Valence Electrons and Chemical Bonds 06 and 08

Valence Electrons and Chemical Bonds 06 and 08 October 2015 Principles of Valence Electrons and Bonds n Ionic Bonds n Metallic Bonds n Covalent Bonds n Intermolecular Forces n Water’s Unique Properties n
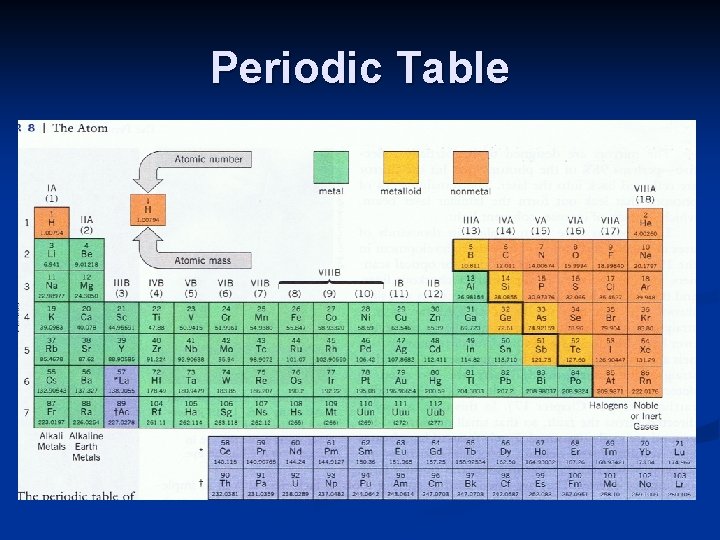
Periodic Table

Two Atoms in Proximity n Observation: when two atoms are brought together, electrons re-arrange to the lowest energy state (i. e. , where valence electrons are most stable) n Consequence: distribution of electrons among atoms re-arranged into bonds n n n Give away electrons Accept electrons Share electrons

Orbits, Shells and Electrons http: //www. colorado. edu/physics/2000/applets/a 2. ht ml

Ionic Bonding n Some atoms give away electrons, whereas other atoms receive electrons n Example of water (two atoms of hydrogen + one atom of oxygen) – H 2 O 1 H + 8 O 1 H + = H 2 O 1 16 1 n Example of lithium (Li) chloride (Cl) 3 Li 6 + 17 35. 5 Cl = Li. Cl

Ionic Bonding n Lithium (Li) Li gives up 1 electron and is left with 2 electrons (-) and 3 protons (+); net positive (+) charge n Chlorine (Cl) Cl has 1 unpaired electron in valence shell, so Cl tends to accept an electron and is left with 18 electrons (-) and 17 protons; net negative (-) charge

Ionic Bonding n n Some atoms tend to give away electrons, whereas other atoms tend to receive electrons Example of lithium chloride Li + Cl = Li. Cl Bonding via electrical attraction between Li+ and Cln Li+ + Cl - = Li+Cl. Consequence: ionic bonds are underpinned by charged ions (atoms with a charge) form crystals of very specific and repeating geometry (very rigid)

Ionic Bonding: Salt

Metallic Bonds n n n Elements that do not give or take electrons (no ionic bonding) but share electrons Valence electrons move freely between adjacent atoms (contrast with ionic bonds) Significance of sharing electrons: compounds tend to show two features n n n Malleability (easily worked or pounded) Conductive of electricity (good conductors) Examples n n Gold jewelry Copper wire

Covalent Bonds (Remember These) n Extremes of behavior in bonding Accept or give away electrons (ionic bonds) n No tendency to share (noble gases) n n Intermediate between these two extremes Do not form ionic bonds n Do not form metallic bonds n Yet share 1, 2, 3 and 4 electrons in unique arrangement called covalent bonds n n Key: orbits of valence electrons are shared so that electrons are shared (and move) between valence shells of adjacent atoms (contrast with metallic bonds)
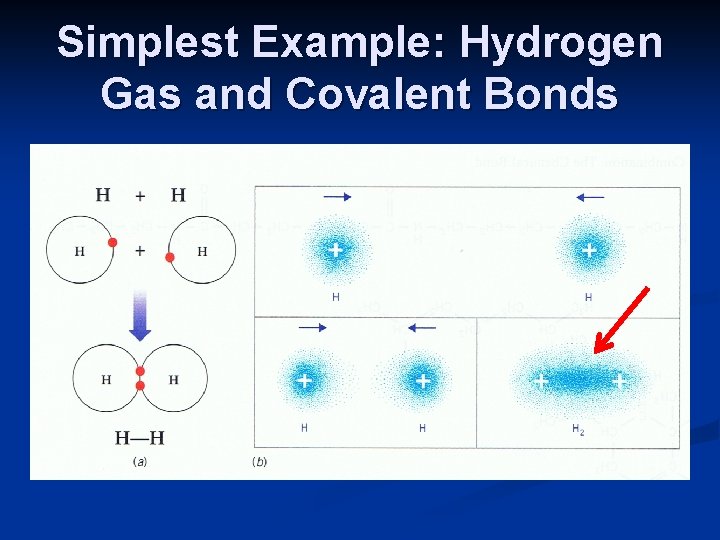
Simplest Example: Hydrogen Gas and Covalent Bonds

Covalent Bonds: Carbon n 6 C 12 is a special case (important in biology & chemistry) n Valence electrons for C are 4 (1 in each orbit) and intermediate between giving and accepting n C - C : single covalent bond (1 orbit) n C - C : two covalent bonds (2 orbits) n Unique behavior of C: four simultaneous C - C bonds C
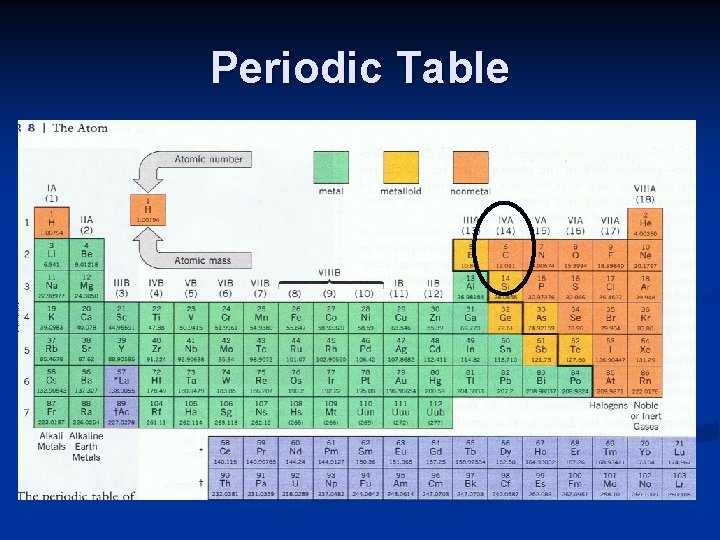
Periodic Table

Behavior of Valence Electrons: Five Options No action (e. g. , inert gases) n Give away one or more electrons in valence state (positive ion leading to ionic bond) n Accept one or more electrons to valence state (negative ion leading to ionic bond) n Share an electron with many other atoms without respect to an orbit/shell (metallic bond) n Share one or more electrons plus their orbits with another atom (covalent bond) n

Valence Electrons and Chemical Bonds 06 and 08 October 2015 Principles of Valence Electrons and Bonds n Ionic Bonds n Metallic Bonds n Covalent Bonds n Inter-molecular Forces n Water’s Unique Properties n

Intermolecular Forces: Polarization & Hydrogen Bonding n Example of water (H 2 O) n When one molecule’s distribution of atoms results in one side of the molecule having either a + or - charge Resulting distribution of charges causes adjoining H 2 O molecule to align with + and charges to be most stable Called “polarity” of molecule (e. g. , magnet) n n

Intermolecular Forces: Van der Waal Forces n In polarity, specific and rigid + and – fields on each molecule that do not change over time n When molecules converge, inevitable that electrons shift and re-distribute (e. g. , planar compound) n In re-distribution, small (weak) net attraction between molecules arise and two molecules form weak bond n Graphite pencil lead

Valence Electrons and Chemical Bonds 06 and 08 October 2015 Principles of Valence Electrons and Bonds n Ionic Bonds n Metallic Bonds n Covalent Bonds n Intermolecular Forces n Water’s Unique Properties n


Water and Its Properties: Liquid State n Water is liquid over broad range of temperatures 0 o. C n to 100 o. C Comparison with other compounds Compound Chemical Formula Water Ammonia Methane H 2 O NH 3 CH 4 Freeze Vapor Range (o. C) 0 -78 -182 100 -33 -164 100 45 18

Water and Its Properties: p. H n n p. H scale: ionization of H 2 O H+ and OHIncrease in H+ results in more acid solution from 7 to 0 Increase in OH- results in more basic solution from 7 to 14 Examples n n n Rainwater of 5. 6 means what? Cell p. H value of 7 -9 means what? See text more discussion of p. H and buffering Importance tofor biological systems

Water and Its Properties: Freezing n Water is unusual in that H 2 O is less dense as a solid than as a liquid (it floats!) n Mechanism n n n H 2 O expands when it solidifies Due to hydrogen bonding Consequence n Ponds, lakes and ocean freeze from the top down

Hierarchy Theory and Emergent Properties of H 2 O Principle of hierarchy theory n Principle of emergent properties n n n a priori: combine one atom of O with two atoms of H and what would you expect? Emergent properties Liquid n Hydrogen bonding and polarity n H+ and OH- in solution n Solvent n Range of temperature at which liquid n Three phases (gas, liquid and solid) n

Importance of Chemical Bonds n Interactions between atoms (i. e. , bonding) responsible for creation of myriad of molecules and compounds (natural and human made) n Interactions between atoms (i. e. , bonding) responsible for all chemical reactions that effect a change in energy (i. e. , kinetic and potential) n Granola bar for lunch n Role of covalent bonds in living systems is basis for most of the uniqueness of life n Water molecule’s unique properties and importance in sciences (physical, chemical and biological)
- Slides: 24