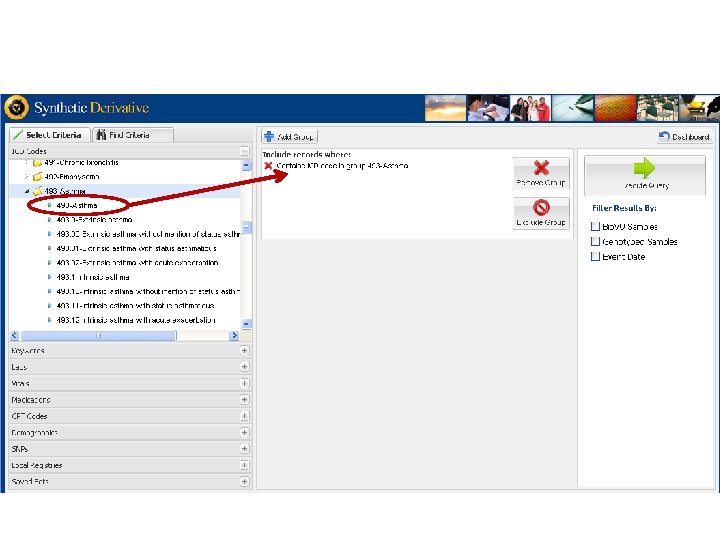
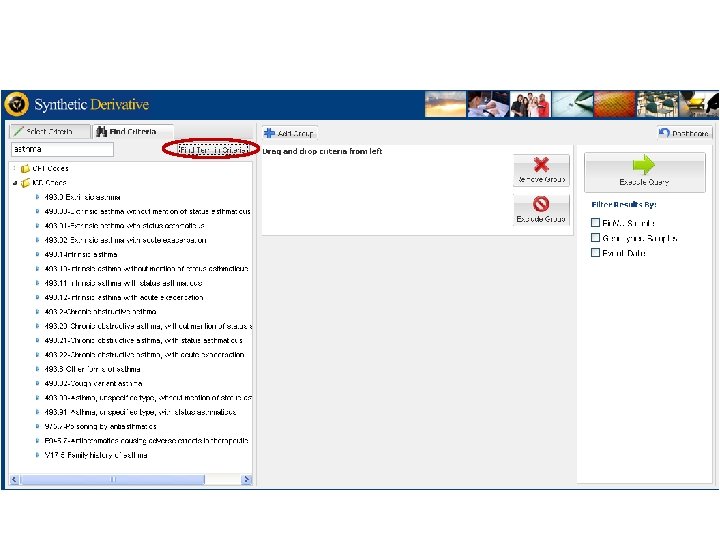
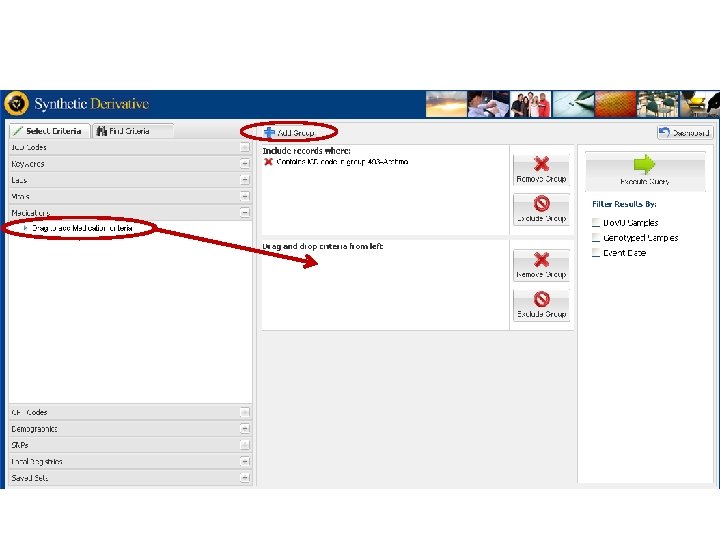
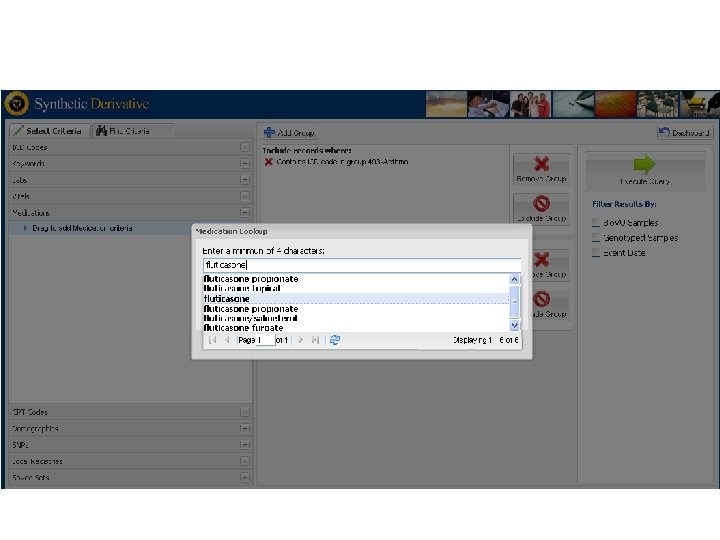
Vanderbilts DNA Databank Bio VU Personalized Medicine Integration

Vanderbilt’s DNA Databank: Bio. VU

Personalized Medicine • Integration of genomic information into clinical decision making • Personalized disease treatment and also preventative therapies

Personalized Medicine • A SNP is a single base-pair mutation that occurs at a specific site in the DNA sequence - occurs in at least 1% of the population • SNPs are responsible for over 80% of the variation between two individuals; they are ideal for establishing correlations between genotype and phenotype • As some SNPs predispose individuals to have a certain disease or trait or react to a drug in a different way, they will be highly useful in diagnostics and drug development

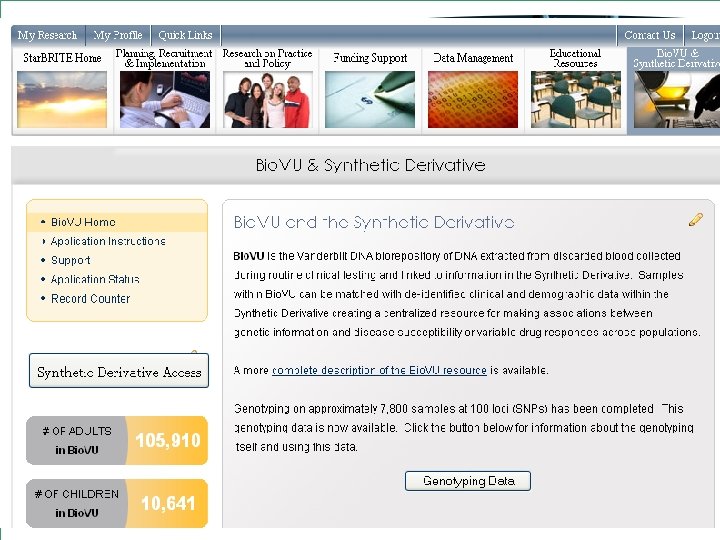
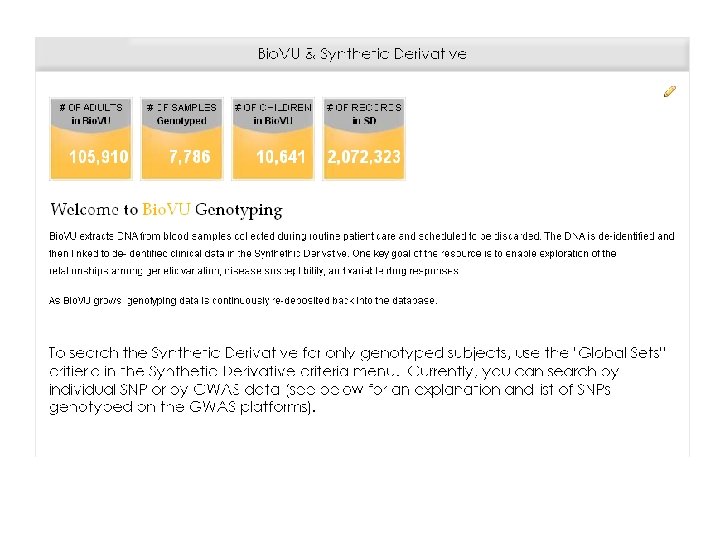
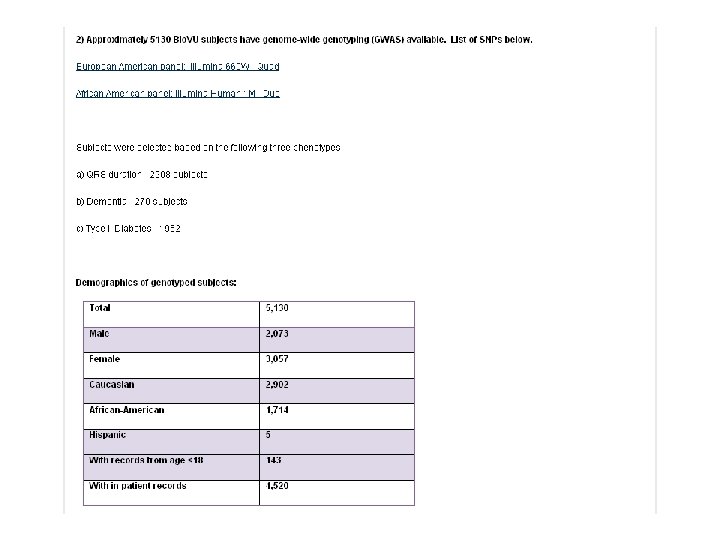
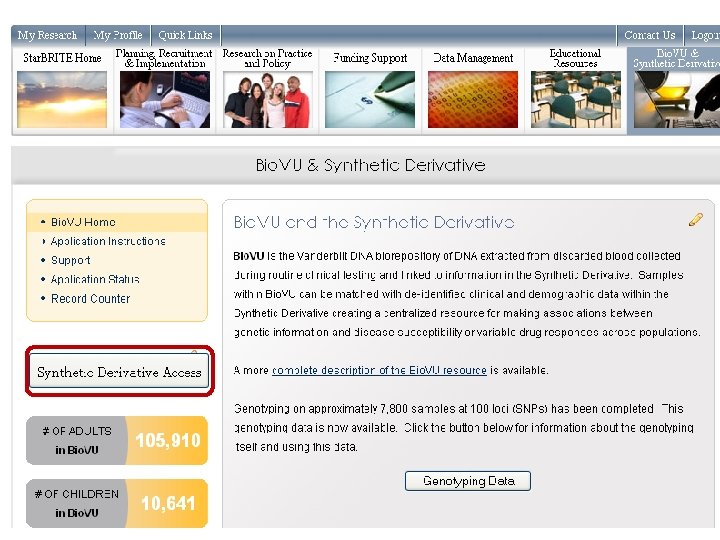
What is Bio. VU? • The move towards personalized medicine requires very large sample sets for discovery and validation • Bio. VU: biobank intended to support a broad view of biology and enable personalized medicine • Contains de-identified DNA extracted from leftover blood after clinically-indicated testing of Vanderbilt patients who have not opted out • Linked to Synthetic Derivative: de-identified EMR • Current sample number: 116, 551 o 105, 910 adult samples o 10, 641 pediatric samples

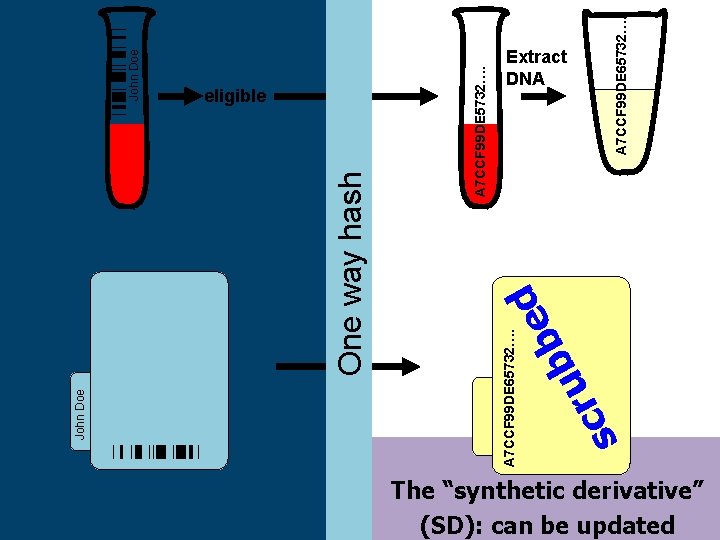
b u r c s d e b John Doe A 7 CCF 99 DE 5732…. One way hash A 7 CCF 99 DE 65732…. John Doe eligible A 7 CCF 99 DE 65732…. Extract DNA The “synthetic derivative” (SD): can be updated
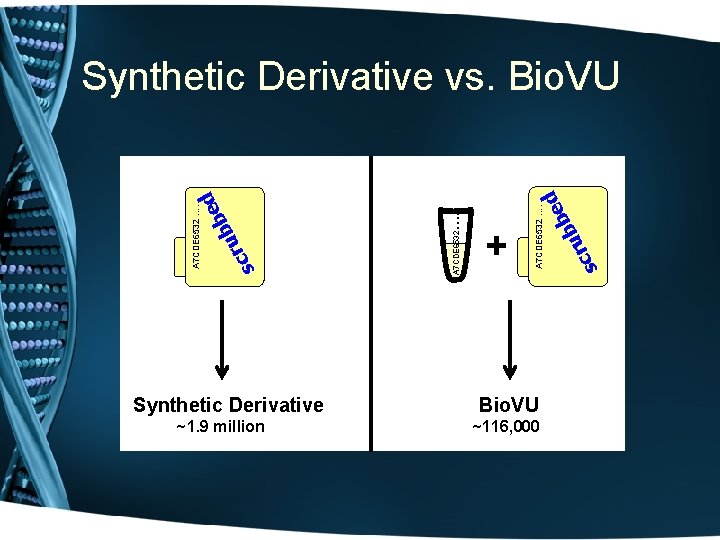
Synthetic Derivative ~1. 9 million Bio. VU ~116, 000 ru c s ed b b + A 7 CDE 6532 …. A 7 CDE 6532 ru sc A 7 CDE 6532 …. ed b b Synthetic Derivative vs. Bio. VU

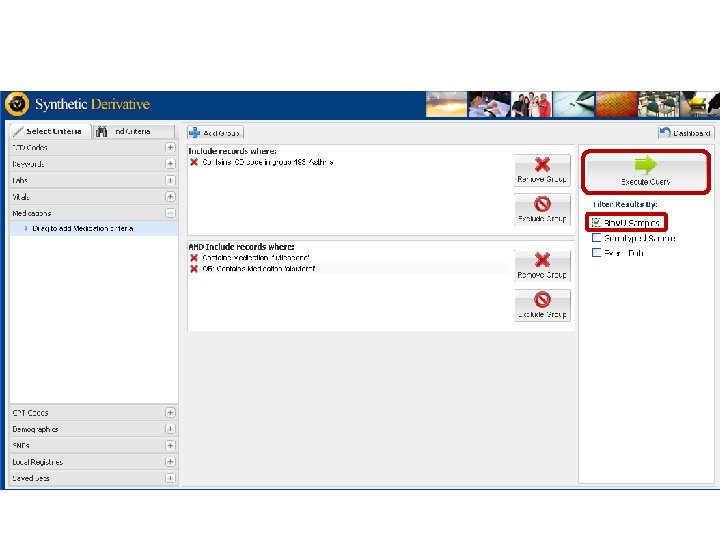
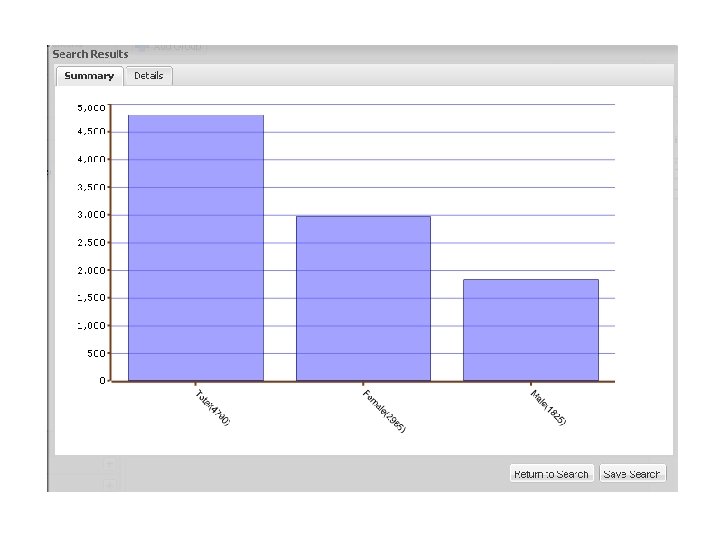
The Synthetic Derivative • A Derivative of the EMR - information content reduced by ‘scrubbing’ identifiers • • Systematically shifted event dates Contains ~1. 9 million records o o o • • ~1 million with detailed longitudinal data averaging 100, 000 bytes in size an average of 27 codes per record Records updated over time and are current through 9/31/09 Can be searched restricting to records for which DNA is available

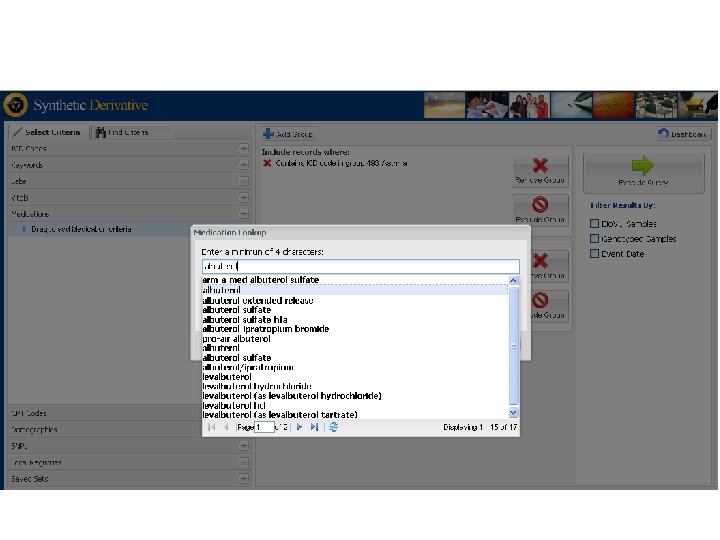
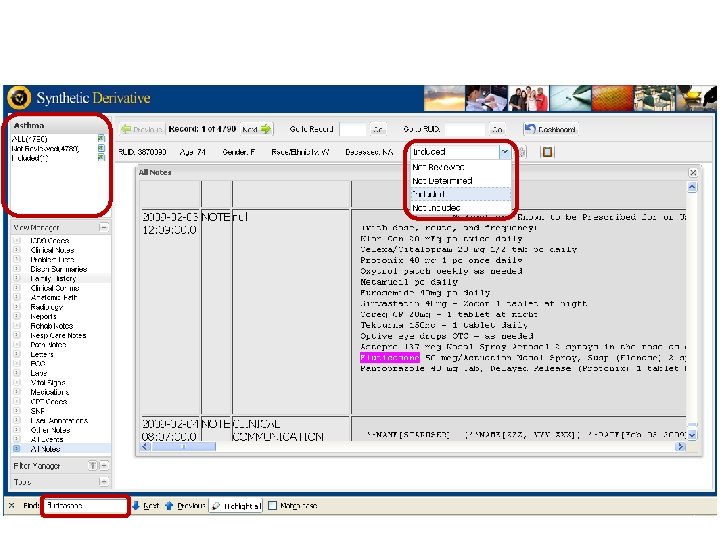
Synthetic Derivative Data Types § Narratives, such as: • Clinical Notes • Discharge Summaries • History and Physicals • Problem Lists • Surgical Reports • Progress Notes • Letters § Diagnostic Codes, Procedural Codes § Forms (intake, assessment) § Reports (pathology, ECGs, echocardiograms) § Clinical Communications § Lab Values and Vital Signs § Medication Orders § Trace. Master (ECGs)
Bio. VU Program Review
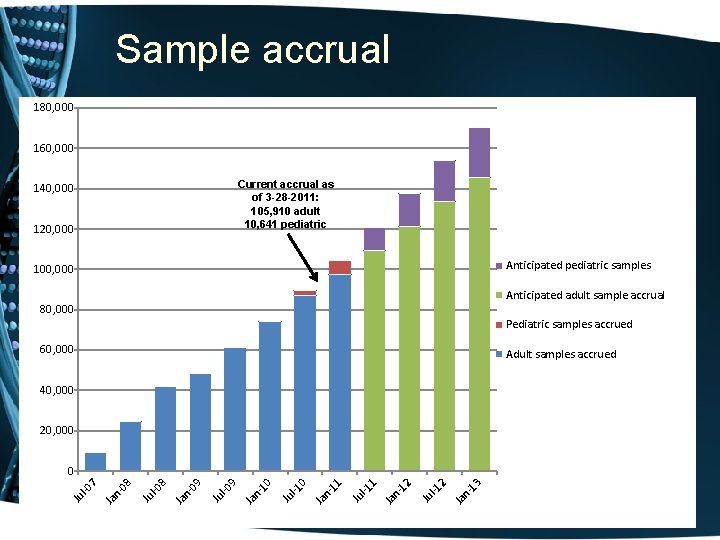
Sample accrual 180, 000 160, 000 Current accrual as of 3 -28 -2011: 105, 910 adult 10, 641 pediatric 140, 000 120, 000 Anticipated pediatric samples 100, 000 Anticipated adult sample accrual 80, 000 Pediatric samples accrued 60, 000 Adult samples accrued 40, 000 20, 000 Ja n 13 2 l-1 Ju Ja n 12 1 l-1 Ju Ja n 11 0 l-1 Ju Ja n 10 9 l-0 Ju Ja n 09 8 l-0 Ju Ja n 08 Ju l-0 7 0

Bio. VU Sample Management RTS Sma. RTStore

Validation in Bio. VU • Sample handling algorithms o o • Ancestry o o o • Gender match 1/384 gender mismatches Characterize sample ancestry, assess usefulness of ‘race’ as defined in EMR Provide a panel of ancestry informative markers that define ancestry No significant difference between the concordance of self-report or observer-report with genetic ancestry Demonstration project – American Journal of Human Genetics o Can known associations between genetic variants and common diseases be identified in the EMR?

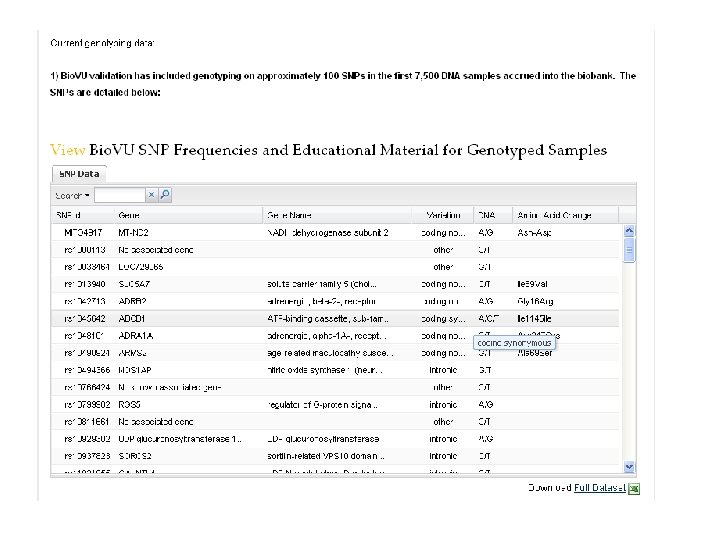
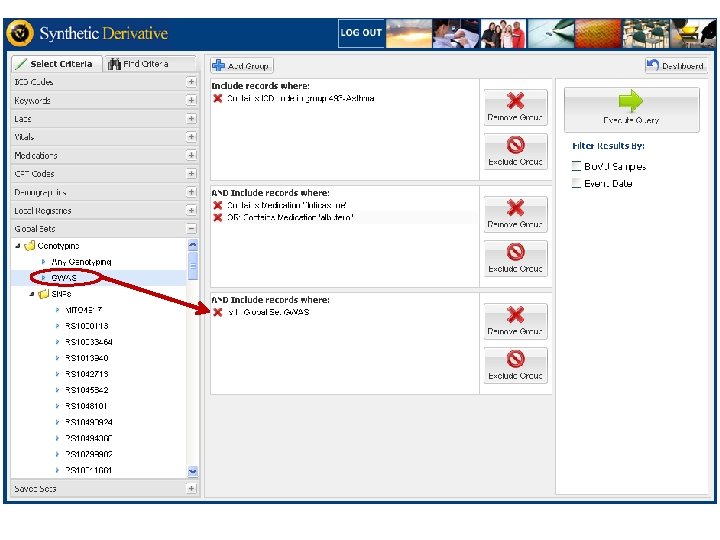
The “demonstration project” • Genotype “high-value” SNPs in the first 8, 000 samples accrued. o including SNPs associated by replicated genome-wide experiments with common diseases & traits 1. 2. 3. 4. 5. Atrial fibrillation Crohn’s disease Multiple Sclerosis Rheumatoid arthritis Type II Diabetes • Develop Natural Language Processing methods to identify cases and controls • Are genotype-phenotype relations replicated?
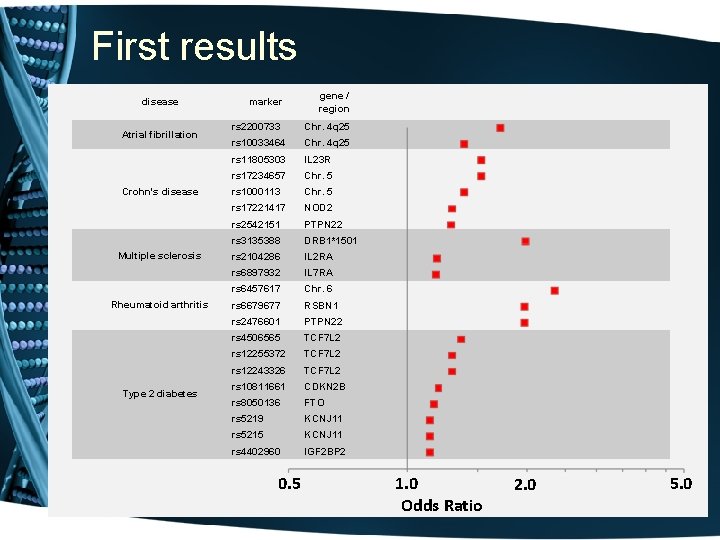
First results disease Atrial fibrillation Crohn's disease Multiple sclerosis Rheumatoid arthritis Type 2 diabetes marker gene / region rs 2200733 Chr. 4 q 25 rs 10033464 Chr. 4 q 25 rs 11805303 IL 23 R rs 17234657 Chr. 5 rs 1000113 Chr. 5 rs 17221417 NOD 2 rs 2542151 PTPN 22 rs 3135388 DRB 1*1501 rs 2104286 IL 2 RA rs 6897932 IL 7 RA rs 6457617 Chr. 6 rs 6679677 RSBN 1 rs 2476601 PTPN 22 rs 4506565 TCF 7 L 2 rs 12255372 TCF 7 L 2 rs 12243326 TCF 7 L 2 rs 10811661 CDKN 2 B rs 8050136 FTO rs 5219 KCNJ 11 rs 5215 KCNJ 11 rs 4402960 IGF 2 BP 2 0. 5 1. 0 Odds Ratio 2. 0 5. 0
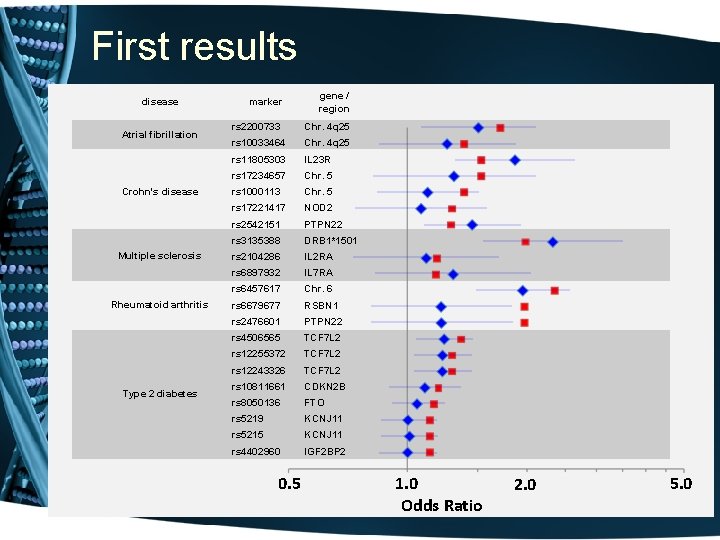
First results disease Atrial fibrillation Crohn's disease Multiple sclerosis Rheumatoid arthritis Type 2 diabetes marker gene / region rs 2200733 Chr. 4 q 25 rs 10033464 Chr. 4 q 25 rs 11805303 IL 23 R rs 17234657 Chr. 5 rs 1000113 Chr. 5 rs 17221417 NOD 2 rs 2542151 PTPN 22 rs 3135388 DRB 1*1501 rs 2104286 IL 2 RA rs 6897932 IL 7 RA rs 6457617 Chr. 6 rs 6679677 RSBN 1 rs 2476601 PTPN 22 rs 4506565 TCF 7 L 2 rs 12255372 TCF 7 L 2 rs 12243326 TCF 7 L 2 rs 10811661 CDKN 2 B rs 8050136 FTO rs 5219 KCNJ 11 rs 5215 KCNJ 11 rs 4402960 IGF 2 BP 2 0. 5 1. 0 Odds Ratio 2. 0 5. 0

Types of projects • Discovery or validation of genotype-phenotype relations for disease susceptibility or drug responses • Discovery of new disease/susceptibility genes resequence in patients (obesity, Cushing's, susceptibility to infection, insomnia, pre-term birth) • Access samples without disease X, or “normals” of specified ancestry, or old normals • Phenome-wide association study (Phe. WAS): in development
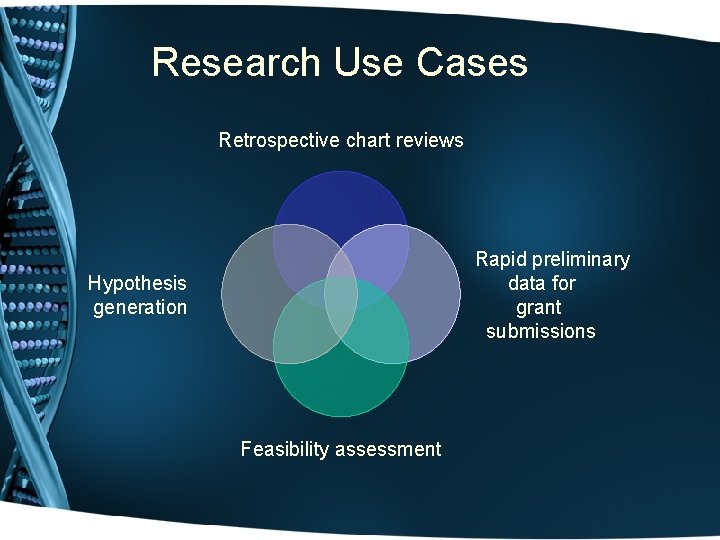
Research Use Cases Retrospective chart reviews Rapid preliminary data for grant submissions Hypothesis generation Feasibility assessment
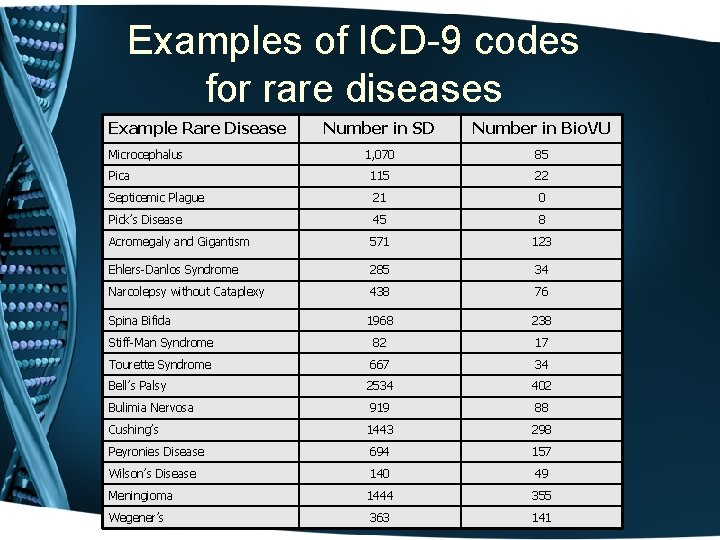
Examples of ICD-9 codes for rare diseases Example Rare Disease Number in SD Number in Bio. VU 1, 070 85 Pica 115 22 Septicemic Plague 21 0 Pick’s Disease 45 8 Acromegaly and Gigantism 571 123 Ehlers-Danlos Syndrome 285 34 Narcolepsy without Cataplexy 438 76 Spina Bifida 1968 238 Stiff-Man Syndrome 82 17 Tourette Syndrome 667 34 Bell’s Palsy 2534 402 Bulimia Nervosa 919 88 Cushing’s 1443 298 Peyronies Disease 694 157 Wilson’s Disease 140 49 Meningioma 1444 355 Wegener’s 363 141 Microcephalus
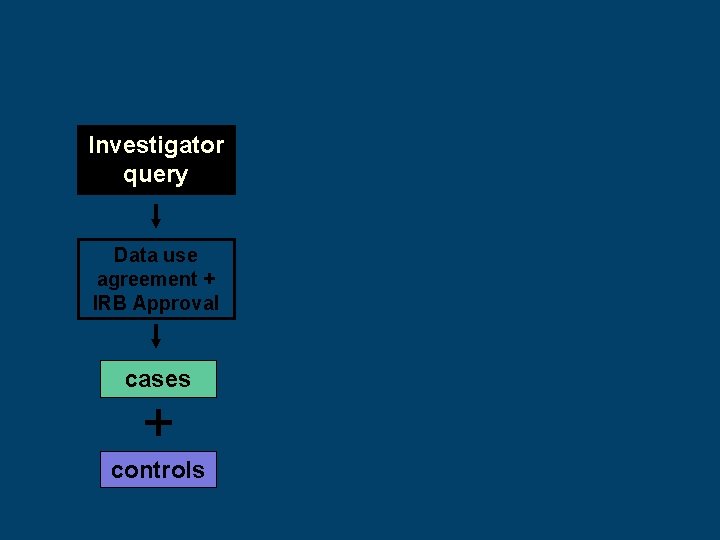
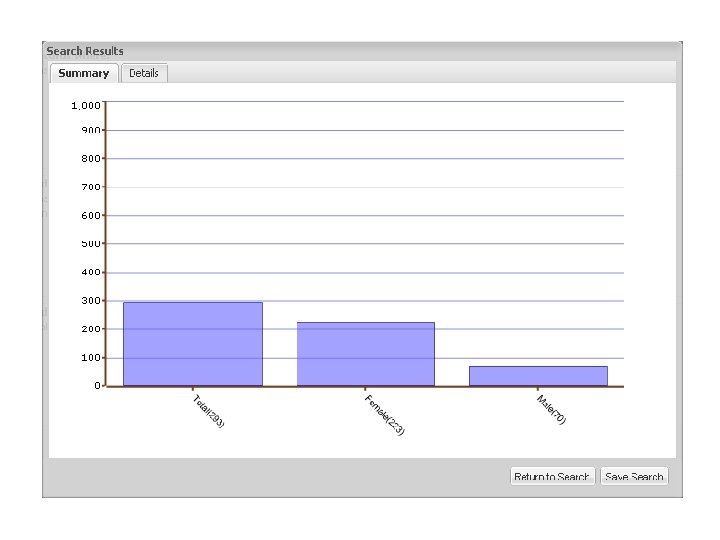
Investigator query Data use agreement + IRB Approval cases + controls
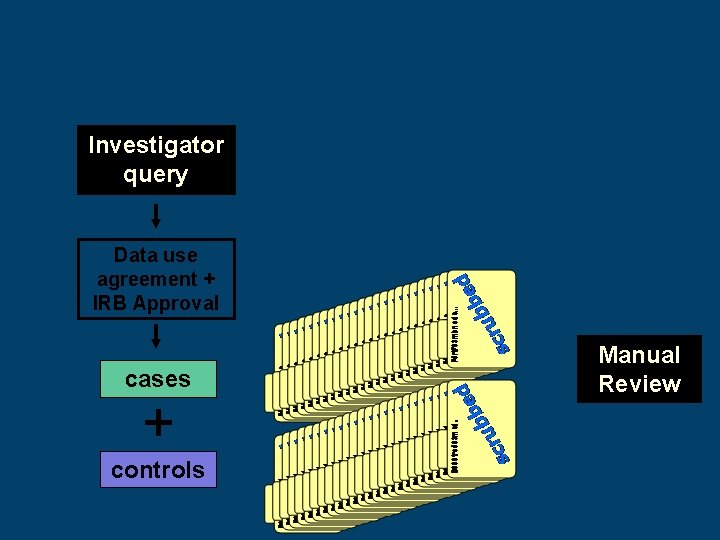
Investigator query Data use agreement + IRB Approval cases + controls Manual Review
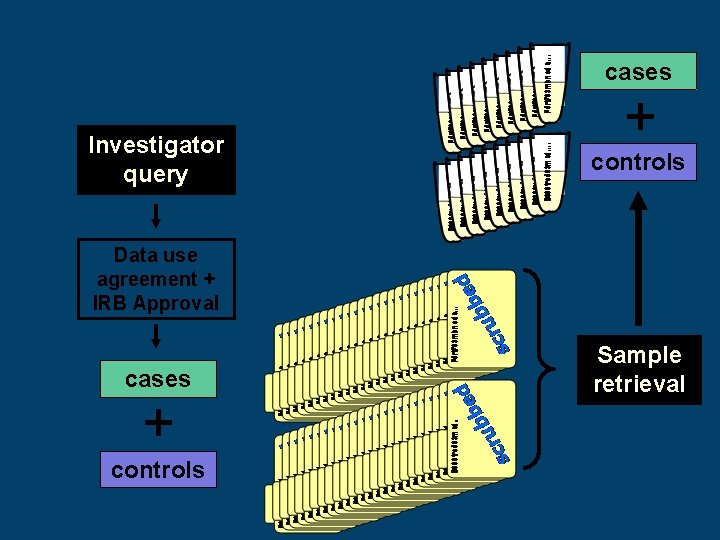
cases Investigator query + controls Data use agreement + IRB Approval cases + controls Sample retrieval
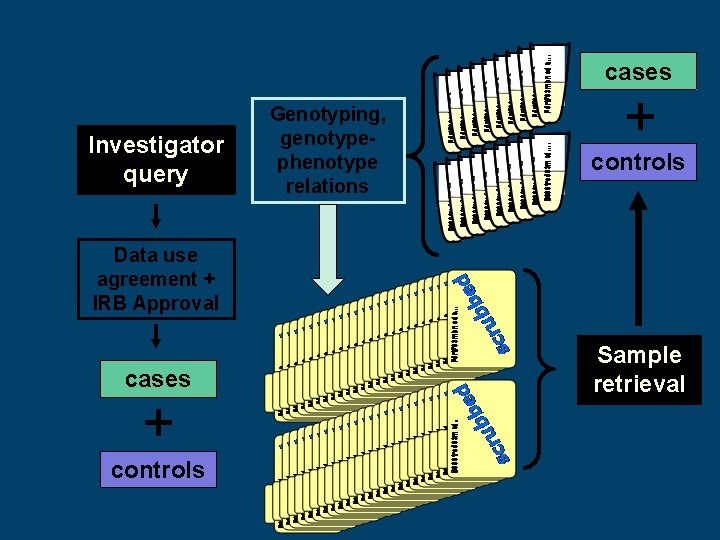
cases Investigator query Genotyping, genotypephenotype relations + controls Data use agreement + IRB Approval cases + controls Sample retrieval

Data Use Agreement
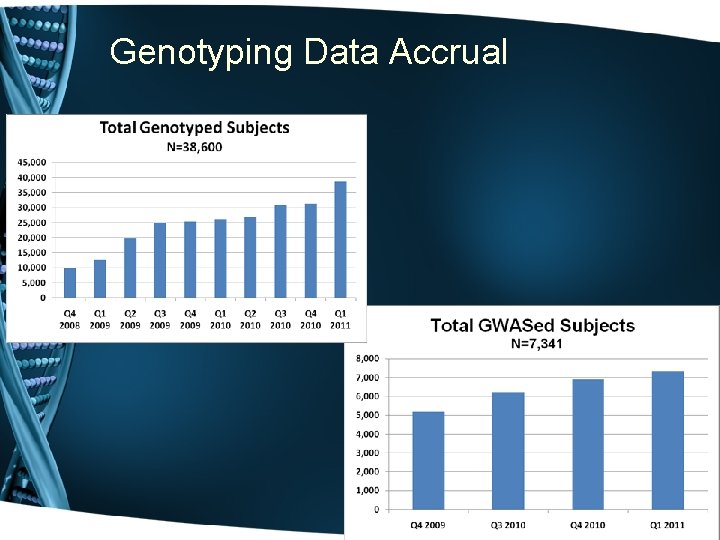
Genotyping Data Accrual

Nationally Prevalent Diseases in the African American Population Disease Bio. VU Count Hypertension 1095 Type 2 Diabetes 714 Coronary Artery Disease 273 Kidney Disease 252 Asthma 210 Pneumonia 193 Stroke 133 Lupus 48 Lung cancer 21

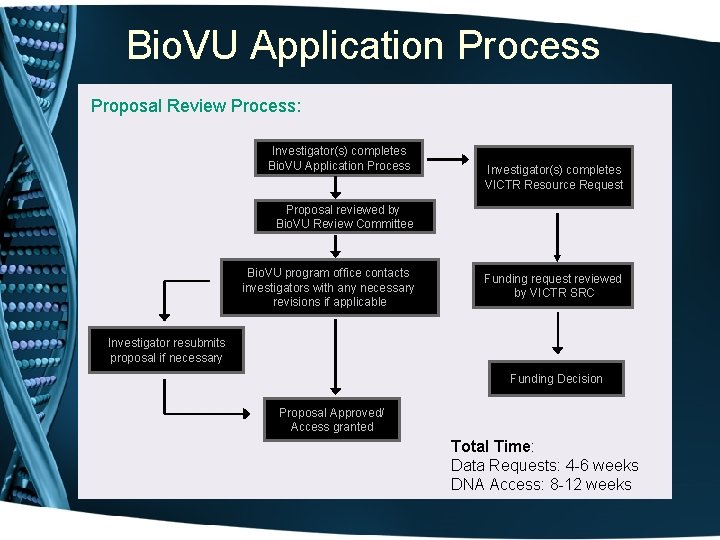
Bio. VU Application Process Proposal Review Process: Investigator(s) completes Bio. VU Application Process Investigator(s) completes VICTR Resource Request Proposal reviewed by Bio. VU Review Committee Bio. VU program office contacts investigators with any necessary revisions if applicable Funding request reviewed by VICTR SRC Investigator resubmits proposal if necessary Funding Decision Proposal Approved/ Access granted Total Time: Data Requests: 4 -6 weeks DNA Access: 8 -12 weeks
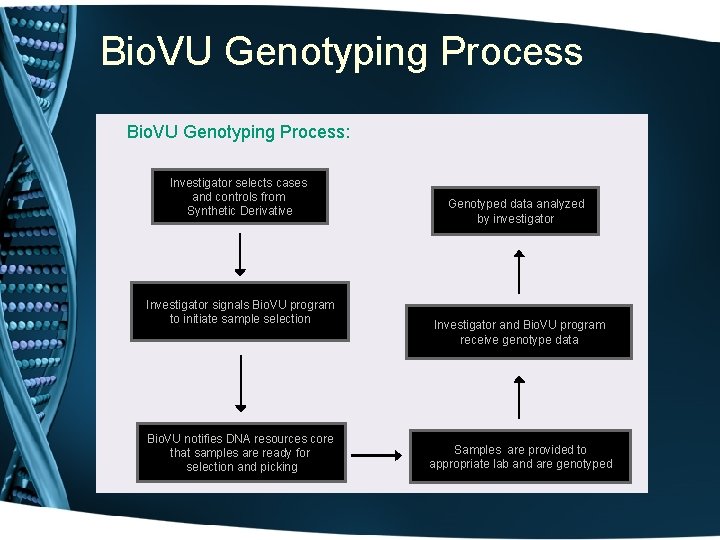
Bio. VU Genotyping Process: Investigator selects cases and controls from Synthetic Derivative Investigator signals Bio. VU program to initiate sample selection Bio. VU notifies DNA resources core that samples are ready for selection and picking Genotyped data analyzed by investigator Investigator and Bio. VU program receive genotype data Samples are provided to appropriate lab and are genotyped
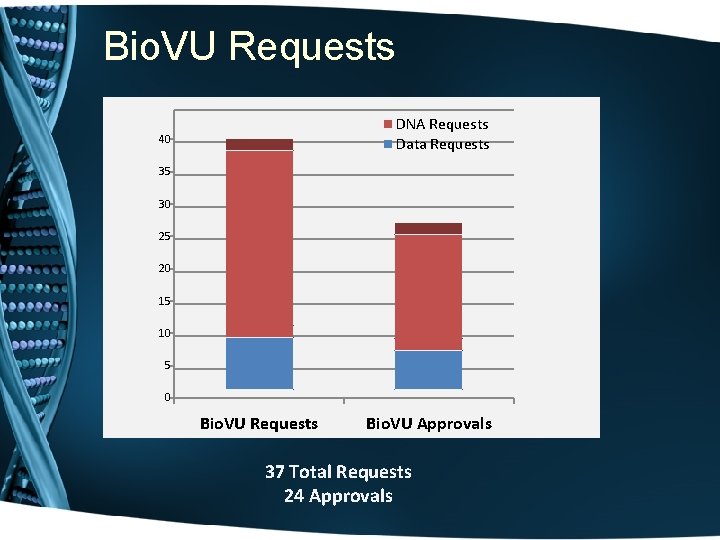
Bio. VU Requests DNA Requests Data Requests 40 35 30 25 20 15 10 5 0 Bio. VU Requests Bio. VU Approvals 37 Total Requests 24 Approvals

FAQ “answers” • SD access: “non-human subjects” IRB review (days) • Current access costs: $4/sample • Genotyping: o Investigator-funded § o Genotyping/sequencing performed in VUMC Core Facilities § o • Consider VICTR as a funding source Justification must be provided for outside genotyping, including quality control plans Genotype “redeposit” part of the data use agreement Anticipate 16, 000 Bio. VU subjects will have GWAS-type genotyping data by fall 2011

Questions? Contact: Erica Bowton Ph. D Bio. VU Program Manager erica. bowton@vanderbilt. edu 322 -1975
- Slides: 57