VALENCE BOND THEORY HOMONUCLEAR DIATOMIC MOLECULES VALENCE BOND

VALENCE BOND THEORY HOMONUCLEAR DIATOMIC MOLECULES: VALENCE BOND (VB) THEORY Chapter 2 Dr. Shuchita Agrawal Government P. G. College, Damoh

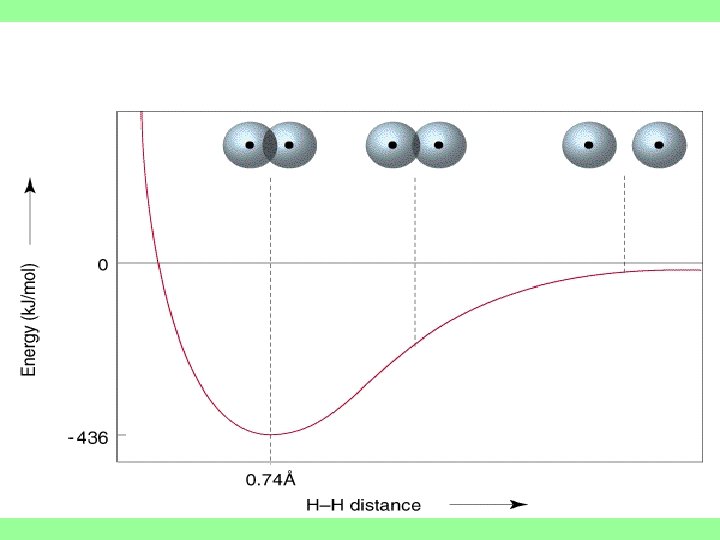
In covalent bonding, as two nuclei approach each other their atomic orbitals overlap • As the amount of overlap increases, the energy of the interaction decreases. • At some distance the minimum energy is reached.

• The minimum energy corresponds to the bonding distance (or bond length). • As the two atoms get closer, their nuclei begin to repel and the energy increases. • At the bonding distance, the attractive forces between nuclei and electrons just balance the repulsive forces (nucleus-nucleus, electron)

A covalent bond forms when the orbitals of two atoms overlap and the overlap region, which is between the nuclei, is occupied by a pair of electrons. A set of overlapping orbitals has a maximum of two electrons that must have opposite spins. The greater the orbital overlap, the stronger (more stable) the bond.
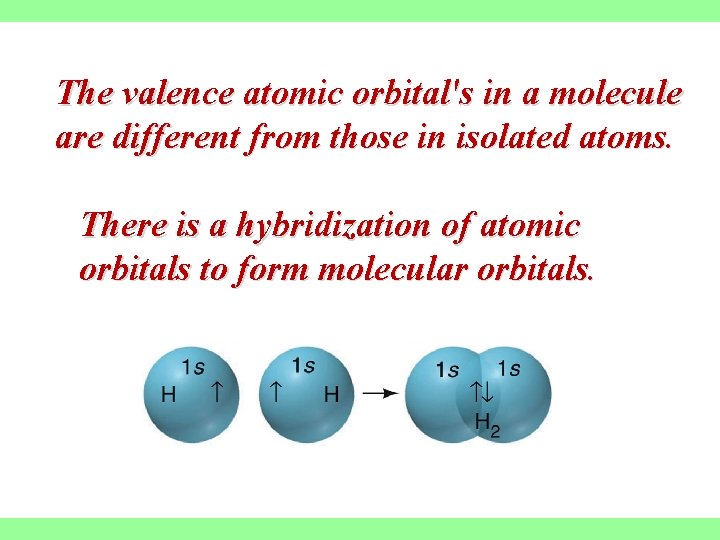
The valence atomic orbital's in a molecule are different from those in isolated atoms. There is a hybridization of atomic orbitals to form molecular orbitals.

HYBRIDIZATION The number of hybrid orbitals obtained equals the number of atomic orbitals mixed. The type of hybrid orbitals obtained varies with the types of atomic orbitals mixed. .
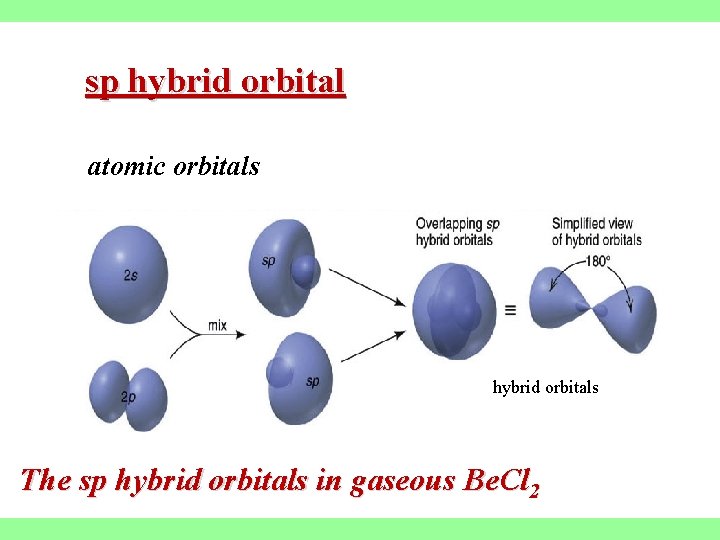
sp hybrid orbital atomic orbitals hybrid orbitals The sp hybrid orbitals in gaseous Be. Cl 2
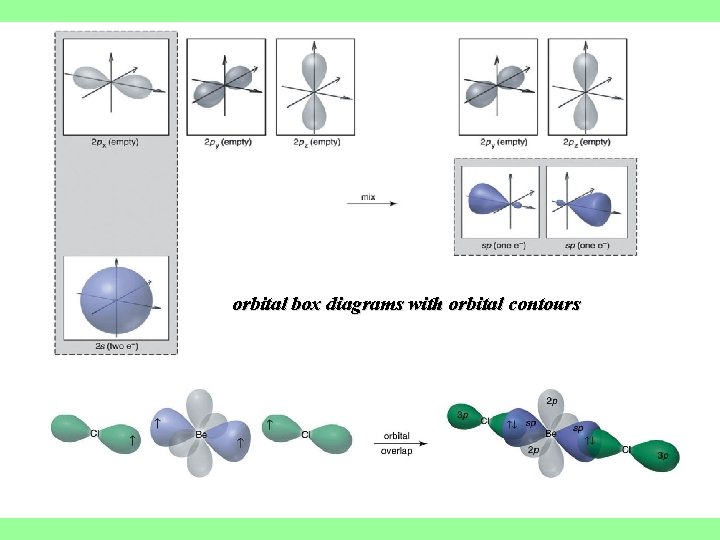
orbital box diagrams with orbital contours

sp 2 hybrid orbitals When we mix n atomic orbitals we must get n hybrid orbitals. sp 2 hybrid orbitals are formed with one s and two p orbitals, therefore, there is one un hybridized p orbital remaining. The large lobes of sp 2 hybrids lie in a trigonal plane All molecules with trigonal planar electron pair geometries have sp 2 orbitals on the central atom
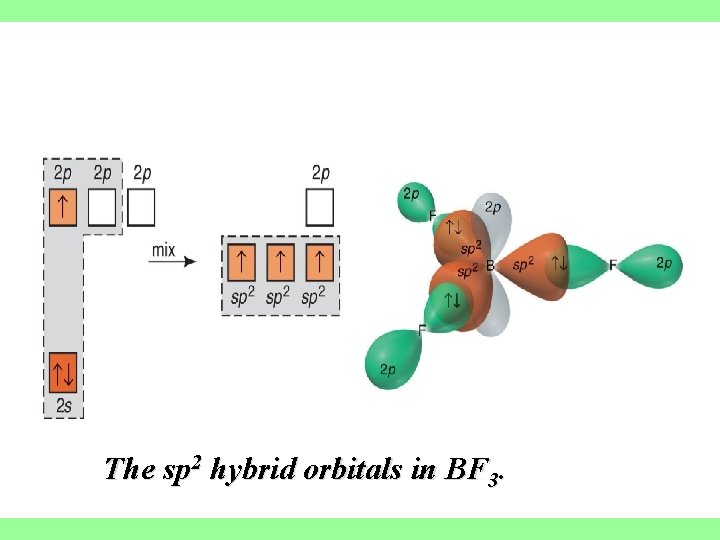
The sp 2 hybrid orbitals in BF 3.

sp 3 hybrid orbitals are formed from one s and three p orbitals, therefore, there are four large lobes. Each lobe points towards the vertex of a tetrahedron. The angle between the large lobes is 109. 5
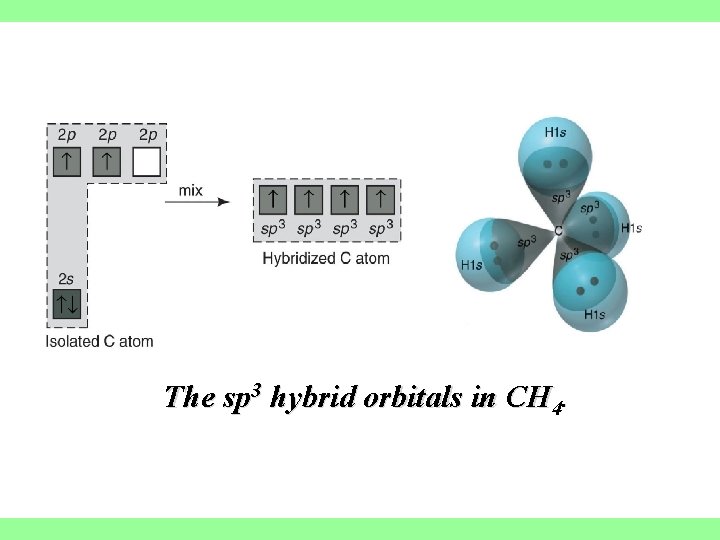
The sp 3 hybrid orbitals in CH 4.
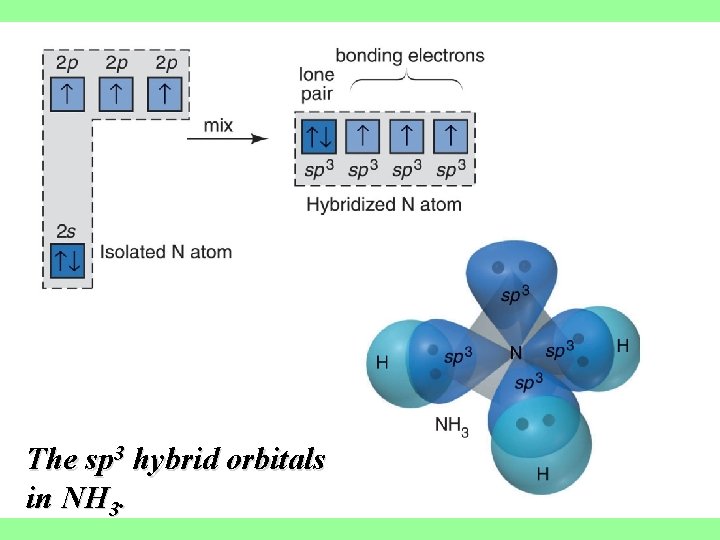
The sp 3 hybrid orbitals in NH 3.
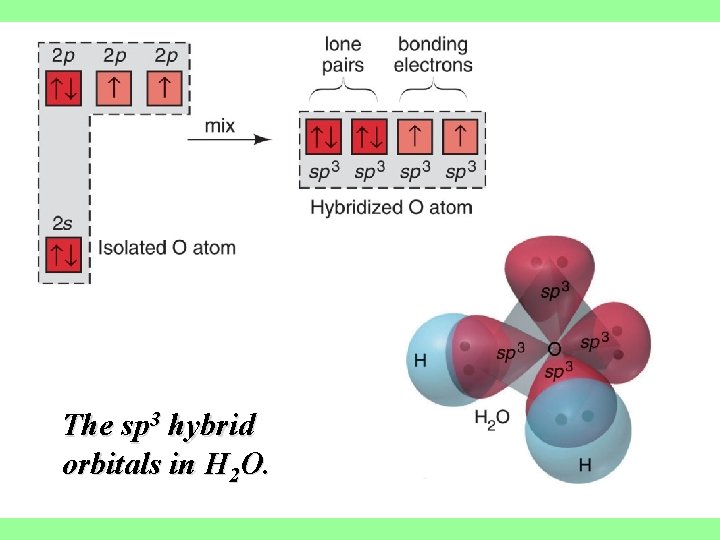
The sp 3 hybrid orbitals in H 2 O.
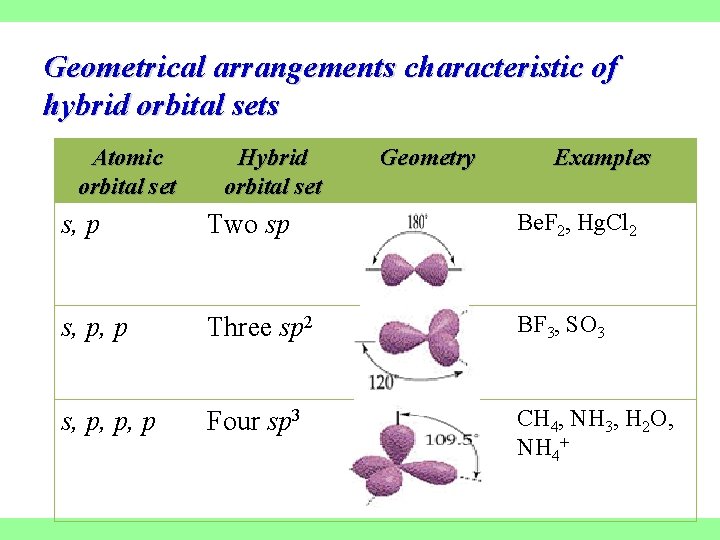
Geometrical arrangements characteristic of hybrid orbital sets Atomic orbital set Hybrid orbital set Geometry Examples s, p Two sp Be. F 2, Hg. Cl 2 s, p, p Three sp 2 BF 3, SO 3 s, p, p, p Four sp 3 CH 4, NH 3, H 2 O, NH 4+
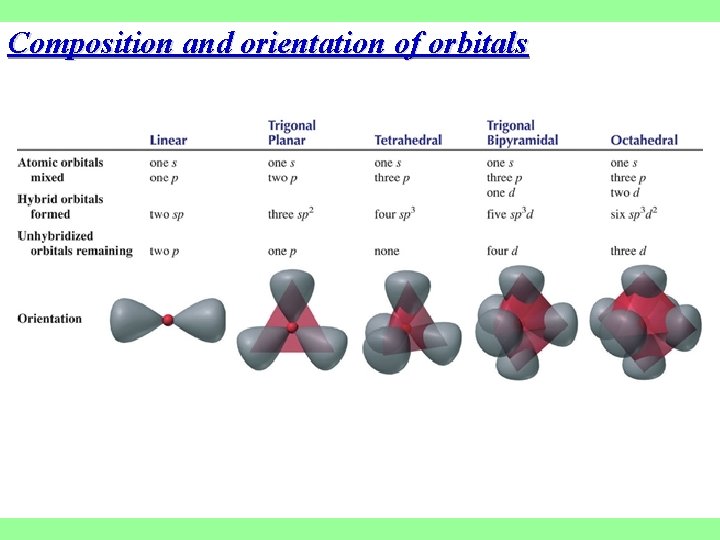
Composition and orientation of orbitals

MULTIPLE BONDS Have and -bonds. In -bonds, the electron density lies on the axis between the nuclei. All single bonds are -bonds. -Bonds: electron density lies above and below the plane of the nuclei. A double bond consists of one -bond and one -bond.
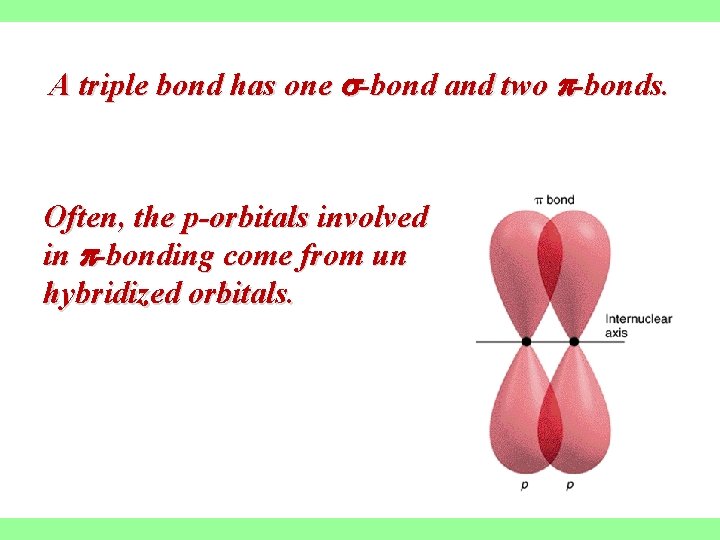
A triple bond has one -bond and two -bonds. Often, the p-orbitals involved in -bonding come from un hybridized orbitals.
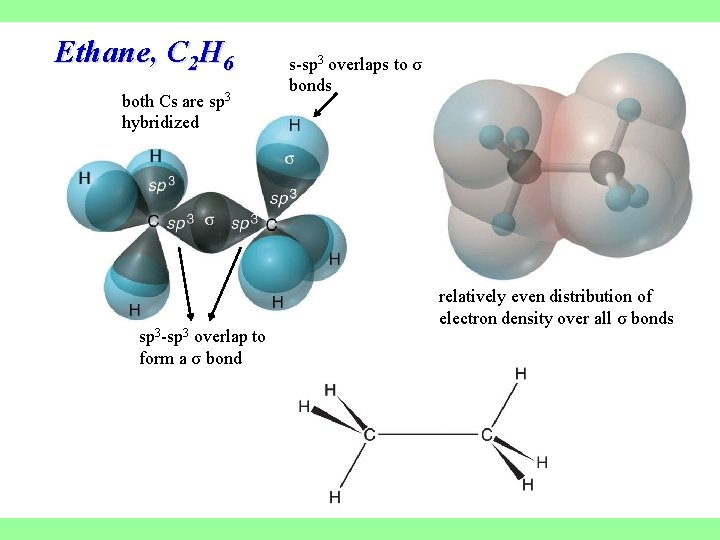
Ethane, C 2 H 6 both Cs are sp 3 hybridized sp 3 -sp 3 overlap to form a σ bond s-sp 3 overlaps to σ bonds relatively even distribution of electron density over all σ bonds
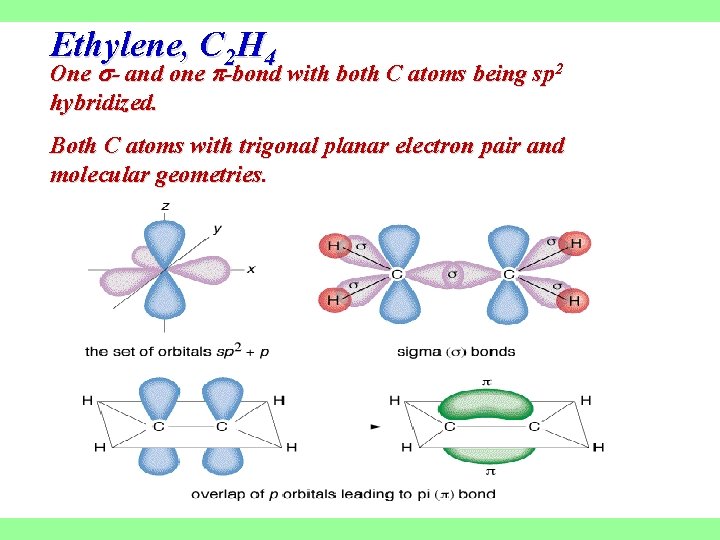
Ethylene, C 2 H 4 One - and one -bond with both C atoms being sp 2 hybridized. Both C atoms with trigonal planar electron pair and molecular geometries.
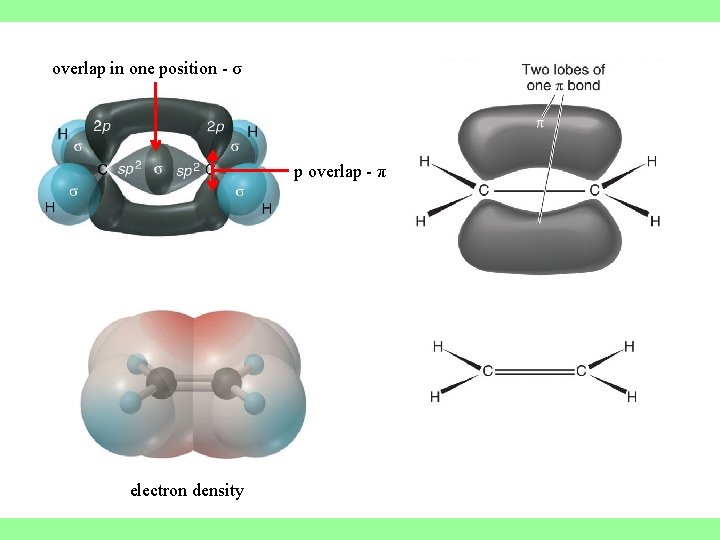
overlap in one position - σ p overlap - π electron density

Acetylene, C 2 H 2 In acetylene: • the electron pair geometry of each C is linear, therefore, the C atoms are sp hybridized. • the sp hybrid orbitals form the C-C and C-H bonds. • there are two unhybridized p-orbitals. • both un hybridized p-orbitals form the two bonds, one -bond is above and below the plane of the nuclei and the other -bond is in front and behind the plane of the nuclei.
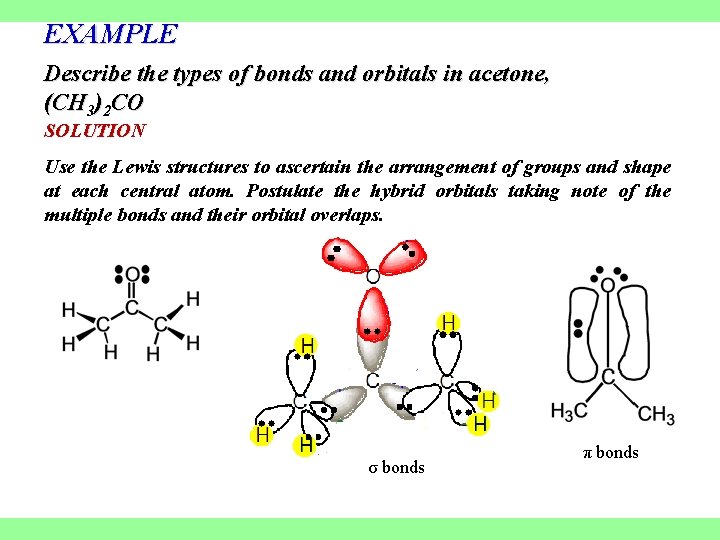
EXAMPLE Describe the types of bonds and orbitals in acetone, (CH 3)2 CO SOLUTION Use the Lewis structures to ascertain the arrangement of groups and shape at each central atom. Postulate the hybrid orbitals taking note of the multiple bonds and their orbital overlaps. σ bonds π bonds
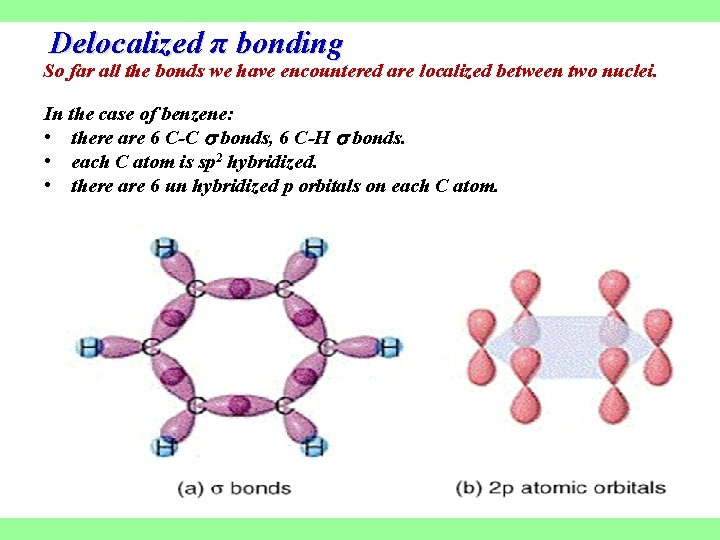
Delocalized π bonding So far all the bonds we have encountered are localized between two nuclei. In the case of benzene: • there are 6 C-C bonds, 6 C-H bonds. • each C atom is sp 2 hybridized. • there are 6 un hybridized p orbitals on each C atom.

In benzene there are two options for the 3 bonds: • localized between C atoms or • delocalized over the entire ring (i. e. the electrons are shared by all 6 C atoms). Experimentally, all C-C bonds are the same length in benzene, therefore, all C-C bonds are of the same type (recall single bonds are longer than double bonds).
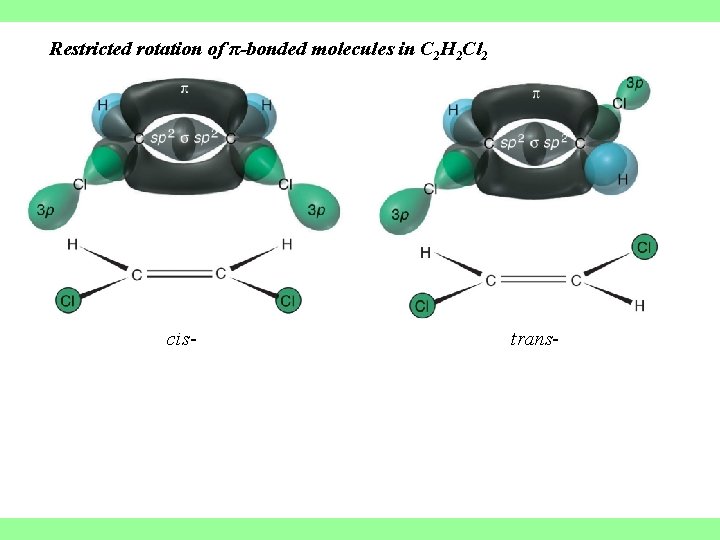
Restricted rotation of π-bonded molecules in C 2 H 2 Cl 2 cis- trans-

General conclusions on multiple bonds • Every two atoms share at least 2 electrons. • Two electrons between atoms on the same axis as the nuclei are bonds. • bonds are always localized. • If two atoms share more than one pair of electrons, the second and third pair form bonds. • When resonance structures are possible, delocalization is also possible.

Thanks • EMAIL ID: mrs. shuchitaagrawal@gmail. com • Ph No : 9977330500 Dr. Shuchita Agrawal Government P. G. College, Damoh 30
- Slides: 30