utritional Biochemistry lker GHAN M D Clinical Biochemistry

utritional Biochemistry İlker GÖÇHAN (M. D) Clinical Biochemistry Specialist

Bioenergetics and Oxidative Phosphorilation WEEK 4



Energy Energy: the capacity to do work In physics, energy is the property that must be transferred to an object in order to perform work on, or to heat the object. It can be converted in form, but not created or destroyed. 5

THERE ARE TWO TYPES OF ENERGY: -kinetic energy: the energy of motion -potential energy: stored energy Energy can take many forms: chemical, mechanical, electric current, heat, light


Living organisms must work to stay alive, to grow and to reproduce All living organisms have the ability to produce energy and to channel it into biological work Living organisms carry out energy transductions, conversions of one form of energy to another form

Modern organisms use the chemical energy in fuels (carbonhydrates, lipids) to bring about the synthesis of complex macromolecules from simple precursors. They also convert the chemical energy into concentration gradients and electrical gradients, into motion and heat, and, in a few organisms into light (fireflies, some deep-sea fishes)

Biological energy transductions obey the same physical laws that govern all other natural processes. Bioenergetics is the quantitative study of the energy transductions that occur in living cells and of the nature and function of the chemical process underlying these transductions

The goals of this lesson Review the laws of thermodynamics Understand the quantitative relationships among free energy, enthalpi and entropy Describe the special role of ATP in biological energy exchanges

What is as light as a feather, but even the world's strongest man couldn't hold it for more than a few minute?

Breath

BIOENERGETICS AND THERMODYNAMICS Biological energy transductions obey the laws of thermodynamics

Laws of thermodynamics 1. For any physical or chemical change, the total amount of energy in the universe remains constant; energy may be changed from or it may be transported from one region to another, but it can not be created or destroyed

First Law of Thermodynamics – energy cannot be created or destroyed -energy can only be converted from one form to another For example: sunlight energy photosynthesis chemical energy 16

2. The universe always tends toward increasing disorder: in all natural processes the entropy of the universe increases

Second Law of Thermodynamics: disorder is more likely than order entropy: disorder in the universe The 2 nd Law of Thermodynamics states that entropy is always increasing. Energy is required to keep order, to do work - keep cells together and organized - perform life processes 18

The reacting system may be an organism, a cell or two reacting compounds. The reacting system and its surroundings together constitute the universe.

In the laboratory some chemical or physical processes can be carried out in closed systems and no material or energy is exchanged with the surroundings However living organisms are open systems. They exchange both material and energy with their surroundings

Living systems are never at equilibrium with their surroundings Gibbs free energy (G): G expresses the amount of energy capable of doing work during a reaction at constant temperature and pressure.

When a reaction proceeds with the release of free energy , (that is, when the system changes so as to posses less free energy) ΔG has a negative value and the reaction is said to be exergonic

In endergonic reactions, the system gains free energy and ΔG is positive The unit of ΔG is joules/mole or calories/mole

Chemical reactions can create changes in free energy: G = H - T S Free energy = entalphy – (entrophy x temp) When products of chemical reactions contain more free energy than reactants – G is positive. When reactants contain more free energy than products – G is negative. 24

Chemical reactions can be described by the transfer of energy that occurs: endergonic reaction: a reaction requiring an input of energy G is positive exergonic reaction: a reaction that releases free energy G is negative 25

Before Mount Everest was discovered, what was the highest mountain on Earth?

Mount Everest

Enthalpy (H): H is the heat content of the reacting system. H reflects the number and kinds of chemical bounds in the reactants and products. When a chemical reaction releases heat, it is said to be exothermic, the heat content of the products is less than that of the reactants and ΔH has a negative value Reacting systems that take up heat from their surroundings are endothermic and have positive values of ΔH

The unit of ΔH is joules/mole or calories/mole Entropy (S): S is a quantitative expression for the randomness or a disorder in a system The unit of ΔS is joules/mole. Kelvin

Enthalpy: All of the energy contained in a molecule’s chemical bonds Free energy: the energy available to do work, to reduce disorder (enthalpy) -denoted by the symbol G (Gibb’s free energy) entropy: disorder in the universe free energy (G) = enthalpy – (entropy x temp. ) G = H - TS 30

Under the constant temperature and pressure changes in free energy, enthalpy and entropy in biological systems are related to each other by the equation : ΔG= ΔH - TΔS ΔG= Change in Gibbs free energy of the reacting system (Gproducts– Greactives) ΔH= Change in enthalpy of the reacting system (Hproducts – Hreactives) T= Absolute temperature ΔS= Change in entropy of the reacting system (Sproducts – Sreactives)

Living organisms preserve their internal order by taking free energy from their surroundings in the form of nutrients or sunlight and returning to their surroundings an equal amount of energy as heat and entropy

Example: The oxidation of glucose Aerobic organisms extract free energy from glucose obtained from their surroundings by oxidizing the glucose with oxygen (also obtained from surroundings). The end products of this oxidation reaction are CO 2 and H 2 O and they are returned to the surroundings. At the end of this process, the surroundings undergo an increase in entropy, whereas the organism itself remains in a steady state and no change occurs in its internal order
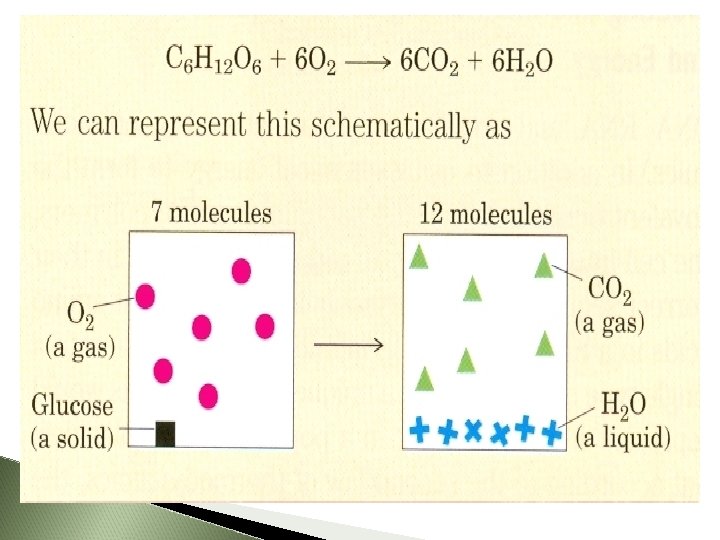


Whenever a chemical reaction results in an increase in the number of molecules the entropy of the surroundings increases. Whenever a solid substance is converted into liquid or gaseous forms the entropy of the surroundings increases. Because, this transformations allow the molecule more freedom for movement

Cells require sources of free energy Living organisms acquire free energy from nutrient molecules. Cells transform this free energy into ATP and other energy-rich compounds. They are capable of providing energy for biological work at constant temparature.

The composition of a reacting system tends to continue changing until equilibrium is reached. At the equilibrium the rates of the forward and revers reactions are equal and no further change occurs in the system.

When a reacting system is not at equilibrium, the tendency to move toward the equilibrium represents a driving force. The magnitude of this driving force is expressed as free energy change (ΔG). Under standard conditions (25 0 C), when reactants and products are initially at the 1 M concentrations the force driving the system toward equilibrium is defined as the standard free energy change (ΔG 0)

ΔG 0 is the difference between the free energy content of the products and the free energy contents of the reactants under standard conditions ΔG 0 = ΔG 0 products– ΔG 0 reactives

When ΔG 0 is negative, the products contain less free energy than the reactants and the reaction will proceed spontaneously under standard conditions When ΔG 0 is positive, the products contain more free energy than the reactants and the reaction will tend to go in the revers direction under standard conditions

Each chemical reaction has a characteristic standard free energy change which may be positive, negative or zero depending on the equilibrium constant of the reaction. ΔG 0 tells us in which direction and how far a given reaction must go to reach equilibrium when the initial concentration of each component is 1 M, the p. H is 7, the temparature is 250 C. Thus ΔG 0 is a constant; a characteristic for a given reaction

Since the two reactions are sequential, we can write the overall reaction as A C ΔG 0 total The ΔG 0 values of sequential reactions are additive. ΔG 0 total = ΔG 01 + ΔG 02

A B ΔG 01 B C ΔG 02 Sum: A C ΔG 01 + ΔG 02 This principle of bioenergetics explains how a thermodynamically unfavorable (endergonic) reaction can be driven in the forward direction by coupling it to a highly exergonic reaction through a common intermediate

The main rule in biochemical reactions in living organisms: All endergonic reactions are coupled to an exergonic reaction. There is an energy cycle in cells that links anabolic and catabolic reactions.

An example: The first step of glycolysis Glucose + Pi Glucose 6 -phosphate+H 2 O ΔG 0 =13, 8 kj/mol ΔG 0 >0 reaction is not spontaneous

Another very exergonic cellular reaction: Hydrolysis of ATP + H 2 O ADP + Pi ΔG 0 = -30, 5 kj/mol ΔG 0 <0 reaction is spontaneous These two reactions share the common intermediates H 2 O and Pi and may be expressed as sequential reactions:

Glucose + Pi Glucose 6 -phosphate+H 2 O ATP + H 2 O ADP + Pi Sum: Glucose+ATP Glucose 6 -phosphate+ADP The overall standard free energy changes: ΔG'0 =13, 8 kj/mol + (-30, 5 kj/mol)= -16, 7 kj/mol Overall reaction is exergonic and spontaneous

Energy stored in ATP is used to drive to synthesis of glycose 6 -phosphate, eventhough its formation from glucose and Pi is endergonic. This strategy works only if compounds such as ATP are continuously available.

FLASHCARDS

the quantitative study of the energy transductions that occur in living cells

Define Bioenergetics

the amount of energy capable of doing work during a reaction at constant temperature and pressure.

What is Gibbs Free Energy “G” ?

How is the Change in Gibbs free energy of the reacting system shown?
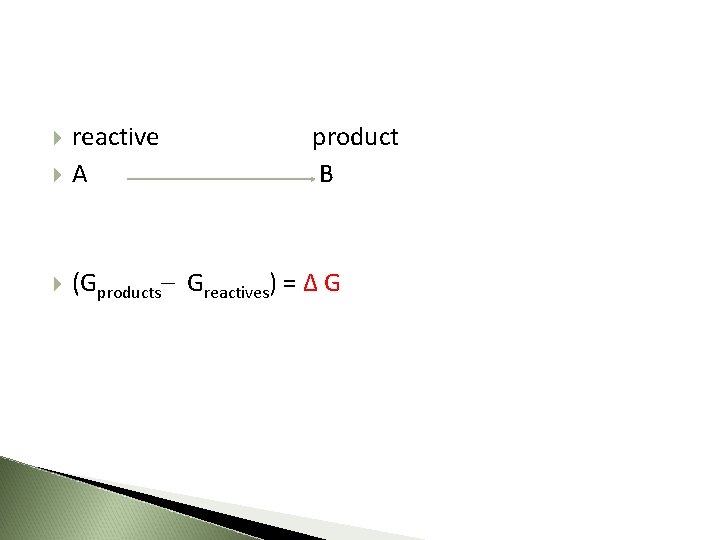
reactive product A B (Gproducts– Greactives) = Δ G

When a reaction proceeds with the release of free energy , (that is, when the system changes so as to posses less free energy) ΔG has a …………………and the reaction is said to be ……………. .

When a reaction proceeds with the release of free energy , (that is, when the system changes so as to posses less free energy) ΔG has a negative value and the reaction is said to be exergonic

ΔG of any reaction proceeding spontaneously toward its equilibrium is ……………, become less negative as the reaction proceeds, and is ……… at the point of equilibrium, indicating that no more work can be done by the reaction.

ΔG of any reaction proceeding spontaneously toward its equilibrium is always negative, become less negative as the reaction proceeds, and is zero at the point of equilibrium, indicating that no more work can be done by the reaction.

In endergonic reactions, the system gains free energy and ΔG is ………………

In endergonic reactions, the system gains free energy and ΔG is positive

The composition of a reacting system tends to continue changing until ……………. . is reached. At the …………………. . the rates of the forward and revers reactions are equal and no further change occurs in the system.

The composition of a reacting system tends to continue changing until equilibrium is reached. At the equilibrium the rates of the forward and revers reactions are equal and no further change occurs in the system.

How is the difference between the free energy content of the products and the free energy contents of the reactants under standard conditions shown ? (standart free energy change)

ΔG 0 is the difference between the free energy content of the products and the free energy contents of the reactants under standard conditions ΔG 0 = ΔG 0 products– ΔG 0 reactives


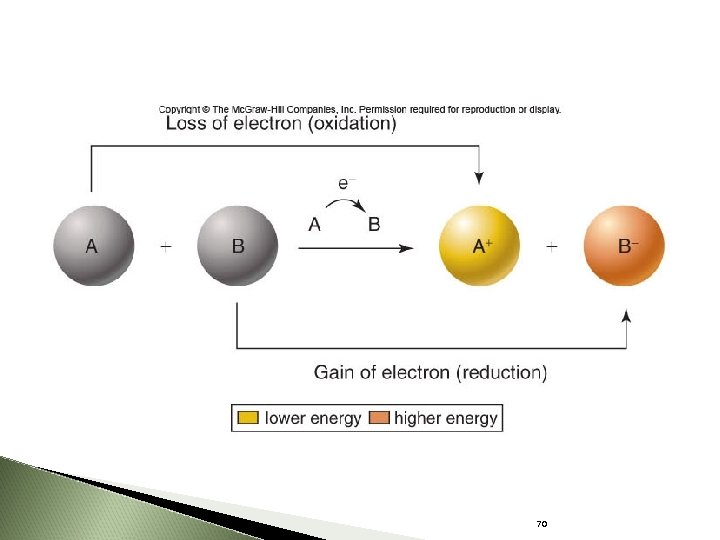
Flow of Energy Potential energy stored in chemical bonds can be transferred from one molecule to another by way of electrons. oxidation: loss of electrons reduction: gain of electrons Redox reactions are coupled to each other. 68

Oxidation-Reduction Reactions A chemical reaction that transfers electrons from one atom to another Oxidation = loss of an electron Reduction = gain of an electron 69
70

Oxidation-Reduction Reactions Oxidation ◦ A chemical reaction in which a molecule gives up electrons ◦ Oxidation releases energy ◦ The molecule loosing the electron is oxidized 79 71

Reduction ◦ A chemical reaction in which a molecule gains electrons and energy ◦ The molecule that accepts electrons is reduced ◦ The molecule being reduced receives energy 80 72
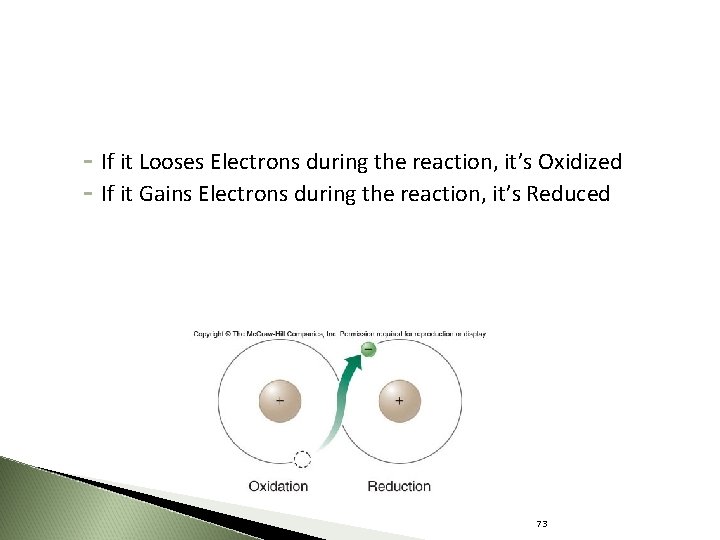
- If it Looses Electrons during the reaction, it’s Oxidized - If it Gains Electrons during the reaction, it’s Reduced 73

Redox Reactions Oxidation and Reduction reactions always occur in pairs ◦ If an atom or molecule is reduced, another atom or molecule must have been oxidized ◦ If an atom or molecule is oxidized, another atom or molecule must have been reduced For this reason Oxidation and Reduction Reactions are known as Redox Reactions 74

ATP

ATP is the molecule that cells use to store, transfer, and provide energy The energy from ATP is used to fuel anabolic reactions ◦ recall: for growth, repair, and reproduction ATP = Adenosine Triphos. Phate ◦ Adenosine (same molecule from DNA and RNA) + ◦ Three inorganic phosphates (functional group PO 4) 76 76

ATP = adenosine triphosphate ATP structure: - ribose, a 5 -carbon sugar - adenine - three phosphates 77
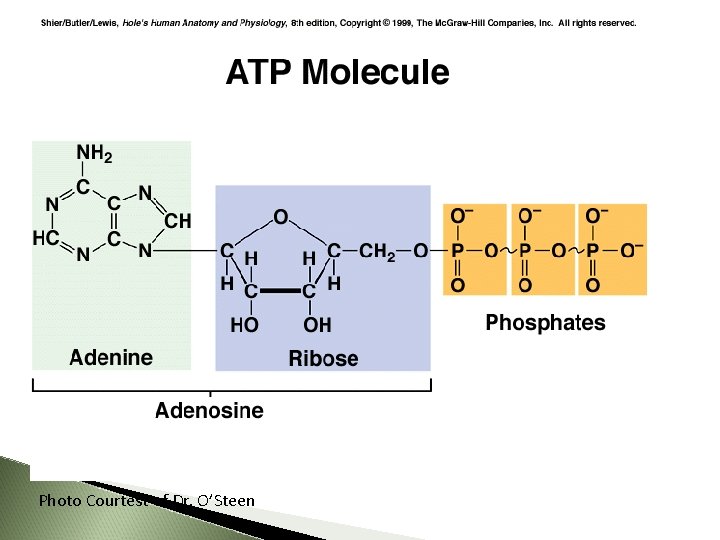
Photo Courtest of Dr. O’Steen

ATP is a molecule that is used as an Energy Currency in cells ◦ ATP’s can be broken down to provide energy for endergonic reactions ◦ Cells use energy to build ATP’s

ATP stores energy in the covalent bonds between phosphates: ◦ Phosphates are highly negative, therefore: the phosphates repel each other much energy is required to keep the phosphates bound to each other Energy is released when the bond between two phosphates is broken 80

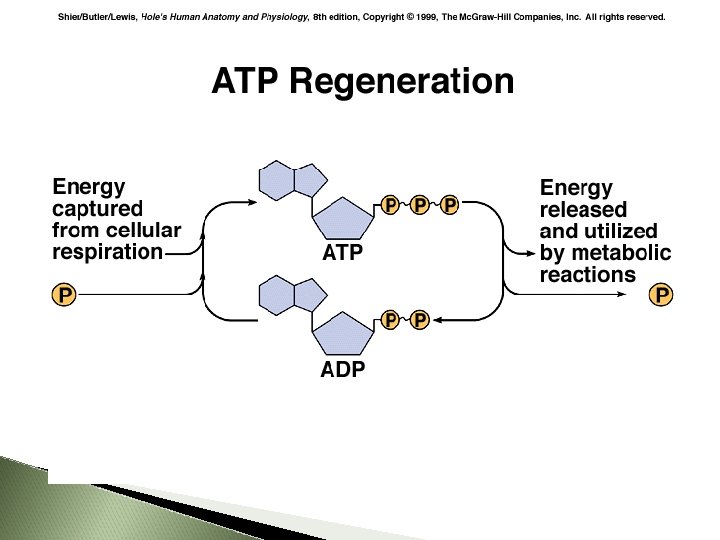
The energy released when ATP is broken down to ADP can be used to fuel endergonic reactions. The energy released from an exergonic reaction can be used to fuel the production of ATP from ADP + Pi. 81
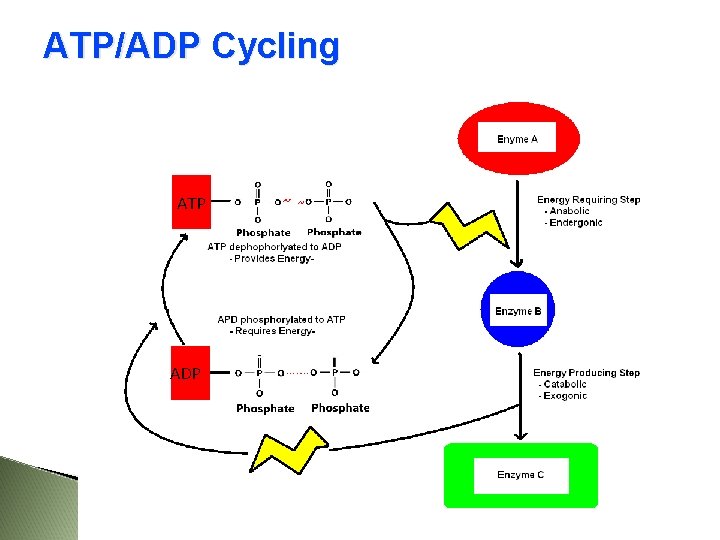
ATP/ADP Cycling ATP ADP

When the bond between phosphates is broken: ATP ADP + Pi energy is released ADP = adenosine diphosphate Pi = inorganic phosphate This reaction is reversible. . . 83
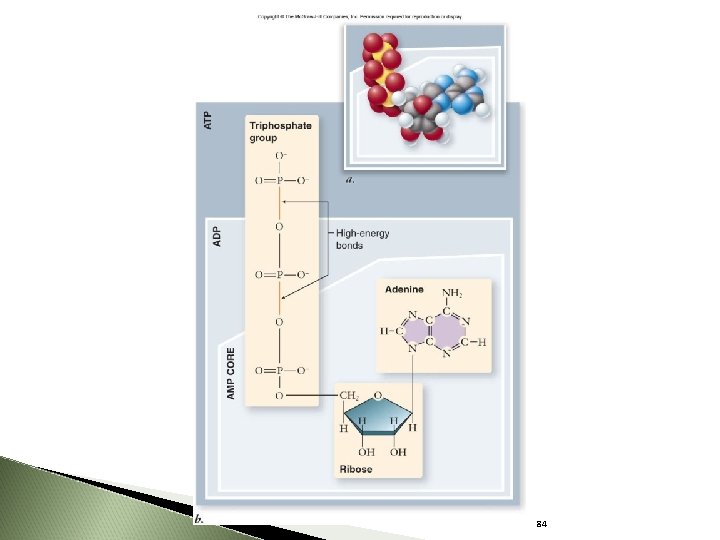
84

ATP - 1 PO 4 = ADP (Adenosine Diphosphate) ADP - 1 PO 4 = AMP (Adenosine Monophosphate) ADP + 1 PO 4 = ATP 85 85

Figure 5_12

Text art 5_06 ATP/ADP Cycling
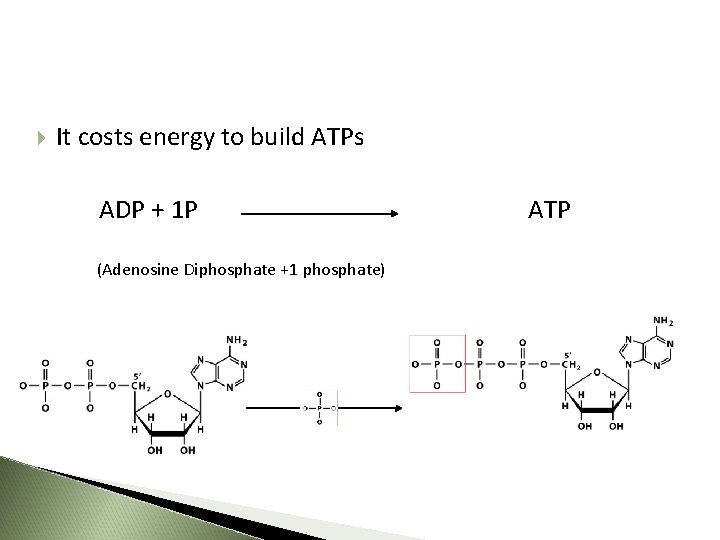
It costs energy to build ATPs ADP + 1 P (Adenosine Diphosphate +1 phosphate) ATP
When the bond between phosphates is formed: ADP + Pi ATP energy is consumed ATP - ADP Cycle 89


92

Other Functions of ATP is used in: l synthesis of new molecules l active transport l muscle contraction l other chemical reactions l regulates enzyme activity 93 93


l Phosphorylation and dephosphorylation - process of adding or removing phosphate groups - can activate or deactivate enzymes l ATP serves as a source of phosphate groups

There are other phosphated molecules providing more energy than ATP these are: Phosphoenolpyruvate 1 3 diphosphoglycerate Phosphocreatinine And less energy than ATP: Glycose -6 - phosphate Glyserole-3 -phosphate AMP

Cellular respiration

Cellular Respiration l Cellular Respiration is a metabolic pathway that breaks down glucose and extracts the energy to produce energy C 6 H 12 O 6 + 6 O 2 6 H 2 O + 6 CO 2 + Energy Glucose Oxygen Water Carbon Dioxide l The Energy is in the form of ATP

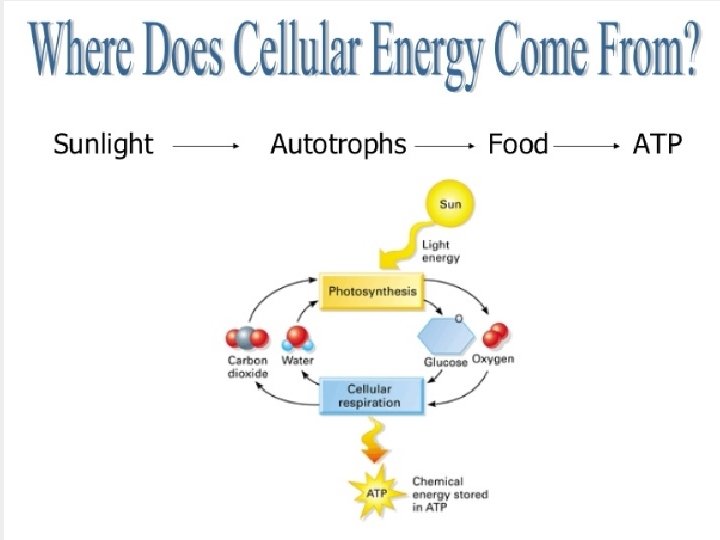


Cellular Respiration C 6 H 12 O 6 + 6 O 2 6 H 2 O + 6 CO 2 + Energy C 6 H 12 O 6 + 6 O 2 + 38 ADP + 38 P 6 H 2 O + 6 CO 2 + 38 ATP

Where do our cells get energy? 10 March 2021

6 -C sugars are the MAJOR source of energy for cell
What type of macromolecule are 6 -C sugars?

◦ Carbohydrates Cells break down glucose a 6 -C sugar to make ATP “energy”

Overall Chemical Process C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + usable energy (ATP) 10 March 2021
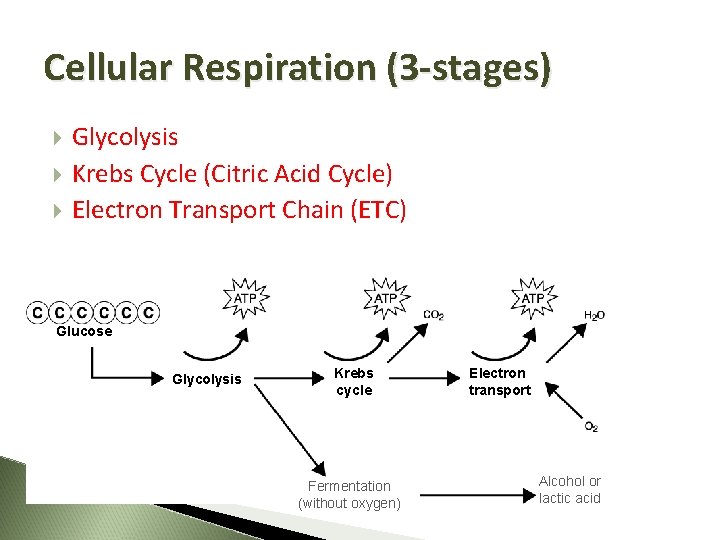
Cellular Respiration (3 -stages) Glycolysis Krebs Cycle (Citric Acid Cycle) Electron Transport Chain (ETC) Glucose Glycolysis Krebs cycle Fermentation (without oxygen) Electron transport Alcohol or lactic acid


Aerobic respiration 10 March 2021

Flowchart Cellular Respiration Glucose (C 6 H 1206) + Oxygen (02) Glycolysis Krebs Cycle Electron Transport Chain Carbon Dioxide (CO 2) + Water (H 2 O) + ATP

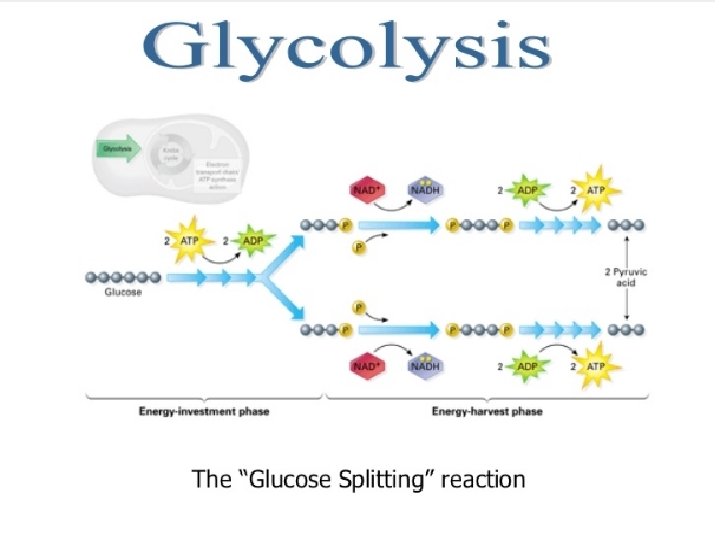
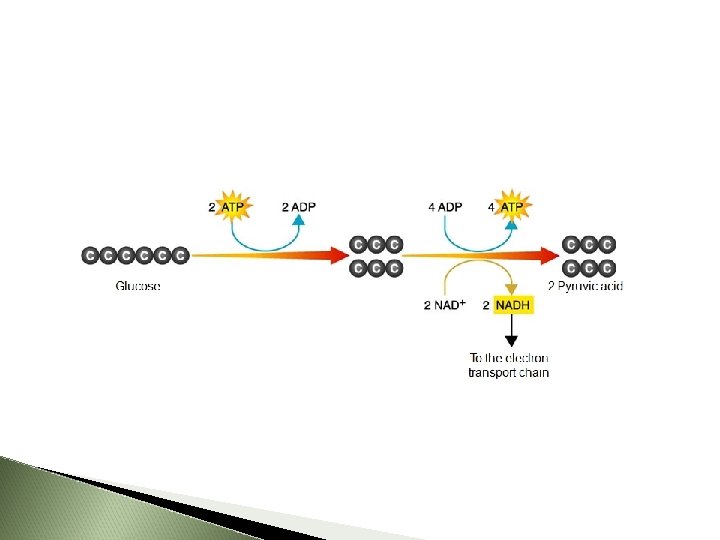
1. Glycolysis




Animation

Where Cytoplasm Energy Yield net gain of 2 ATP at the expense of 2 ATP 6 -C glucose Two 3 -C pyruvates Free e- and H+ combine with organic ion carriers called NAD+ NADH + H+ (NADH 2) (nicotinamide dinucleotide) 10 March 2021

Summary In Glucose (6 -C) 2 ATP Out 2 pyruvate; 2(3 -C) 2 NADH a net of 2 ATP

Questions Where does glycolysis take place? 10 March 2021

Cytoplasm

Glycolysis energy yield?

2 ATP

Breaks glucose into TWO ____.

pyruvate

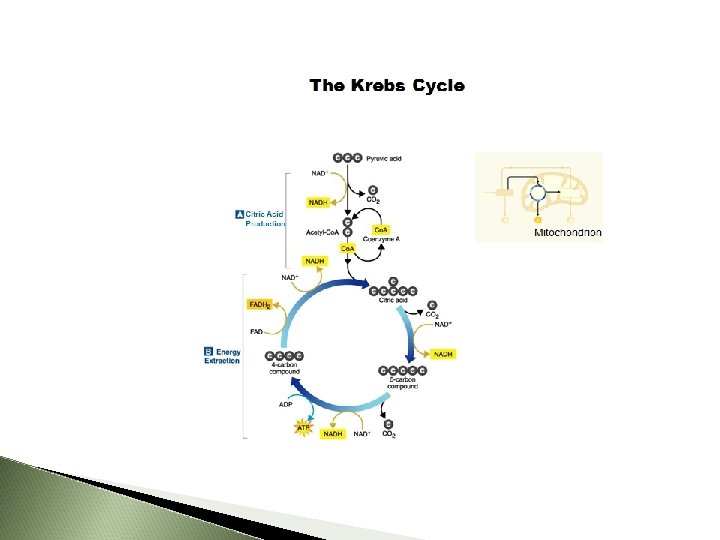
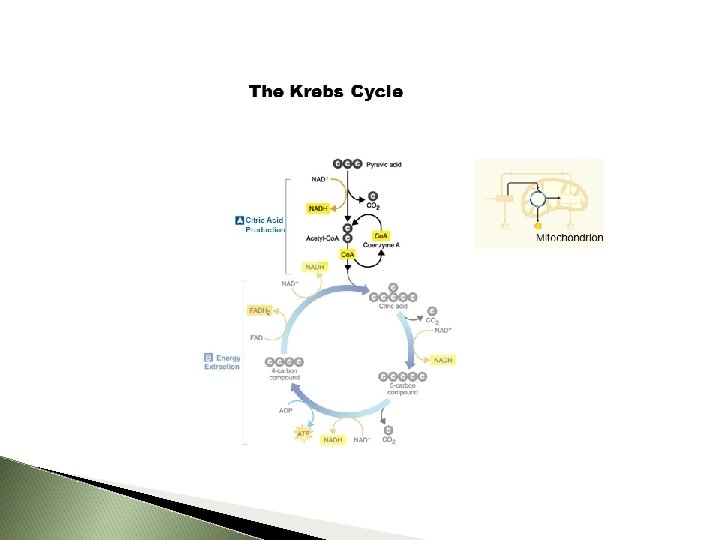
Krebs Cycle 10 March 2021

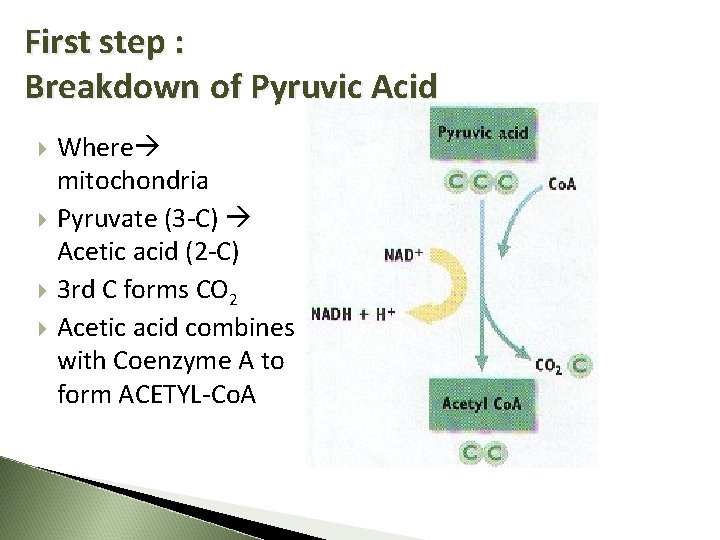
First step : Breakdown of Pyruvic Acid Where mitochondria Pyruvate (3 -C) Acetic acid (2 -C) 3 rd C forms CO 2 Acetic acid combines with Coenzyme A to form ACETYL-Co. A


Summary In Pyruvate NAD Co. A Out CO 2 (as waste) NADH Acetyl-Co. A 10 March 2021

Second Step: Citric Acid Cycle (Krebs Cycle) Where Mitochondrial matrix Energy Yield 2 ATP and more e. Acetyl-Co. A (2 -C) combines with 4 -C to form 6 -C CITRIC ACID Citric Acid (6 -C) changed to 5 -C then to a 4 -C Gives off a CO 2 molecule NAD+ and FAD pick up the released e. FAD becomes FADH 2 NAD+ becomes NADH + H+ Cycle ALWAYS reforming a 4 -C molecule 10 March 2021
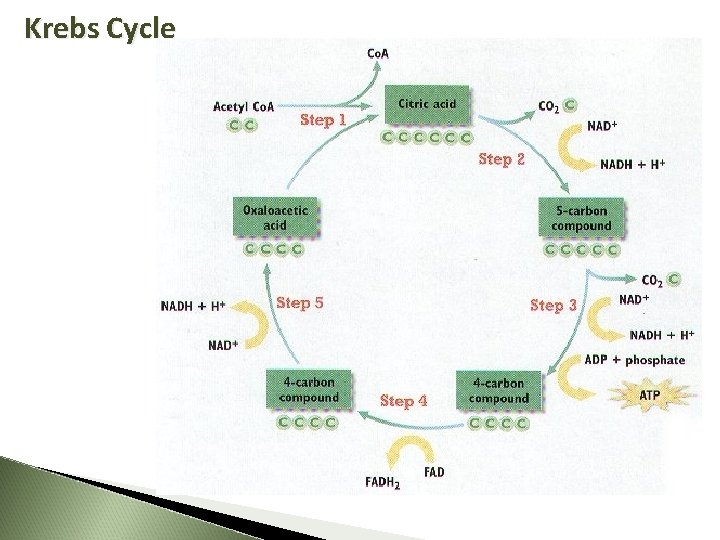
Krebs Cycle

animation



animation
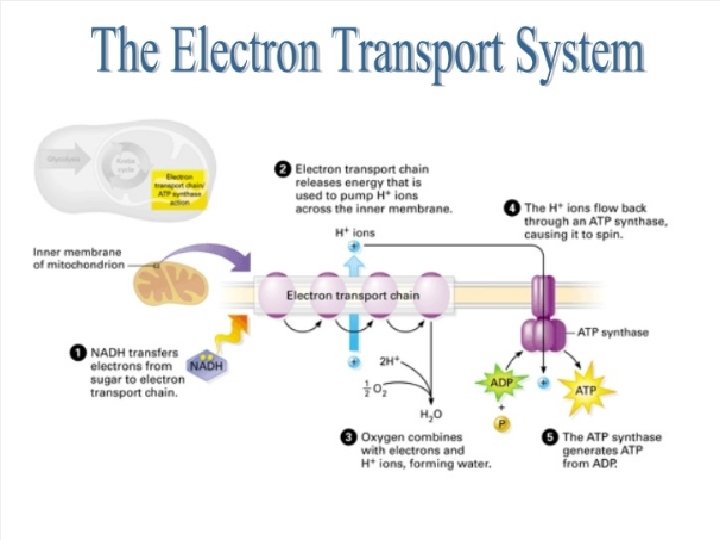
Electron Transport Chain Where inner membrane of mitochondria Energy Yield Total of 32 ATP O 2 combines with TWO H+ to form H 2 O Exhale - CO 2, H 2 O comes from cellular respiration 10 March 2021


ETC Where inner membrane of mitochondria Energy Yield Total of 32 ATP O 2 combines with TWO H+ to form H 2 O Exhale - CO 2, H 2 O comes from cellular respiration


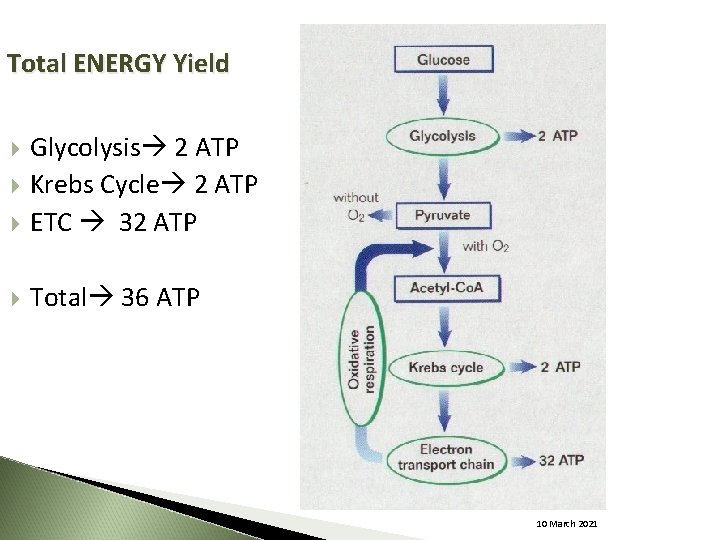
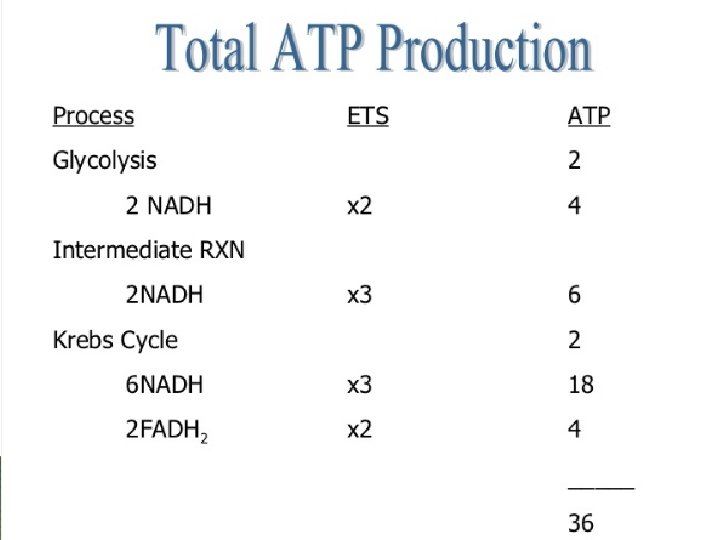
Total ENERGY Yield Glycolysis 2 ATP Krebs Cycle 2 ATP ETC 32 ATP Total 36 ATP 10 March 2021

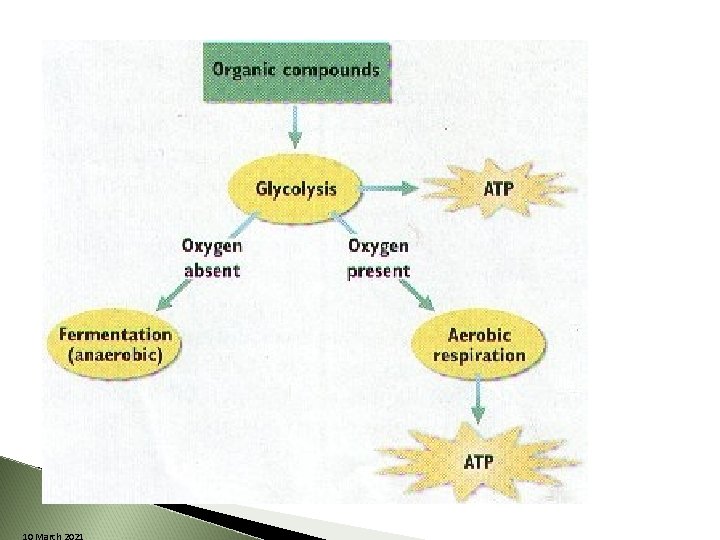
What happens if there is no O 2? 10 March 2021 Cellular respiration process STOPS

Aerobic vs. Anaerobic DOES Aerobic requires NOT require oxygen ◦ Simple ◦ fast ◦ produces smaller amounts of energy (ATP) ◦ Yields large amounts of energy ◦ What is this energy molecule? ATP, 10 March 2021
10 March 2021

Releasing Energy w/out Oxygen Anaerobic Respiration NO Additional ATP is Formed NO O 2 leads to Fermentation ◦ Two Types Lactic Acid Fermentation Alcoholic Fermentation 10 March 2021

Lactic Acid Fermentation bacteria, plants and most animals After glycolysis ◦ 2 pyruvic acid changed to lactic acid Sometimes happens in your muscles, cramps----Exercise 10 March 2021
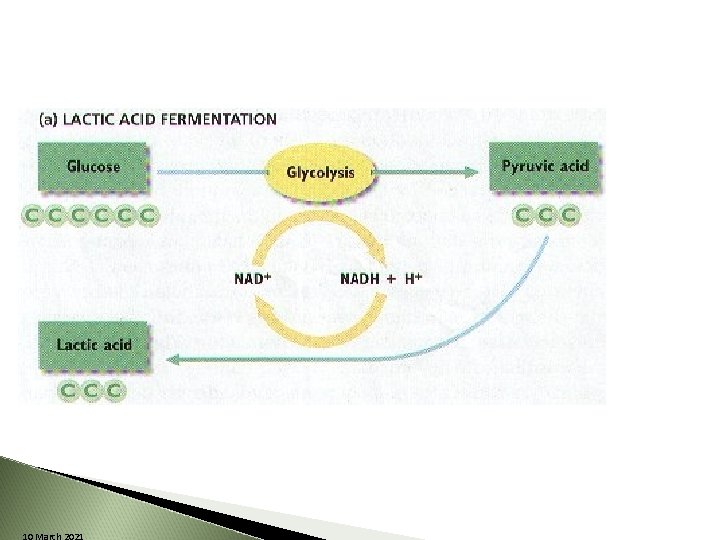
10 March 2021

Alcoholic Fermentation Bacteria and fungi (yeast) Ethyl alcohol and carbon dioxide are the end products Process used to form beer, wine, and other alcoholic beverages Also used to raise dough, bread 10 March 2021

10 March 2021

Cellular Respiration Review Three Main Stages ◦ Glycolysis (2 ATP) ◦ Kreb’s Cycle (2 ATP) ◦ Electron Transport Chain (32 ATP) 10 March 2021





- Slides: 160