Utah Life Science Summit State of the Industry

























- Slides: 33

Utah Life Science Summit State of the Industry - Growth and Success through Outsourcing/Partnering Regulated Bioanalysis for the Pharmaceutical Industry Scott A. Reuschel, M. S. F. S Tandem Labs – Salt Lake City, UT A Labcorp Company

Who are we? Who or what is Tandem Labs and what do we do?

Company Overview • Our 33 rd year of operation • 1981 Northwest Toxicology established in SLC, UT (clinical toxicology) • 1985 Certified drugs-of-abuse testing lab (NIDA, Do. D, SAMSHA) – GC/MS • 1994 Established bioanalytical division, SLC, UT – GLP bioanalytical, GC/MS, LC/MS • 1998 Established 2 nd site in NJ – Discovery PK/GLP bioanalytical, LC/MS • 2004 Divested drug testing division – Renamed company to Tandem Labs • 2008 Acquired by Laboratory Corporation of America® Holdings • 2009 Established 3 rd site in San Diego, CA – GLP Immunoanalytical • 2010 Acquired BA division of Enthalpy Analytical - RTP, NC (4 th Tandem site) – GLP bioanalytical, LC/MS • 200+ scientific/support staff; ~50 mass spectrometers across all sites • Contract Research Organization (CRO) – Regulated Bioanalysis

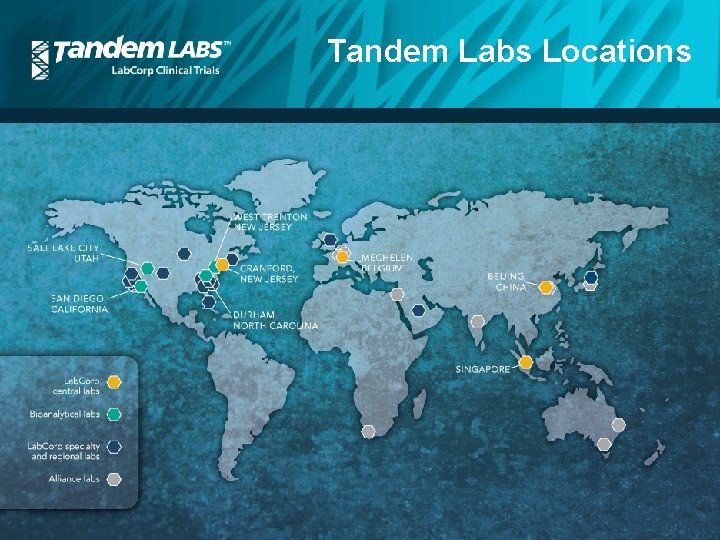
Tandem Labs Locations

Regulated Bioanalysis What regulatory agencies have authority over the work performed at Tandem Labs? • Not regulated by Clinical Laboratory Improvement Amendments (CLIA)

Regulated Bioanalysis What regulatory agencies have authority over the work performed at Tandem Labs? • Not regulated by Clinical Laboratory Improvement Amendments (CLIA) • Are regulated by the Food and Drug Administration (FDA)

Regulated Bioanalysis What regulatory agencies have authority over the work performed at Tandem Labs? • Not regulated by Clinical Laboratory Improvement Amendments (CLIA) • Are regulated by the Food and Drug Administration (FDA) • Are subject to the Code of Federal Regulations (CFR) • 21 CFR Part 58 – Good Laboratory Practice for Non-Clinical Laboratory Studies • 21 CFR Part 11 – Electronic Records, Electronic Signatures

Regulated Bioanalysis What regulatory agencies have authority over the work performed at Tandem Labs? • Not regulated by Clinical Laboratory Improvement Amendments (CLIA) • Are regulated by the Food and Drug Administration (FDA) • Are subject to the Code of Federal Regulations (CFR) • 21 CFR Part 58 – Good Laboratory Practice for Non-Clinical Laboratory Studies • 21 CFR Part 11 – Electronic Records, Electronic Signatures • Also subject to: • ICH (International Conference on Harmonization) - Guideline for Good Clinical Practice (GCP) – informed consent, patient confidentiality, blinding

Regulated Bioanalysis What regulatory agencies have authority over the work performed at Tandem Labs? • Are subject to additional regulations and guidelines from various international regulatory authorities, including:

Regulated Bioanalysis What regulatory agencies have authority over the work performed at Tandem Labs? • Are subject to additional regulations and guidelines from various international regulatory authorities, including: • US FDA – Guidance for Industry: Bioanalytical Method Validation

Regulated Bioanalysis What regulatory agencies have authority over the work performed at Tandem Labs? • Are subject to additional regulations and guidelines from various international regulatory authorities, including: • US FDA – Guidance for Industry: Bioanalytical Method Validation • European Medicines Agency (EMA): Guideline on Bioanalytical Method Validation

Regulated Bioanalysis What regulatory agencies have authority over the work performed at Tandem Labs? • Are subject to additional regulations and guidelines from various international regulatory authorities, including: • US FDA – Guidance for Industry: Bioanalytical Method Validation • European Medicines Agency (EMA): Guideline on Bioanalytical Method Validation • Brazilian Health Surveillance Agency (ANVISA): Bioanalytical Guidance RDC 27/2012

Regulated Bioanalysis What regulatory agencies have authority over the work performed at Tandem Labs? • Are subject to additional regulations and guidelines from various international regulatory authorities, including: • US FDA – Guidance for Industry: Bioanalytical Method Validation • European Medicines Agency (EMA): Guideline on Bioanalytical Method Validation • Brazilian Health Surveillance Agency (ANVISA): Bioanalytical Guidance RDC 27/2012 • Japanese Ministry of Health, Labour and Welfare (MHLW): Draft Guideline on Bioanalytical Method Validation in Pharmaceutical Development

Regulated Bioanalysis What is the purpose of all this regulated bioanalysis?

Pharmacodynamics (PD) and Pharmacokinetics (PK) • Tandem Labs helps pharmaceutical companies obtain the necessary information to make assessments regarding PD/PK of new drugs that are being developed. • Pharmacodynamics (PD) is the study of the biochemical and physiological effects of drugs on the body (i. e. what the drug does to a body). • Pharmacokinetics (PK) describes the drug concentration/time course in body fluids resulting from administration of a certain drug dose (i. e. what the body does to a drug). • Tandem Labs use mass spectrometry to provide both qualitative and quantitative information to our pharmaceutical partners during all phases of the drug development process.

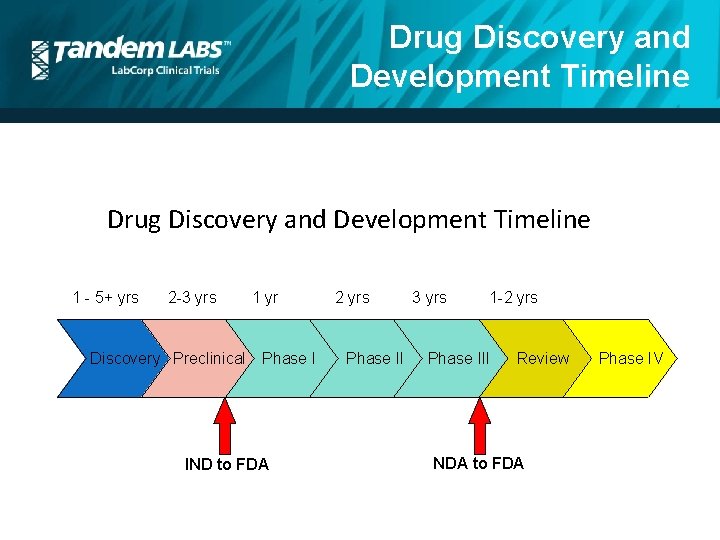
Drug Discovery and Development Timeline 1 - 5+ yrs 2 -3 yrs Discovery Preclinical 1 yr Phase I IND to FDA 2 yrs Phase II 3 yrs 1 -2 yrs Phase III Review NDA to FDA Phase IV

Discovery Drug Development • Qualitative Analysis (by Mass Spectrometry) • In Vitro • High-throughput screening (lead generation, identification, and optimization) • Metabolite Identification (cytochrome P 450 enzymes) • Quantitative Analysis (by Mass Spectrometry) • In Vitro • Solubility, plasma protein binding, permeability, plasma stability, metabolic stability • In Vivo (preliminary animal studies) • Determine basic PK parameters (e. g. half-life, oral bioavailability , clearance and tissue distribution) • Qualified Assays / “Fit for Purpose”; less stringent acceptance criteria, “quick and dirty”

Preclinical Drug Development • Quantitative Analysis (by Mass Spectrometry) • Exploratory Toxicology (non-GLP) • Dose range finding studies • Different species and methods of administration • Multiple dosing regimens • Definitive Toxicology (GLP) – validated assays • General toxicology studies • Different species and methods of administration • Immunogenicity studies with non-human primates (NHPs) • Dose formulation analysis • Safety is the key focus

Clinical Drug Development • Clinical (Phases I-IV) • Quantitative Analysis (by Mass Spectrometry) • Validated Assays – not GLP; however, conducted under the principles of GLP • First in Human • Single Ascending Dose (SAD) • Multiple Ascending Dose (MAD) • Food Effects (fed vs. fasted) • Special Populations • Elderly, renal impaired, hepatic impaired • Drug-Drug Interaction (DDI) • Bioequivalence (BE)

Quantitative Analysis by LC/MS • Method Development • Extraction conditions, chromatography conditions, MS parameters • Very challenging; dictated by the chemistry of the analytes, maximum sensitivity often required, instability, non-specific binding, tight time pressures. • Method Validation • Highly regulated; A/P, stability, robustness, selectivity, matrix effects, etc. • Constantly evolving requirements; additional tests • Sample Analysis • Tight time pressures, sample dilution, sample discrepancies with clinics, ISR

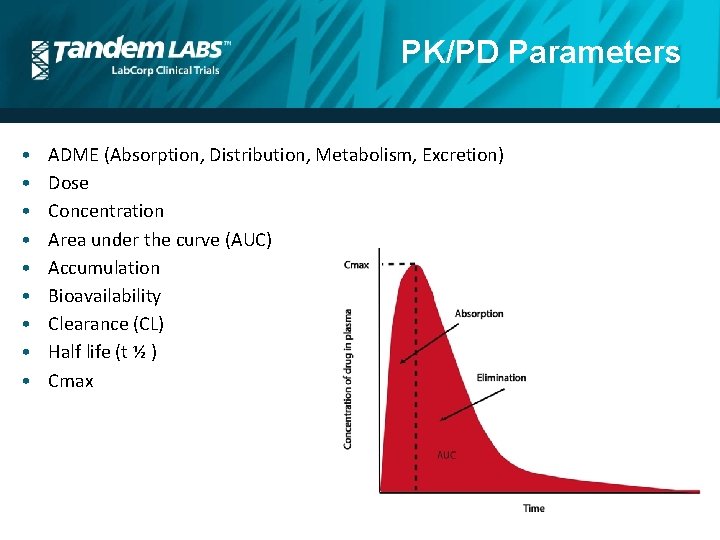
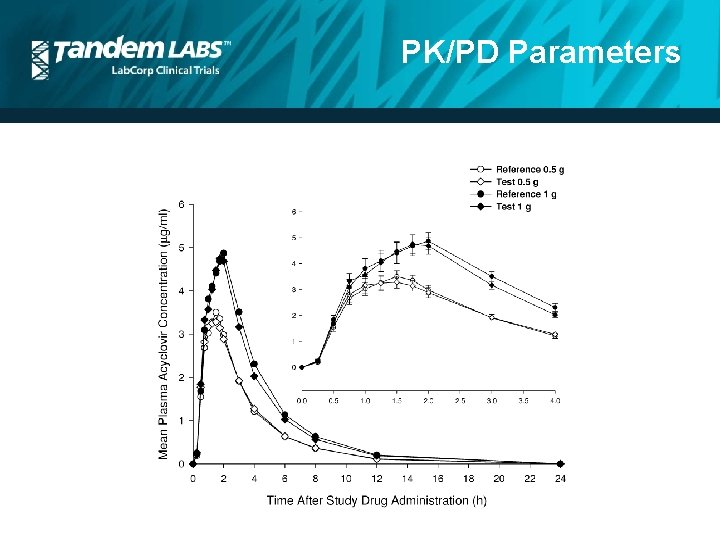
PK/PD Parameters • • • ADME (Absorption, Distribution, Metabolism, Excretion) Dose Concentration Area under the curve (AUC) Accumulation Bioavailability Clearance (CL) Half life (t ½ ) Cmax

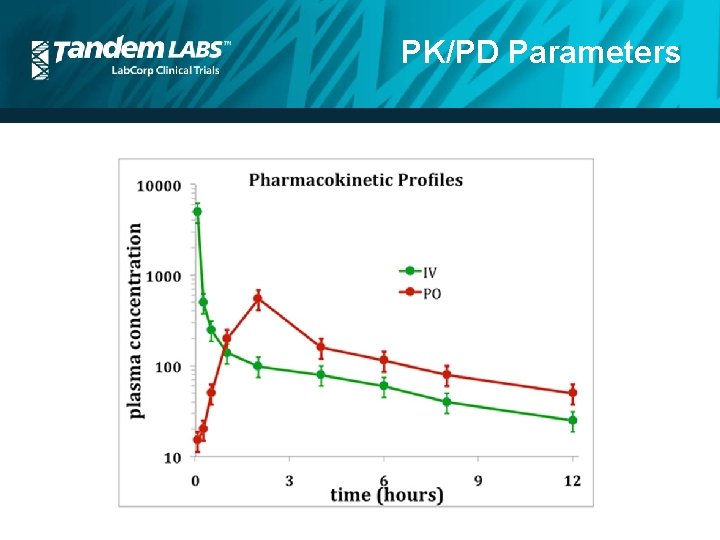
PK/PD Parameters

PK/PD Parameters

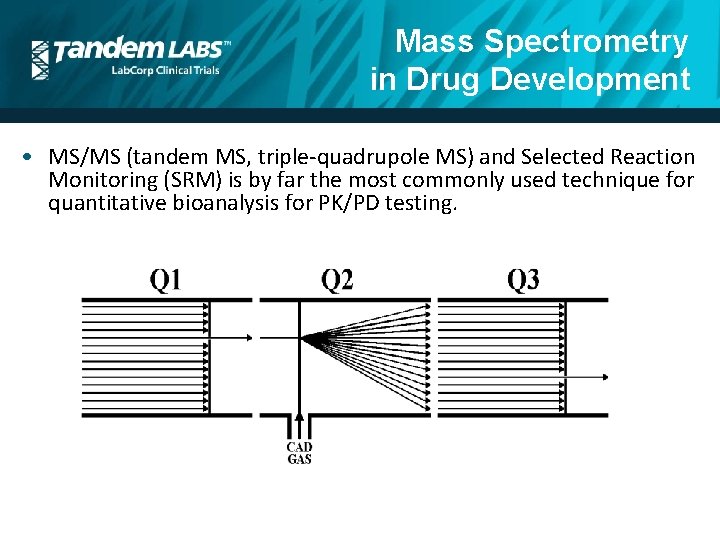
Mass Spectrometry in Drug Development Why is Mass Spectrometry such a good tool for these applications? • Selective • Allows the discrimination of a target analyte to the exclusion of other interferences. • Sensitive • Can routinely detect analytes at ng/m. L, pg/m. L and sub pg/m. L levels. • Compatible with other separation techniques • “hyphenated methods” • GC-MS, LC-MS, UPLC-MS, CE-MS, etc. • Robust / High throughput • Versatile

Mass Spectrometry in Drug Development Why is Mass Spectrometry such a good tool for these applications? • Selectivity • Biological samples are first extracted (PPE, SPE, LLE, SLE) • Chromatographic Separation (GC, HPLC, UPLC, Microflow LC) • Multiple MS Techniques • Full Scan • SIM • MS/MS (SRM, Product Ion, Precursor Ion, Neutral Loss) • TOF/MS • HRAM

Mass Spectrometry in Drug Development • MS/MS (tandem MS, triple-quadrupole MS) and Selected Reaction Monitoring (SRM) is by far the most commonly used technique for quantitative bioanalysis for PK/PD testing.

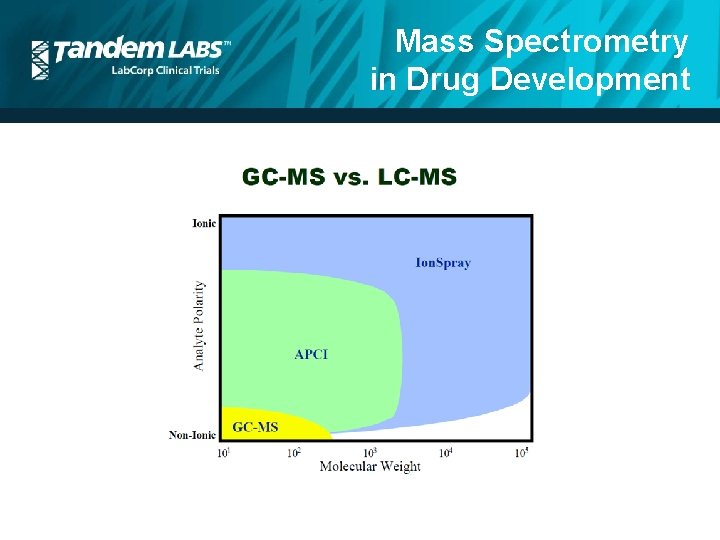
Mass Spectrometry in Drug Development • LC ionization techniques have also revolutionized quantitative MS bioanalysis for PK/PD testing. • Ionization is required for mass spectrometry (charged species; m/z) • GC/MS applications were limited to volatile, thermally stable compounds for ionization. • Atmospheric ionization techniques (ESI, APCI) removed these limitations, allowing for LC/MS applications to expand to a wider variety of compounds.

Mass Spectrometry in Drug Development

Mass Spectrometry in Drug Development • Types of compounds analyzed with LC/MS techniques: • Small Molecules (MW < 800 amu) • Traditional drugs, NCEs • Large Molecules (MW > 800 amu; up to 10 -40, 000 Da) • Biologics • Peptides • Oligonuclieotides • Si. RNA • Lipids • Biomarkers • Proteins

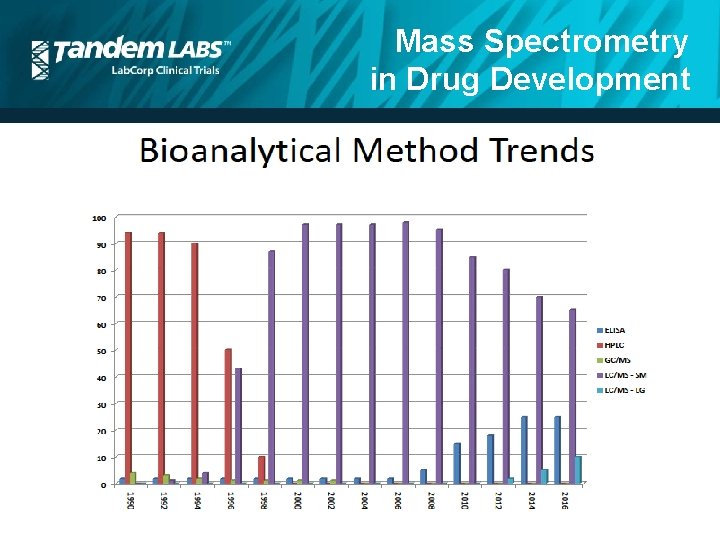
Mass Spectrometry in Drug Development

Mass Spectrometry in Drug Development

Acknowledgements Tandem Labs - Salt Lake City, UT • • Laixin Wang, Ph. D Min Meng, Ph. D Troy Voelker, Ph. D Juan Rogness, MS Life Science Cluster, Utah Governor's Office of Economic Development • Kevin Jessing

Q & A