Using FRAP to Study the KinetochoreMicrotubule Interaction C

Using FRAP to Study the Kinetochore-Microtubule Interaction C. G. Pearson, P. S. Maddox, E. D. Salmon and K. Bloom
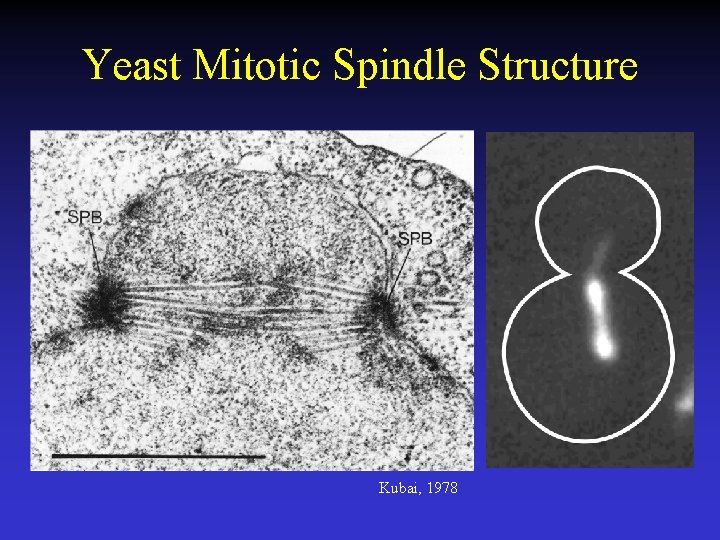
Yeast Mitotic Spindle Structure Kubai, 1978
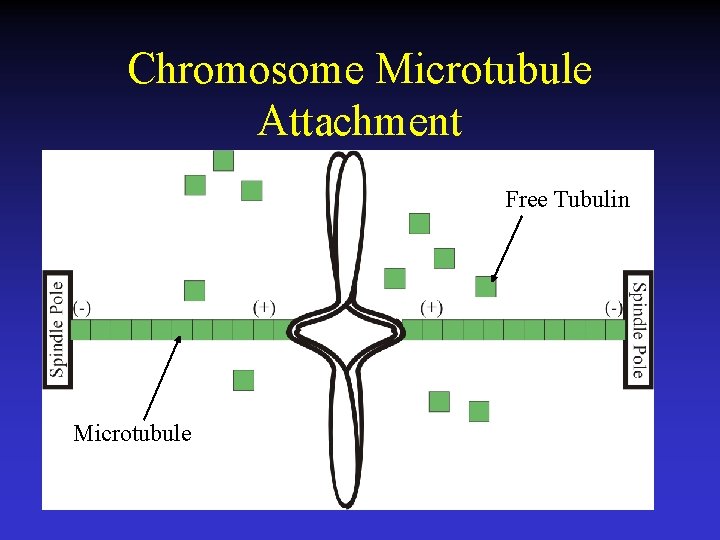
Chromosome Microtubule Attachment Free Tubulin Microtubule

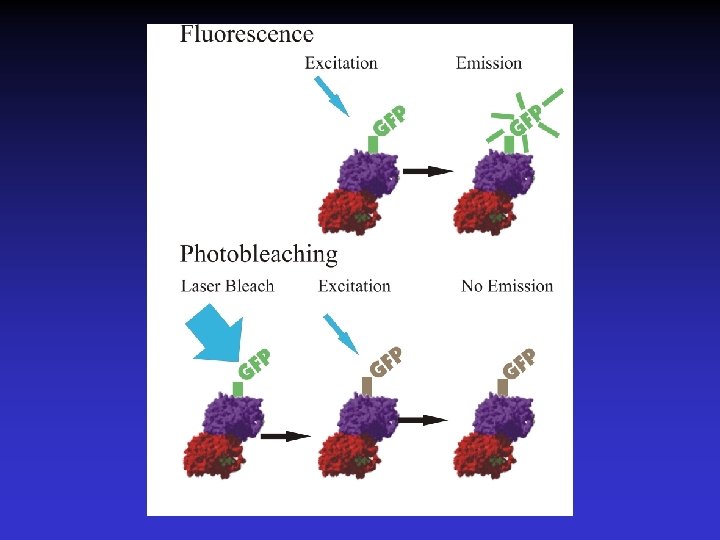
Fluorescence Recovery After Photobleaching (FRAP) • Fluorescence based assay to determine protein dynamics (localized and/or diffusive). • Photobleaching of GFP tagged proteins without destruction of protein function. • Determine tubulin turnover within the microtubule by measuring rate and extent of fluorescence recovery.

FRAP Microscope Argon Laser Hamamatsu Orca ER CCD Camera Metamorph Acquisition System Nikon E 300 Inverted Microscope For more detail: www. bio. unc. edu/faculty/bloom/lab www. bio. unc. edu/faculty/salmon/lab
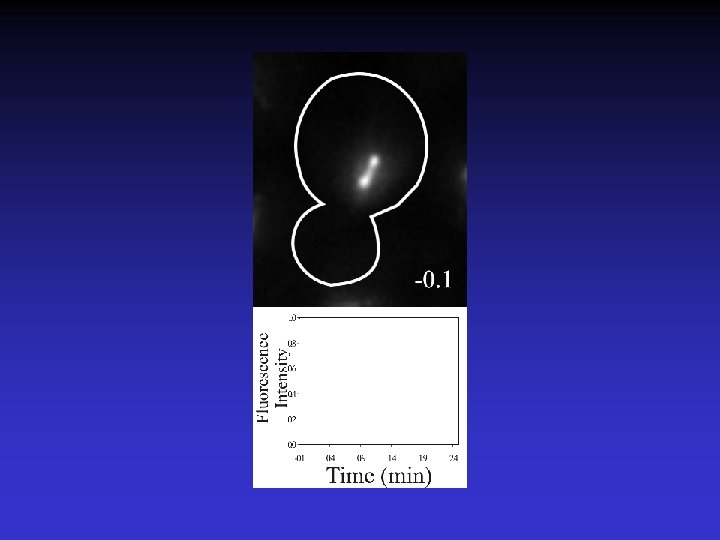

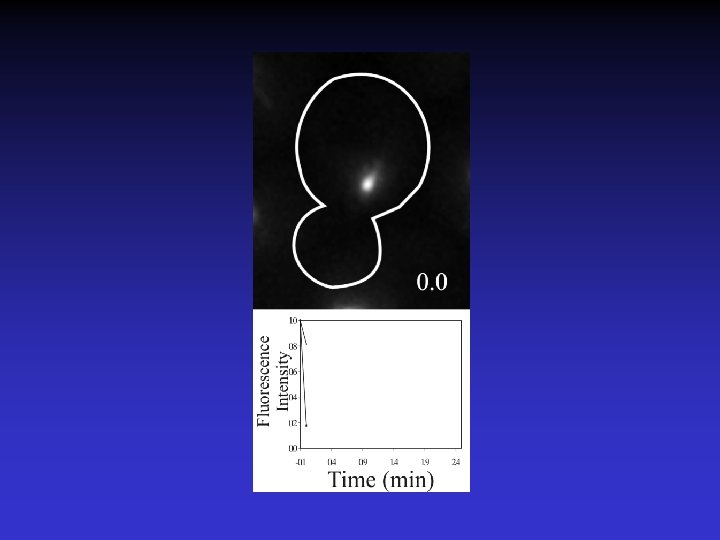
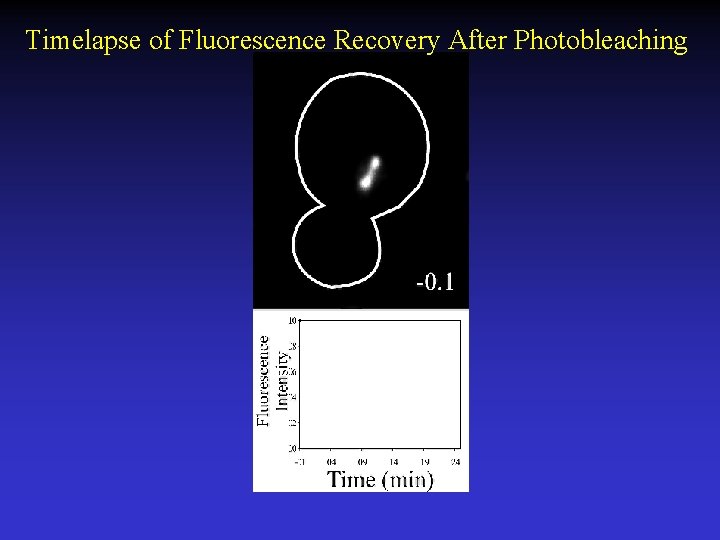
Timelapse of Fluorescence Recovery After Photobleaching
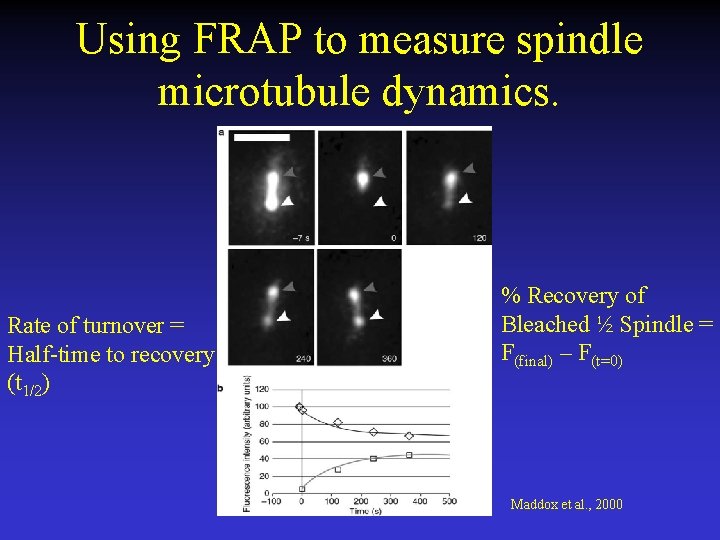
Using FRAP to measure spindle microtubule dynamics. Rate of turnover = Half-time to recovery (t 1/2) % Recovery of Bleached ½ Spindle = F(final) – F(t=0) Maddox et al. , 2000
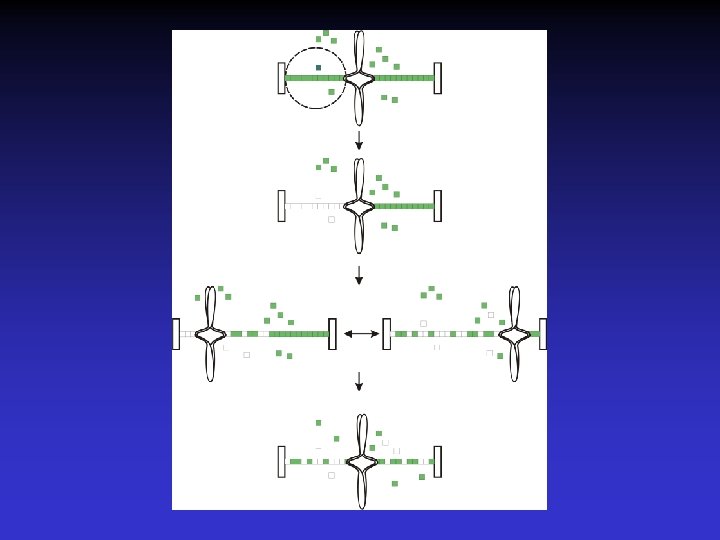

What are the dynamic properties of microtubules in the Metaphase spindle?

66 % of metaphase spindle microtubules turnover with a half -life of 53 sec. 33% are much more stable.

There are 24 microtubules per half spindle. 16 (66 % ) are kinetochore microtubules. While 8 (33 %) are overlapping interpolar microtubules. Winey et al. (1995) Journal of Cell Biology. 129(6): 1601 -1615.

Therefore we conclude that the kinetochore microtubules are dynamic while the interpolar microtubules are stable.
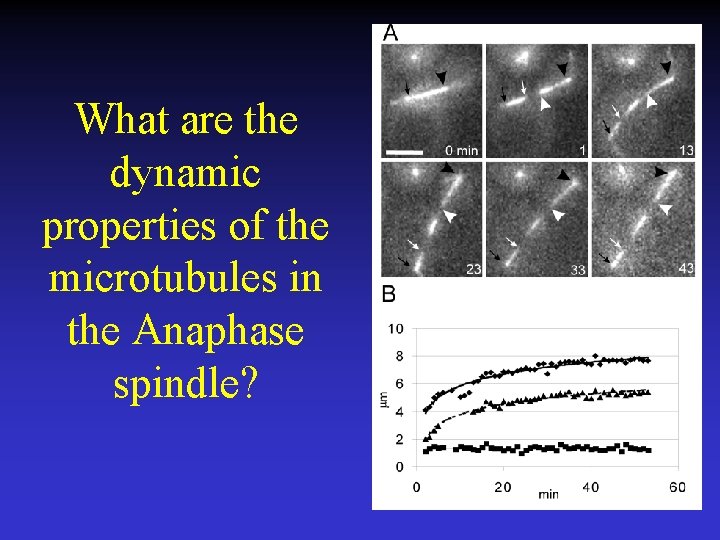
What are the dynamic properties of the microtubules in the Anaphase spindle?

Microtubule turnover in kinetochore protein mutants. CTF 13 and STU 2 - Essential, mutants delay in metaphase by the spindle checkpoint, chromosome loss mutant, localize to CEN. CTF 13 (ctf 13 -30) - Core kinetochore component STU 2 (stu 2 td) –Microtubule binding protein.
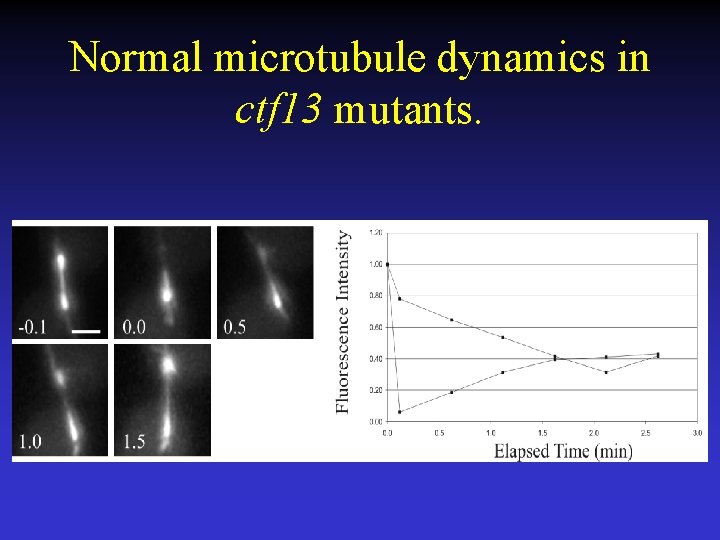
Normal microtubule dynamics in ctf 13 mutants.
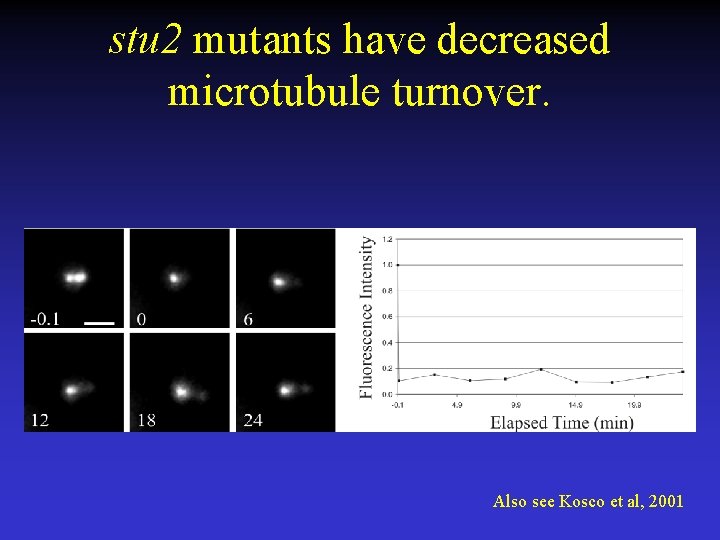
stu 2 mutants have decreased microtubule turnover. Also see Kosco et al, 2001

Microtubule turnover in kinetochore protein mutants. • FRAP allowed us to discern differences in mutants that show similar morphological phenotypes.

What is Fluorescent Speckle Microscopy (FSM)? • Fluorescent discontinuities, “speckles” in biological polymers (e. g. microtubules, actin filaments) • Caused by stochastic incorporation of fluorescently tagged subunits into the polymer • Allows visualization of assembly dynamics and motility of the polymer

C. M. Waterman-Storer and E. D. Salmon. (1998). How microtubules get fluorescent speckles. Biophys Journal 75, 2059 -2069.

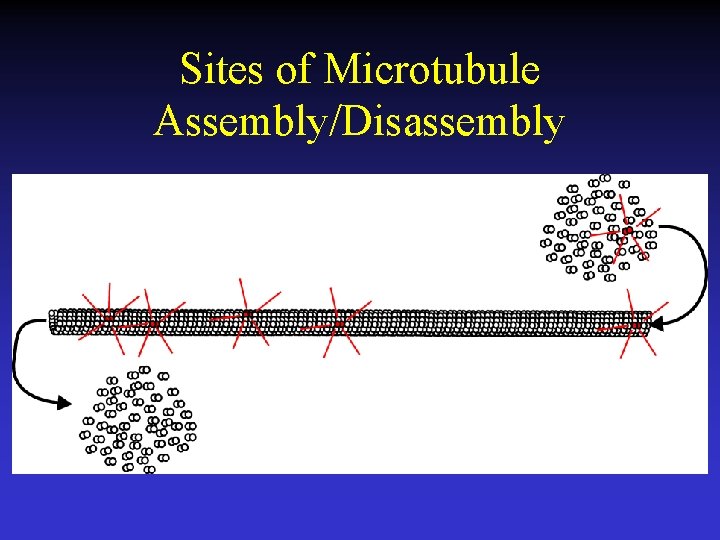
Sites of Microtubule Assembly/Disassembly

Microtubule Translocation

How Do Microtubules get Fluorescent Speckles?
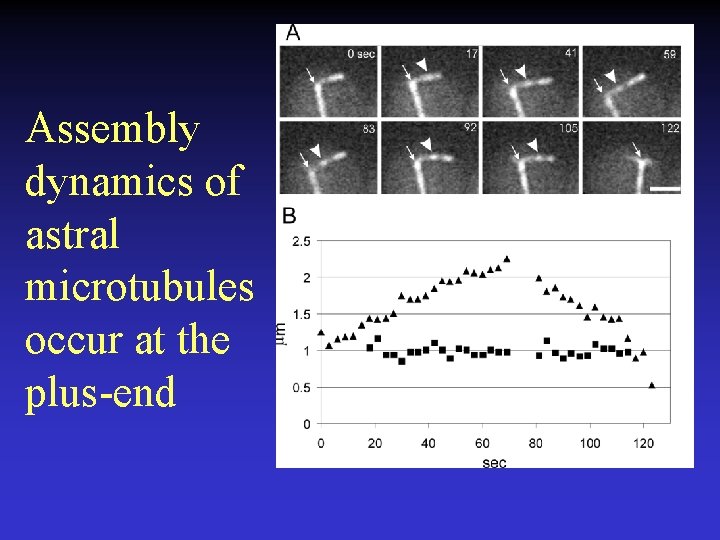
Assembly dynamics of astral microtubules occur at the plus-end

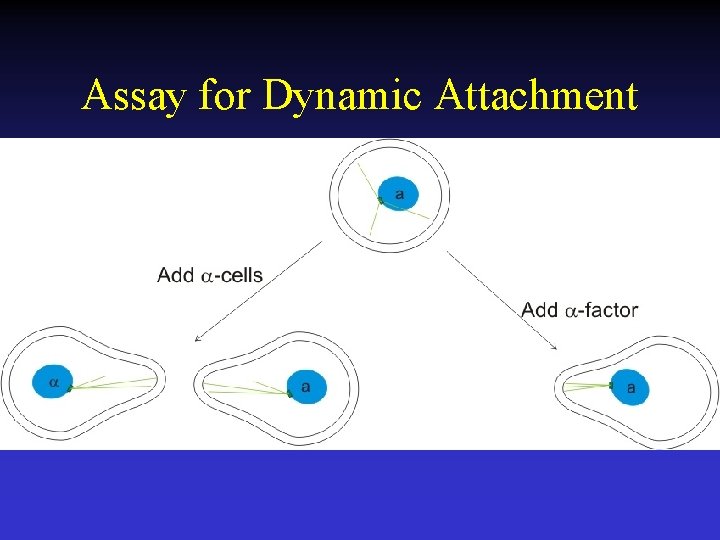
Assay for Dynamic Attachment

Assay for Dynamic Attachment

Assay for Dynamic Attachment

Assay for Dynamic Attachment
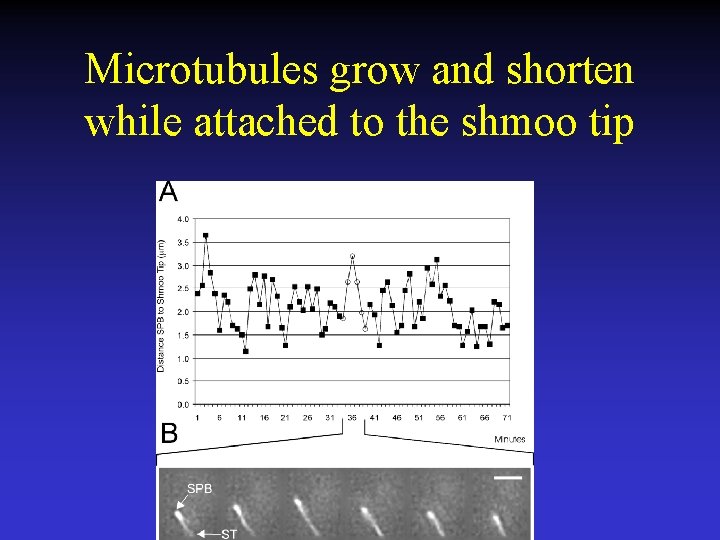
Microtubules grow and shorten while attached to the shmoo tip
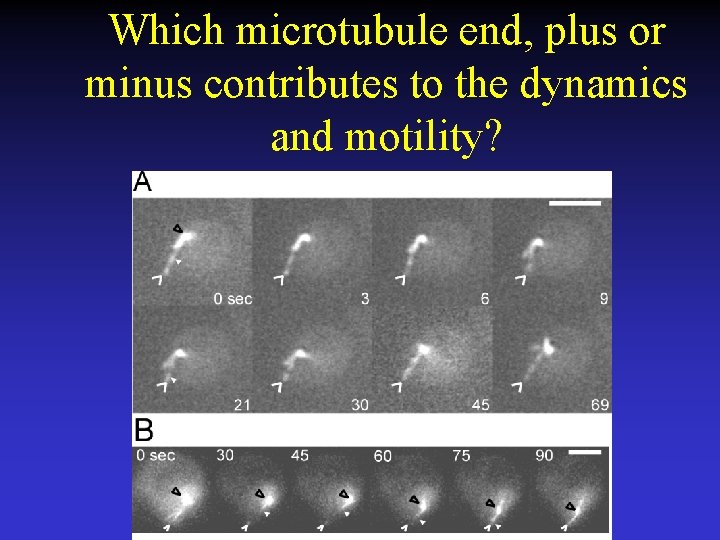
Which microtubule end, plus or minus contributes to the dynamics and motility?
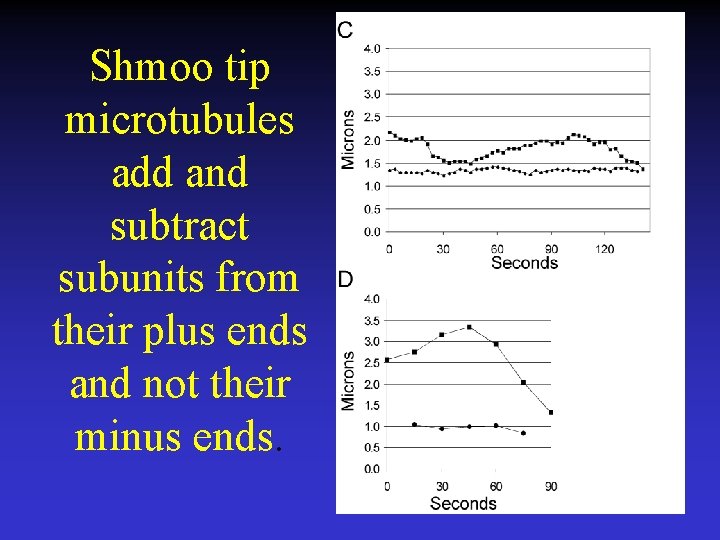
Shmoo tip microtubules add and subtract subunits from their plus ends and not their minus ends.

Analysis of Protein Dynamics Using FRAP. • Dynamics of localized and diffuse proteins in live cells. • Spindle MT FRAP to study microtubule dynamics and their regulation by chromosomes.

Bloom Lab Elaine Yeh Dale Beach Mythreye Karthikeyan Leanna Topper Ted Zarzar Jennifer Stemple David Bouck Goldstein Lab Thank You Kerry Bloom Ted Salmon Paul Maddox Collaborators Tim Huffaker Karena Kosco Salmon Lab Julie Canman Bonnie Howell Katie Shannon Jennifer Deluca Daniela Cimini Lisa Cameron Jeff Molk Ben Moree
- Slides: 39