Utilizes relationship between chemical potential energy electrical energy

Utilizes relationship between chemical potential energy & electrical energy

Redox Reactions • • battery to start car prevent corrosion cleaning with bleach (oxidizing agent) Na, Al, Cl prepared or purified by redox reactions • breathing • O 2 H 2 O and CO 2

Redox Reactions • Synthesis • Decomposition • Single Replacement • DR rxns NOT redox rxn! Redox rxns

Predicting Redox Reactions • Table J: predict if given redox reaction will occur • metals donate electrons to ion of metals below itself • nonmetal steals electrons from ion of nonmetal below itself

Predicting Single Replacement Redox Reactions Element + Compound New Element + New Compound If element above swapable ion, reaction is spontaneous If element below swapable ion, reaction is not spontaneous

Predicting Redox Reactions A + BX B + AX If metal A above metal B (Table J): reaction is spontaneous X + AY Y + AX If nonmetal X above nonmetal Y (Table J): reaction is spontaneous

Spontaneous or not? • Li + Al. Cl 3 Yes • Cs + Cu. Cl 2 Yes • I 2 + Na. Cl No • Cl 2 + KBr Yes • Fe + Ca. Br 2 No • Mg + Sr(NO 3)2 No • F 2 + Mg. Cl 2 Yes

Started with: 1. Zn(NO 3)2 & Cu 2. Ag. NO 3 & Cu Which beaker had Zn ions & which had Ag ions?

Overview of Electrochemistry • TWO kinds of cells: 1. Galvanic or Voltaic (NYS – Electrochemical) Electrochemical Use spontaneous rxn to produce flow of electrons (electricity) = Exothermic 2. Electrolytic Use flow of electrons (electricity) to force nonspontaneous rxn to occur = Endothermic

Vocabulary • • Redox Half-reaction Oxidation Reduction Cell Half-Cell Electrode • • Anode Cathode Galvanic Voltaic Electrochemical Electrolytic Salt bridge

Electrochemical Cells • spontaneous SR redox rxn: produces flow of electrons • Electrons flow from oxidized substance to reduced substance • Names: Galvanic cells, voltaic cells, or electrochemical cells (NYS)

Electrochemical Cells • Redox rxn arranged so electrons forced to flow through wire • When electrons travel through a wire, can make them do work - light a bulb, ring a buzzer • oxidation & reduction reactions must be separated physically

Half-Cell • Place where each half-reaction takes place – ½ cells: • 2 needed for complete redox rxn • connected by wire so electrons flow through • connected by salt bridge to maintain electrical neutrality
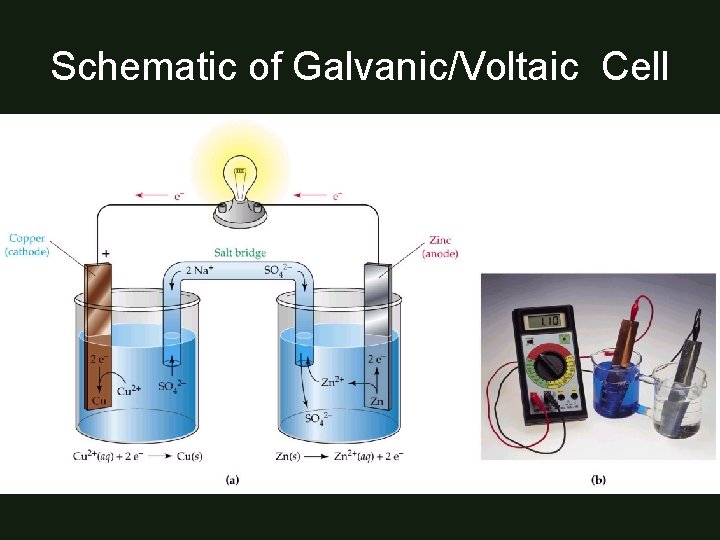
Schematic of Galvanic/Voltaic Cell

Parts of a Galvanic/Voltaic Cell • 2 half-cells: – One for oxidation rxn & – One for reduction rxn – Each consists of: • container with aqueous solution & electrode (surface where electron transfer takes place) – Wire connects electrodes – Salt bridge connects solutions

How much work can you get out of this reaction? • can measure voltage by allowing electrons to travel through voltmeter • galvanic cell is a battery – not easy battery to transport or use in real-life applications

Electrode Surface at which oxidation or reduction half-reaction occurs: Anode & Cathode

An Ox Ate a Red Cat • Anode – Oxidation – the anode = location for the oxidation half-reaction • Reduction – Cathode – the cathode = location for the reduction half-reaction

Anode / Cathode • How know which electrode is which? • Table J: predict which electrode and which electrode is cathode

Anode • Anode = Oxidation = Electron Donor – anode is metal higher on Table J

Cathode • Cathode = Reduction = Electron Acceptor – cathode is metal lower on Table J
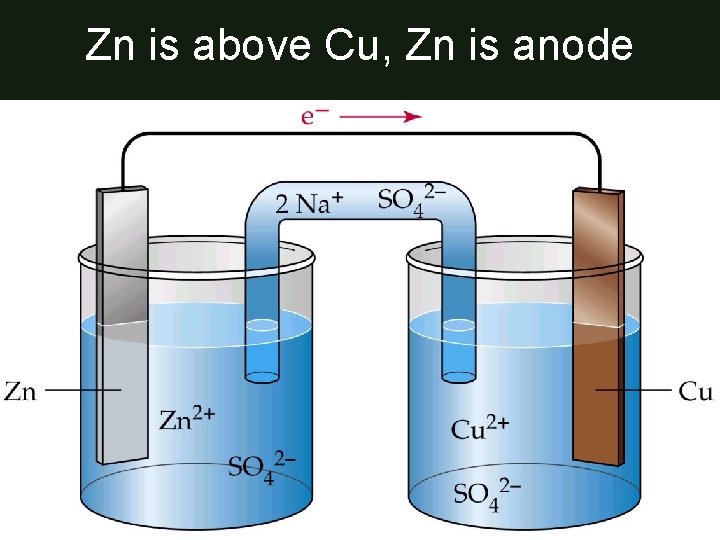
Zn is above Cu, Zn is anode

Direction of Electron Flow (through wire): Anode → Cathode Direction of Positive Ion Flow (salt bridge): Anode → Cathode

Positive & Negative Electrode • Negative electrode (anode): where electrons originate – here it’s Zn electrode • Positive electrode (cathode): electrode that attracts electrons – here it’s the Cu electrode

Aqueous Solution • Solution containing ions of same element as electrode • Cu electrode: – Solution: Cu(NO 3)3 or Cu. SO 4 • Zn electrode: – Solution: Zn(NO 3)2 or Zn. SO 4

Salt Bridge • migration of ions between half-cells • necessary to maintain electrical neutrality • reaction can not proceed without salt bridge

A(s) + BX(aq) B(s) + AX(aq) • SR rxn occurs during operation of galvanic/voltaic cell • One electrode gains mass (B) and one electrode dissolves (A) • concen of metal ions ↑ in one soln (making AX) & ↓ in other soln (using up BX)

Half-Reactions Zn Zn+2 + 2 e. Cu+2 + 2 e- Cu _____________ Zn + Cu+2 Zn+2 + Cu Which electrode is dissolving? Zn Which species is increasing its mass? Zn+2
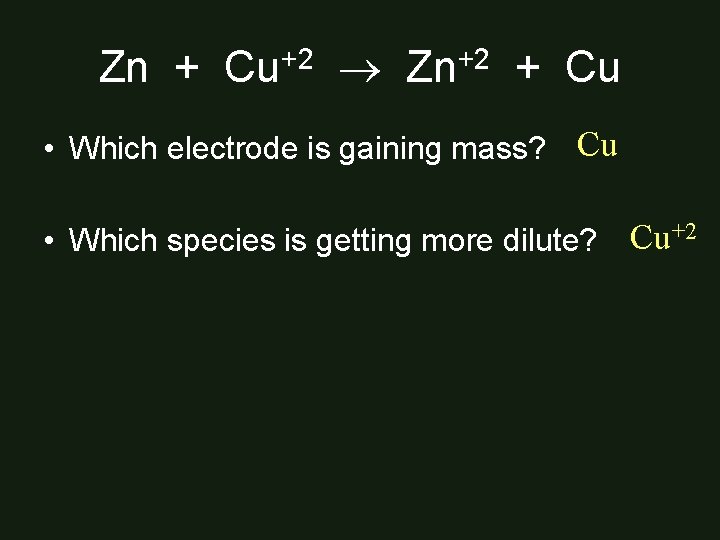
Zn + Cu+2 Zn+2 + Cu • Which electrode is gaining mass? Cu • Which species is getting more dilute? Cu+2

When the reaction reaches equilibrium • voltage is 0! –electrons no longer flow

Construct Galvanic Cell with Al & Pb • Use Table J to identify anode & cathode • Draw Cell: – put in electrodes & solutions • Label: – anode, cathode, direction of electron flow in wire, direction of positive ion flow in salt bridge, positive electrode, negative electrode • Negative electrode: where electrons originate Positive electrode: attracts electrons
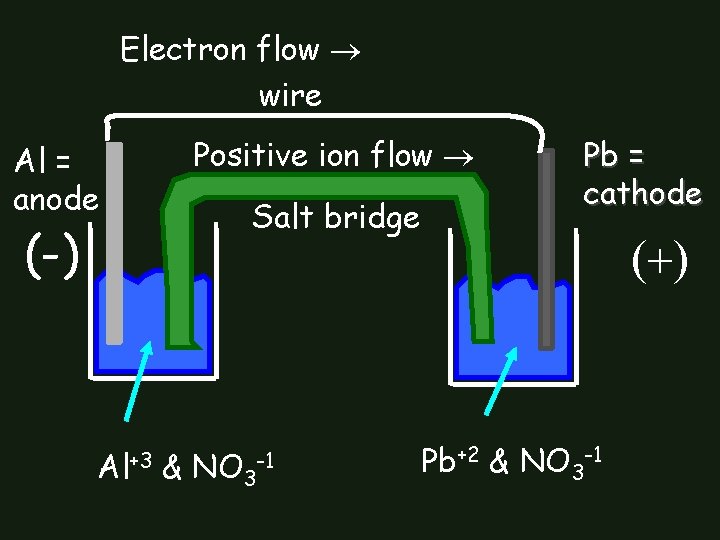
Electron flow wire Al = anode (-) Al+3 Positive ion flow Salt bridge & NO 3 -1 Pb = cathode Pb+2 & NO 3 -1

What are half-reactions? Al Al+3 + 3 e • Al metal is electrode that’s dissolving • Al+3 ions go into solution Pb+2 + 2 e- Pb • Pb+2 ions are in the solution • Ions pick up 2 electrons & plate together on surface of Pb electrode as Pb 0

Overall Rxn 2(Al Al+3 + 3 e-) + 3(Pb+2 + 2 e- Pb) _______________ 2 Al + 3 Pb+2 + 6 e- 2 Al+3 + 3 Pb + 6 e 2 Al + 3 Pb+2 2 Al+3 + 3 Pb

2 Al + 3 Pb+2 2 Al+3 + 3 Pb • • Al Which electrode is losing mass? Which electrode is gaining mass? Pb What’s happening to the [Al+3]? Increasing What’s happening to the [Pb+2]? Decreasing
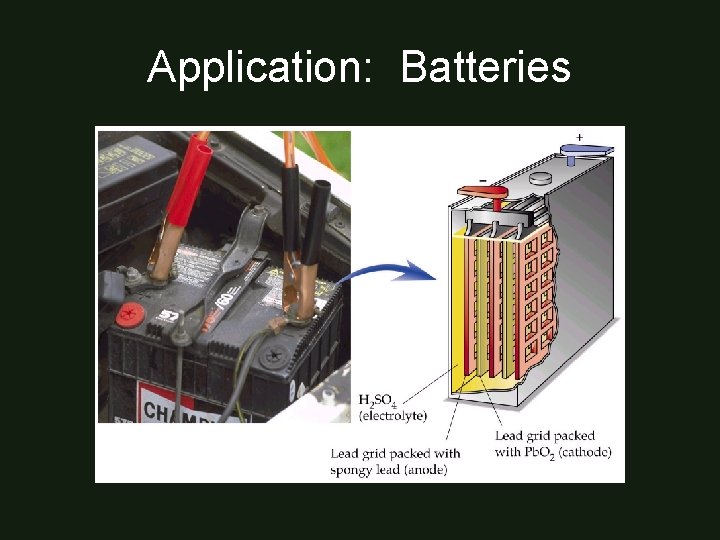
Application: Batteries

Dry Cell

Mercury battery
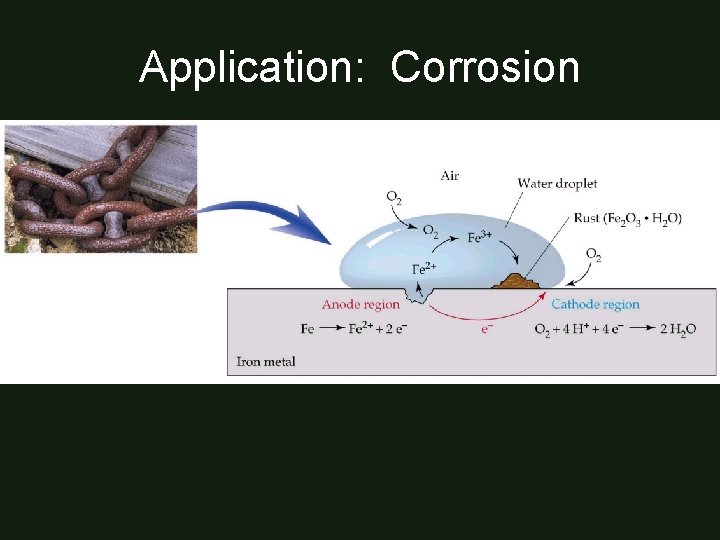
Application: Corrosion
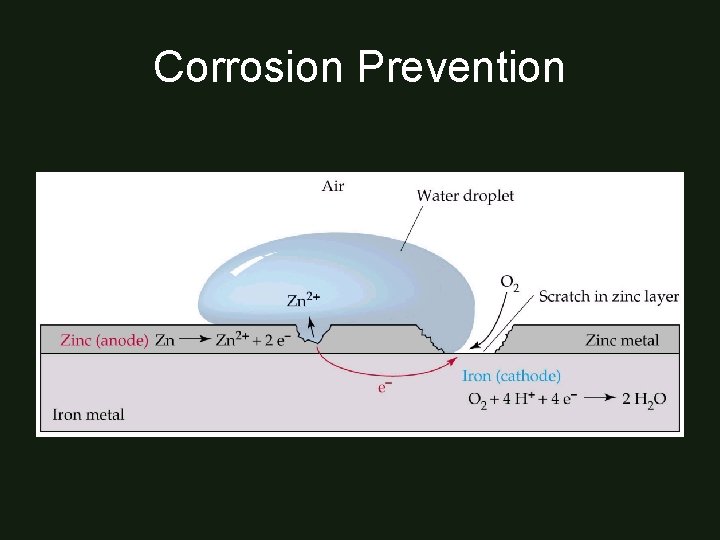
Corrosion Prevention

What’s wrong with this picture?
- Slides: 41