Using Surveillance Indicators for VaccinePreventable Diseases VPDs National

Using Surveillance Indicators for Vaccine-Preventable Diseases (VPDs): National Notifiable Diseases Surveillance System (NNDSS) 1998 -2007 Sandra W. Roush, MT, MPH National Immunization Conference March 2008

Vaccine-Preventable Diseases Surveillance Indicators Team National Center for Immunization and Respiratory Diseases Centers for Disease Control and Prevention Linda M. Baldy, MPH Albert Barskey, MPH Kristin H. Brown, MPH Kashif Iqbal, MPH Susan B. Redd Sandra W. Roush, MT, MPH

Purpose of Surveillance Indicators Assess national surveillance and data quality for measles, mumps, rubella, pertussis, and Haemophilus influenzae, in terms of: – – surveillance infrastructure timeliness of reporting adequacy of case investigation appropriateness of laboratory testing and diagnostic effort.

National Surveillance Background • Diseases/conditions under national public health surveillance, as determined by the Council of State and Territorial Epidemiologists (CSTE), are included in the National Notifiable Diseases Surveillance System (NNDSS) • NNDSS is a passive system, designed to monitor epidemiologic trends and assess programmatic impact • Cases are reported by health care providers and laboratories to local/state public health officials • NNDSS data are transmitted electronically to CDC through the National Electronic Telecommunications System for Surveillance (NETSS) or the National Electronic Disease Surveillance System (NEDSS) – http: //www. cdc. gov/epo/dphsi/nndsshis. htm – http: //www. cdc. gov/nedss/

Factors Related to Variations in Reporting for VPDs • Disease/condition • Jurisdiction (laws, regulations) • Patient and provider awareness – Symptoms – Incidence • • Clinical severity Transmission setting Availability of laboratory diagnostics Capacity for electronic data transmission

General Methods • NNDSS data, including weekly (provisional) and annual (provisional and final) data, are published in CDC's MMWR • The surveillance data from MMWR (19982006 final, 2007 provisional) were analyzed to assess surveillance indicators for measles, mumps, rubella, pertussis, and Haemophilus influenzae

Measles, Mumps, and Rubella Shared Surveillance Indicators Measles, mumps, and rubella have four indicators in common: • • The proportion of confirmed cases reported to NNDSS with complete information The median interval between symptom onset and notification of a public health authority The proportion of confirmed cases that is laboratory-confirmed The proportion of cases that has an imported source

Measles and Rubella Disease. Specific Surveillance Indicators • Measles: – – • The proportion of cases for which at least one clinical specimen for virus isolation was collected and submitted to CDC The number of discarded measles-like illness (MLI) reports (discontinued 1/1/2006) Rubella: The proportion of confirmed cases among women of child-bearing age with known pregnancy status

Pertussis Surveillance Indicators • • The proportion of cases reported to NNDSS with complete information The mean interval between date of symptom onset and date of public health notification The proportion of cases meeting clinical case definition that is laboratory tested The proportion of cases with complete vaccine history

Haemophilus influenzae Surveillance Indicators • • • The proportion of cases reported to NNDSS with complete information The proportion of cases among children < 5 years of age with complete vaccine history The proportion of cases among children < 5 years of age with serotyped isolate

Assessment of Measles, Mumps, and Rubella Surveillance Indicators • Cases reported with confirmed or unknown case status (measles, rubella) or confirmed, probable, and unknown (mumps) case status were included in analyses • Reporting interval is median number of days from date of onset to date of health department notification • Assessment of completeness of reporting included clinical case definition, hospitalization, lab testing, vaccine history, date reported to health department, transmission setting, outbreak related, epidemiologic linkage, date of birth, and onset date • Missing values were considered invalid • Unknown values were considered valid

Assessment of Pertussis Surveillance Indicators • Cases reported with confirmed, probable, or unknown case status were included in analyses • Assessment of completeness of reporting included clinical case definition, complications, antibiotic treatment, lab testing, vaccine history, and epidemiologic data (outbreak, epidemiologic linkage) • Reporting interval is median number of days from date of onset to date of health department notification • Complete vaccination history included vaccine date and type for all reported doses • Missing and unknown values were considered invalid

Assessment of Haemophilus influenzae Surveillance Indicators • Cases reported with confirmed, probable, or unknown case status were included in analyses • Assessment of completeness of reporting included clinical case definition (species, specimen type), vaccine history, and serotype • Missing values were considered invalid • Unknown values were considered valid

Results: Disease Epidemiology and Surveillance Indicators
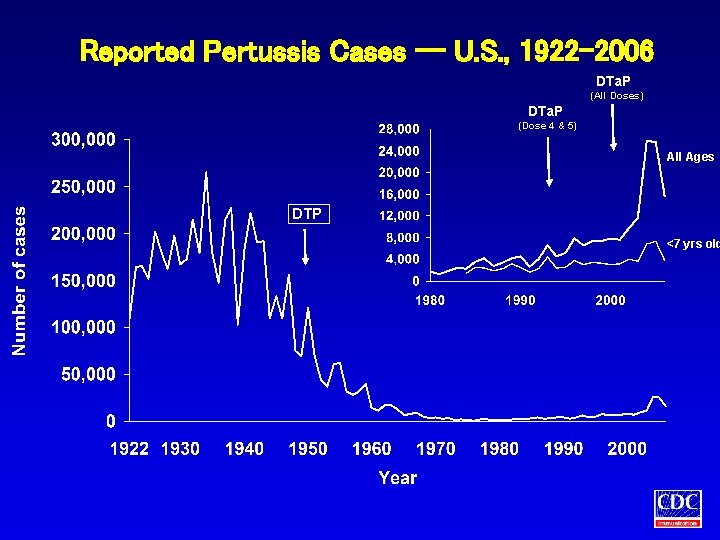
Reported Pertussis Cases -- U. S. , 1922 -2006 DTa. P (All Doses) DTa. P (Dose 4 & 5) All Ages DTP <7 yrs old
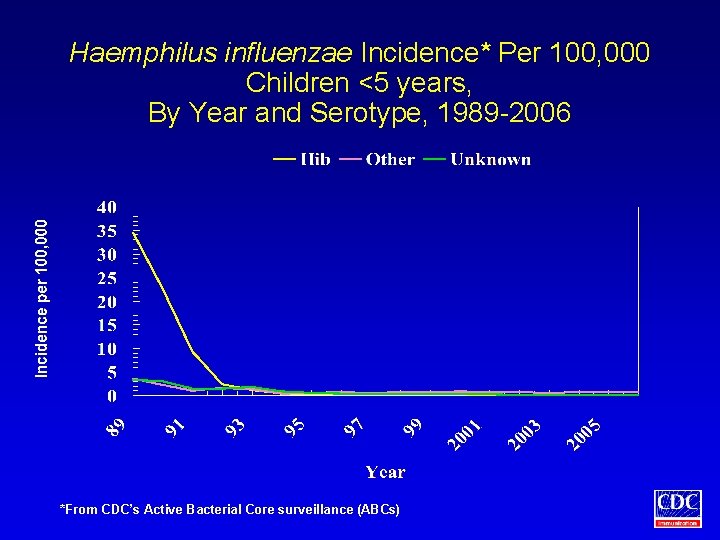
Haemphilus influenzae Incidence* Per 100, 000 Children <5 years, By Year and Serotype, 1989 -2006 *From CDC’s Active Bacterial Core surveillance (ABCs)
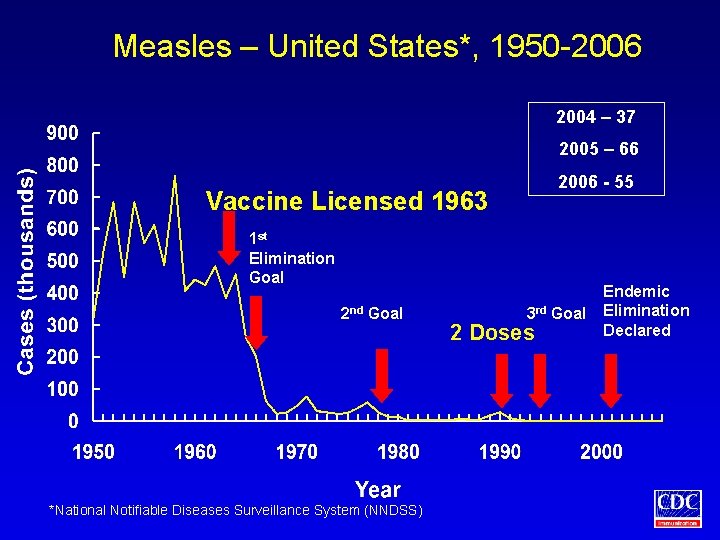
Measles – United States*, 1950 -2006 2004 – 37 2005 – 66 2006 - 55 Vaccine Licensed 1963 1 st Elimination Goal 2 nd Goal *National Notifiable Diseases Surveillance System (NNDSS) 3 rd Goal 2 Doses Endemic Elimination Declared
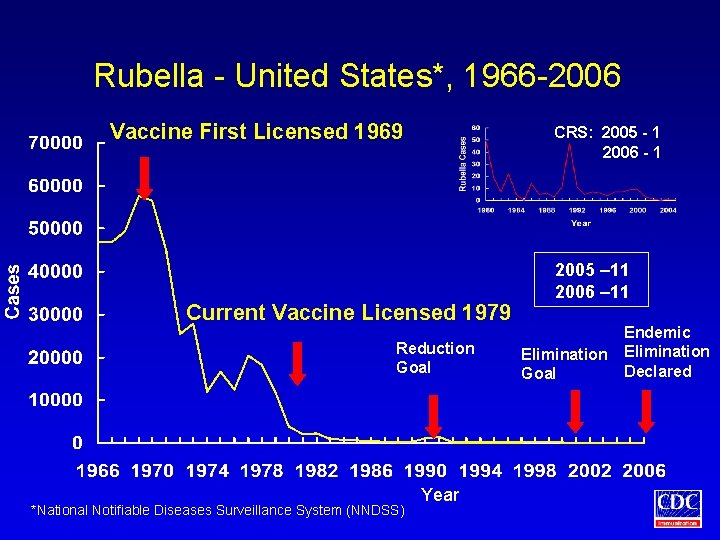
Rubella - United States*, 1966 -2006 Vaccine First Licensed 1969 CRS: 2005 - 1 2006 - 1 Current Vaccine Licensed 1979 Reduction Goal *National Notifiable Diseases Surveillance System (NNDSS) Year 2005 – 11 2006 – 11 Elimination Goal Endemic Elimination Declared
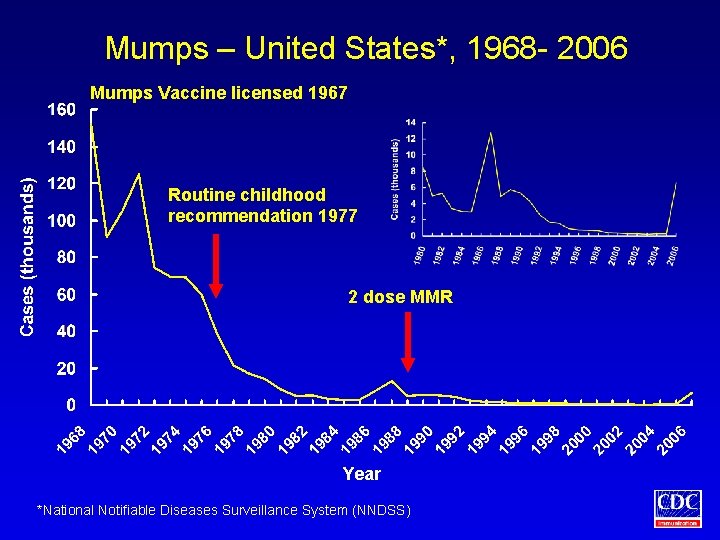
Mumps – United States*, 1968 - 2006 Mumps Vaccine licensed 1967 Routine childhood recommendation 1977 2 dose MMR Year *National Notifiable Diseases Surveillance System (NNDSS)
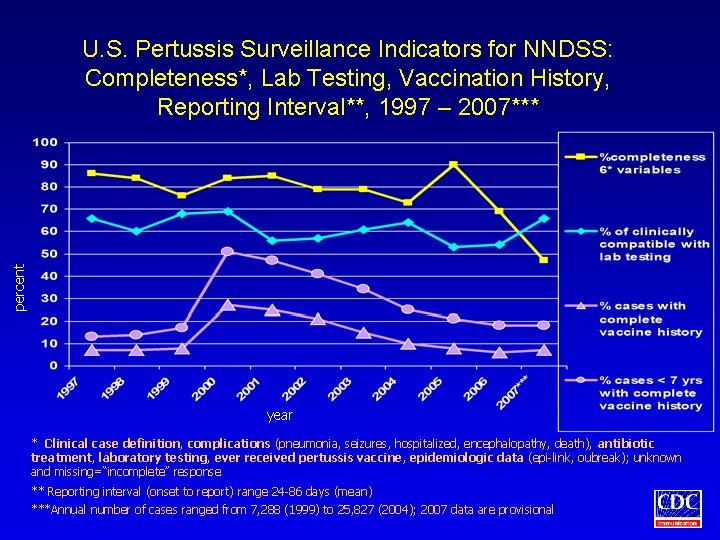
percent U. S. Pertussis Surveillance Indicators for NNDSS: Completeness*, Lab Testing, Vaccination History, Reporting Interval**, 1997 – 2007*** year * Clinical case definition, complications (pneumonia, seizures, hospitalized, encephalopathy, death), antibiotic treatment, laboratory testing, ever received pertussis vaccine, epidemiologic data (epi-link, oubreak); unknown and missing=“incomplete” response ** Reporting interval (onset to report) range 24 -86 days (mean) ***Annual number of cases ranged from 7, 288 (1999) to 25, 827 (2004); 2007 data are provisional
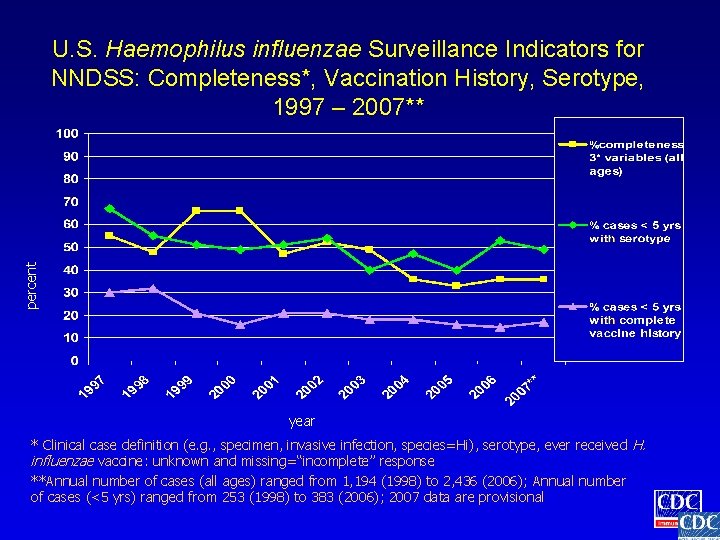
percent U. S. Haemophilus influenzae Surveillance Indicators for NNDSS: Completeness*, Vaccination History, Serotype, 1997 – 2007** year * Clinical case definition (e. g. , specimen, invasive infection, species=Hi), serotype, ever received H. influenzae vaccine: unknown and missing=“incomplete” response **Annual number of cases (all ages) ranged from 1, 194 (1998) to 2, 436 (2006); Annual number of cases (<5 yrs) ranged from 253 (1998) to 383 (2006); 2007 data are provisional
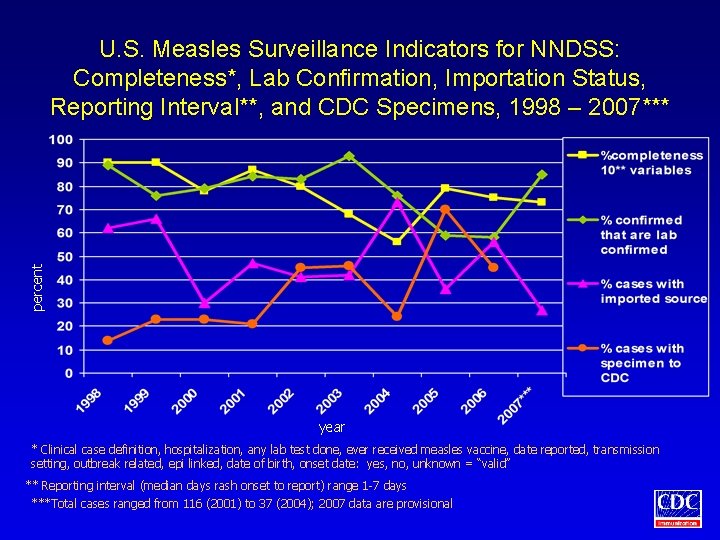
percent U. S. Measles Surveillance Indicators for NNDSS: Completeness*, Lab Confirmation, Importation Status, Reporting Interval**, and CDC Specimens, 1998 – 2007*** year * Clinical case definition, hospitalization, any lab test done, ever received measles vaccine, date reported, transmission setting, outbreak related, epi linked, date of birth, onset date: yes, no, unknown = “valid” ** Reporting interval (median days rash onset to report) range 1 -7 days ***Total cases ranged from 116 (2001) to 37 (2004); 2007 data are provisional
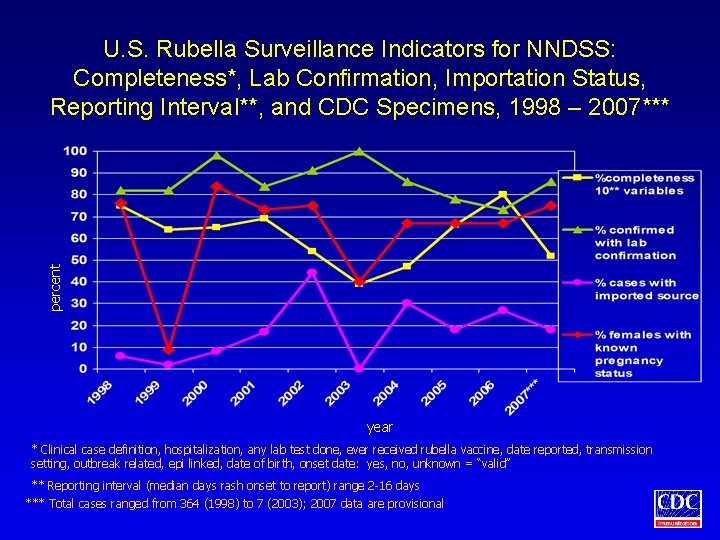
percent U. S. Rubella Surveillance Indicators for NNDSS: Completeness*, Lab Confirmation, Importation Status, Reporting Interval**, and CDC Specimens, 1998 – 2007*** year * Clinical case definition, hospitalization, any lab test done, ever received rubella vaccine, date reported, transmission setting, outbreak related, epi linked, date of birth, onset date: yes, no, unknown = “valid” ** Reporting interval (median days rash onset to report) range 2 -16 days *** Total cases ranged from 364 (1998) to 7 (2003); 2007 data are provisional
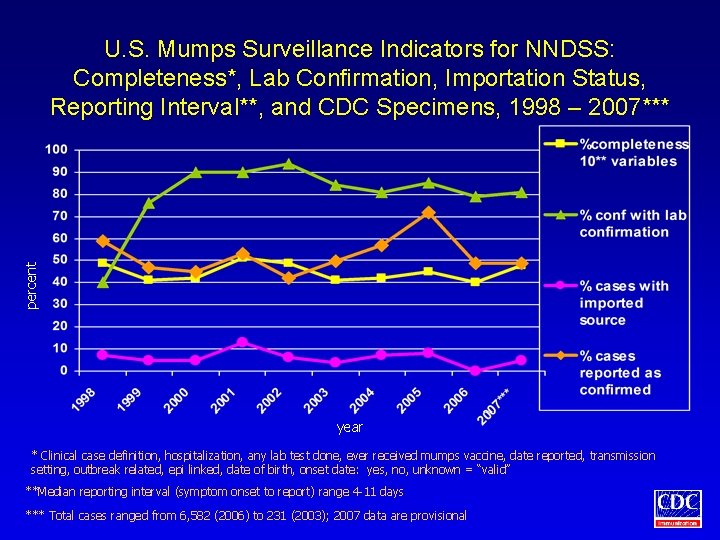
percent U. S. Mumps Surveillance Indicators for NNDSS: Completeness*, Lab Confirmation, Importation Status, Reporting Interval**, and CDC Specimens, 1998 – 2007*** year * Clinical case definition, hospitalization, any lab test done, ever received mumps vaccine, date reported, transmission setting, outbreak related, epi linked, date of birth, onset date: yes, no, unknown = “valid” **Median reporting interval (symptom onset to report) range 4 -11 days *** Total cases ranged from 6, 582 (2006) to 231 (2003); 2007 data are provisional

Conclusions • Surveillance indicators can be monitored using passive surveillance data collected electronically. • For H. influenzae, data completeness is very low, especially the percent of cases <5 years with serotype and with complete vaccine history. • For measles, effort must be maintained to ensure data completeness, determination of importation status, and laboratory testing at CDC. • For pertussis, strategies must be implemented to enhance documentation of (adult and child) case vaccination history, while building laboratory testing infrastructure. • For rubella, effort must be enhanced to achieve data completeness, focusing on pregnancy status for females and importation status for all cases. • For mumps, effort must be enhanced to achieve data completeness, including specimen collection, laboratory testing, clinical description, vaccination history, and epi linkage.

Recommendations • Continue annual assessment of VPD surveillance indicators • Communicate results to partners • Apply results in setting surveillance goals and strategies • Explore application of surveillance indicators to other VPDs

Limitations of Analyses • Phased implementation of data systems (NETSS mid-1990’s, NEDSS/NBS ongoing) • Published data possibly different from electronic data set prior to mid-1990’s • Incomplete data possibly due to data system (transmission errors, coding errors) in addition to investigative effort • Few external standards available to monitor completeness of case reporting
- Slides: 27