valence electrons electrons in the outermost principal energy











- Slides: 20


valence electrons – electrons in the outermost principal energy level Example: 6 electrons Oxygen has 6 valence electrons since its electron configuration is 1 s 2 2 p 4 Inner shell Outer shell

Octet Rule Atoms tend to form an octet of electrons in the valence shell by gaining, losing or sharing electrons. Example: Oxygen has 6 valence electrons 1 s 2 2 p 4 Oxygen atoms have a tendency to gain 2 electrons to achieve an octet (8).

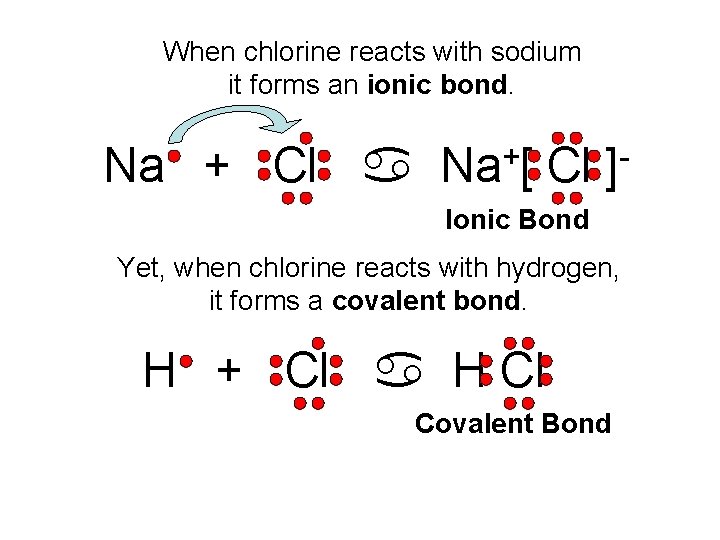
When chlorine reacts with sodium it forms an ionic bond. Na + Cl a + Na [ Cl ] Ionic Bond Yet, when chlorine reacts with hydrogen, it forms a covalent bond. H + Cl a H Cl Covalent Bond

How are you supposed to know whether a compound like HCl or Na. Cl is ionic or covalently bonded?

An ionic bond will form only if one of the atoms has a much greater attraction of electrons than the other atom. What measures electron attraction is electronegativity.

Table of Electronegativities (pg 151) r Inc s ea

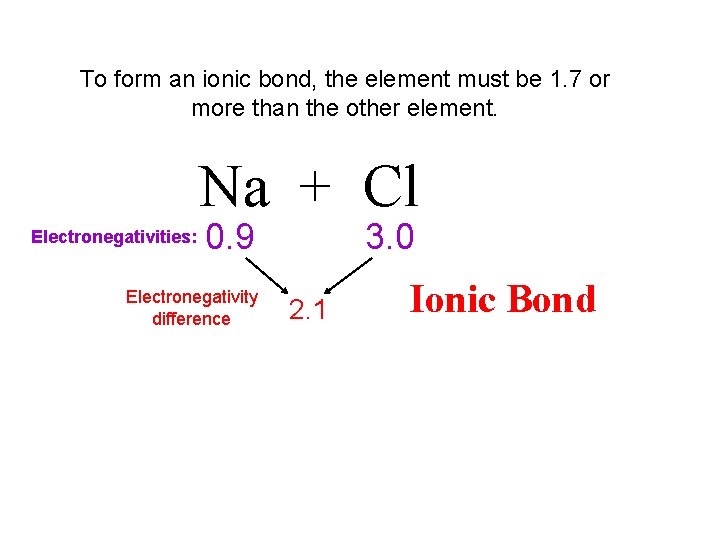
To form an ionic bond, the element must be 1. 7 or more than the other element. Na + Cl Electronegativities: 0. 9 Electronegativity difference 3. 0 2. 1 Ionic Bond

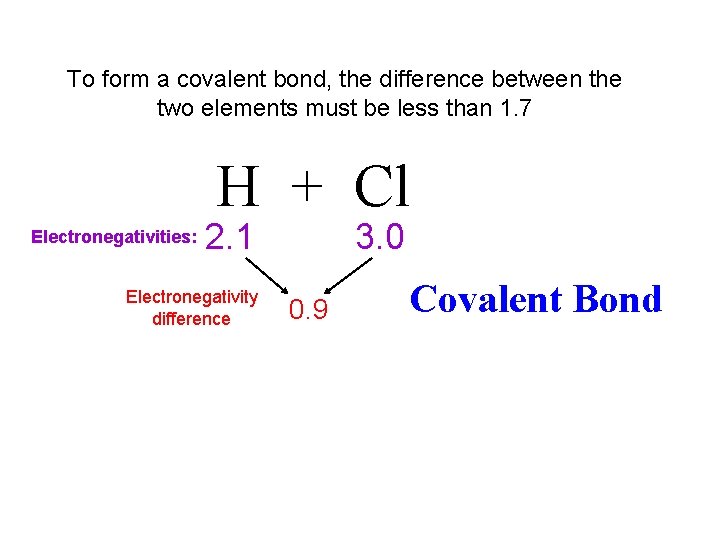
To form a covalent bond, the difference between the two elements must be less than 1. 7 H + Cl Electronegativities: 2. 1 Electronegativity difference 3. 0 0. 9 Covalent Bond

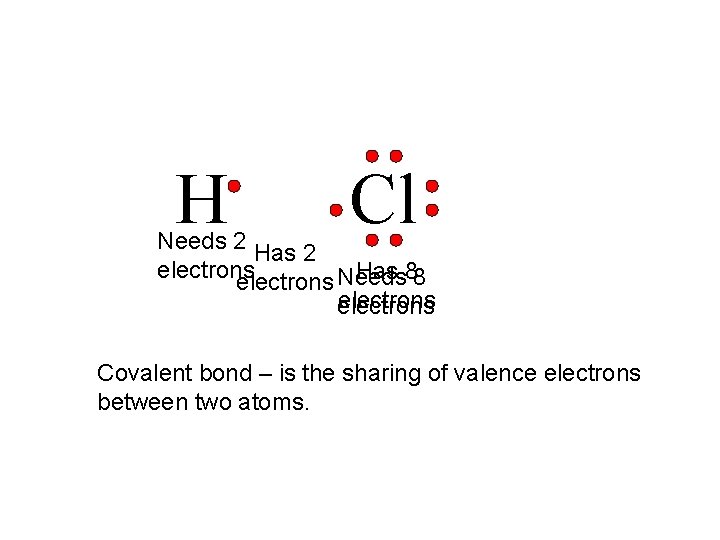
H Cl Needs 2 Has 2 electrons Has 88 electrons Needs electrons Covalent bond – is the sharing of valence electrons between two atoms.

0. 9 d HCl 2. 1 3. 0 d + - Polar Covalent bond This bond is considered to be covalent with slight ionic character because of the slight positive and negative charges at each end.

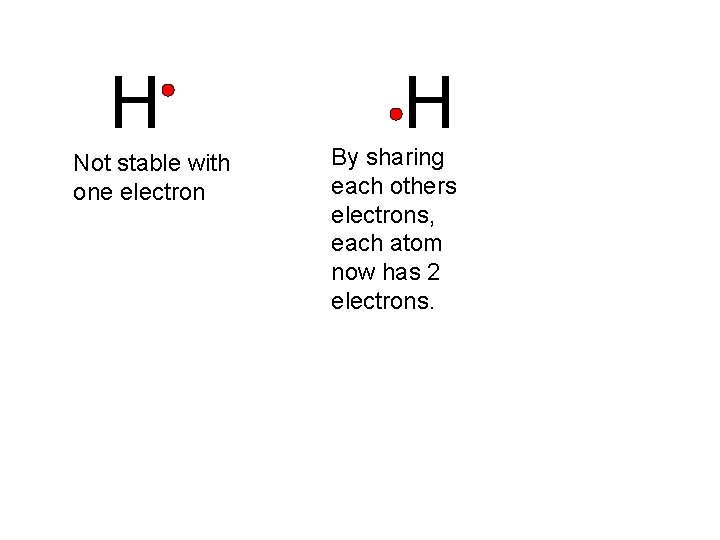
H Not stable with one electron H By sharing each others electrons, each atom now has 2 electrons.

HH Electronegativities: 2. 1 Electronegativity difference 0. 0 Nonpolar Covalent bond

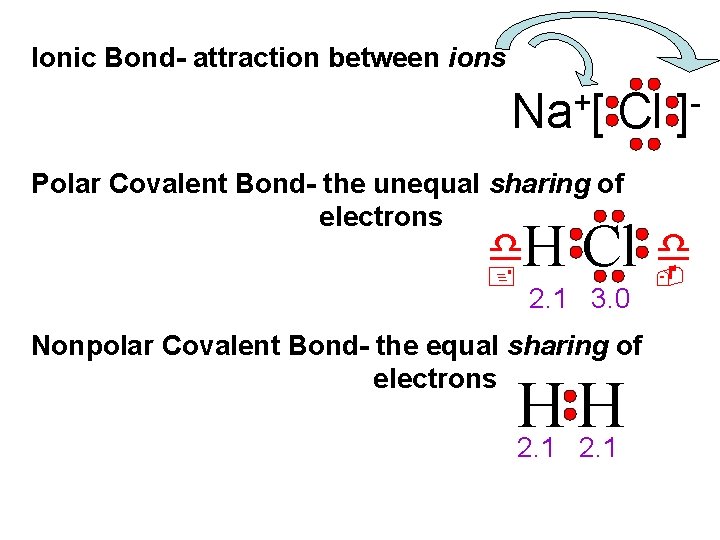
Ionic Bond- attraction between ions + Na [ Cl Polar Covalent Bond- the unequal sharing of electrons d. H + ] Cl d - 2. 1 3. 0 Nonpolar Covalent Bond- the equal sharing of electrons H H 2. 1

d type Ionic Bon. Electronegativity difference 1. 7 or more Polar Covalent 0. 3 to 1. 6 Nonpolar Covalent 0. 0 to 0. 2

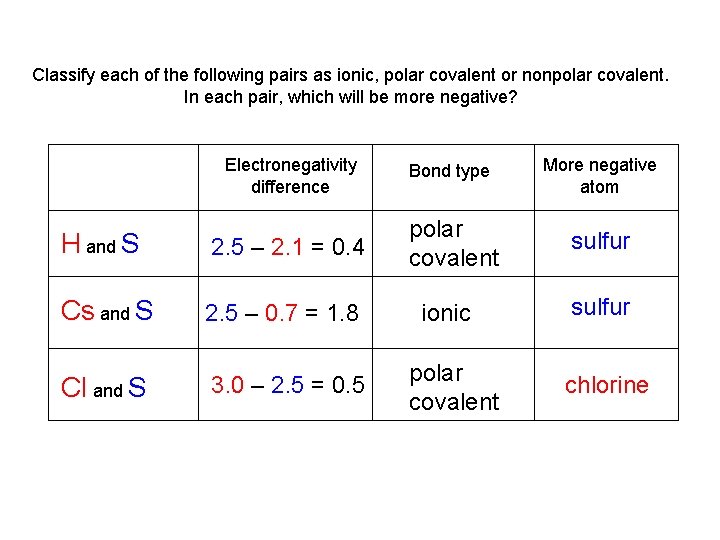
Examples from you textbook pg 163 Classify each of the following pairs as ionic, polar covalent or nonpolar covalent. In each pair, which will be more negative? H and S Cs and S Cl and S

Classify each of the following pairs as ionic, polar covalent or nonpolar covalent. In each pair, which will be more negative? Electronegativity difference Bond type More negative atom H and S 2. 5 – 2. 1 = 0. 4 polar covalent sulfur Cs and S 2. 5 – 0. 7 = 1. 8 Cl and S 3. 0 – 2. 5 = 0. 5 ionic polar covalent sulfur chlorine

Examples from you textbook pg 163 Classify each of the following pairs as ionic, polar covalent or nonpolar covalent. In each pair, which will be more negative? Cl and Ca Cl and O Cl and Br

Classify each of the following pairs as ionic, polar covalent or nonpolar covalent. In each pair, which will be more negative? Cl and Ca Electronegativity difference Bond type More negative atom 3. 0 – 1. 0 = 2. 0 ionic chlorine Cl and O 3. 5 – 3. 0 = 0. 5 polar covalent Cl and Br 3. 0 – 2. 8 = 0. 2 nonpolar covalent oxygen chlorine

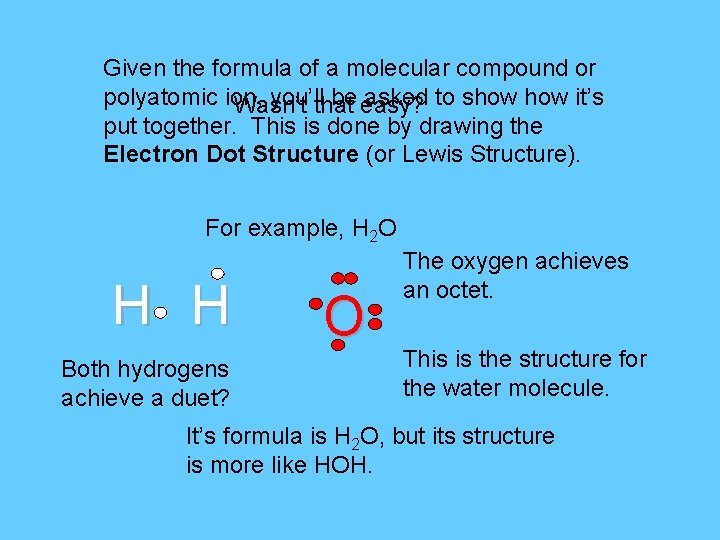
Given the formula of a molecular compound or polyatomic ion, you’llthat be easy? asked to show it’s Wasn’t put together. This is done by drawing the Electron Dot Structure (or Lewis Structure). For example, H 2 O H H Both hydrogens achieve a duet? O The oxygen achieves an octet. This is the structure for the water molecule. It’s formula is H 2 O, but its structure is more like HOH.