UVVIS Spectrometry And Atomic Absorption By Morgan Biehn

UV/VIS Spectrometry And Atomic Absorption By: Morgan Biehn

What’s Ahead… § Introduction to UV/VIS and AA § Procedure § Results § Error Analysis § Conclusions and Recommendations § Q&A

Introduction Ultraviolet/Visible (UV/VIS) Spectrometry: § A form of colorimetric analysis § Passes light through a cuvette containing solution § Referenced to a solution that absorbs no light (distilled water) § Beer-Lambert Law: A = αcl § In absence of α, use calibration curve

Introduction (contd. ) Atomic Absorption (AA) Spectrometry: § Samples must undergo desolvation and vaporization in a flame § When atoms absorb light, they transition to higher electronic energy levels § Concentration determined from amount of absorption § Flame AA can increase path length which increases absorption by Beer-Lambert Law

Procedure § Objective: Determine concentration of an unknown solution. § Potassium Permanganate (KMn. O 4) solutions § 5 standards prepared with concentrations of 0. 1, 0. 09, 0. 08, 0. 07, and 0. 06 g/L. Each standard tested twice § Perkin Elmer UV/VIS SP Spectrometer with 1 cm long cuvettes § Perkin Elmer AA spectrometer § Unknown solution tested five times for each method
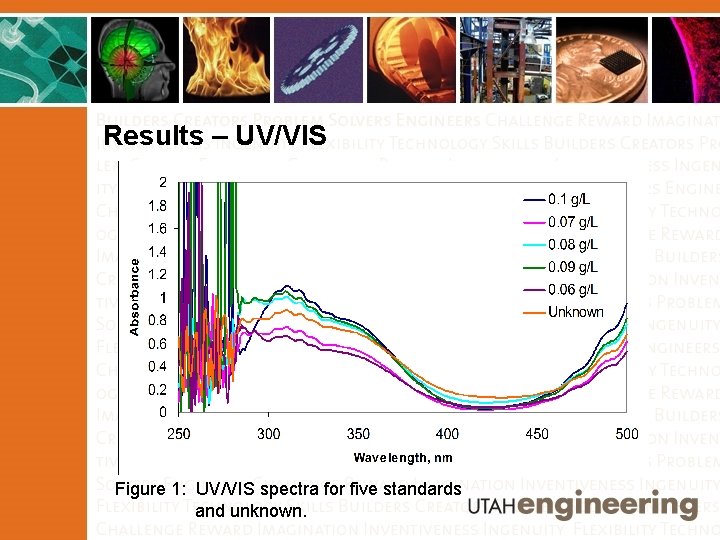
Results – UV/VIS Figure 1: UV/VIS spectra for five standards and unknown.
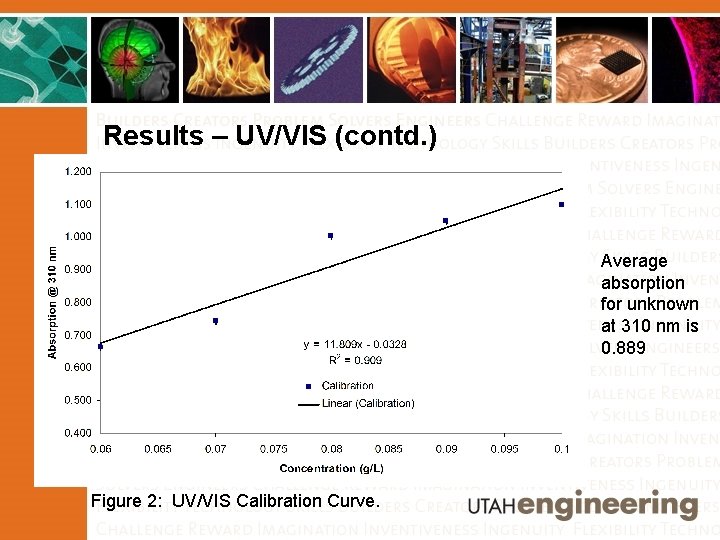
Results – UV/VIS (contd. ) Average absorption for unknown at 310 nm is 0. 889 Figure 2: UV/VIS Calibration Curve.
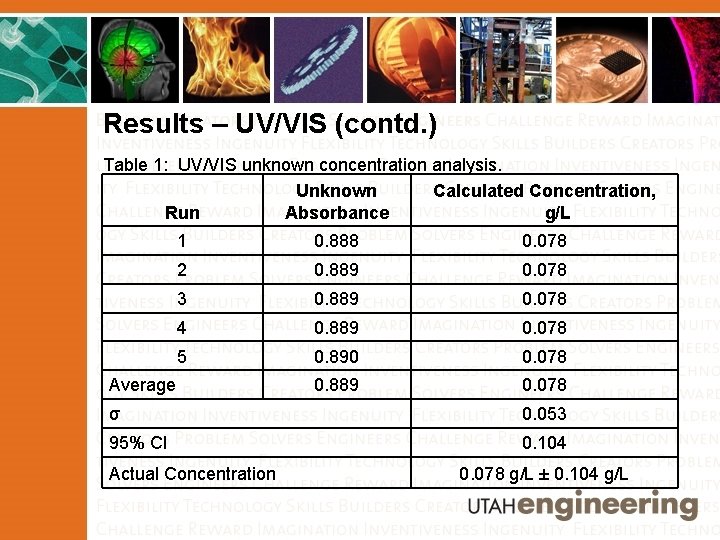
Results – UV/VIS (contd. ) Table 1: UV/VIS unknown concentration analysis. Unknown Calculated Concentration, Run Absorbance g/L 1 0. 888 0. 078 2 0. 889 0. 078 3 0. 889 0. 078 4 0. 889 0. 078 5 0. 890 0. 078 0. 889 0. 078 Average σ 0. 053 95% CI 0. 104 Actual Concentration 0. 078 g/L ± 0. 104 g/L
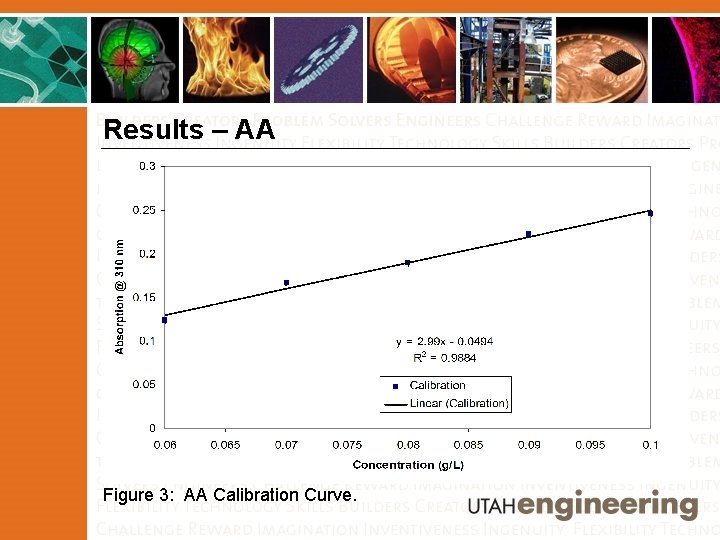
Results – AA Figure 3: AA Calibration Curve.
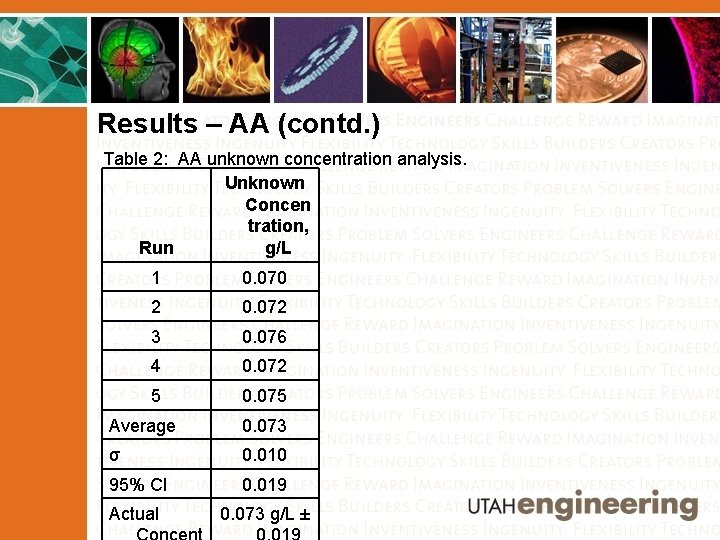
Results – AA (contd. ) Table 2: AA unknown concentration analysis. Unknown Concen tration, g/L Run 1 0. 070 2 0. 072 3 0. 076 4 0. 072 5 0. 075 Average 0. 073 σ 0. 010 95% CI 0. 019 Actual 0. 073 g/L ±

Error Analysis Four types of error: 1. Instrument error 2. Fit error 3. Dilution error 4. Operator error

Error Analysis (contd. ) 1. Instrument Error: Sample standard deviation 2. Fit Error: Use 3. calibration curve 4. linear regression 5.

Error Analysis (contd. ) 3. Dilution Error: Combine final concentration equation with error propagation equation 4. Operator Error: From comparison of previous users’ data and use of sample standard deviation
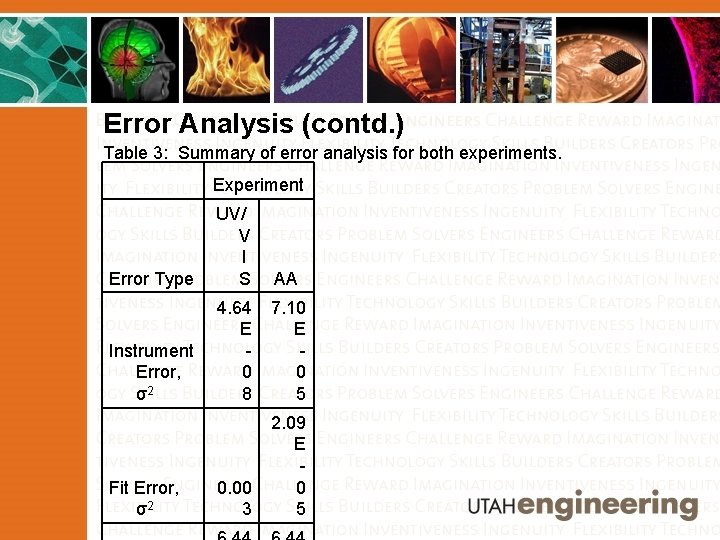
Error Analysis (contd. ) Table 3: Summary of error analysis for both experiments. Experiment Error Type UV/ V I S AA 4. 64 E Instrument 0 Error, σ2 8 7. 10 E 0 5 Fit Error, σ2 2. 09 E 0 5 0. 00 3

Conclusions § UV/VIS and AA spectra were compared § Bad UV/VIS calibration curve produced large error § AA data provided smaller standard deviation than the UV/VIS § Unknown has concentration of 0. 073 g/L ± 0. 019 g/L (95% confidence) § AA provides a quick, easy, and relatively painless method for determining concentrations

Recommendations § Monitor standards to reduce the risk of contamination § Store standards in a dark place § Try to conduct both experiments on the same day § Do as many tests as possible

Where We’ve Been… § Background information on UV/VIS and AA experimental methods § Objective for this specific experiment and procedure to obtain objective § Discussion of Results § Discussion of error and which method produced more accurate results § Provided recommendations

Questions?

References Schwedt, George. The Essential Guide to Analytical Chemistry. Wiley and Sons: Hoboken, NJ, 1997. Tissue, Brian M. “Atomic-Absorption Spectroscopy (AA). ” http: //elchem. kaist. ac. kr/vt/chem. -ed/spec/atomic/aa. htm. Last updated 8/21/96. Walpole, Ronald E. , Myers, Raymond H. , Myers Sharon L. Probability and Statistics for Engineers and Scientists, 6 th Edition. Prentice Hall: NJ, 1998.
- Slides: 19