an ideal solution. Where ɣi is an activity coefficient (deviation from solution ideality in liquid phase).

4. VLE by Modified Raoult’s Law • For bubblepoint calculation, (assuming • For dewpoint calculation, (assuming ) )

5. VLE from K-value Correlations • Equilibrium ratio, Ki • When Ki > 1, species exhibits a higher concentration of vapor phase • When Ki < 1, species exhibits a higher concentration of liquid phase (is considered as heavy constituent. )

5. VLE from K-value Correlations • K value for Raoult’s Law since • K value for modified Raoult’s Law since

5. VLE from K-value Correlations • For bubblepoint calculations, • For dewpoint calculations

Example For a mixture of 10 mol-% methane, 20 mol-% ethane, and 70 mol-% propane at 50°F, determine: (a) The dewpoint pressure (b) The bubblepoint pressure

Example For a mixture of 10 mol-% methane, 20 mol-% ethane, and 70 mol-% propane at 50°F, determine: (a) The dewpoint pressure When the system at its dewpoint, only an insignificant amount of liquid is present. Thus 10 mol-% methane, 20 mol-% ethane, and 70 mol-% propane are the values of yi. assuming, thus, By trial, find the value of pressure that satisfy

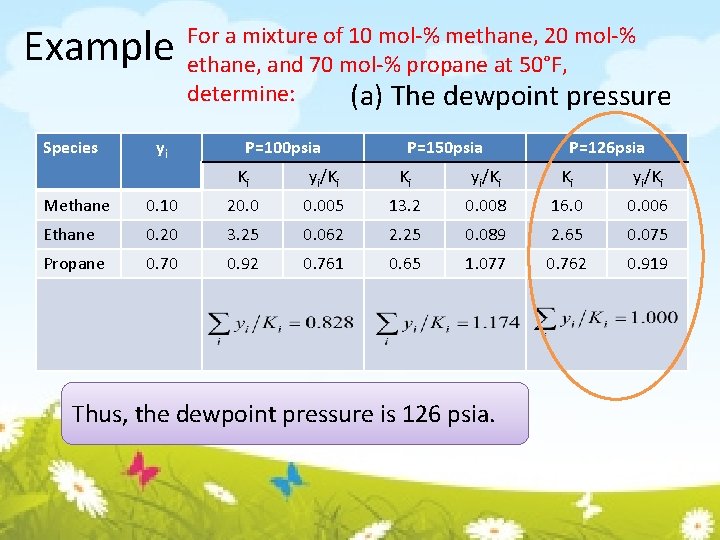
Example Species yi For a mixture of 10 mol-% methane, 20 mol-% ethane, and 70 mol-% propane at 50°F, determine: (a) The dewpoint pressure P=100 psia P=150 psia P=126 psia Ki yi/Ki Methane 0. 10 20. 005 13. 2 0. 008 16. 0 0. 006 Ethane 0. 20 3. 25 0. 062 2. 25 0. 089 2. 65 0. 075 Propane 0. 70 0. 92 0. 761 0. 65 1. 077 0. 762 0. 919 Thus, the dewpoint pressure is 126 psia.

Example For a mixture of 10 mol-% methane, 20 mol-% ethane, and 70 mol-% propane at 50°F, determine: (b)The bubblepoint pressure assuming , thus By trial, find the value of pressure that satisfy


Example For a mixture of 10 mol-% methane, 20 mol-% ethane, and 70 mol-% propane at 50°F, (b) The bubble point pressure determine: Species xi P=380 psia P=400 psia P=385 psia Ki K i xi Methane 0. 10 5. 60 0. 560 5. 25 0. 525 5. 49 0. 549 Ethane 0. 20 1. 11 0. 222 1. 07 0. 214 1. 10 0. 220 Propane 0. 70 0. 335 0. 235 0. 32 0. 224 0. 33 0. 231 Thus, the bubblepoint pressure is 385 psia.
- Slides: 51