utritional Biochemistry lker GHAN M D Clinical Biochemistry

utritional Biochemistry İlker GÖÇHAN (M. D) Clinical Biochemistry Specialist

Nucleic acids, DNA and RNA structures and functions WEEK 10

OVERVIEW Nucleic acids are required for the storage and expression of genetic information. There are two chemically distinct types of nucleic acids: Deoxyribonucleic acid (DNA) and Ribonucleic acid (RNA)

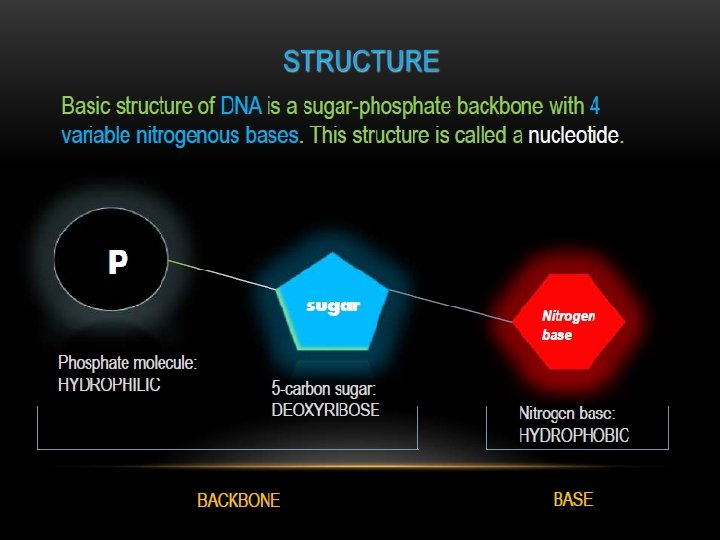
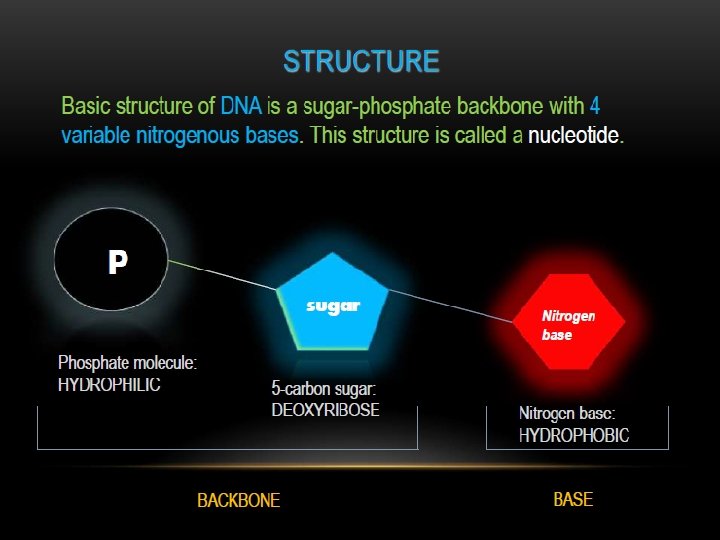
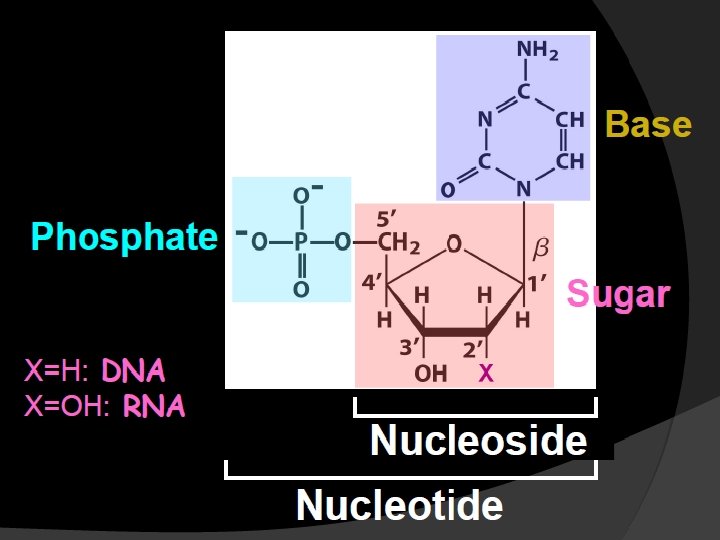
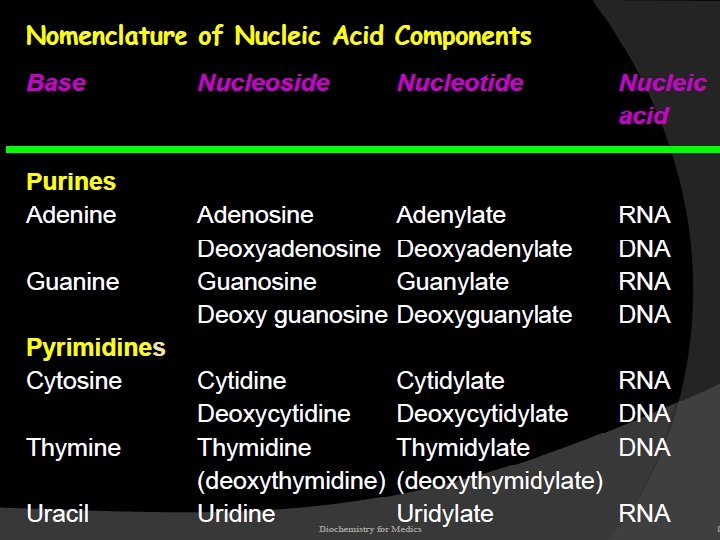
Nucleotides contain; Base + sugar + phosphoryl group
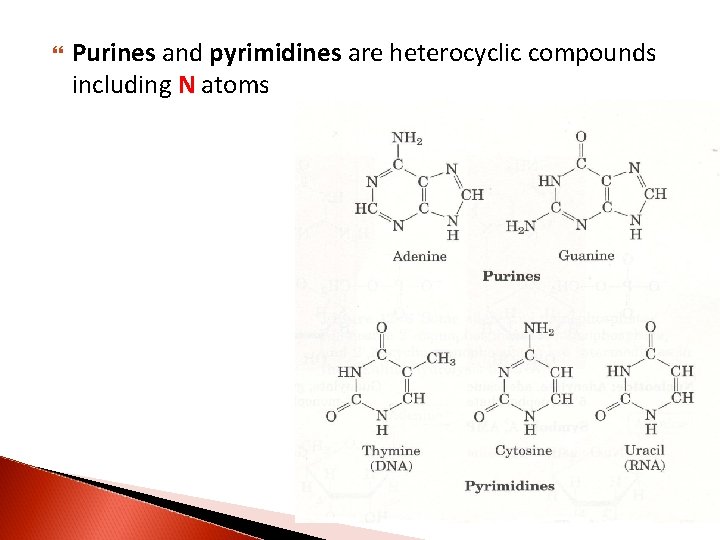
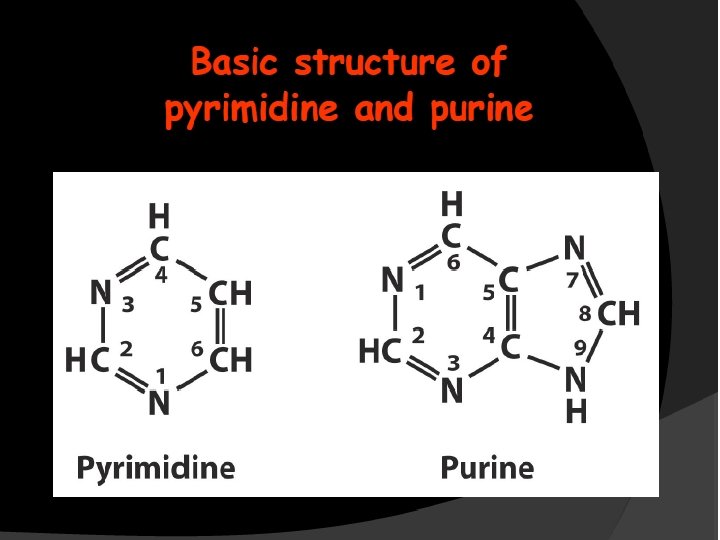
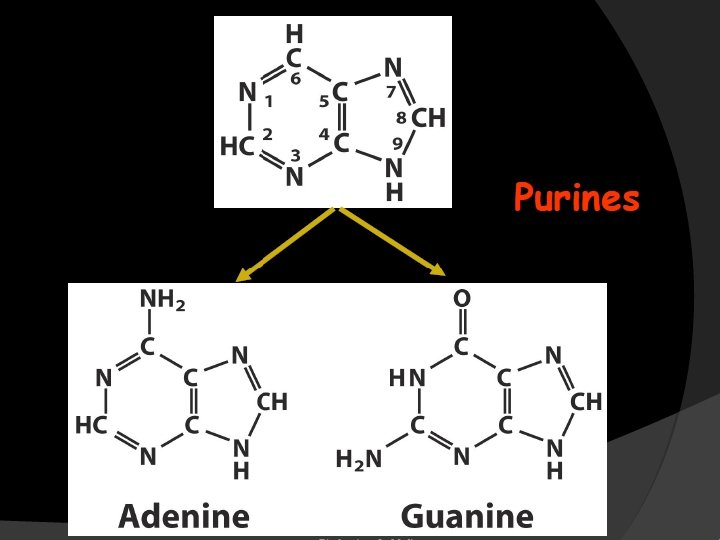
Purines and pyrimidines are heterocyclic compounds including N atoms
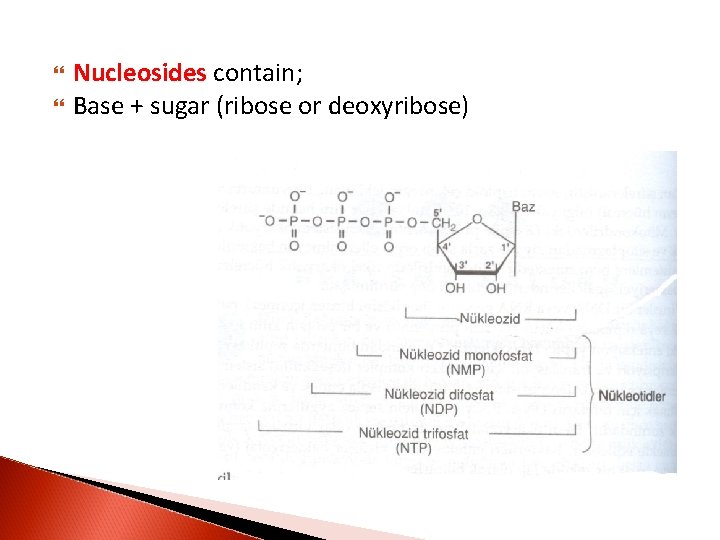
Nucleosides contain; Base + sugar (ribose or deoxyribose)
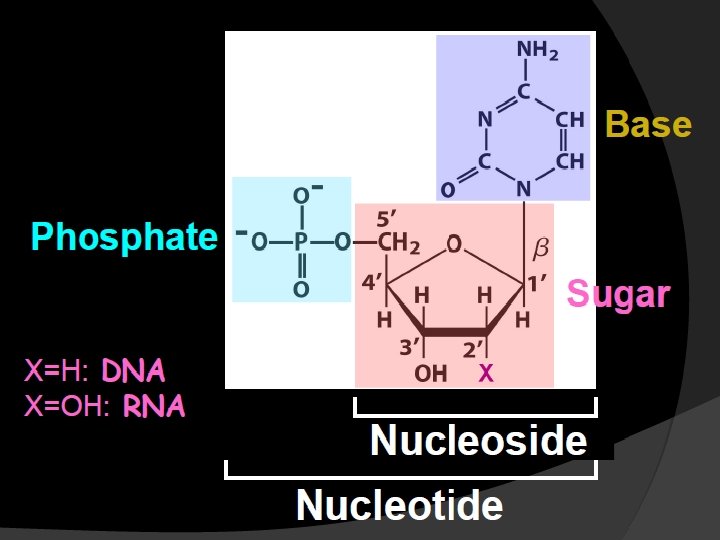
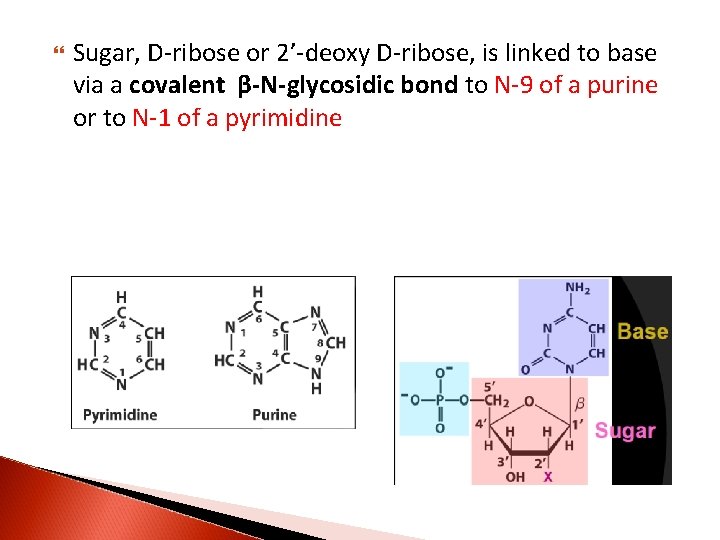
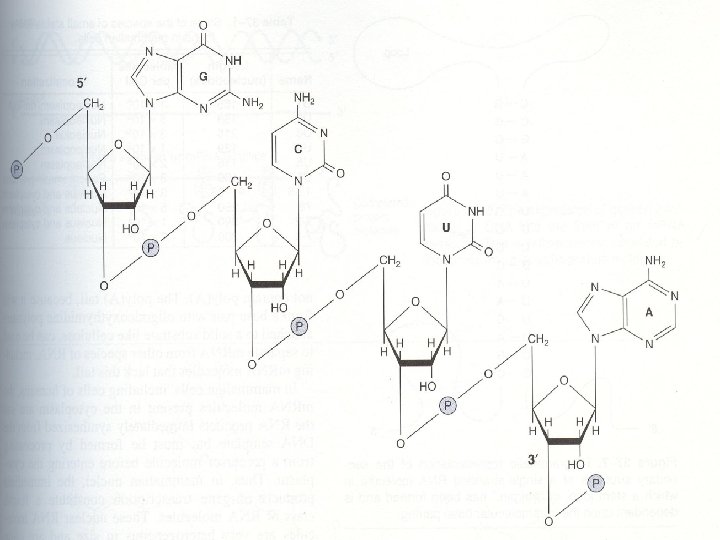
Sugar, D-ribose or 2’-deoxy D-ribose, is linked to base via a covalent β-N-glycosidic bond to N-9 of a purine or to N-1 of a pyrimidine
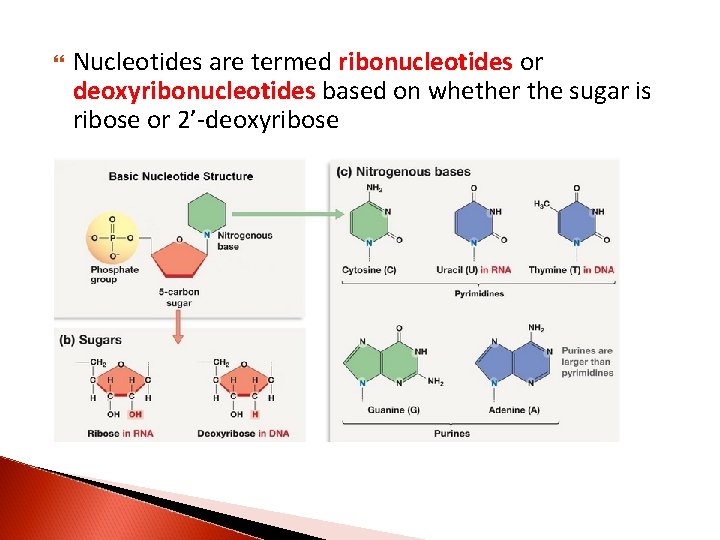
Nucleotides are termed ribonucleotides or deoxyribonucleotides based on whether the sugar is ribose or 2’-deoxyribose
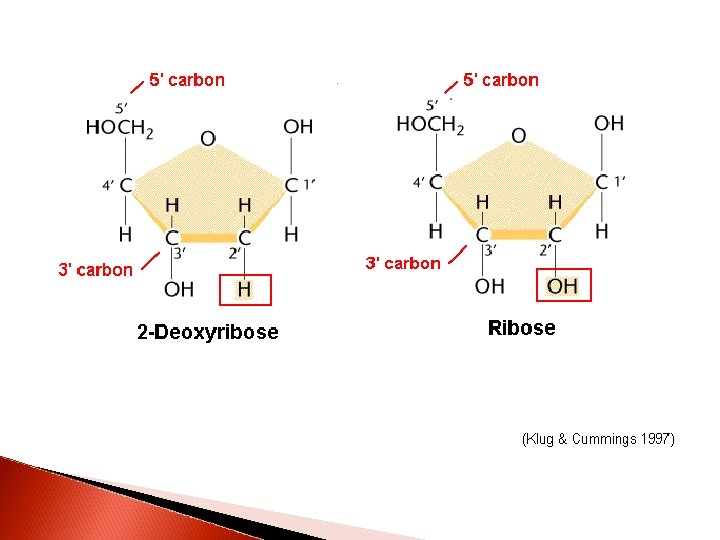
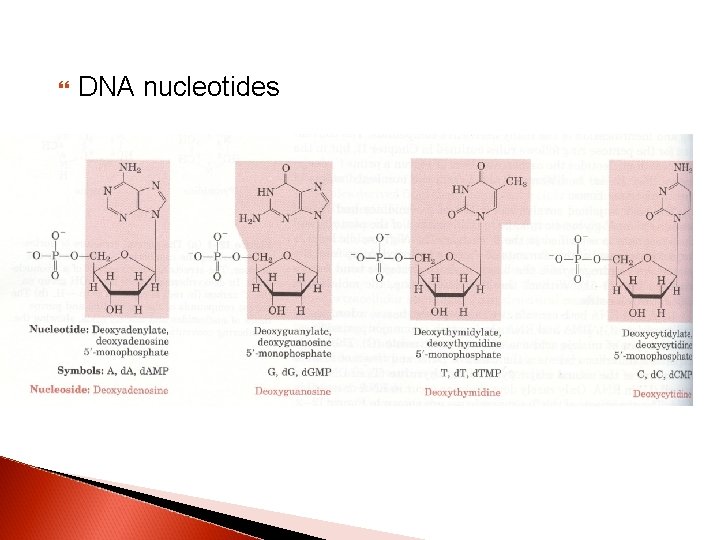
DNA nucleotides
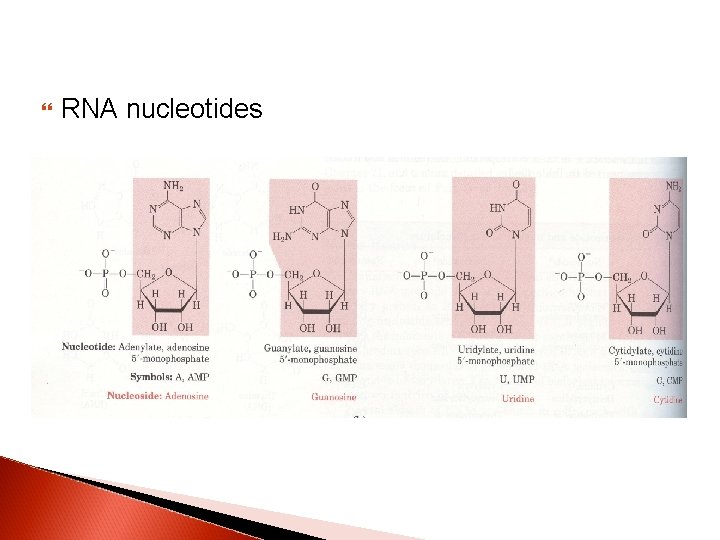
RNA nucleotides

Nucleotides are phosphorolated nucleosides Mononucleotides are nucleotides singly phosphorolated on hydroxyl group of the sugar AMP → Adenosine monophosphate Adenine + ribose +phosphate Additional phosphates linked by acid anhydride bonds to the existing phosphate of a mononucleotide form nucleoside di- and triphosphates
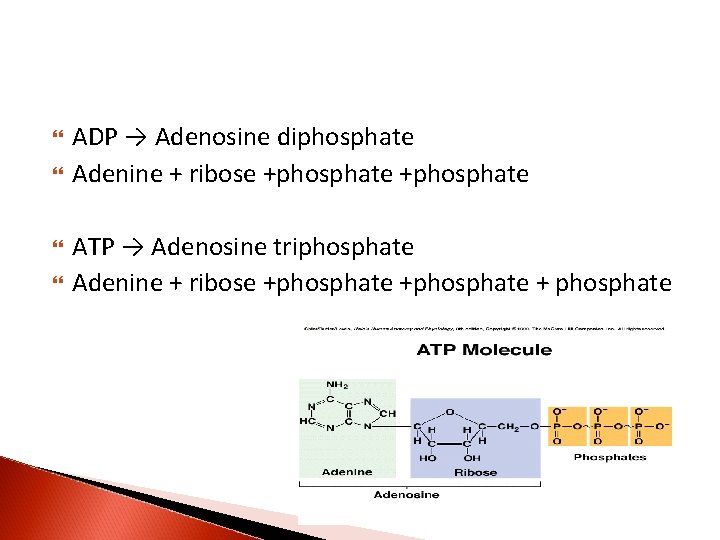
ADP → Adenosine diphosphate Adenine + ribose +phosphate ATP → Adenosine triphosphate Adenine + ribose +phosphate + phosphate

Functions of nucleotides *Nucleic acid biosynthesis *Energy production and transduction *Protein biosynthesis *Regulatory cascades *Intra- and intercellular signal transduction *Biosynthesis some biomolecules

Some properties of nucleotides 1. Mononucleotides have a negative charge at physiological p. H 2. Nucleotides absorb UV light 3. Many coenzymes are nucleotide derivatives 4. Synthetic nucleotide analogs are used in chemotherapy 5. Nucleoside triphosphates have high group transfer potential 6. Some nucleotides are involved in signal transduction

1. Mononucleotides have a negative charge at physiological p. H. The p. Ks of the primary and secondary phosphoryl groups are about 1. 0 and 6. 2, respectively Nucleosides and or free purine or pyrimidine bases are uncharged at physiological p. H

2. Nucleotides absorb UV light. The conjugated double bonds of the heterocyclic bases of purines and pyrimidines ensure that nucleosides, nucleotides and polynucleotides absorb UV

3. Many coenzymes are nucleotide derivatives AMP is present in many coenzymes
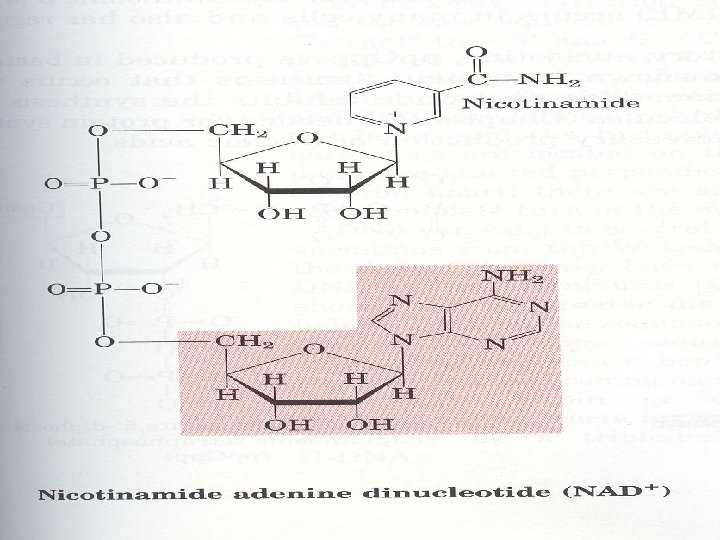
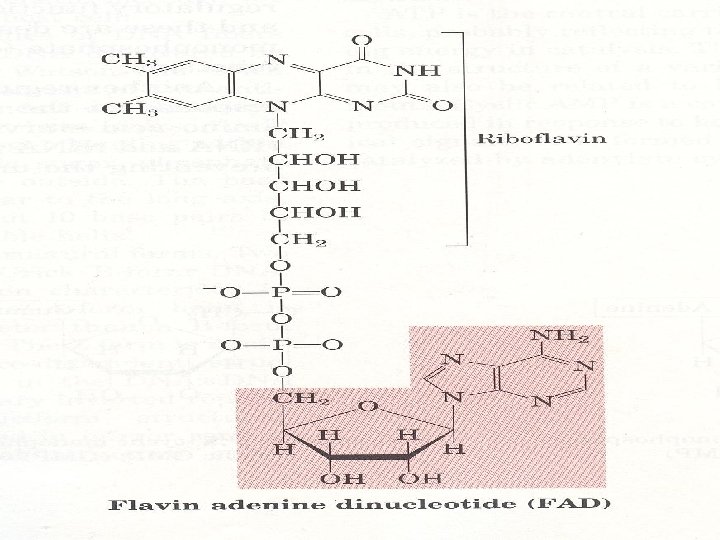

4. Synthetic nucleotide analogs are used in chemotherapy Chemically synthesized analogs of purines and pyrimidines, their nucleosides and their nucleotides find numerious applications in clinical medicine
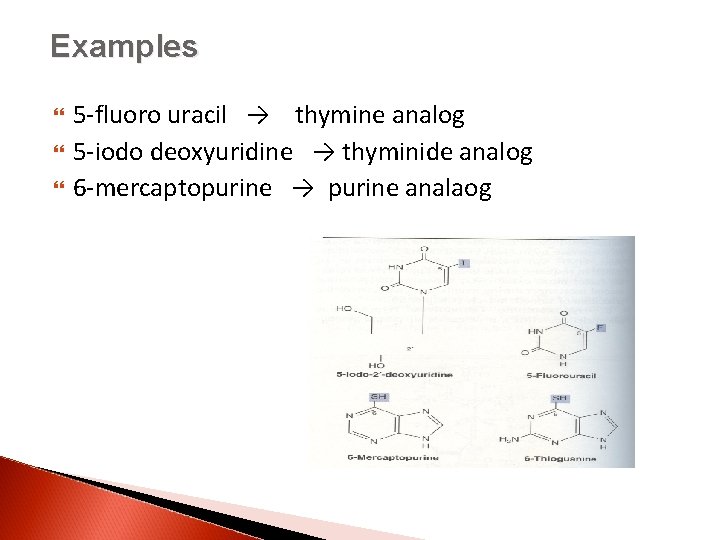
Examples 5 -fluoro uracil → thymine analog 5 -iodo deoxyuridine → thyminide analog 6 -mercaptopurine → purine analaog
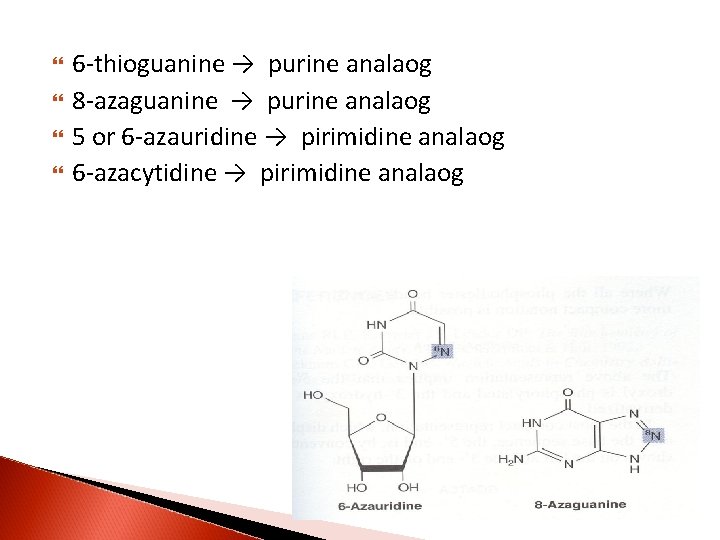
6 -thioguanine → purine analaog 8 -azaguanine → purine analaog 5 or 6 -azauridine → pirimidine analaog 6 -azacytidine → pirimidine analaog
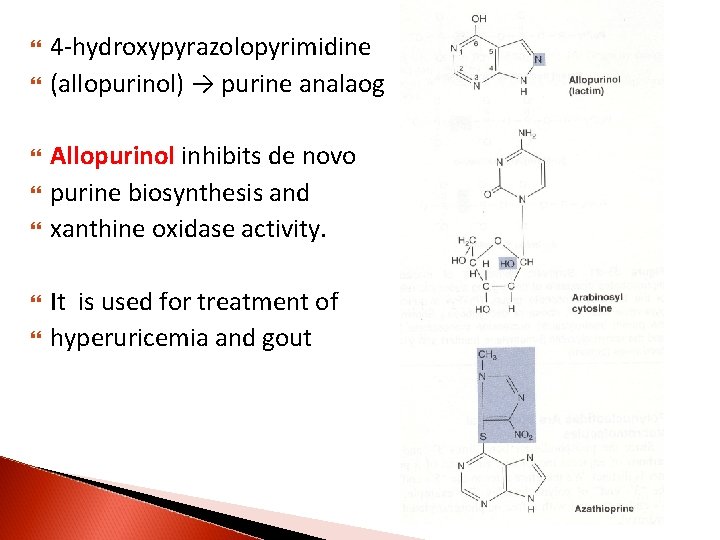
4 -hydroxypyrazolopyrimidine (allopurinol) → purine analaog Allopurinol inhibits de novo purine biosynthesis and xanthine oxidase activity. It is used for treatment of hyperuricemia and gout

The nucleoside, cytarabine (arabinosyl cytosine), in which arabinose replaces ribose, is used in chemotherapy and in treatment of viral infections. Azathioprine is catabolized to 6 -mercaptopurine and is used during organ transplantation to suppress immunological rejection

5. Nucleoside triphosphates have high group transfer potential because of acid anhydride bonds High group transfer potential of nucleoside triphosphates allows them to participate as group transfer reagents in various reactions.

ADP and ATP are substrates and products, respectively, for oxidative phosphorylation ATP serves as the major biologic transducer of free energy ATP donates some of its chemical energy by hydrolysis of the terminal phosphoanhydride bond

6. Some nucleotides are involved in signal transduction c. AMP and c. GMP
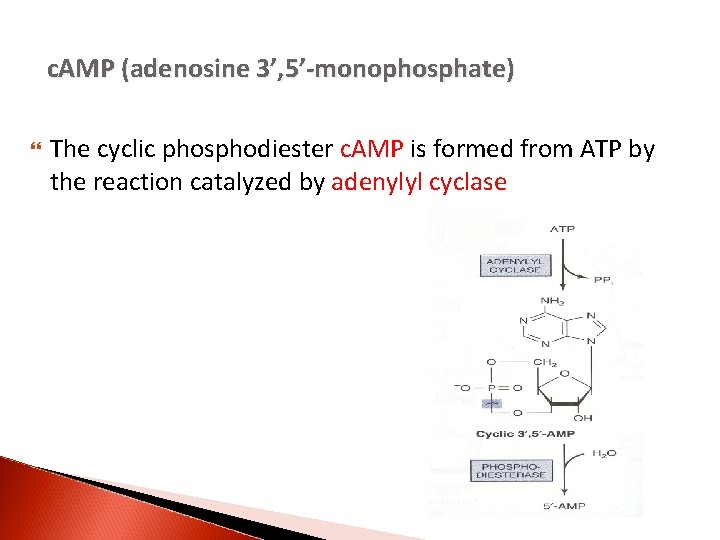
c. AMP (adenosine 3’, 5’-monophosphate) The cyclic phosphodiester c. AMP is formed from ATP by the reaction catalyzed by adenylyl cyclase

c. AMP is a second messenger in signal transduction Adenylyl cyclase activity is regulated by complex interactions, many of which involve hormon receptors As a second messenger, c. AMP participates numerous regulatory functions by activating c. AMP dependent protein kinases c. AMP is broken down by c. AMP phosphodiesterase

c. GMP (guanosine 3’, 5’-monophosphate) c. GMP is a second messenger in signal transduction that can act antagonistically to c. AMP c. GMP is formed from GMP by guanylyl cyclase Both adenylyl and guanylyl cyclases are regulated by effectors that include hormones A phosphodiesterase hydrolyzes c. GMP to GMP

An increase in the level of the c. GMP as response to the nitric oxide serves as the main second messenger during events that characterize the relaxation of smooth muscle

Nucleases Nucleic acids are digested by nucleases Nucleases exhibit specifity to deoxyribonucleic acids are referred to as deoxyribonucleases Those which specifically hydrolyze ribonucleic acids are ribonucleases Enzymes capable of cleaving internal phosphodiester bonds are referred to as endonucleases

Some nucleases are capable of hydrolyzing a nucleotide only when it is present at a terminal of a nucleic acid. These enzymes are referred to as exonucleases Exonucleases can act in one direction only; 3’→ 5’ or 5’→ 3’

DNA

What is DNA ? 5: 23

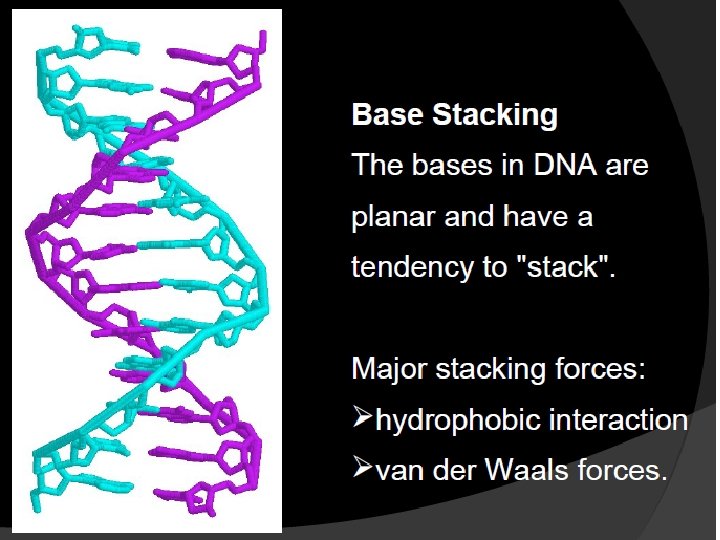
STRUCTURE OF DNA With the exception of a few viruses that contain singlestranded (ss) DNA, DNA exists as a double stranded (ds) molecule, in which the two strands wind around each other, forming a double helix.

THE DISCOVERY OF DNA DOUBLE HELIX IS ONE OF THE GREATEST FINDINGS OF ALL TIME, BUT IT’S ALSO ONE OF THE MOST CONTROVERSIAL.

KEY SCIENTISTS INVOLVED :



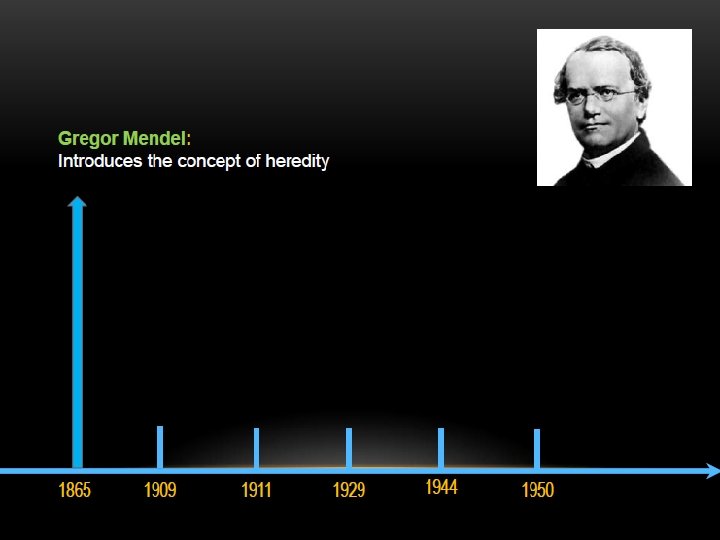
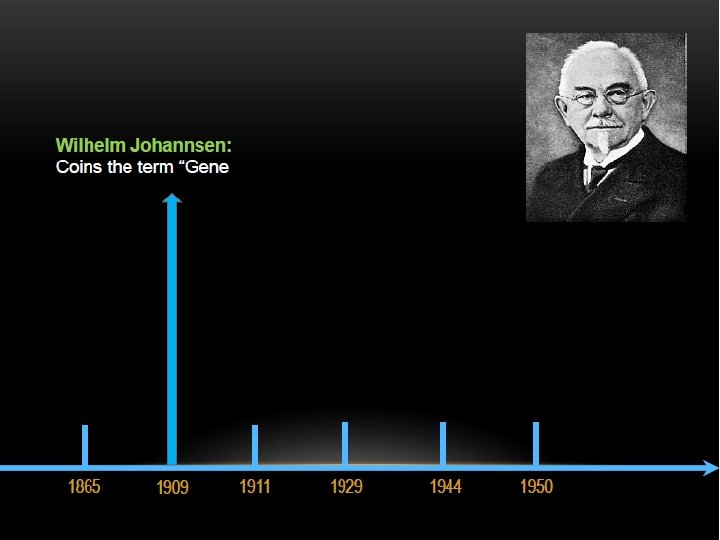
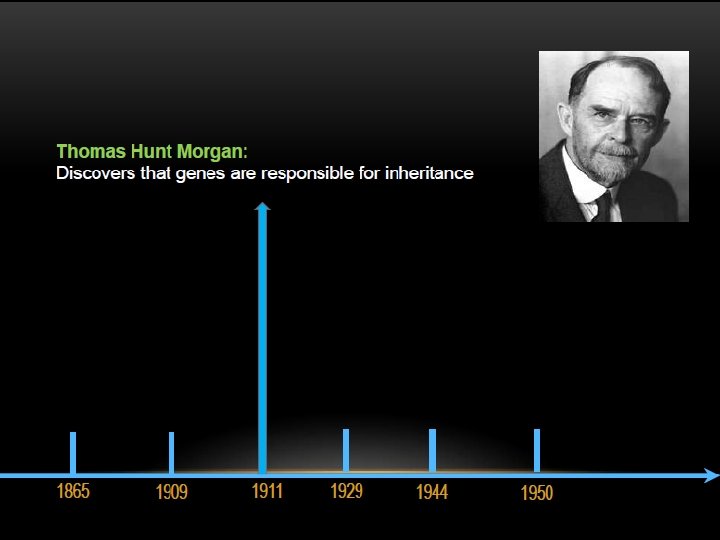
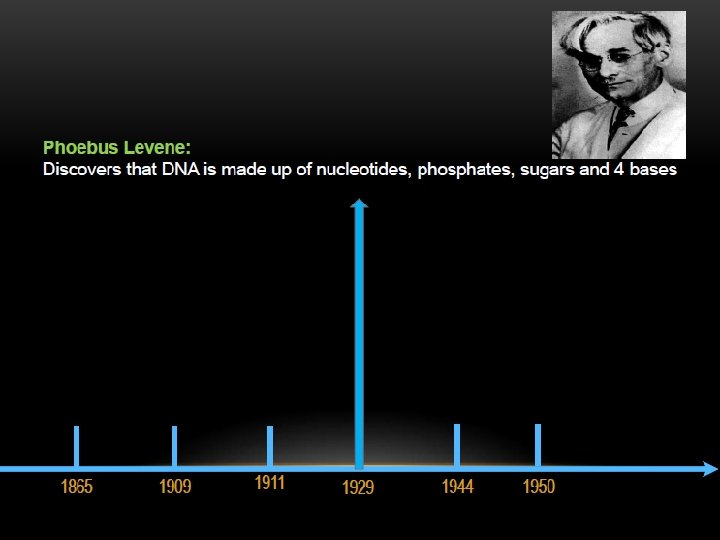
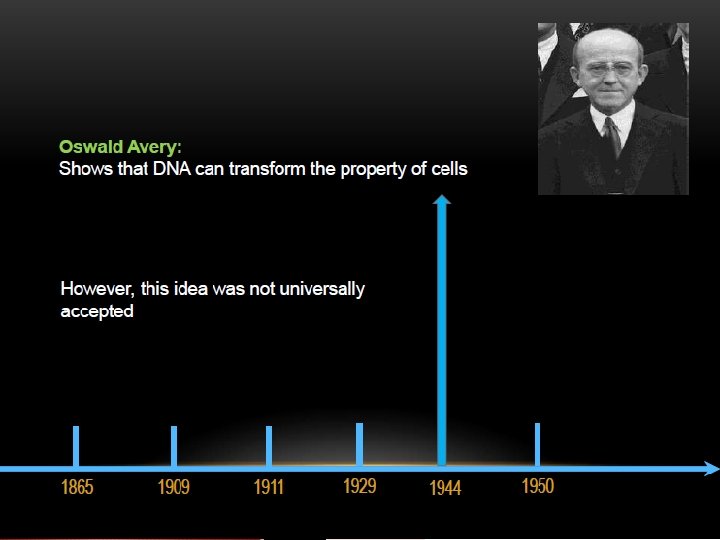
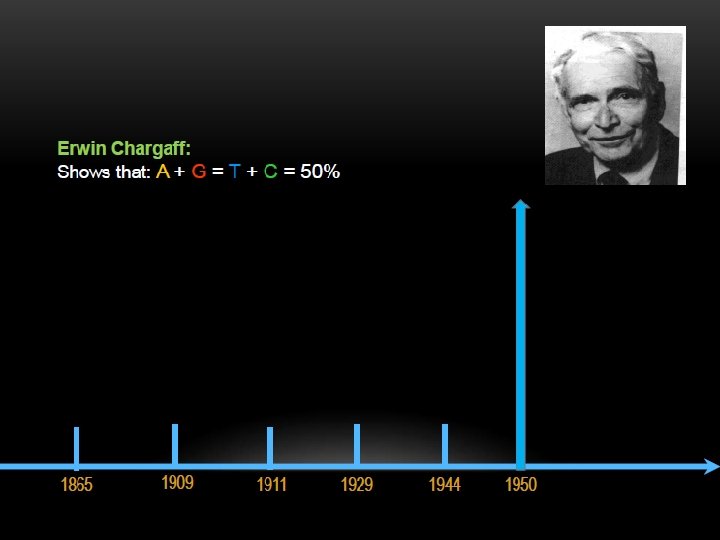






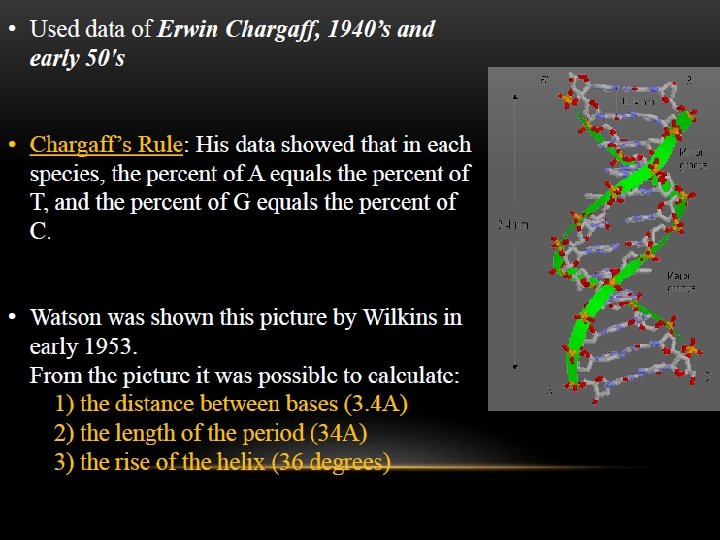





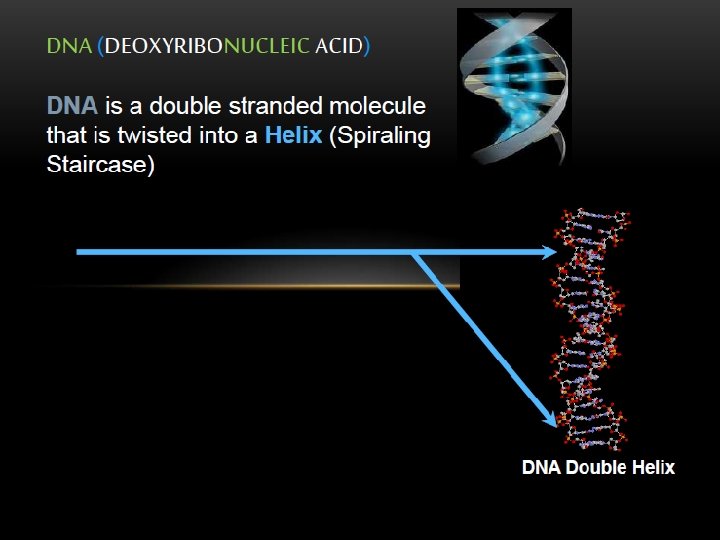
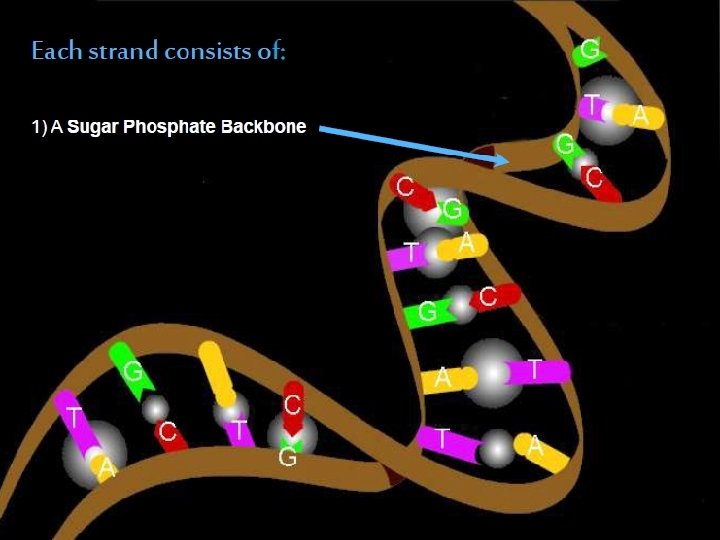
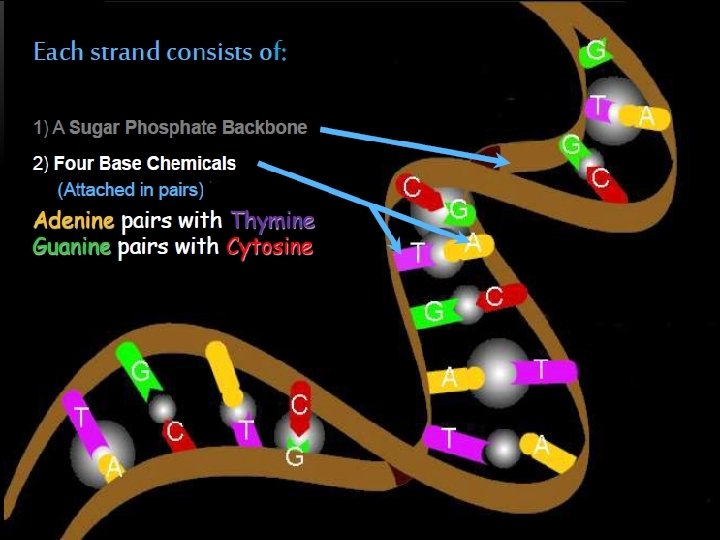



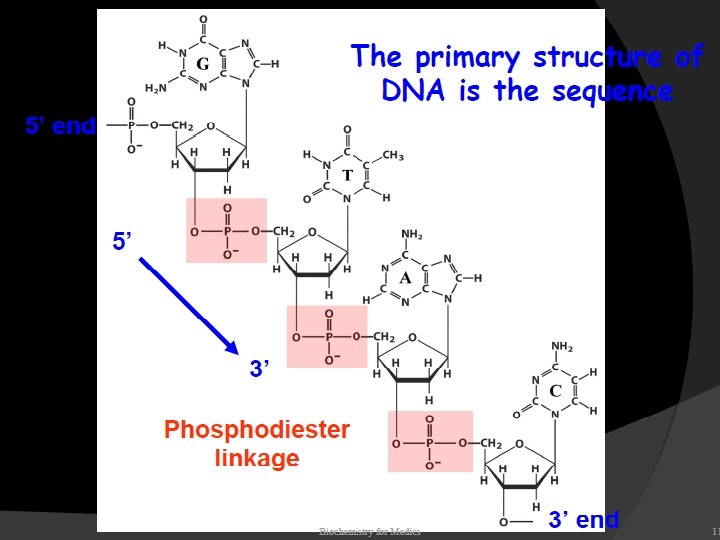
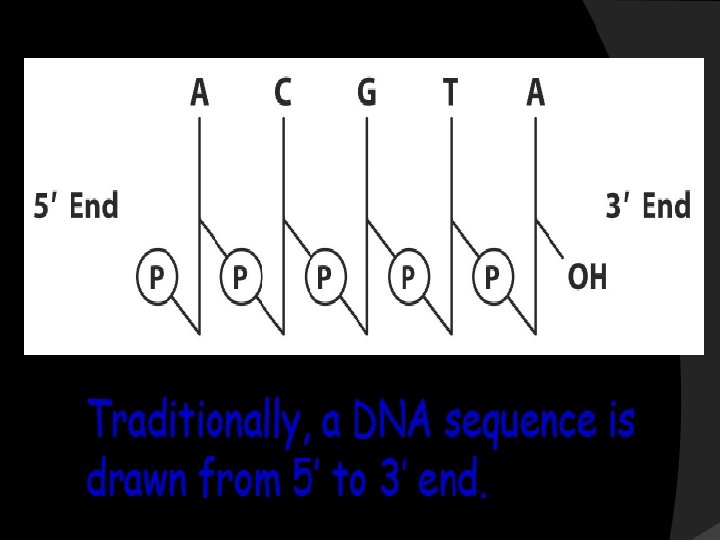

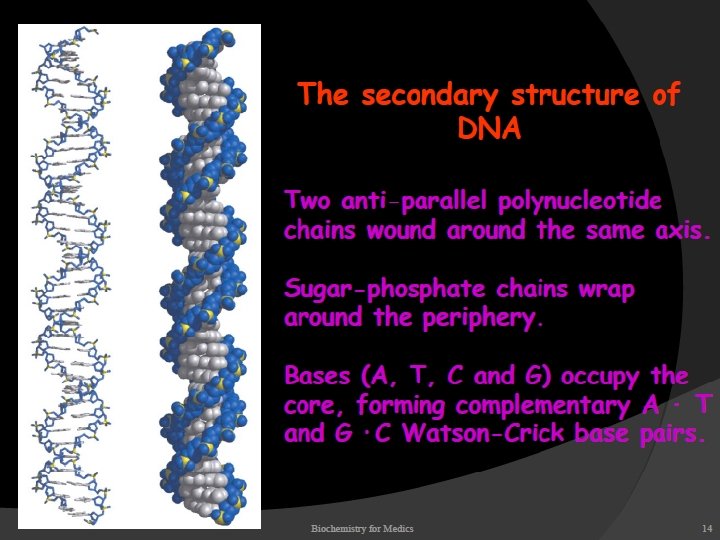

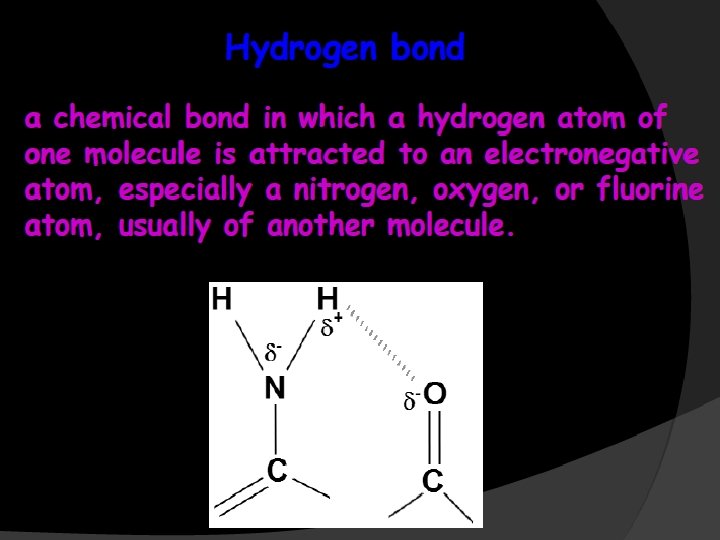
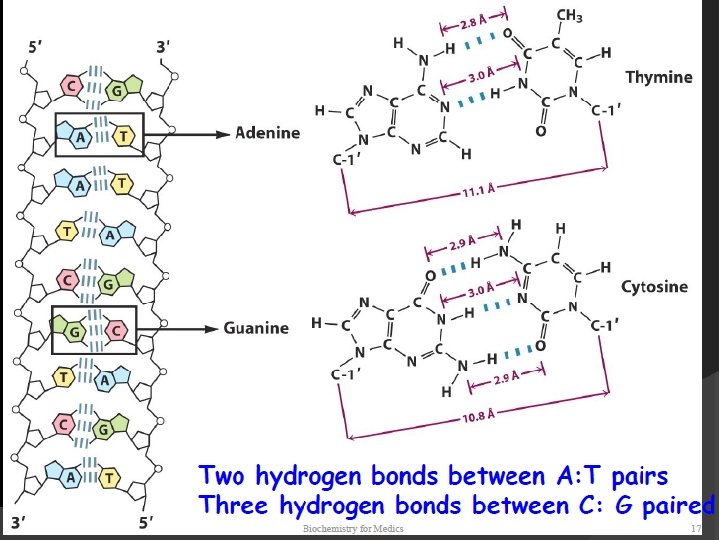

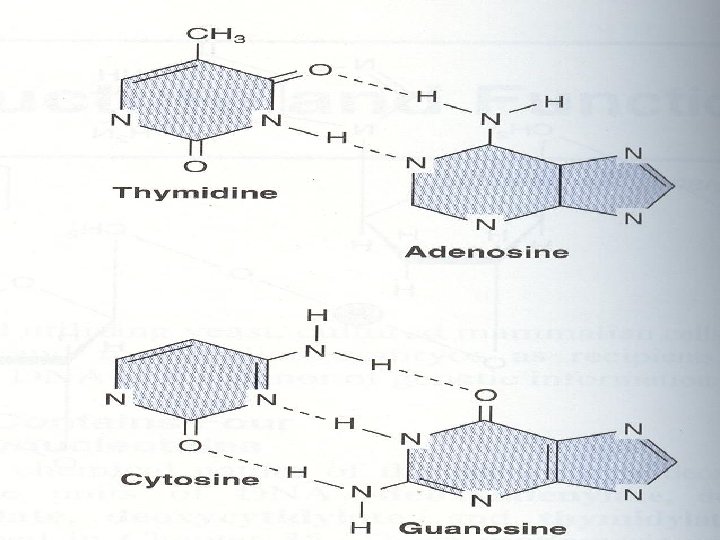
The two strands of this double-stranded helix are held in register by hydrogen bonds between the purine and pyrimidine bases of the respective linear molecules The pairings between the purine and pyrimidine nucleotides on the opposite strands are very specific and are present between A and T; C and G


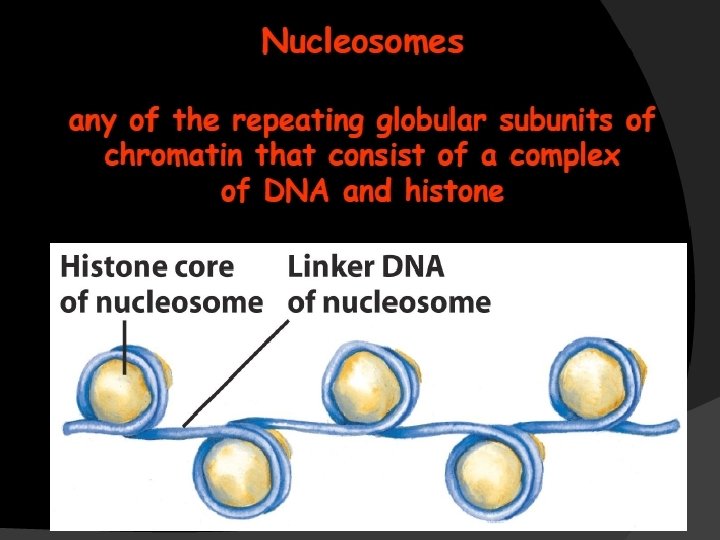
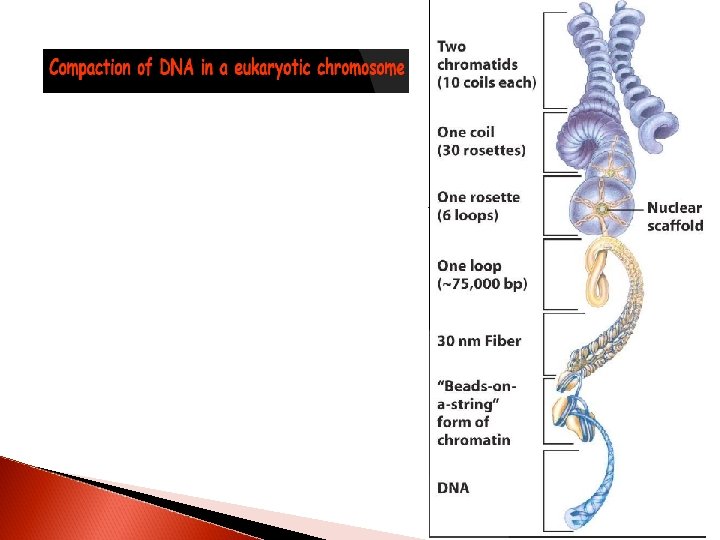

DNA replication 3: 23

What is a choromosome ? 5: 02

RNA

Chemical structure of RNA The chemical nature of RNA differs from that of DNA RNA is also formed by purines and pyrimidines linked by 3’-5’ phosphodiester bonds

Although sharing many features with DNA , RNA possesses several specific differences: 1. In RNA, the sugar to which the phosphates and purine and pyrimidines are attached is ribose instead of deoxyribose of DNA

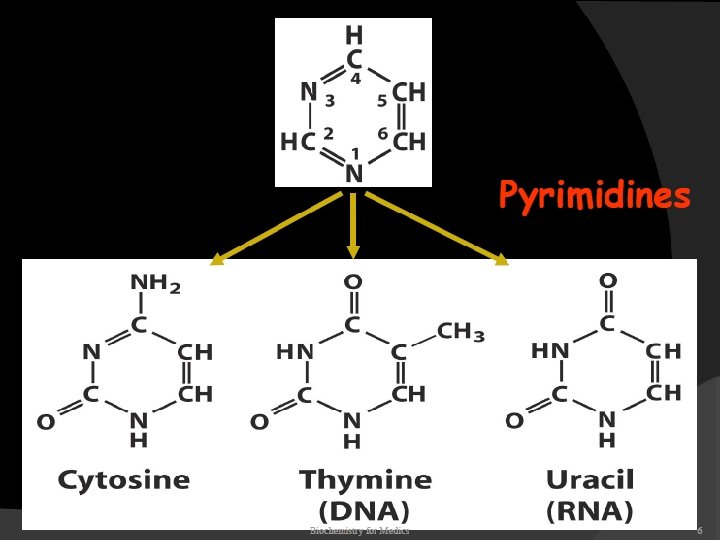
2. The pyrimidine components of RNA is differ from those of DNA. RNA contains the A, G, C but does not contain T (with a rare exception), instead of T, U is present in RNA

3. RNA exist as a single strand. However, given the proper complementary base sequence with opposite polarity, single strand RNA is capable of folding back on itself like a hairpin thus acquiring double-stranded structure
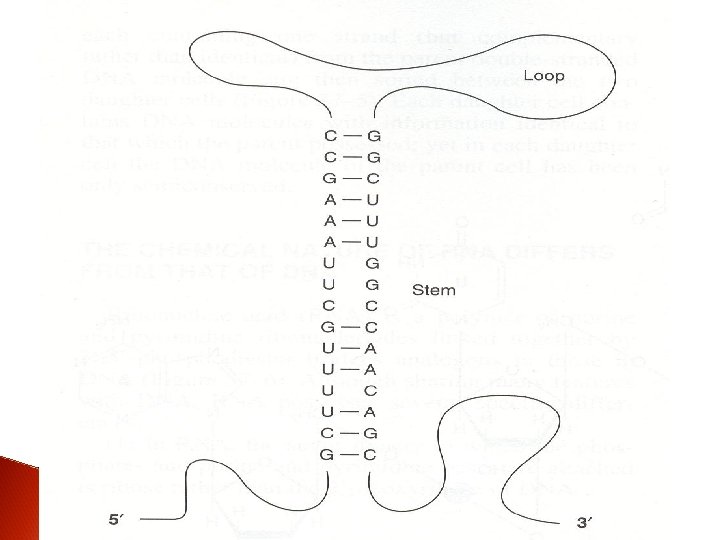

4. Since the RNA molecule is a single strand complementary to only one of the two strands of a gene, its guanine content does not necessarily eaqual its cytosine content; and its adenine content does not necessarily eaqual its uracil content.

5. RNA can be hydrolyzed by alkali to 2’, 3’cyclic diesters of mononucleotides but those molecules can not be formed from alkali-treated DNA because of the absence of a 2’- OH group

Information within the RNA is contained in its sequence of purine and pyrimidine nucleotides. The sequence is complementary to the template strand of the gene that was transcribed

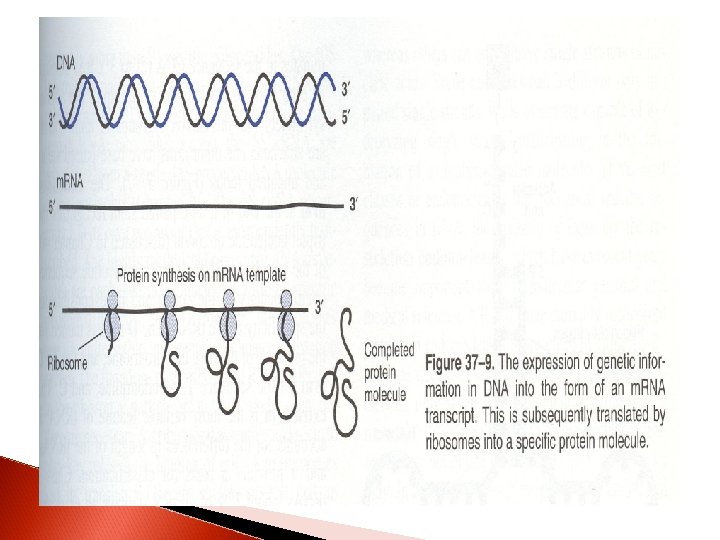
RNA types and their funtions Cytoplasmic RNA molecules that serve as a templates for protein synthesis are designated as messenger RNAs (m. RNA). m. RNA molecules transfer genetic information from DNA to protein-synthesizing machinary

Many other cytoplasmic RNA molecules have structural roles. They contribute to the formation of ribosomes (ribosomal RNA, r. RNA) or serve as adapter molecules (transfer RNA, t. RNA) for the translation of RNA information into specific sequences of polymerized amino acids


Some RNA molecules have intrinsic catalytic activity. The activity of these ribozymes often involved in the cleavage of a nucleic acid In human cells there are small nuclear RNA (sn. RNA) species. These are not directly involved in protein synthesis but that may have roles in RNA processing These relatively small molecules vary in size from 90 to 300 nucleotides

The genetic material for some animal and plant viruses is RNA rather than DNA

Protein synthesis 5: 07

1. Messenger RNA (m. RNA) This is the most heterogenous class in size and stability All members of the class function as messengers conveying the information in a gene to the proteinsynthesizing machinery Each serves as a template for a specific sequence of amino acids that is polimerized to form a specific protein

2. Transfer RNA (t. RNA) t. RNA molecules vary in lenght from 74 to 95 nucleotides t. RNA molecules serve as a adapters for the translation of the information in the sequence of nucleotides of m. RNA into specific amino acids There at least 20 species of t. RNA molecules in every cell. At least one, often several corresponding to each of the 20 amino acid are required for protein synthesis

Although each specific t. RNA differs from the others in its sequence of nucleotides, the t. RNA molecules as a class have many features in common The primary structure of all t. RNA molecules allows extensive folding and intrastrand complementarity to generate a secondary structure This structure appears like a cloverleaf


All t. RNA molecules contain four main arms The acceptor arm consists of a base-paired stem that terminates in the sequence CCA (5’ to 3’). It is through an ester bond to the 3’ hydroxyl group of the adenosyl moiety that the carboxyl groups of amino acids are attached
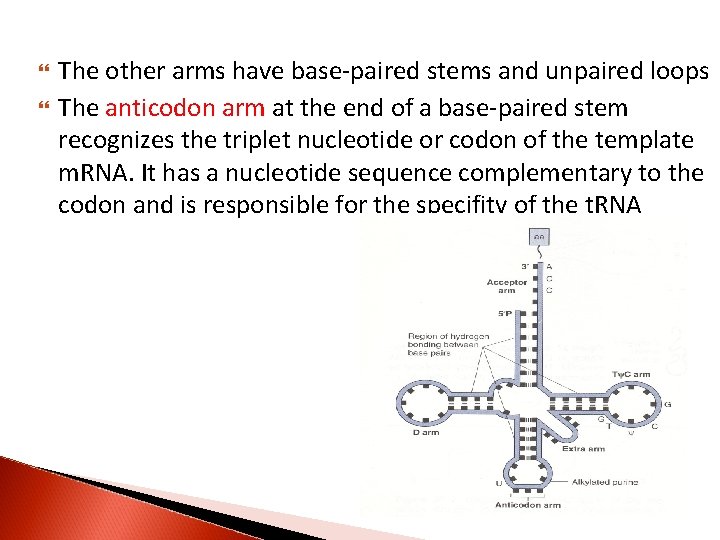
The other arms have base-paired stems and unpaired loops The anticodon arm at the end of a base-paired stem recognizes the triplet nucleotide or codon of the template m. RNA. It has a nucleotide sequence complementary to the codon and is responsible for the specifity of the t. RNA

The D arm is named for the presence of the base dihydrouridine, and the TΨC arm for the sequence T, pseudouridine, and C

The extra arm is the most variable feature of t. RNA. It accounts for the differences in lenght of the t. RNAs; and it provides a basis for classification. Class 1 t. RNAs have an extra arm that is 3 -5 bp long Class 2 t. RNAs have an extra arm that is 13 -21 bp long

The secondary structure of t. RNA molecules is maintained by the base pairing in these arms and this is a consistent feature : The TΨC and anticodon arms have 5 bp The D arm has 3 -4 bp The acceptor arm has 7 bp
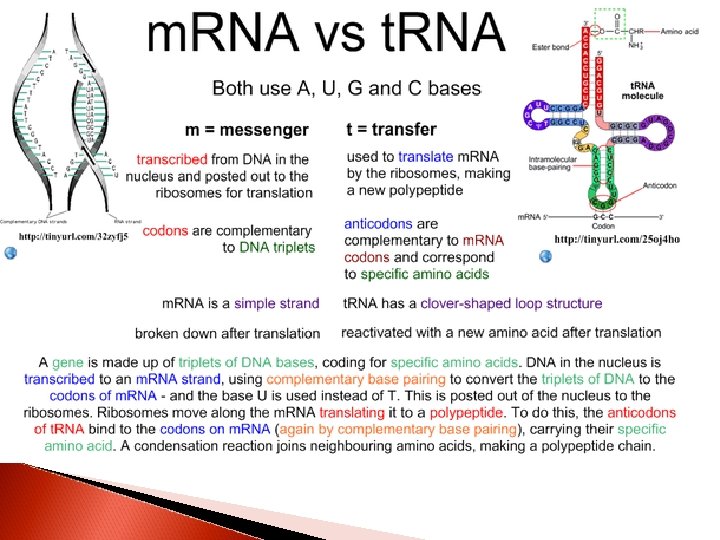

3. Ribosomal RNA (r. RNA) A ribosome is a cytoplasmic nucleoprotein structure that acts as the machinery for the synthesis of proteins from the m. RNA templates On the ribosomes, the m. RNA and t. RNA molecules interact to translate into a specific protein molecule information transcribed from the gene During the active protein synthesis, many ribosomes are associated with an m. RNA molecule in an assembly called the polysome

The mammalian ribosome contains two major nucleoprotein subunits, a larger one with a molecular weight of 2. 8 x 106 (sedimentation velocity is 60 S*) and a smaller subunit with a molecular weight of 1. 4 x 106 (40 S) The 60 S subunit contains a 5 S ribosomal RNA, a 5. 8 S r. RNA and a 28 S r. RNA; there also probably more than 50 specific polypeptides *Svedberg unit (sedimentation coefficient)

The 40 S subunit is smaller and contains 18 S r. RNA and approximately 30 polypeptide chains

Ribosomal RNA molecules are necessary for ribosomal assembly and seem to play key roles in the binding of m. RNA to ribosomes and its translation

Small stable RNA (sn. RNA) A large number of discrete, highly conserved and small stable RNA species are found in the mammalian cells The majority of these molecules exist as ribonucleoproteins and are distributed in the nucleus, cytoplasm or in both They are involved in m. RNA processing and gene regulation

Sources 1. lippincott - illustrated reviews - biochemistry, 3 rd ed 2. Color atlas of biochemistry (Koolman J. , Roehm K. H. - 2005 - 2 nd ed. Thieme) 3. General, Organic, and Biochemistry 9 th ed - Katherine J. Denniston et al. (Mc. Graw-Hill, 2017) 4. Clinical Biochemistry - An Illustrated Colour Text (5 th Ed)
- Slides: 126