Valence Bond Theory Overlap of Atomic Orbitals s
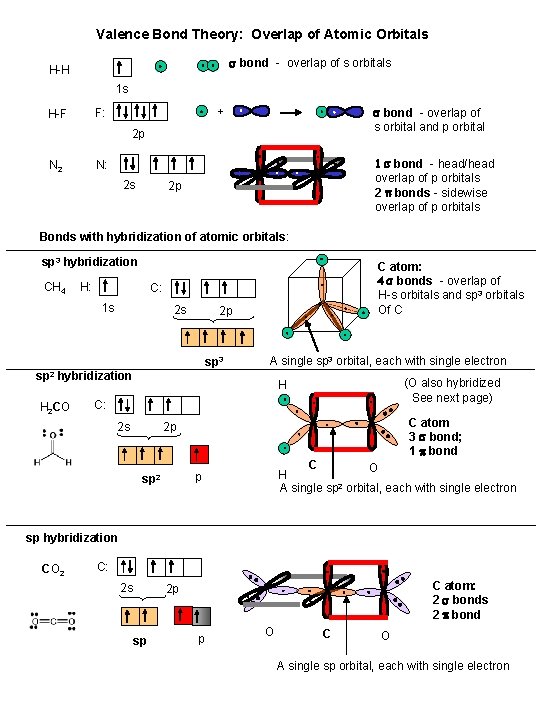
Valence Bond Theory: Overlap of Atomic Orbitals s bond - overlap of s orbitals H-H 1 s s bond - overlap of s orbital and p orbital + F: H-F 2 p N 2 1 s bond - head/head overlap of p orbitals 2 p bonds - sidewise overlap of p orbitals N: 2 s 2 p Bonds with hybridization of atomic orbitals: sp 3 hybridization CH 4 H: C atom: 4 s bonds - overlap of H-s orbitals and sp 3 orbitals Of C C: 1 s 2 s 2 p sp 3 A single sp 3 orbital, each with single electron sp 2 hybridization H 2 CO (O also hybridized See next page) H C: 2 s C atom 3 s bond; 1 p bond 2 p C O H A single sp 2 orbital, each with single electron p sp 2 sp hybridization CO 2 C: 2 s C atom: 2 s bonds 2 p bond 2 p sp p O C O A single sp orbital, each with single electron
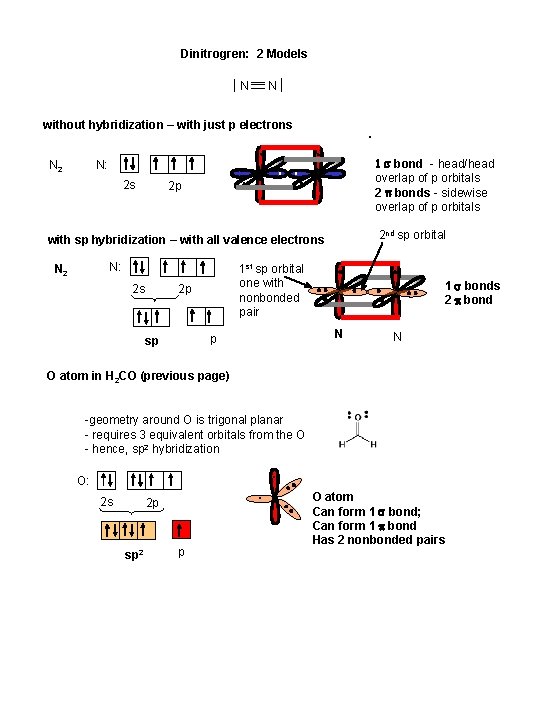
Dinitrogren: 2 Models N N without hybridization – with just p electrons N 2 1 s bond - head/head overlap of p orbitals 2 p bonds - sidewise overlap of p orbitals N: 2 s 2 p 2 nd sp orbital with sp hybridization – with all valence electrons N: N 2 2 s 1 st sp orbital one with nonbonded pair 2 p p sp 1 s bonds 2 p bond N N O atom in H 2 CO (previous page) -geometry around O is trigonal planar - requires 3 equivalent orbitals from the O - hence, sp 2 hybridization O: 2 s 2 p sp 2 p O atom Can form 1 s bond; Can form 1 p bond Has 2 nonbonded pairs
- Slides: 2