UV Spectra Interpretation Lecture 2 Dapatkan kedua senyawa




- Slides: 15

UV Spectra Interpretation Lecture #2

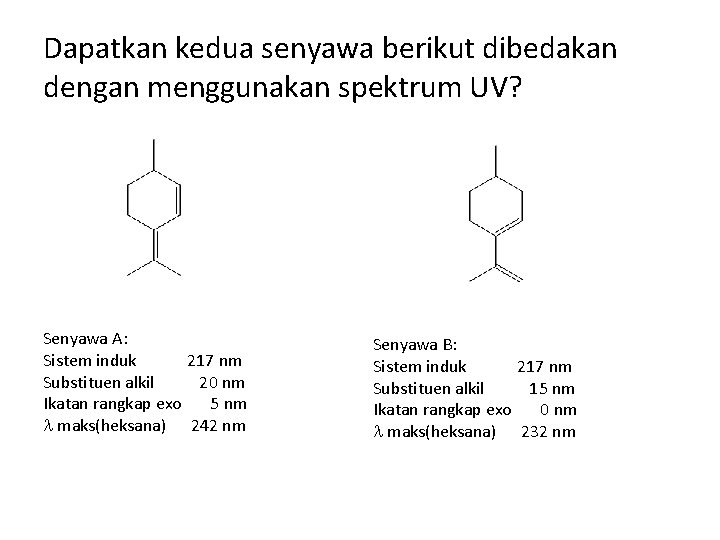
Dapatkan kedua senyawa berikut dibedakan dengan menggunakan spektrum UV? Senyawa A: Sistem induk 217 nm Substituen alkil 20 nm Ikatan rangkap exo 5 nm maks(heksana) 242 nm Senyawa B: Sistem induk 217 nm Substituen alkil 15 nm Ikatan rangkap exo 0 nm maks(heksana) 232 nm

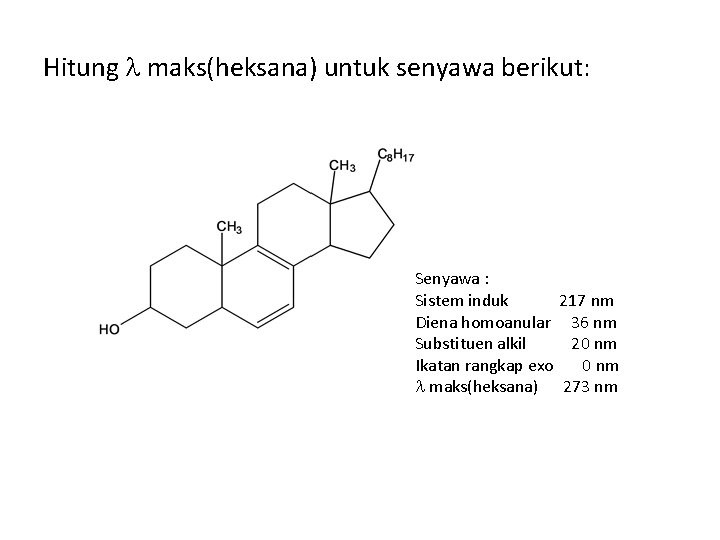
Hitung maks(heksana) untuk senyawa berikut: Senyawa : Sistem induk 217 nm Diena homoanular 36 nm Substituen alkil 20 nm Ikatan rangkap exo 0 nm maks(heksana) 273 nm

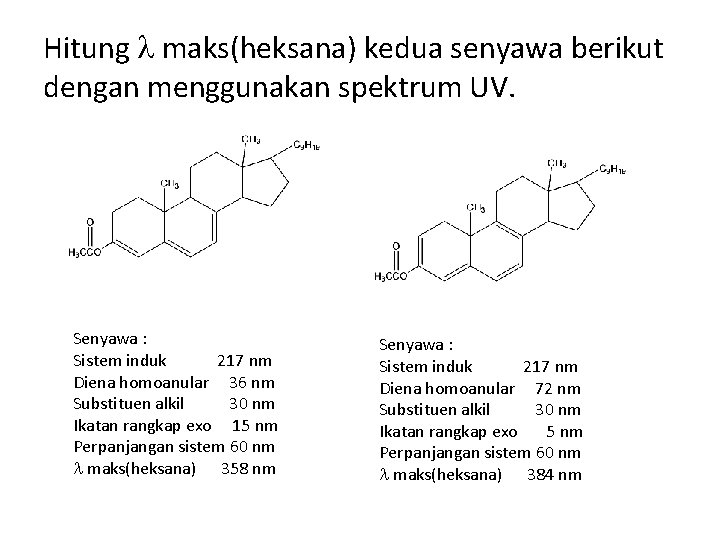
Hitung maks(heksana) kedua senyawa berikut dengan menggunakan spektrum UV. Senyawa : Sistem induk 217 nm Diena homoanular 36 nm Substituen alkil 30 nm Ikatan rangkap exo 15 nm Perpanjangan sistem 60 nm maks(heksana) 358 nm Senyawa : Sistem induk 217 nm Diena homoanular 72 nm Substituen alkil 30 nm Ikatan rangkap exo 5 nm Perpanjangan sistem 60 nm maks(heksana) 384 nm

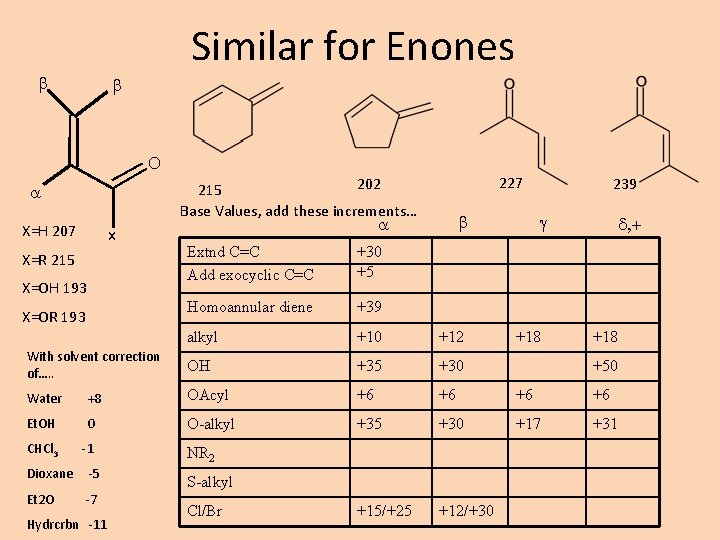
Similar for Enones b b O 227 202 215 Base Values, add these increments… X=H 207 x b 239 g d, + Extnd C=C Add exocyclic C=C +30 +5 Homoannular diene +39 alkyl +10 +12 With solvent correction of…. . OH +35 +30 Water +8 OAcyl +6 +6 Et. OH 0 O-alkyl +35 +30 +17 +31 CHCl 3 -1 +15/+25 +12/+30 X=R 215 X=OH 193 X=OR 193 Dioxane -5 Et 2 O -7 Hydrcrbn -11 NR 2 S-alkyl Cl/Br +18 +50

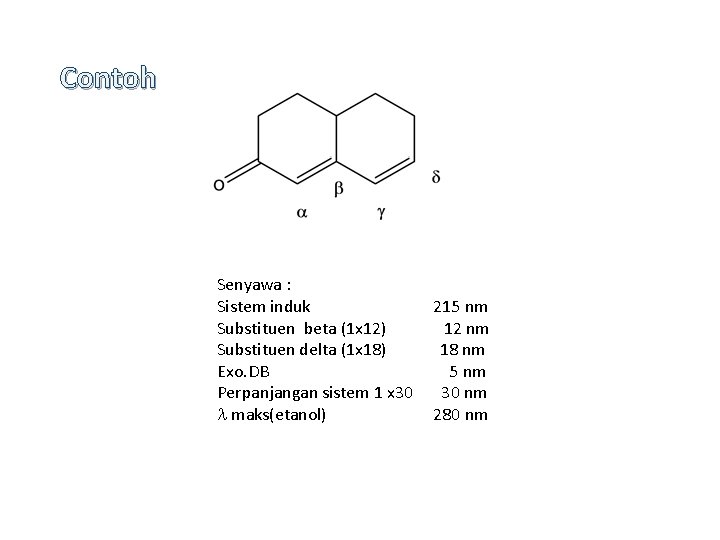
Contoh Senyawa : Sistem induk Substituen beta (1 x 12) Substituen delta (1 x 18) Exo. DB Perpanjangan sistem 1 x 30 maks(etanol) 215 nm 12 nm 18 nm 5 nm 30 nm 280 nm

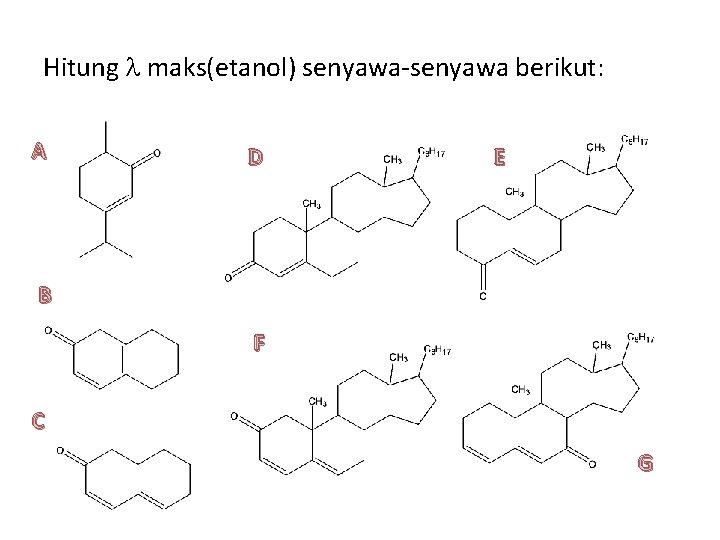
Hitung maks(etanol) senyawa-senyawa berikut: A D E B F C G

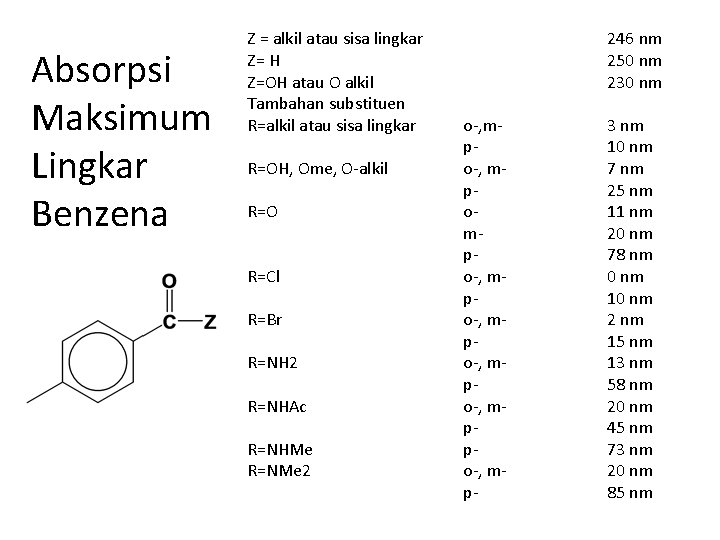
Absorpsi Maksimum Lingkar Benzena Z = alkil atau sisa lingkar Z= H Z=OH atau O alkil Tambahan substituen R=alkil atau sisa lingkar R=OH, Ome, O-alkil R=O R=Cl R=Br R=NH 2 R=NHAc R=NHMe R=NMe 2 246 nm 250 nm 230 nm o-, mpompo-, mppo-, mp- 3 nm 10 nm 7 nm 25 nm 11 nm 20 nm 78 nm 0 nm 10 nm 2 nm 15 nm 13 nm 58 nm 20 nm 45 nm 73 nm 20 nm 85 nm

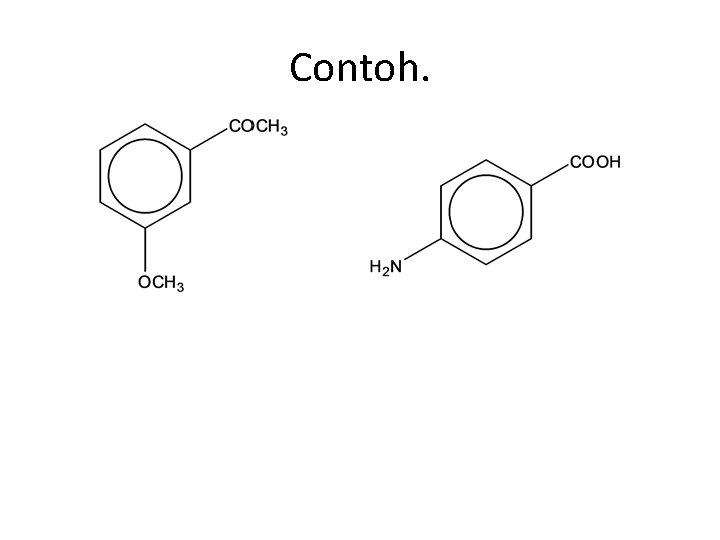
Contoh.

Generally, extending conjugation leads to red shift “particle in a box” QM theory; bigger box Substituents attached to a chromophore that cause a red shift are called “auxochromes” Strain has an effect… max 253 239 256 248

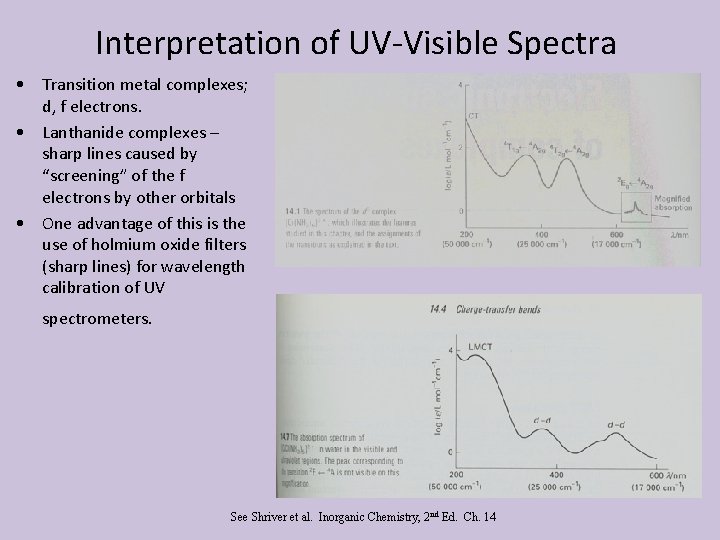
Interpretation of UV-Visible Spectra • Transition metal complexes; d, f electrons. • Lanthanide complexes – sharp lines caused by “screening” of the f electrons by other orbitals • One advantage of this is the use of holmium oxide filters (sharp lines) for wavelength calibration of UV spectrometers. See Shriver et al. Inorganic Chemistry, 2 nd Ed. Ch. 14

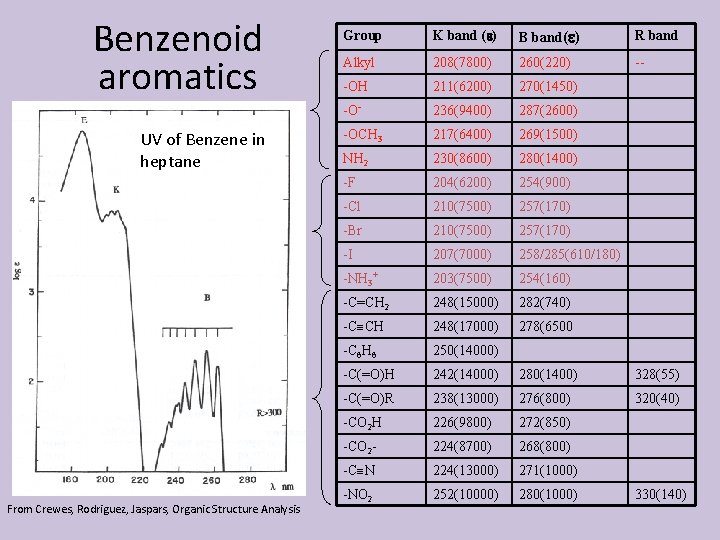
Benzenoid aromatics UV of Benzene in heptane From Crewes, Rodriguez, Jaspars, Organic Structure Analysis Group K band ( ) B band( ) R band Alkyl 208(7800) 260(220) -- -OH 211(6200) 270(1450) -O- 236(9400) 287(2600) -OCH 3 217(6400) 269(1500) NH 2 230(8600) 280(1400) -F 204(6200) 254(900) -Cl 210(7500) 257(170) -Br 210(7500) 257(170) -I 207(7000) 258/285(610/180) -NH 3+ 203(7500) 254(160) -C=CH 2 248(15000) 282(740) -C CH 248(17000) 278(6500 -C 6 H 6 250(14000) -C(=O)H 242(14000) 280(1400) 328(55) -C(=O)R 238(13000) 276(800) 320(40) -CO 2 H 226(9800) 272(850) -CO 2 - 224(8700) 268(800) -C N 224(13000) 271(1000) -NO 2 252(10000) 280(1000) 330(140)

Substituent effects don’t really add up Can’t tell any thing about substitution geometry Exception to this is when adjacent substituents can interact, e. g hydrogen bonding. E. g the secondary benzene band at 254 shifts to 303 in salicylic acid In p-hydroxybenzoic acid, it is at the phenol or benzoic acid frequency

Heterocycles Nitrogen heterocycles are pretty similar to the benzenoid anaologs that are isoelectronic. Can study protonation, complex formation (charge transfer bands)

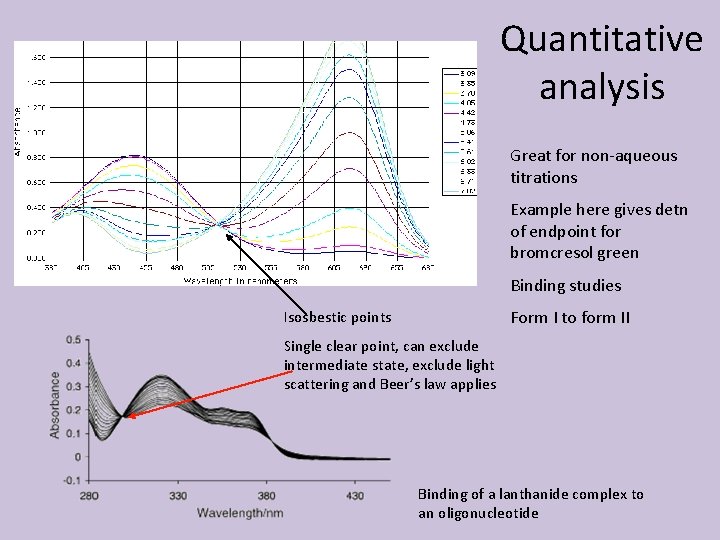
Quantitative analysis Great for non-aqueous titrations Example here gives detn of endpoint for bromcresol green Binding studies Form I to form II Isosbestic points Single clear point, can exclude intermediate state, exclude light scattering and Beer’s law applies Binding of a lanthanide complex to an oligonucleotide