Vacuum Fundamentals Basic Concepts 1 5991 9443 EN

Vacuum Fundamentals Basic Concepts 1 5991 -9443 EN Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018
Agilent Technologies is committed to the educational community and is willing to provide access to company-owned material contained herein. This slide set is created by Agilent Technologies. The usage of the slides is limited to teaching purpose only. These materials and the information contained herein are accepted “as is” and Agilent makes no representations or warranties of any kind with respect to the materials and disclaims any responsibility for them as may be used or reproduced by you. Agilent will not be liable for any damages resulting from or in connection with your use, copying or disclosure of the materials contained herein. You agree to indemnify and hold Agilent harmless for any claims incurred by Agilent as a result of your use or reproduction of these materials. In case pictures, sketches or drawings should be used for any other purpose please contact Agilent Technologies. 2 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

Table of Contents Introduction Conductance • History of Vacuum Development • General • Overview • Conductance in Viscous Flow Definition of Vacuum Ranges • Conductance in Molecular Flow Pressure Examples • Molecular Collisions • Conductance kills pump speed • In a Chamber • Moving Particles • Measuring Atmospheric Pressure • Surface Cleanliness & Electrical Isolation • Altitude versus Pressure Summary Flow Further Information • What is Flow? • Gas Flow Regimes • Transition Flow

Introduction History of Vacuum Development 1600 Otto von Guericke establishes the physics of vacuums. 1920 s F. Holweck recommends “molecular drag” pumps as well 1643 Evangelista Torricelli invents the mercury barometer. The “Torr” (unit) is named after him. 1948 Varian Associates was founded by Varian brothers, W. W. Hansen and E. Ginzton 1650 s Blaise Pascal studies hydraulic fluids. The name Pascal has been given to the SI unit of pressure, to a program-ing language, and Pascal's law 1956 Varian Associates invents the “sputter-ion” pump (tube life) 1660 s Robert W. Boyle is a philosopher, chemist and physicist, best known for Boyle’s law. 1738 Daniel Bernoulli publishes Hydrodynamica describing the kinetic theory of gases. 1915 1800 s Gay-Lussac`s law refers to thermal expansion of gases. Wolfgang Gaede invents the mercury vapor “diffusion” pump and proposes pumps based on “molecular drag”. 1980 s First commercialization of “drag stage” turbo-molecular pumps. See notes for details To. C 4 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

Introduction Overview The word “vacuum” comes from the Latin adjective vacuus which means “vacant” or “void”. It is the absence of matter. It is the state of a gas where the density of matter is lower than atmospheric pressure at the earth surface. Vacuum is needed for many technical processes and instruments such as • Move particles over a long distance – High Energy Physics (Particle Accelerators) – Mass Spectrometry & Electron Microscopes • Create and maintain clean surfaces – Surface coating (glass, film, semi-conductor) – Food Packaging • Prevent electrical breakdown – Electrical transformers – Vacuum Tubes To. C 5 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

Definition of Vacuum Ranges The ranges are determined by the vacuum quality which is defined by the amount of matter in the system. The higher the vaccum range, the less matter is around. Atmospheric Pressure 760 Torr Pick-up & Conveyance Rough Vacuum 25 Torr Food Packaging Medium Vacuum 7. 5 e-4 Torr Freeze Drying Incandescentt Lamps High Vacuum 7. 5 e-7 Torr Heat Treatment Surface Coating Semiconductors To. C 6 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018 Ultra High Vacuum 7. 5 e-10 Torr Thin Film Deposition Mass Spectrometry Electron Microscopy Extreme High Vacuum 7. 5 e-13 Torr Nanotechnology Sub-Atomic Research Space Research

Pressure Molecular Collisions P 1 Momentum transfer from a particle hitting a fixed surface creates a force on the wall. The resulting pressure is calculated by: P = Pressure F 1 Px FX P 2 F = Force A = Surface area F 2 F 3 Gauge pressure is the pressure relative to the ambient pressure. P 3 Schematic showing pressure as exerted by particle collision inside a closed environment. To. C 7 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018
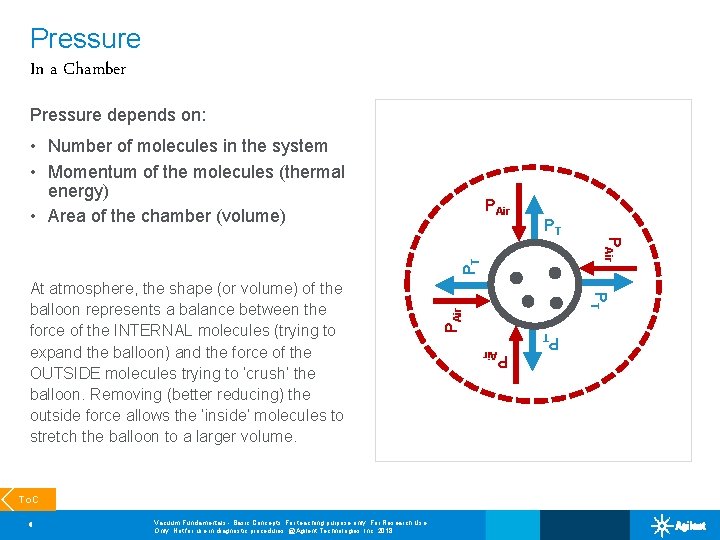
Pressure In a Chamber Pressure depends on: • Number of molecules in the system • Momentum of the molecules (thermal energy) • Area of the chamber (volume) Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018 PAir 8 PT To. C PT At atmosphere, the shape (or volume) of the balloon represents a balance between the force of the INTERNAL molecules (trying to expand the balloon) and the force of the OUTSIDE molecules trying to ‘crush’ the balloon. Removing (better reducing) the outside force allows the ‘inside’ molecules to stretch the balloon to a larger volume. PAir PT PT PAir

Pressure Measuring Atmospheric Pressure Vacuum is also measured on the barometric scale or as a percentage of atmospheric pressure in bars or atmospheres. To. C 9 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018
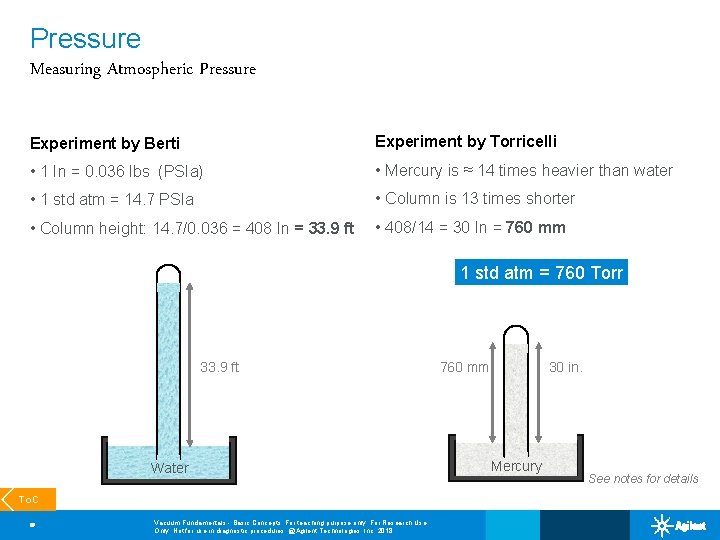
Pressure Measuring Atmospheric Pressure Experiment by Berti Experiment by Torricelli • 1 In = 0. 036 lbs (PSIa) • Mercury is ≈ 14 times heavier than water • 1 std atm = 14. 7 PSIa • Column is 13 times shorter • Column height: 14. 7/0. 036 = 408 In = 33. 9 ft • 408/14 = 30 In = 760 mm 1 std atm = 760 Torr 33. 9 ft Water To. C 10 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018 760 mm 30 in. Mercury See notes for details

Pressure Altitude versus Pressure Altitude 1. 000 miles 1609 km Pressure (Torr) Particles/cm 3 10 -10 4 x 106 25 miles 40 km 2 8 x 1016 10 miles 16 km 78 3 x 1018 27. 500 ft 8. 3 km 253 1 x 1019 20. 000 ft 6 km 350 1. 5 x 1019 5. 000 ft 1. 5 km 632 2. 5 x 1019 2. 000 ft 0. 6 km 709 2. 8 x 1019 760 1019 Sea level Less air molecules 3. 0 x More air molecules To. C 11 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

Flow What is Flow? Unlike solids, liquids and gases do not retain their shape as soon as they are filled into a vessel or room. Liquids will ‘flow’ from one place to another with relatively low forces. While liquids are constrained by ‘body forces’ (gravity and centrifugal), gases are relatively immune to these. Gas flow type Rough vacuum Viscous flow Medium vaccum Knudsen flow High vaccum Molecular flow Ultrahigh vacuum Molecular flow Liquid moves at meters per second while gas can move at hundreds of meters per second with relatively small pressure differences. To. C 12 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

Flow Gas Flow Regimes The flow of gases in a vacuum has three defined regimes. As gas diffuses from an area of high pressure to one of lower pressure the movement can be described with terms used in fluid mechanics. Gas behavior will be different depending on the pressure. Viscous flow (P>100 m. Torr) • Gas behaves like liquid in a tube • Distance between molecules is small • Molecule-molecule collisions dominate • Flow through momentum transfer • Flow profile: turbulent, laminar Transition flow Molecular flow (P< 1 m. Torr) • Gas molecules behave like discrete particles • Molecule-wall collisions dominate • Minimal collisions between gas particles • Flow profile: molecular PRESSURE See notes for details To. C 13 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

Flow Transition Flow The region between viscous flow (100 m. Torr) and molecular flow (1 m. Torr) is called transition flow (also referred to as Knudsen flow). Gas flow behavior is less predictable here. In reality, flow characteristics are based on the relationship between the pressure and the dimensions of the vessel. Estimates of vacuum pressure in transition flow can vary by as much as 50% because there at least three different methods of calculating the flow in transition flow and the answers will vary by up to 50%. Characterizing flow based on pressure only is technically not valid. To. C 14 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

Conductance General Conductance (C) is the ability of gas (fluid) to transmit through material. It is the gas volume per unit (Q) time that can move through a notch like an orifice, pipe or tube. Q P 2 The term conductance is assigned to passive system components. It is the reciprocal of the resistance to gas flow. Conductance is expressed in volume units per unit time (L/s). P 2 > P 1 The gas flow depends on the pressure (Px) difference and on the geometry of the connection (e. g. orifice or pipe). To. C 15 P 1 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

Conductance Viscous flow The viscous flow conductance in a tube is calculated by: (a) D = Tube diameter (cm) L = Length (cm) P = Pressure (Torr) Conductance depends to the 4 th power of the tube diameter and is inversely related to the pipe length (L). (b) Viscous flow profile: turbulent (a) and laminar (b) Orifice conductance in viscous flow is extremely complicated to calculate. Therefore, this is rarely done in practice. To. C 16 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

Conductance Molecular Flow A = Orifice area Molecular flow profile See notes for details To. C 17 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018
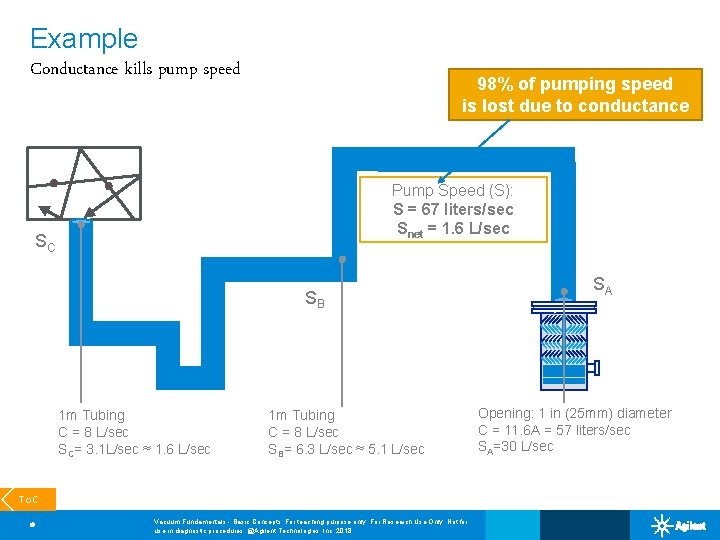
Example Conductance kills pump speed 98% of pumping speed is lost due to conductance Pump Speed (S): S = 67 liters/sec Snet = 1. 6 L/sec SC SA SB SD 1 m Tubing C = 8 L/sec SC= 3. 1 L/sec ≈ 1. 6 L/sec 1 m Tubing C = 8 L/sec SB= 6. 3 L/sec ≈ 5. 1 L/sec To. C 18 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018 Opening: 1 in (25 mm) diameter C = 11. 6 A = 57 liters/sec SA=30 L/sec

Example Moving Particles CERN Large Hadron Collider (LHC) • • 11, 100 laps/sec around 27 km (17 mile) path Beam pipe at approx. 10 -11 Torr (same as on moon surface) Particles travel at 99. 9999% of speed of light Vacuum also used as thermal shield for cryogenic magnets AGILENT Time-of-Flight (TOF) Mass Spectrometry • • • Approx. 2 m flight path (TOF section) TOF section pressure ≈10 -6 Torr Vacuum also required for high voltages to accelerate and collect ions Flight tube View of the LHC tunnel sector 3 -4 © 2009 -2017 CERN To. C 19 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018 See notes for details
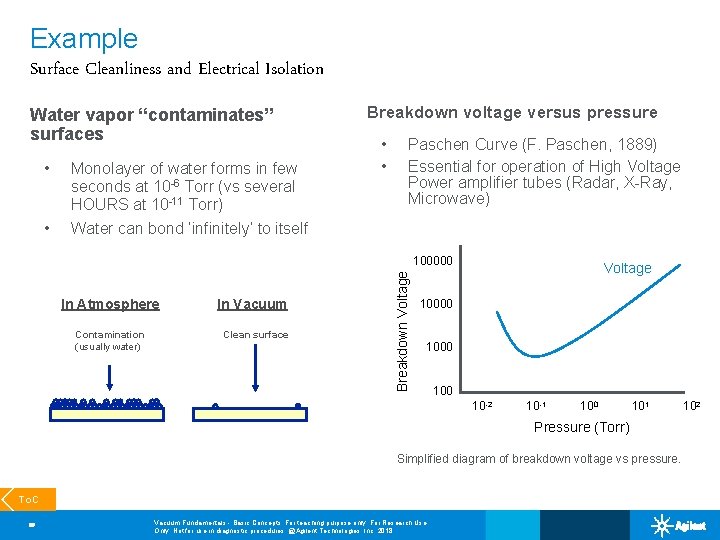
Example Surface Cleanliness and Electrical Isolation Water vapor “contaminates” surfaces • Monolayer of water forms in few seconds at 10 -6 Torr (vs several HOURS at 10 -11 Torr) • Water can bond ‘infinitely’ to itself Breakdown voltage versus pressure • • Paschen Curve (F. Paschen, 1889) Essential for operation of High Voltage Power amplifier tubes (Radar, X-Ray, Microwave) In Atmosphere Contamination (usually water) In Vacuum Clean surface Breakdown Voltage 100000 Voltage 10000 100 10 -2 10 -1 100 101 Pressure (Torr) Simplified diagram of breakdown voltage vs pressure. To. C 20 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018 102

Summary Remember Much of the pioneering work in Vacuum was done hundreds of years ago (in the time of Galileo). Vacuum is used in a huge variety of industries - from quantum calculators, space exploration, manufacturing of thin film displays and special alloys of metal. Pressure is the force exerted on a surface through impact of (gas) molecule collisions. The gas behavior in a vessel varies significantly as the pressure changes, and this has an impact on how pressure is measured and the type of vacuum pumps needed to achieve lower pressure. Conductance (losses) reduce the ability to pump gas. To. C 21 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018
Further Information For more information on products from Agilent, visit www. agilent. com or www. agilent. com/chem/academia Have questions or suggestions to this presentation? Contact academia. team@agilent. com Publication Title Web Technology teaching slides Web Vacuum Solutions Notes To. C 22 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018

THANK YOU www. agilent. com/chem/academia 23 Vacuum Fundamentals - Basic Concepts; For teaching purpose only; For Research Use Only. Not for use in diagnostic procedures. @Agilent Technologies, Inc. 2018
- Slides: 23