Using Skills Based Rubrics in Area of Study

Using Skills Based Rubrics in Area of Study 3 Reflections from the classroom STAV VCE Chemistry Conference 2017 Cristy Herron Adele Hudson cherron@aitkencollege. edu. au ahudson@aitkencollege. edu. au www. mychemhub. com Aitken College 2017

AOS 3 Research Task - AOS 3 Investigation Task Assessment Rubrics based on the key science skills Connect learning through all year levels and across Sciences Aitken College 2017
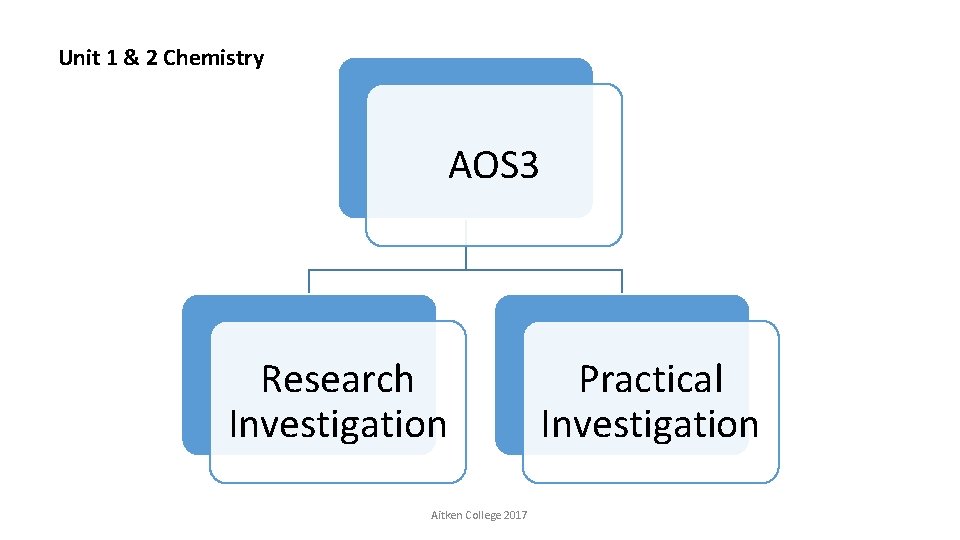
Unit 1 & 2 Chemistry AOS 3 Research Investigation Aitken College 2017 Practical Investigation

Aitken College 2017
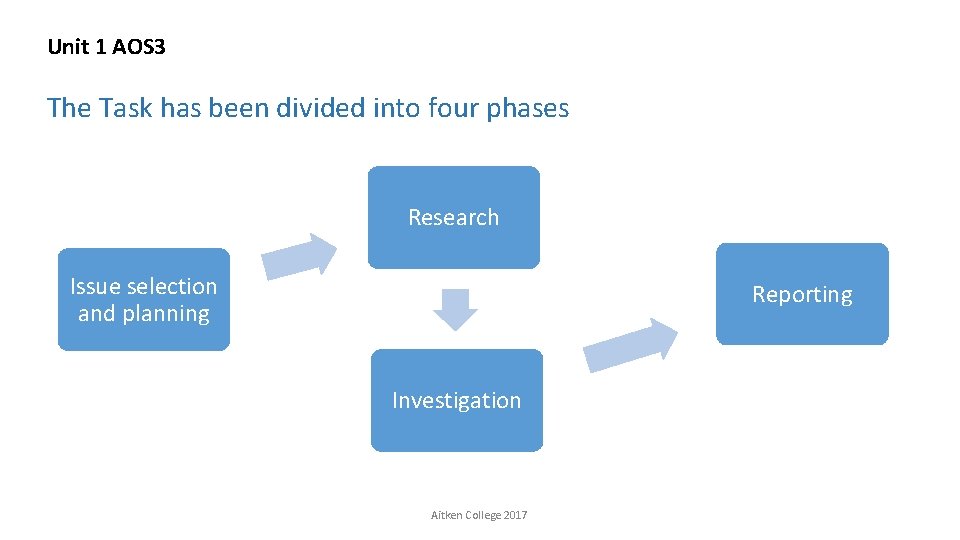
Unit 1 AOS 3 The Task has been divided into four phases Research Issue selection and planning Reporting Investigation Aitken College 2017

Unit 1 AOS 3 Aitken College 2017

Unit 1 AOS 3 Aitken College 2017
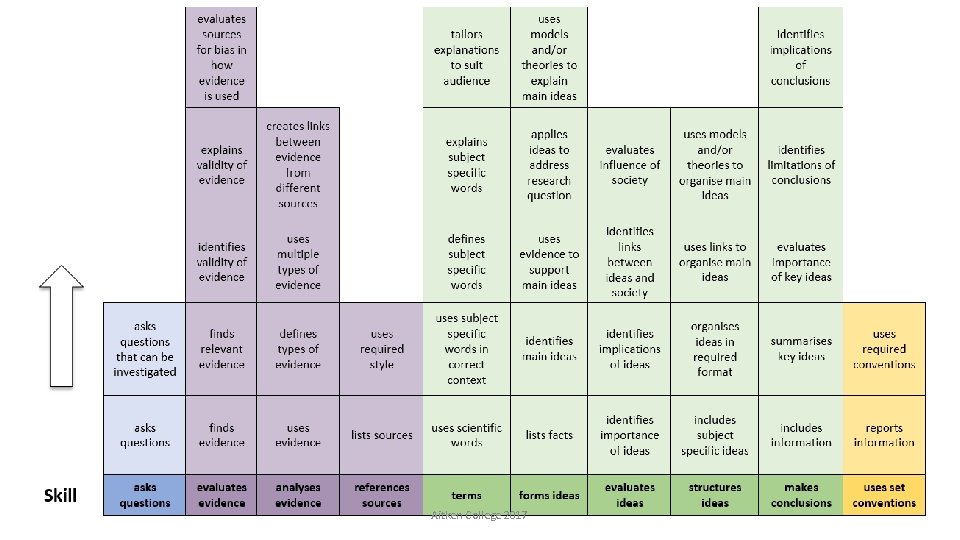
Aitken College 2017
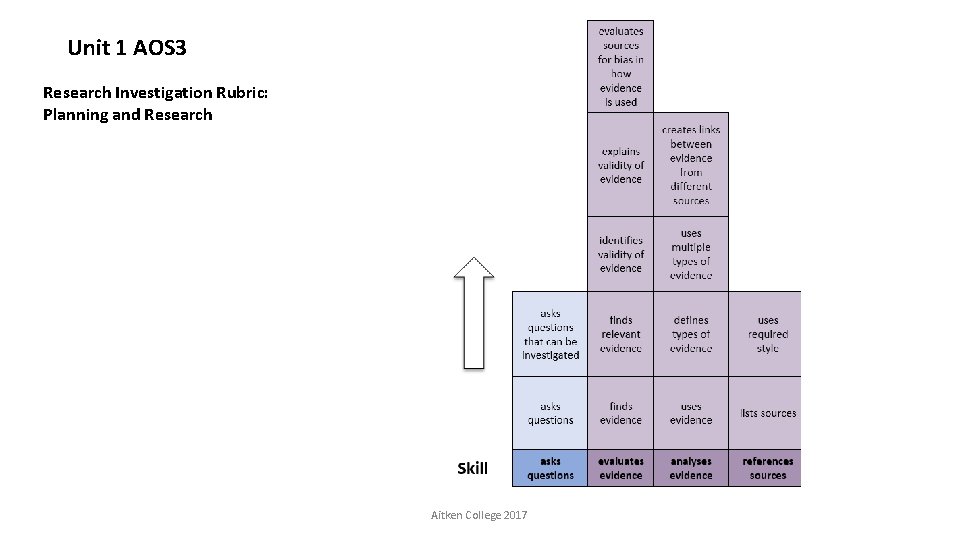
Unit 1 AOS 3 Research Investigation Rubric: Planning and Research Aitken College 2017
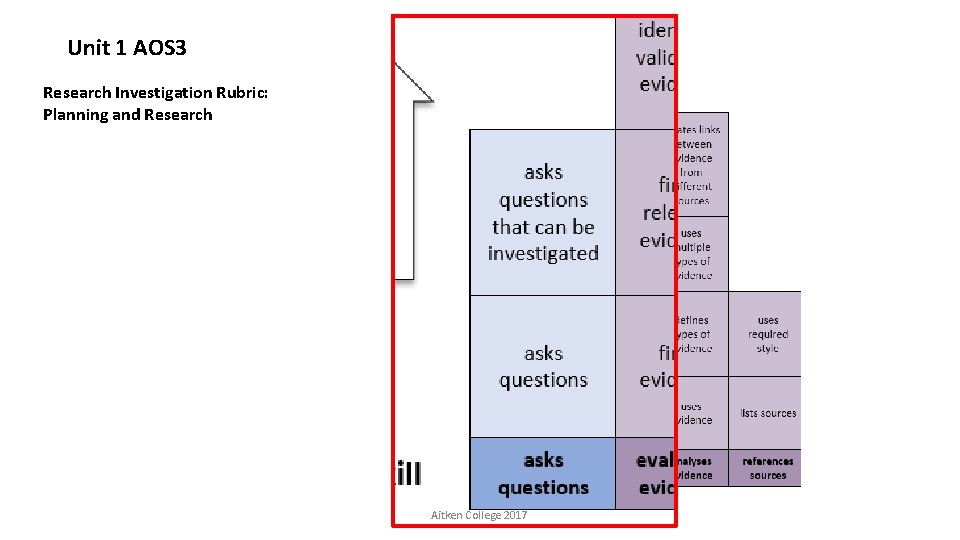
Unit 1 AOS 3 Research Investigation Rubric: Planning and Research Aitken College 2017
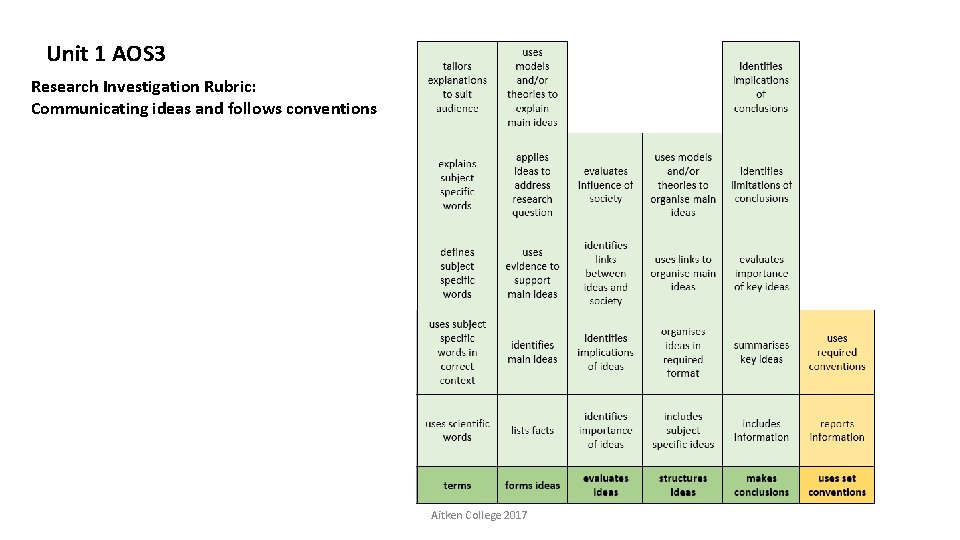
Unit 1 AOS 3 Research Investigation Rubric: Communicating ideas and follows conventions Aitken College 2017
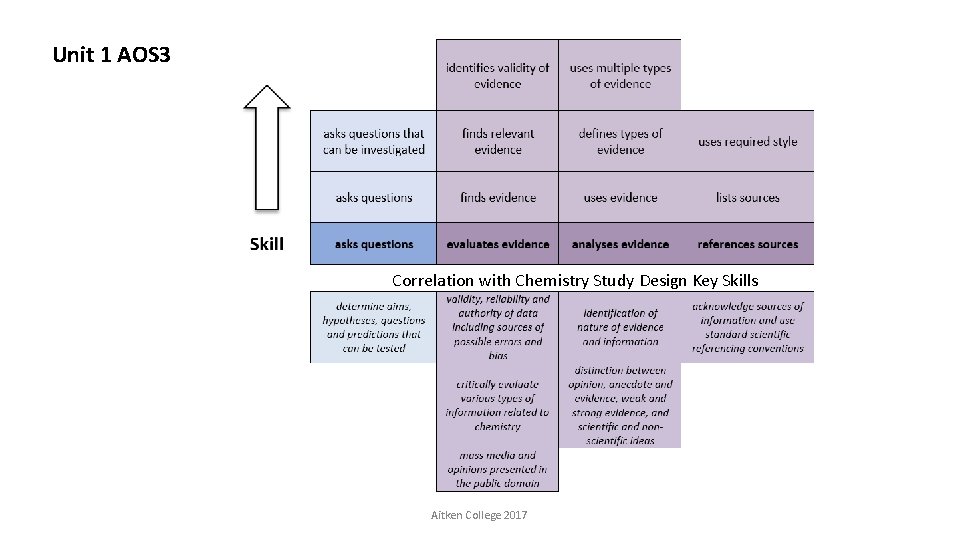
Unit 1 AOS 3 Correlation with Chemistry Study Design Key Skills Aitken College 2017
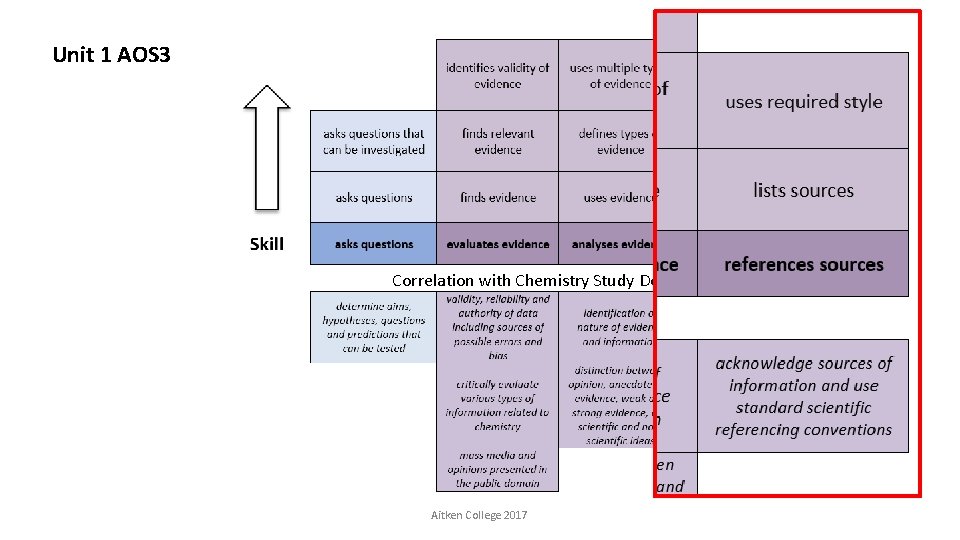
Unit 1 AOS 3 Correlation with Chemistry Study Design Key Skills Aitken College 2017
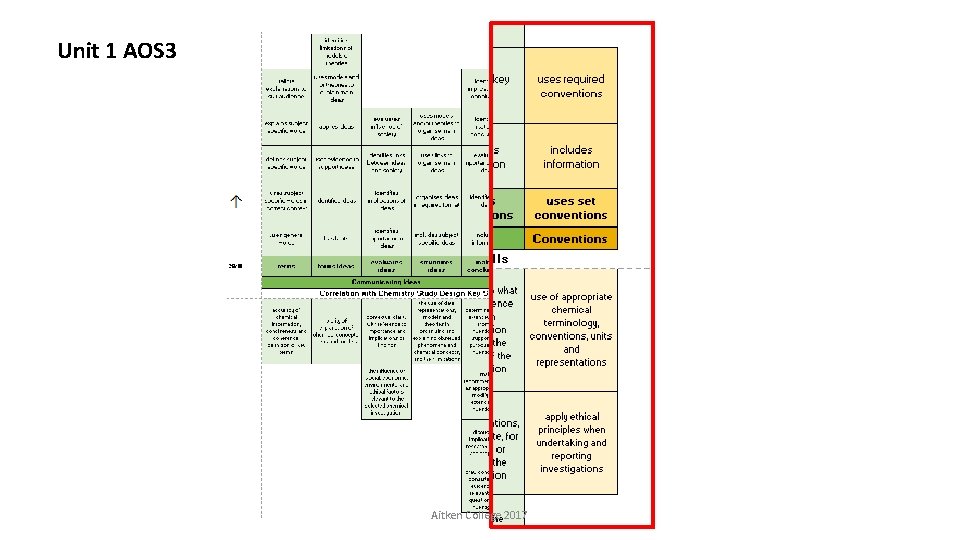
Unit 1 AOS 3 Aitken College 2017

Aitken College 2017

Unit 2 AOS 3 Aitken College 2017

Unit 2 AOS 3 Aitken College 2017

Unit 2 AOS 3 Aitken College 2017
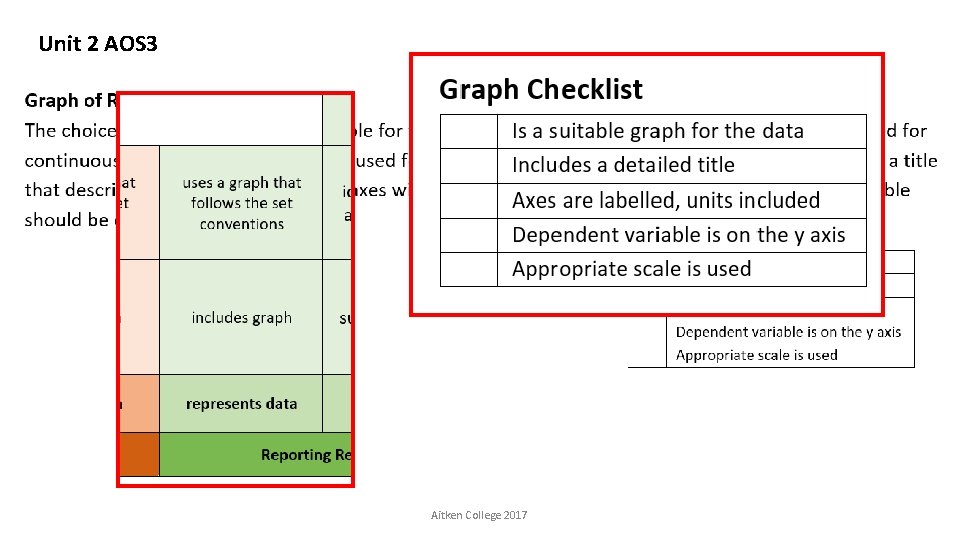
Unit 2 AOS 3 Aitken College 2017
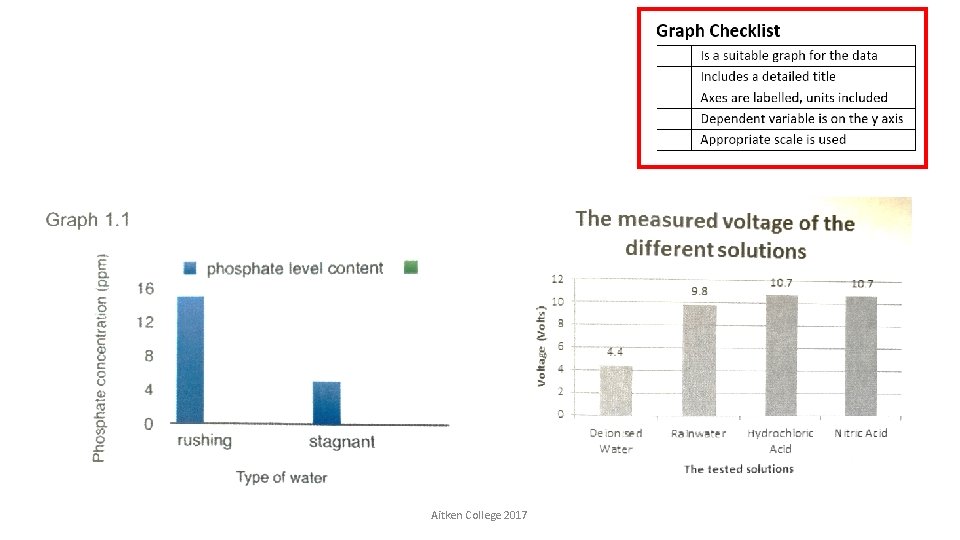
Aitken College 2017

Unit 2 Chemistry Practical Investigation Rubric: Planning, conducting and reporting results Aitken College 2017
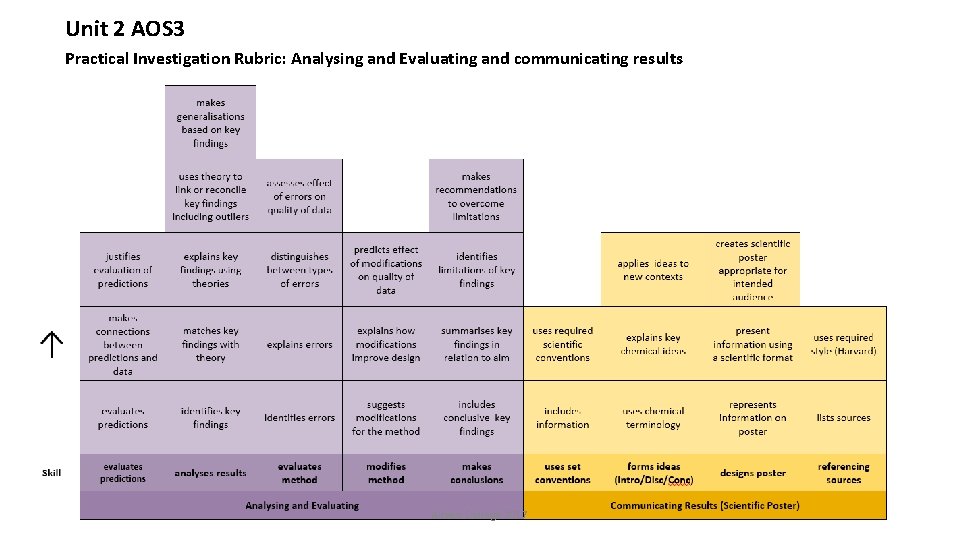
Unit 2 AOS 3 Practical Investigation Rubric: Analysing and Evaluating and communicating results Aitken College 2017

Unit 2 AOS 3 Aitken College 2017
Aitken College 2017

Aitken College 2017

Aitken College 2017

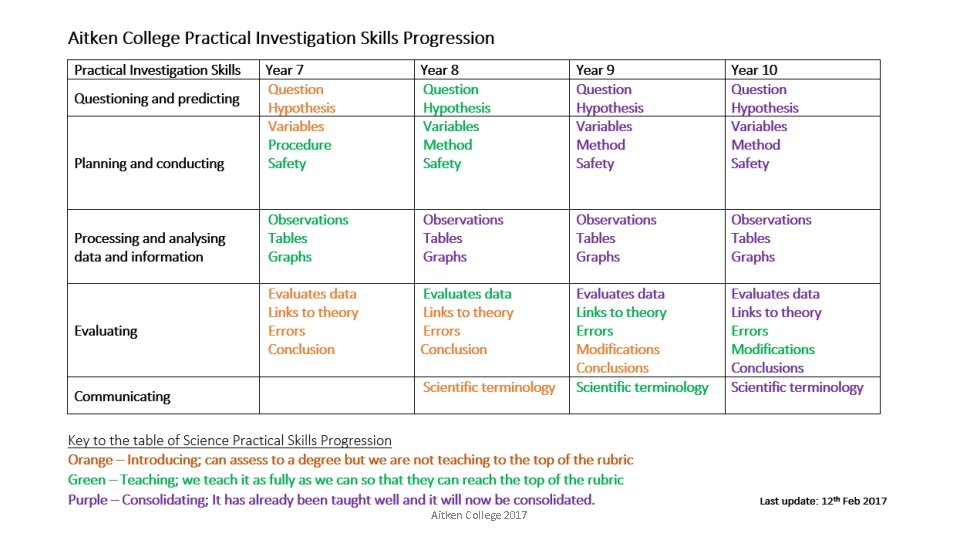
Year 7 Student Design Forces Investigation Aitken College 2017

Year 7 Student Design Forces Investigation Aitken College 2017

Year 7 Student Design Forces Investigation Aitken College 2017

Year 7 Student Design Forces Investigation Aitken College 2017
Aitken College 2017
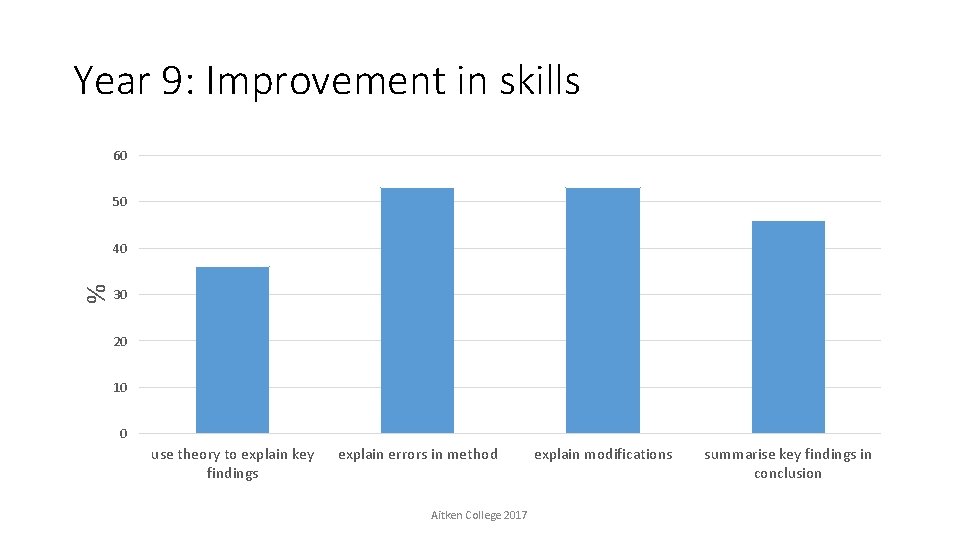
Year 9: Improvement in skills 60 50 % 40 30 20 10 0 use theory to explain key findings explain errors in method Aitken College 2017 explain modifications summarise key findings in conclusion

Year 12 - Determination of the enthalpy changes in chemical reactions by use of calorimetry This investigation involved the calibration of calorimeter and the use calorimetry to measure the enthalpy changes in a number of chemical reactions. In part A the students determined the calibration factor for the calorimeter using two different techniques with the first being electrical calibration and the second being chemical calibration. In Part B the students determined the enthalpy change in an exothermic reaction, dissolution of calcium chloride while in Part C, the students determined the enthalpy change in an endothermic reaction, the dissolution of potassium nitrate. Student 1 Student 2 In Part A the temperature changes were 4. 8 o. C for trial 1 and 4. 6 o. C for trial 2. The temperature changed in a linear manner as the system as electrical energy is being transformed into thermal energy. Technique 2 also showed similar changed in temperature 4. 9 o. C and 4. 8 o. C. Using the electric heating method the calculated calibration factor was 357 J. o. C-1 while using the chemical calibration technique gave a calibration factor of 526. 8 J. o. C-1. The heat of dissolution of calcium chloride was -42. 7 k. J. mol-1 using the first calibration factor and -63. 08 k. J. mol-1 using the second calibration factor. Part B had differences in temperature of 3. 5 o. C, 3. 4 o. C and 3. 5 o. C. Part C had a change in temperature of 1. 8 o. C, 1. 8 o. C and 1. 7 o. C. The heat of dissolution of potassium nitrate was +24. 3 k. J. mol-1 using the first calibration factor and +35. 9 k. J. mol-1 using the second. The heat of dissolution values calculated using chemical calibration for both calcium chloride and potassium nitrate were more accurate in that they were closer to the known value of -82. 8 k. J. mol-1 for dehydrated calcium chloride and +35. 0 k. J. mol-1 for potassium nitrate. Aitken College 2017 evaluates quality of data summarises data lists data reports data

Year 12 - Determination of the enthalpy changes in chemical reactions by use of calorimetry Student 1 The two calibration factors from part A did not result in an enthalpy of dissolution for calcium chloride that was close to the expected value of -89. 9 k. J. mol-1. This may have been due to the calcium chloride becoming hydrated which would have lowered the enthalpy value. Modifications to the design of the experiment would have been to heat the calcium chloride before use to make sure that it was dehydrated before use. Student 2 A modification to the design of the experiment is to specify that the calcium chloride be heated before use to ensure that only dehydrated calcium chloride was present. Monohydrated calcium chloride has an enthalpy of dissolution of -49 k. J. mol-1 compared to -82. 9 k. J. mol-1 for dehydrated calcium chloride as energy is required to break the ion-dipole bonds between the water and calcium chloride. By ensuring that all of the calcium chloride was dehydrated before the reaction then this would mean that there would be more energy released when it was dissolved improving the accuracy of the result. Aitken College 2017 predicts how modifications improve the quality of the data explains how modifications improve the design identifies modifications modifies method

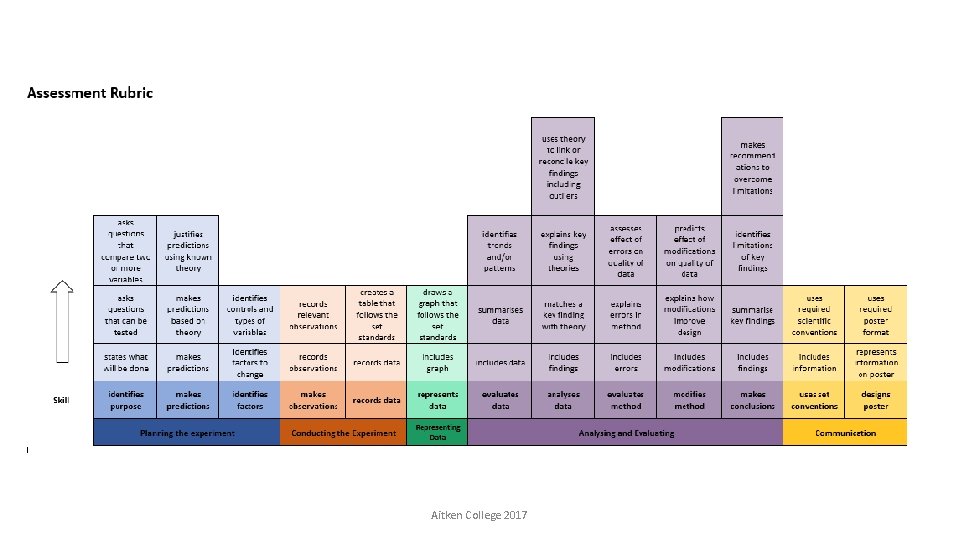
Key Ideas Rubrics based on developmental progressions enables • introduction of key skills from year 7 in preparation for VCE • skill progression to be made visible to students • consistent assessment across all year levels and sciences • tracking of student progress Aitken College 2017

Using Skills Based Rubrics in Area of Study 3 Reflections from the classroom STAV VCE Chemistry Conference 2017 Cristy Herron Adele Hudson cherron@aitkencollege. edu. au ahudson@aitkencollege. edu. au Resources can be obtained from www. mychemhub. com Aitken College 2017
- Slides: 36