Valence Bond hybridization Googleyoutube referrences 1 Tro pages

Valence Bond & hybridization Google-youtube referrences: 1. Tro pages 408 and 411 2. Valence bond: molecular shape and Orbital hybridization. 3. Section 7 5 sigma and pi bonds. 4. Sp 3 Atomic orbital Hybridization – Chemistry Sigma and Pi bond symmetry 5. Major Intermolecular Forces 6. Shapes and polarity of molecules

Valence Bond & hybridization The orbital pattern valence electrons for 6 C are: __ __ 2 s 2 px 2 py 2 pz The 4 bonds in CH 4 are identical. The explanation is that valence electrons hybridize into an sp 3 hybrid: __ __ sp 3
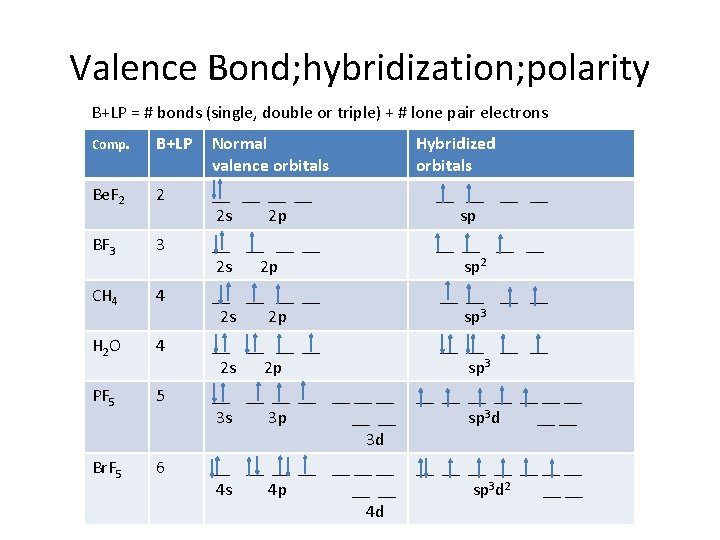
Valence Bond; hybridization; polarity B+LP = # bonds (single, double or triple) + # lone pair electrons Comp. B+LP Normal valence orbitals Hybridized orbitals Be. F 2 2 __ __ 2 s 2 p __ __ sp BF 3 3 __ __ 2 s 2 p __ __ sp 2 CH 4 4 __ __ 2 s 2 p __ __ sp 3 H 2 O 4 __ __ 2 s 2 p __ __ sp 3 PF 5 5 __ __ 3 s 3 p __ __ 3 d __ __ sp 3 d __ __ Br. F 5 6 __ __ 4 s 4 p __ __ 4 d __ __ sp 3 d 2 __ __
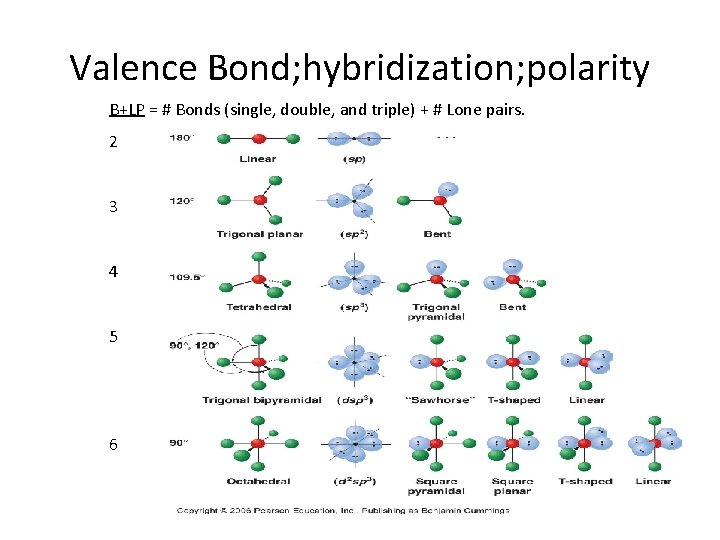
Valence Bond; hybridization; polarity B+LP = # Bonds (single, double, and triple) + # Lone pairs. 2 3 4 5 6
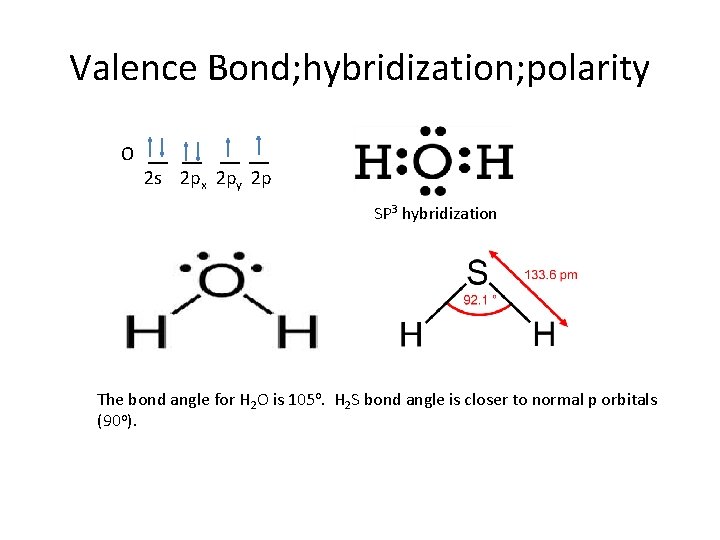
Valence Bond; hybridization; polarity O __ __ 2 s 2 px 2 py 2 p SP 3 hybridization The bond angle for H 2 O is 105 o. H 2 S bond angle is closer to normal p orbitals (90 o).
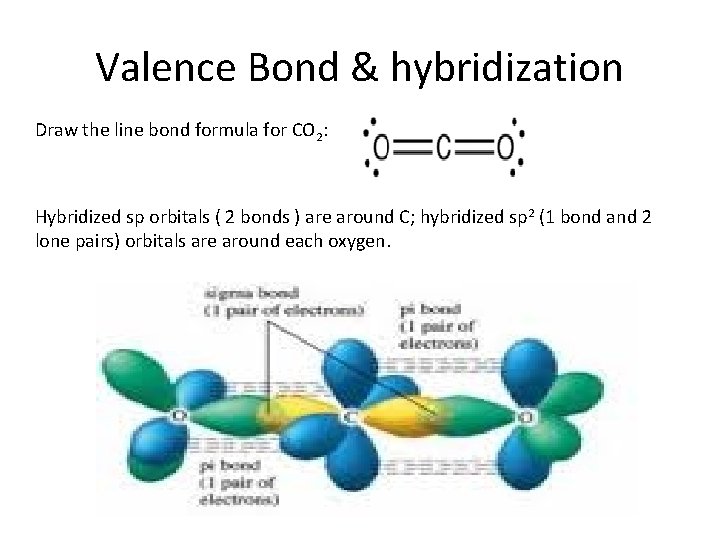
Valence Bond & hybridization Draw the line bond formula for CO 2: Hybridized sp orbitals ( 2 bonds ) are around C; hybridized sp 2 (1 bond and 2 lone pairs) orbitals are around each oxygen.
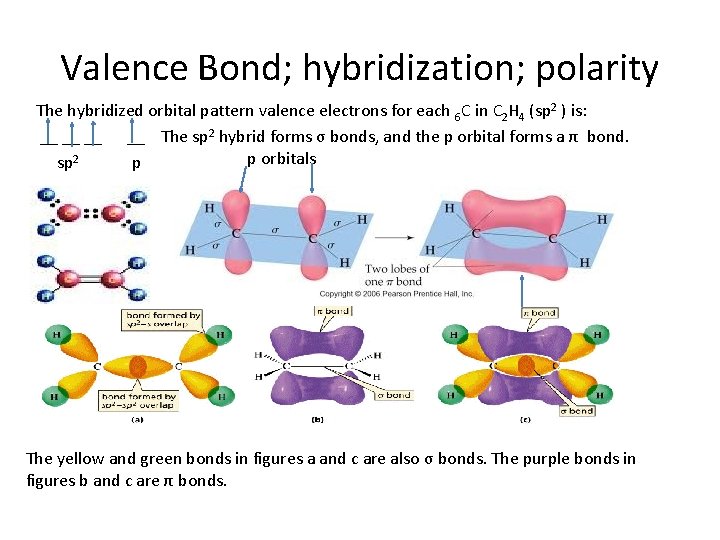
Valence Bond; hybridization; polarity The hybridized orbital pattern valence electrons for each 6 C in C 2 H 4 (sp 2 ) is: __ __ The sp 2 hybrid forms σ bonds, and the p orbital forms a π bond. p orbitals sp 2 p The yellow and green bonds in figures a and c are also σ bonds. The purple bonds in figures b and c are π bonds.
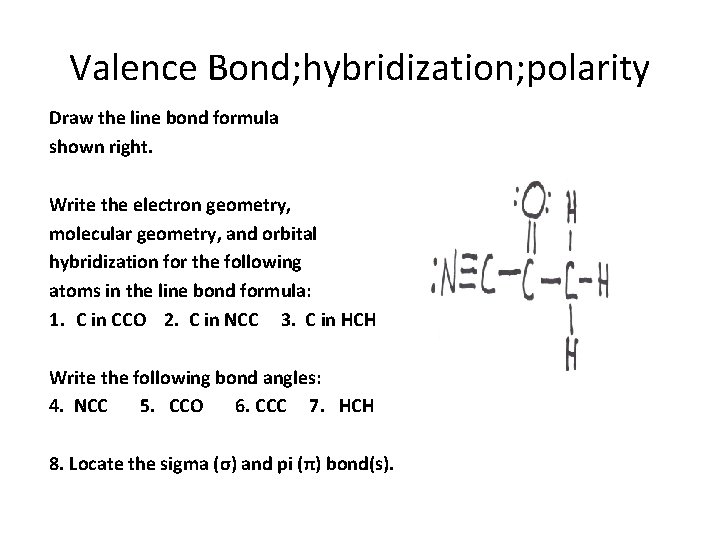
Valence Bond; hybridization; polarity Draw the line bond formula shown right. Write the electron geometry, molecular geometry, and orbital hybridization for the following atoms in the line bond formula: 1. C in CCO 2. C in NCC 3. C in HCH Write the following bond angles: 4. NCC 5. CCO 6. CCC 7. HCH 9. CCH 8. Locate the sigma (σ) and pi (π) bond(s).
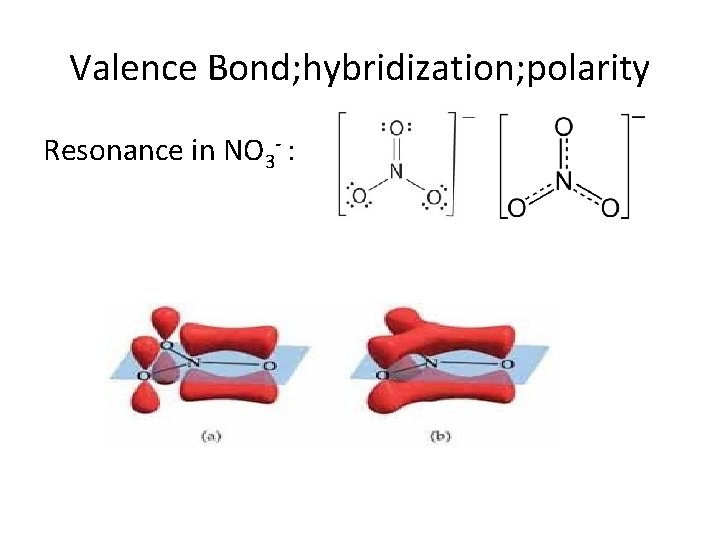
Valence Bond; hybridization; polarity Resonance in NO 3 - :
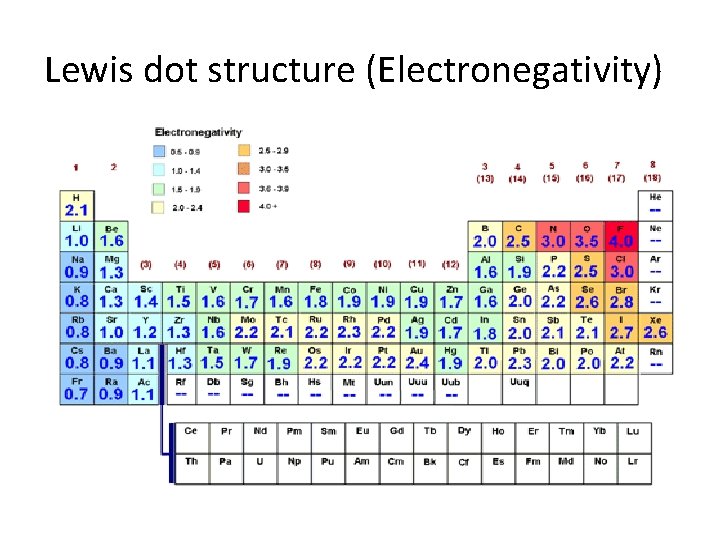
Lewis dot structure (Electronegativity)
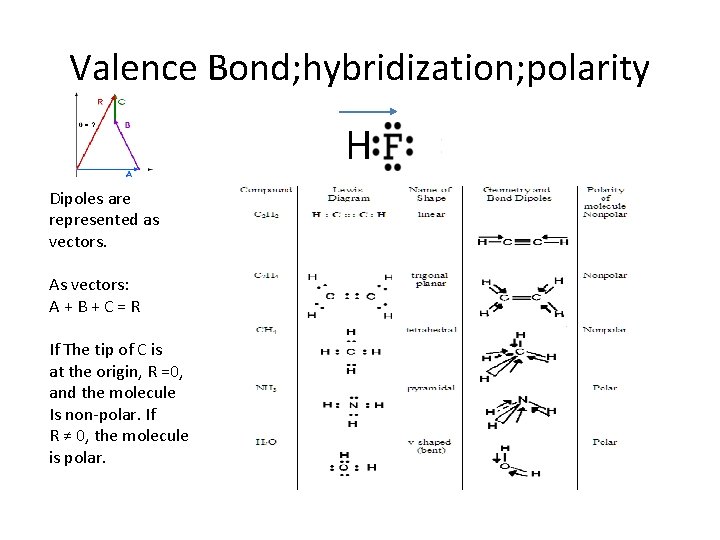
Valence Bond; hybridization; polarity H Dipoles are represented as vectors. As vectors: A+B+C=R If The tip of C is at the origin, R =0, and the molecule Is non-polar. If R ≠ 0, the molecule is polar.

Valence Bond; hybridization; polarity Identify the electronic and molecular geometries. Draw the molecular formulas. Identify the hybridized orbitals and locate the sigma and pi bonds (if any). Predict the polarity (polar or nonpolar)for the following molecules (not ions): The first non-hydrogen atom is central. 1. CO 2 2. CH 2 O 3. CH 4 O 4. C 2 H 2 5. NO 2 - 6. HCN 7. Xe. F 4 8. C 2 H 4 O 9. CO 32 - 10. NO 3 - 11. Br. F 5 12. IF 4+ 13. C 6 H 6 ( See next slide. ) 14. I 3 -
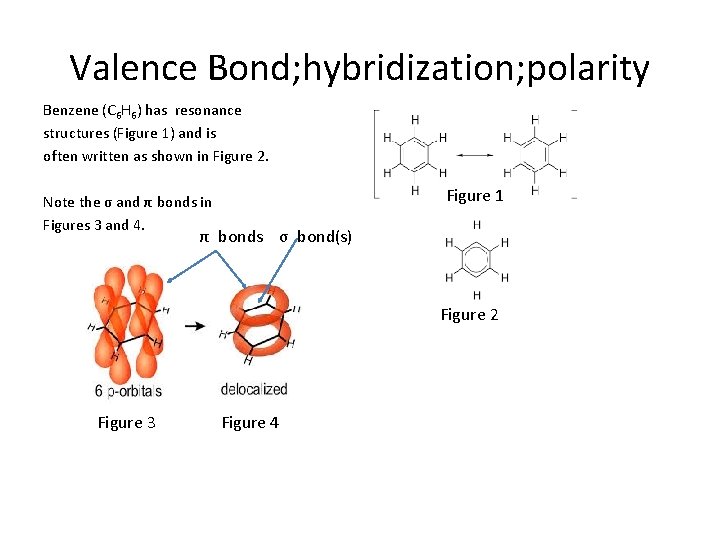
Valence Bond; hybridization; polarity Benzene (C 6 H 6) has resonance structures (Figure 1) and is often written as shown in Figure 2. Figure 1 Note the σ and π bonds in Figures 3 and 4. π bonds σ bond(s) Figure 2 Figure 3 Figure 4
- Slides: 13