Valence Electrons and Bonding The number of valence
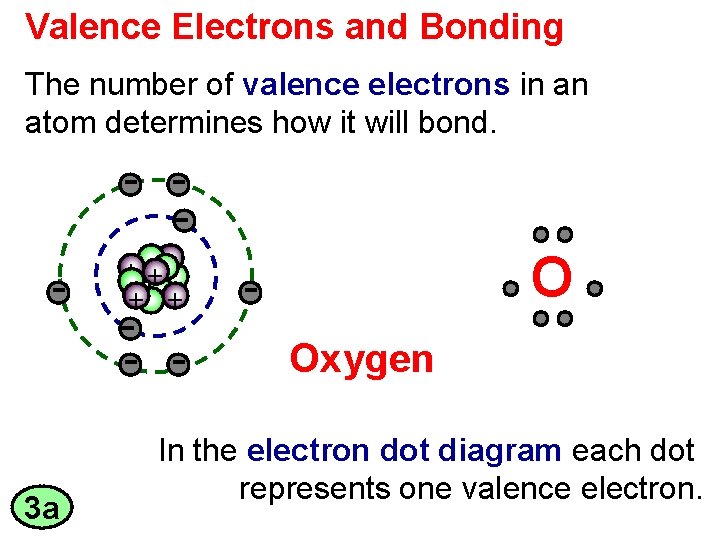
Valence Electrons and Bonding The number of valence electrons in an atom determines how it will bond. + ++ + + O Oxygen 3 a In the electron dot diagram each dot represents one valence electron.
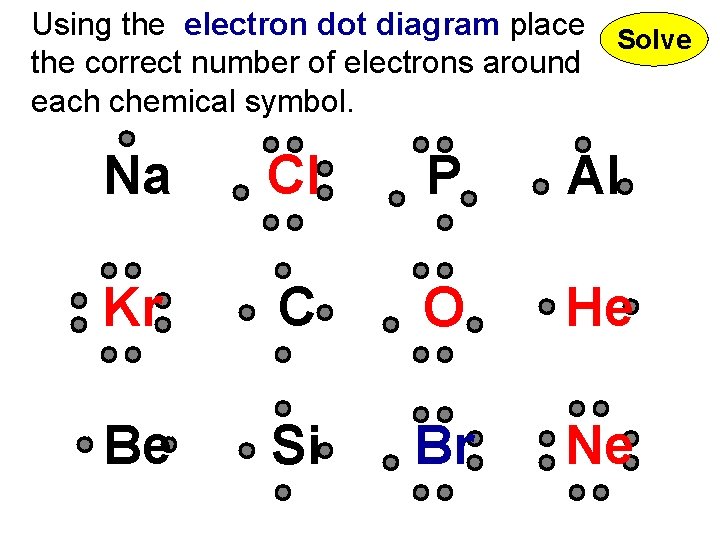
Using the electron dot diagram place Solve the correct number of electrons around each chemical symbol. Na Cl P Al Kr C O He Be Si Br Ne
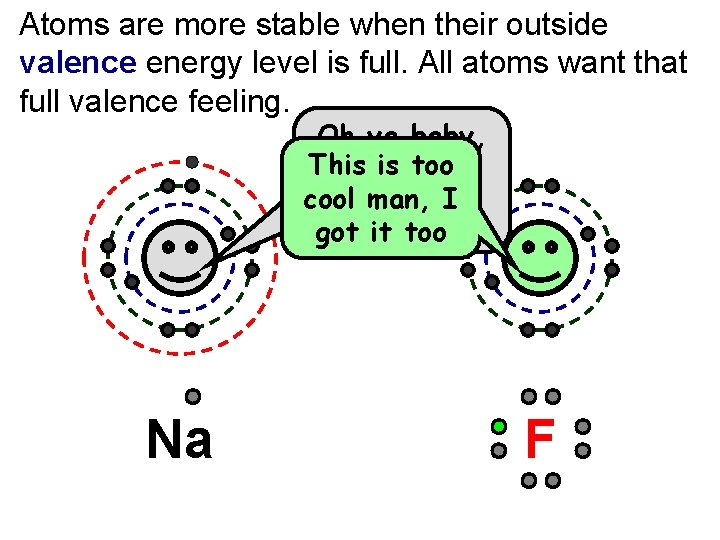
Atoms are more stable when their outside valence energy level is full. All atoms want that full valence feeling. P 11 N 12 Na Oh ya baby, This is too I got that cool I fullman, valence gotfeeling! it too P 9 N 10 F
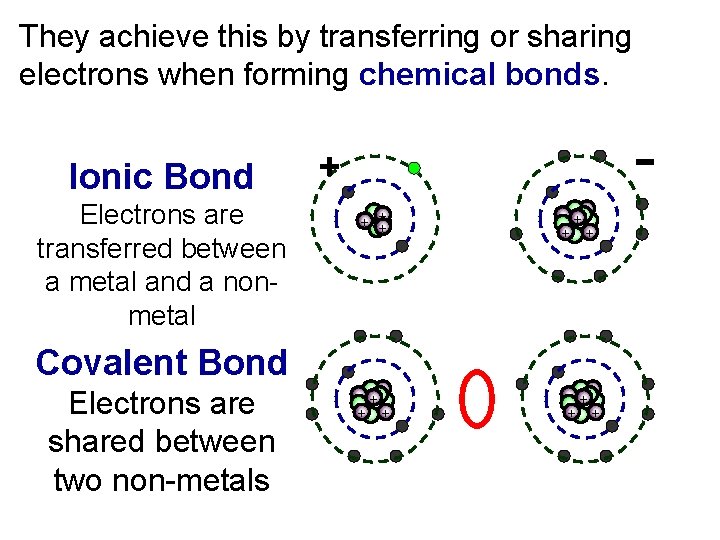
They achieve this by transferring or sharing electrons when forming chemical bonds. Ionic Bond Electrons are transferred between a metal and a nonmetal + + ++ +++ Covalent Bond Electrons are shared between two non-metals ++ +++

Ionic Bonds Chapter 5 Section 2 Pg. 184 -189
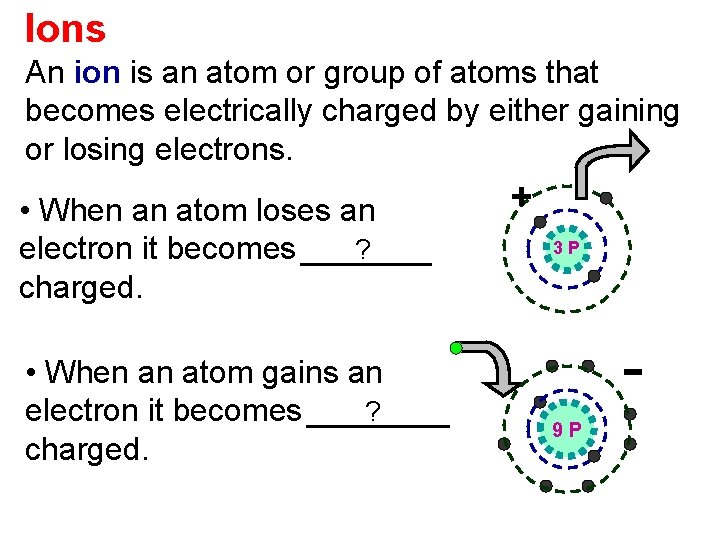
Ions An ion is an atom or group of atoms that becomes electrically charged by either gaining or losing electrons. • When an atom loses an ? electron it becomes positively charged. • When an atom gains an ? electron it becomes negatively charged. + + 3 P + + P ++9++ +
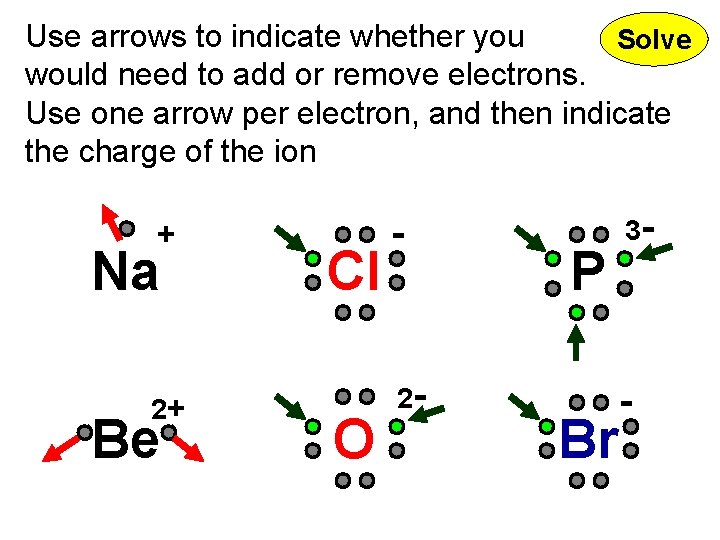
Use arrows to indicate whether you Solve would need to add or remove electrons. Use one arrow per electron, and then indicate the charge of the ion + Na 2+ Be Cl O - 2 - P Br 3 - -
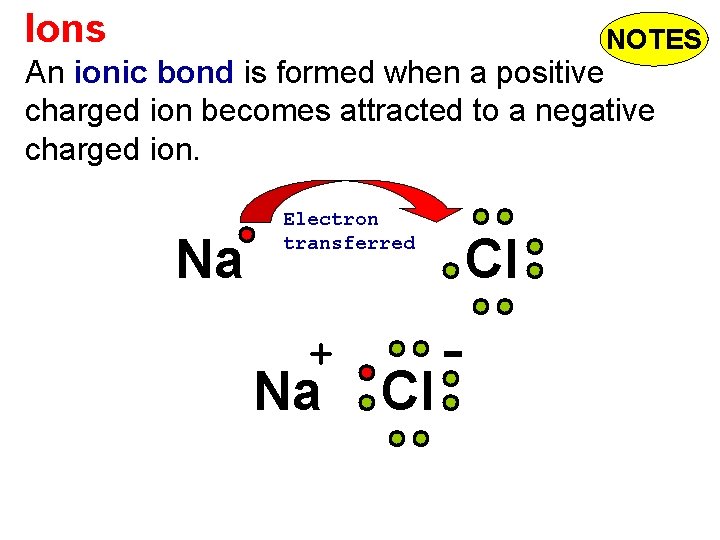
Ions NOTES An ionic bond is formed when a positive charged ion becomes attracted to a negative charged ion. Na Electron transferred + Na Cl Cl
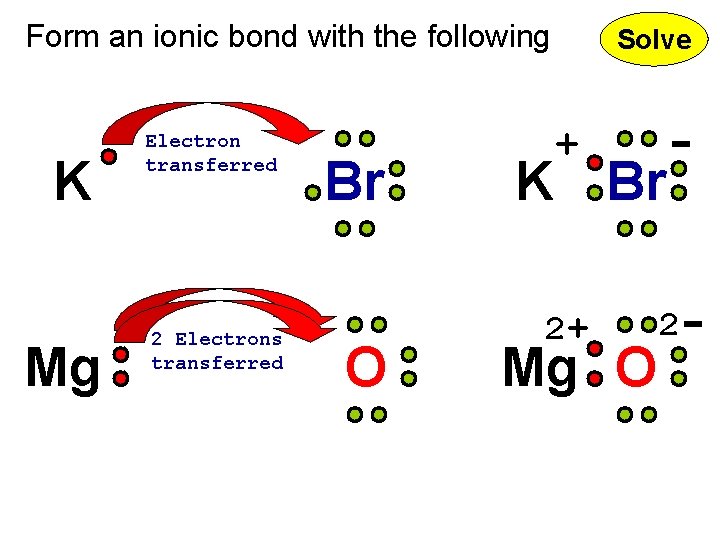
Form an ionic bond with the following K Mg Electron transferred 2 Electrons transferred Br O Solve + K Br 2+ Mg O 2
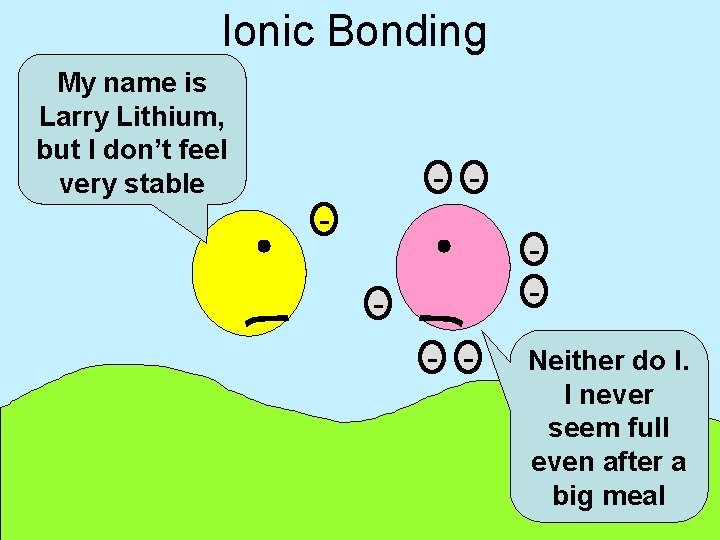
Ionic Bonding My name is Larry Lithium, but I don’t feel very stable - - - Neither do I. I never seem full even after a big meal
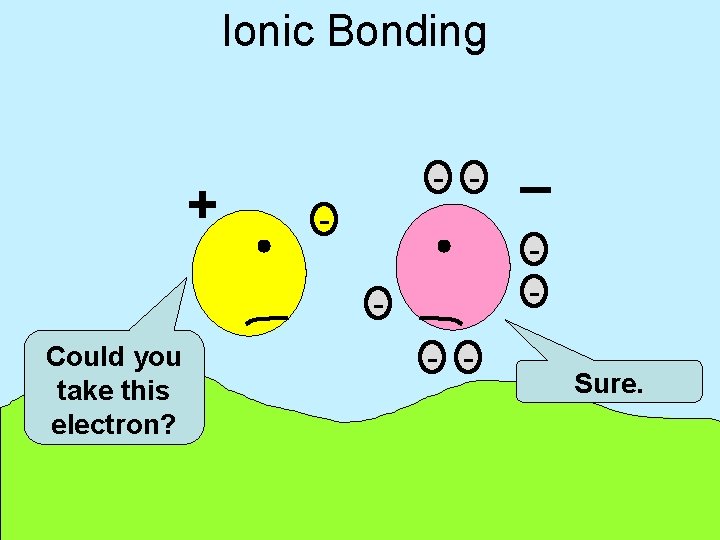
Ionic Bonding + - - - Could you take this electron? _ - - Sure.

Ionic Bonding + - - _ - - That’s much better, but I don’t feel like my old self. I’m much more Positive I’m feeling more stable myself. And I like your new charge!
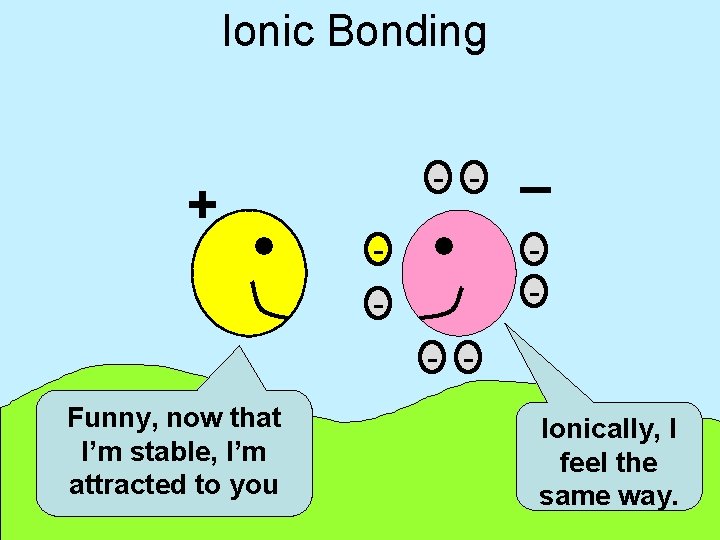
Ionic Bonding + - - _ - - Funny, now that I’m stable, I’m attracted to you Ionically, I feel the same way.


Covalent Bonds Chapter 5 Section 3 Pg. 192 -197
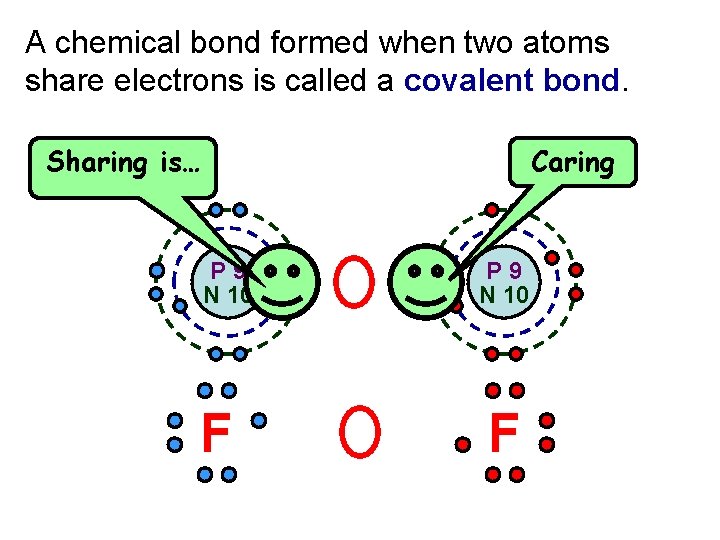
A chemical bond formed when two atoms share electrons is called a covalent bond. Caring Sharing is… P 9 N 10 F
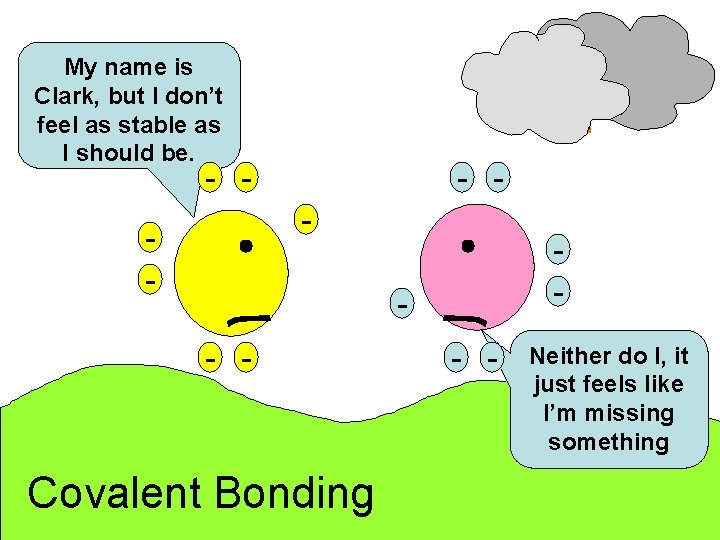
My name is Clark, but I don’t feel as stable as I should be. - - - - Covalent Bonding - - Neither do I, it just feels like I’m missing something
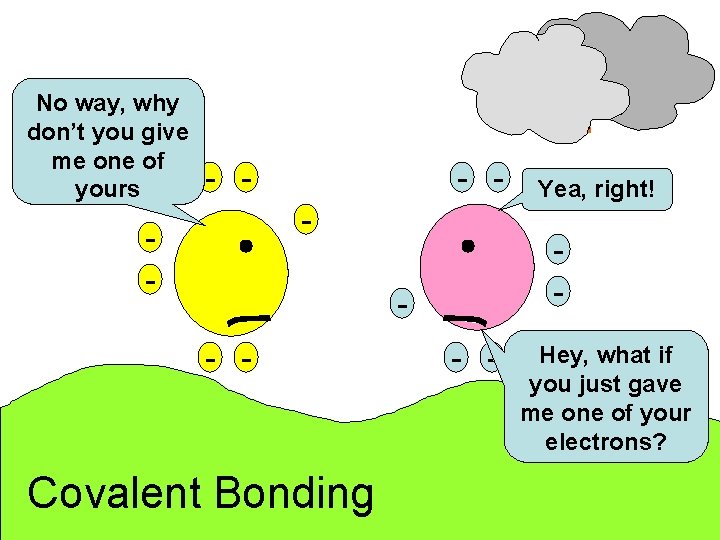
No way, why don’t you give me one of yours - - - - Covalent Bonding Yea, right! - - Hey, what if you just gave me one of your electrons?
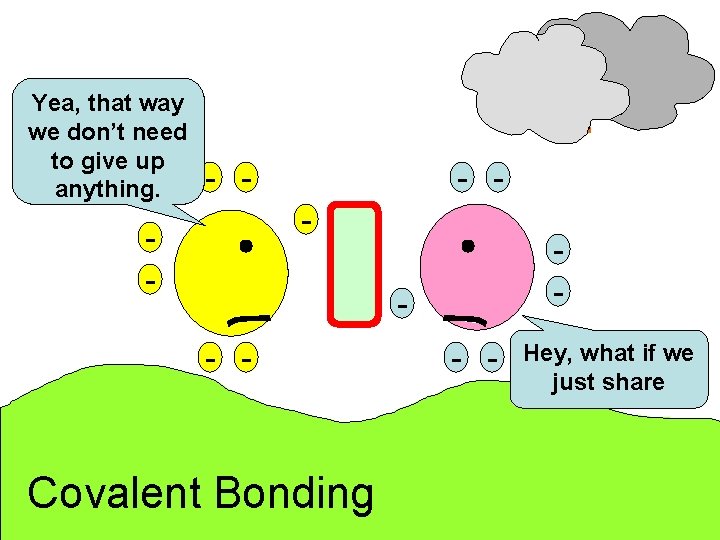
Yea, that way we don’t need to give up anything. - - - - Covalent Bonding - - Hey, what if we just share
Lois, I think this is the beginning of a super Covalent bond - - - - Covalent Bonding - -
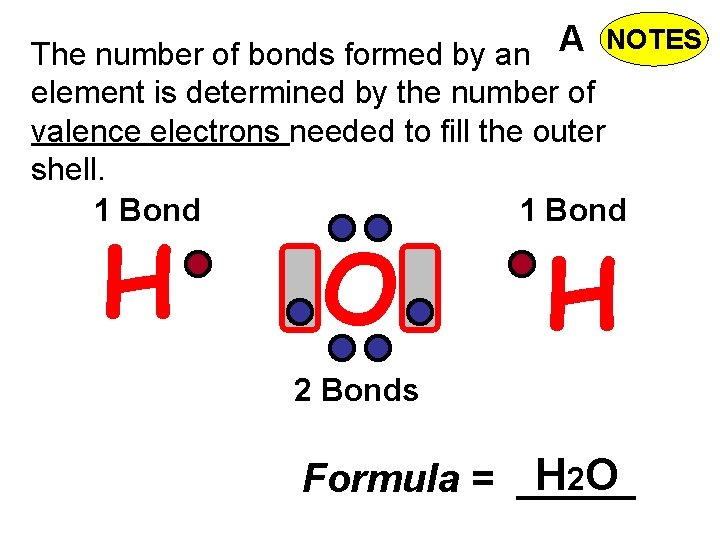
A The number of bonds formed by an NOTES element is determined by the number of valence electrons needed to fill the outer shell. 1 Bond H O H 2 Bonds Formula = H 2 O
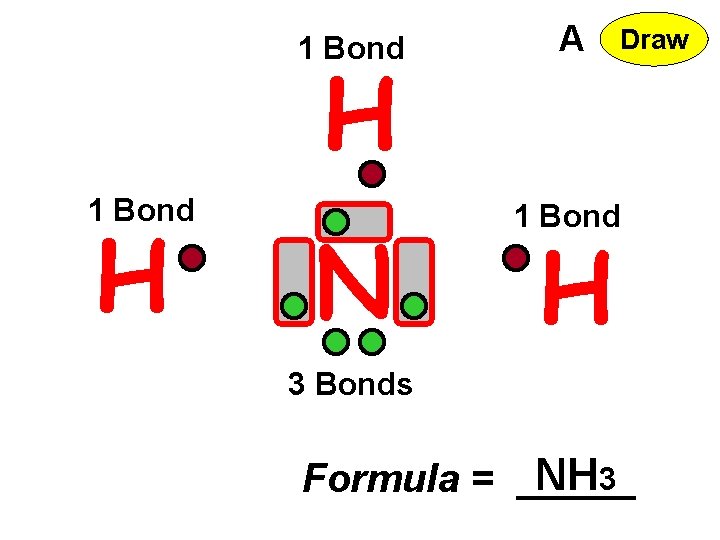
1 Bond A N 1 Bond H H 3 Bonds Formula = NH 3 Draw
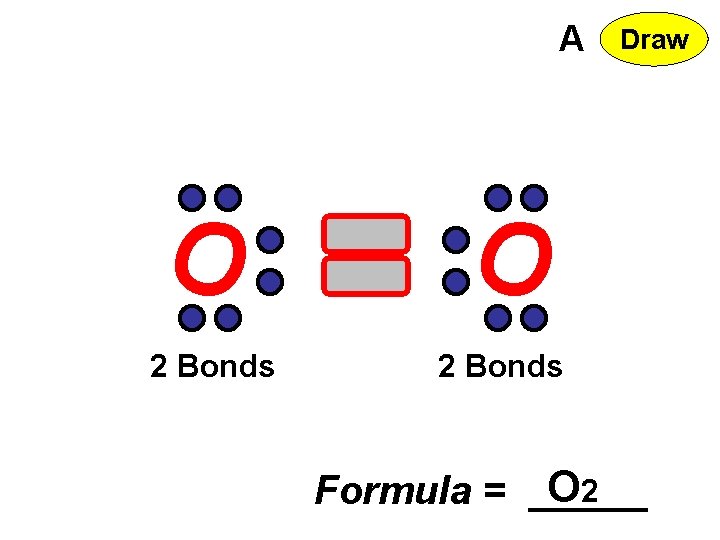
A O 2 Bonds Formula = O 2 Draw
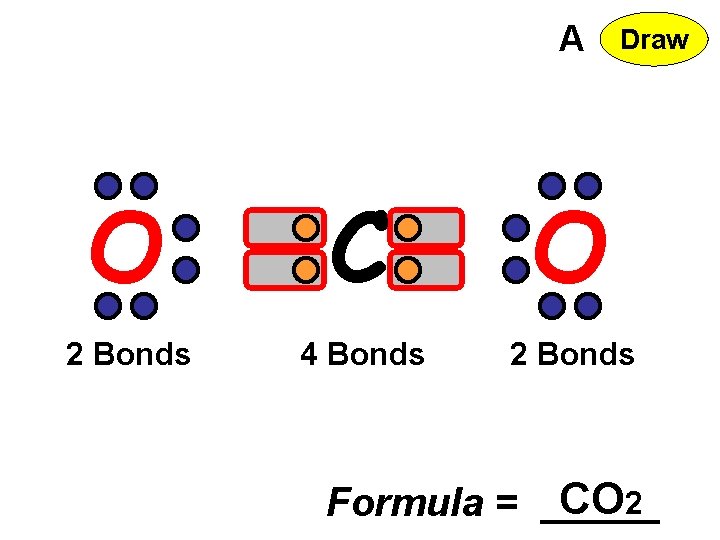
A Draw O C O 2 Bonds 4 Bonds 2 Bonds Formula = CO 2
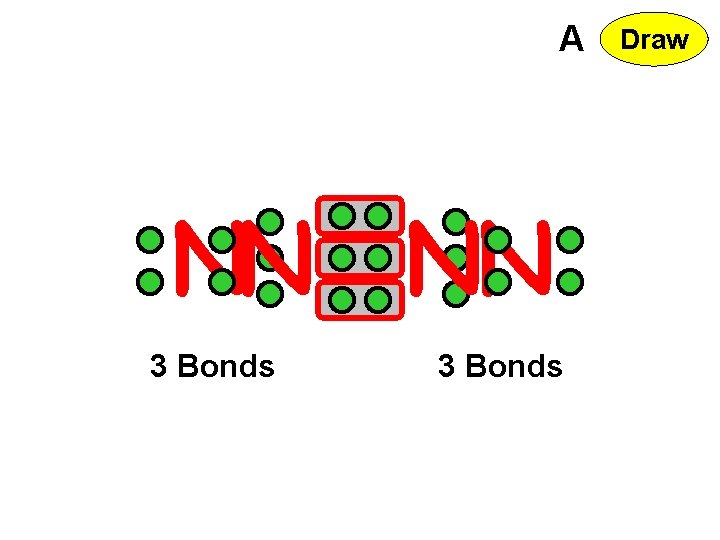
A NN NN 3 Bonds Draw
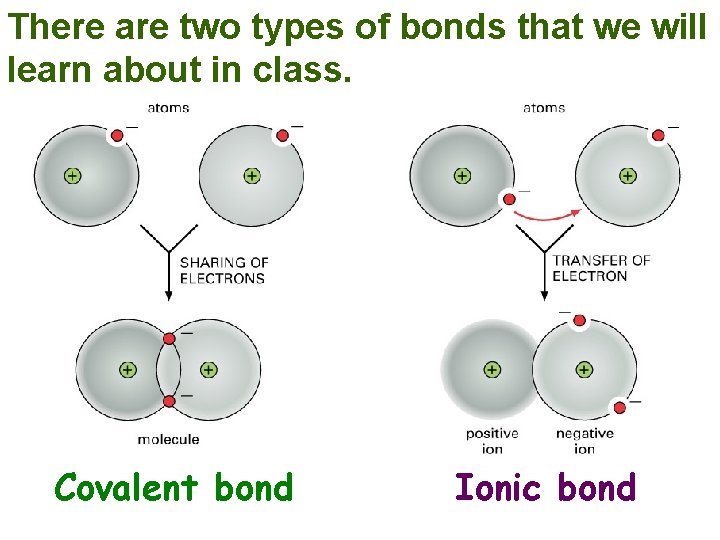
There are two types of bonds that we will learn about in class. Covalent bond Ionic bond
- Slides: 27