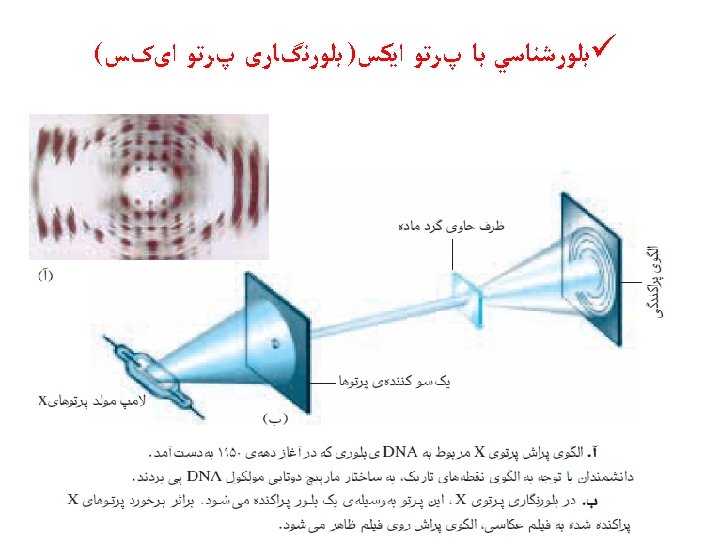
Use partial orbital diagrams and Lewis symbols to

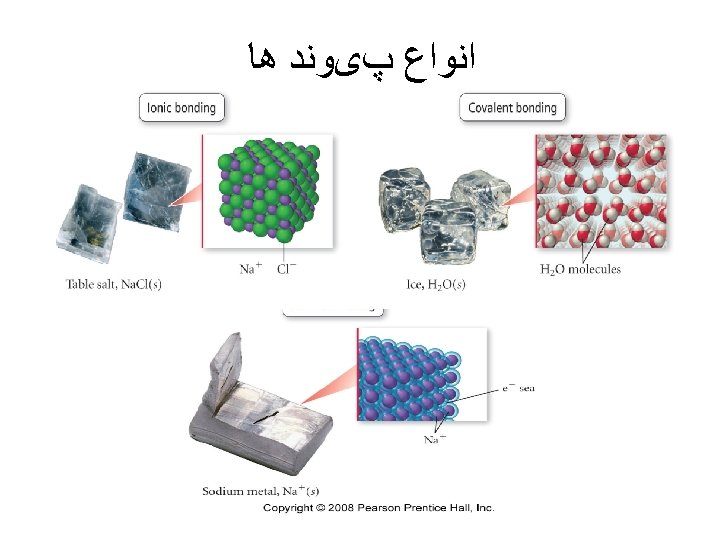

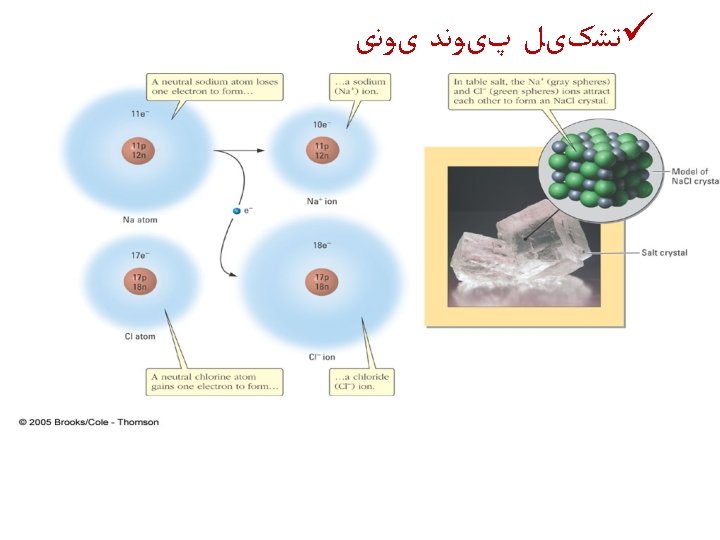
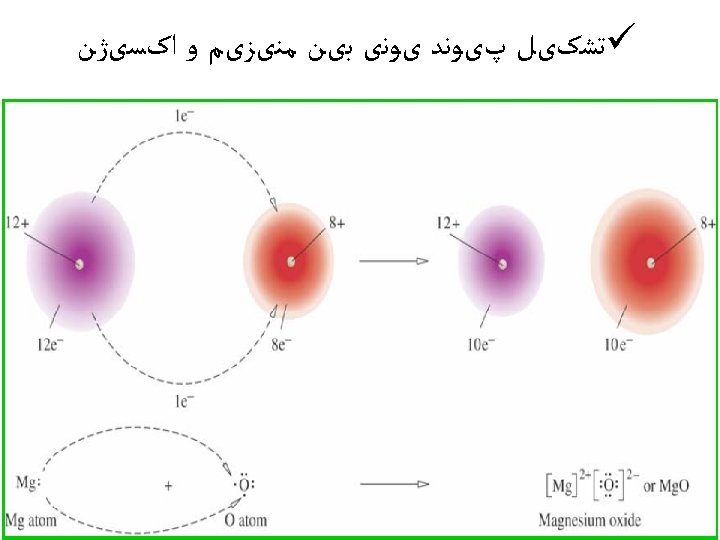
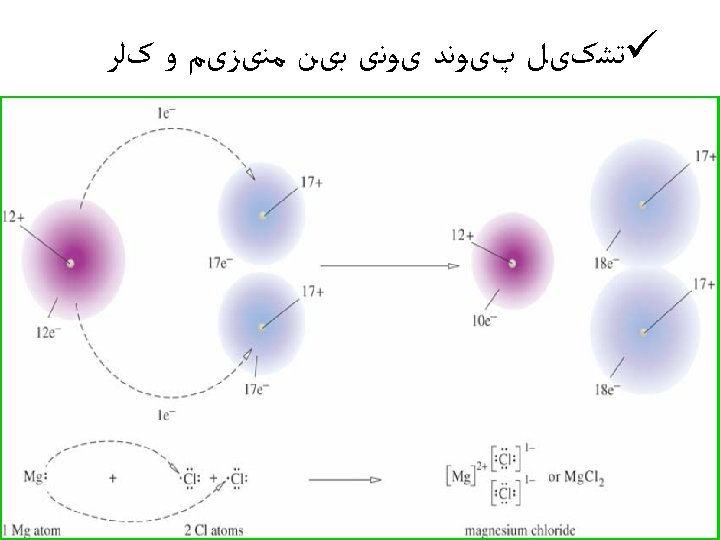

ﺳﻮﺍﻝ ü Use partial orbital diagrams and Lewis symbols to depict the formation of ions from the atoms and determine the formula of the compound. Example: Na + O
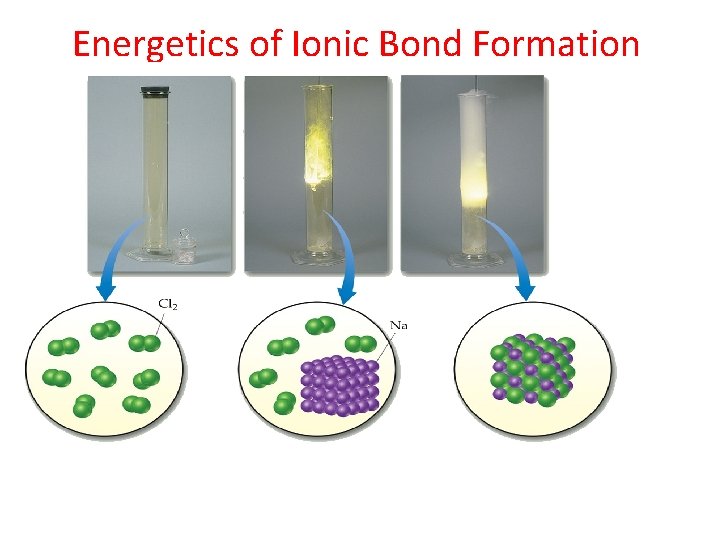
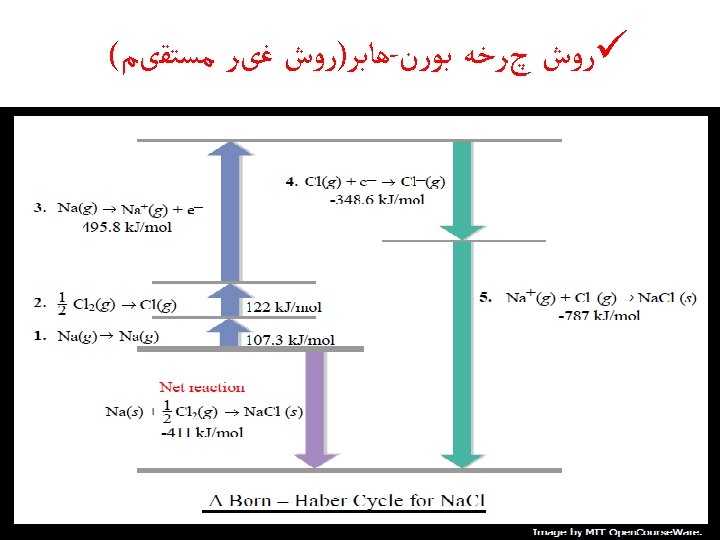
Energetics of Ionic Bond Formation


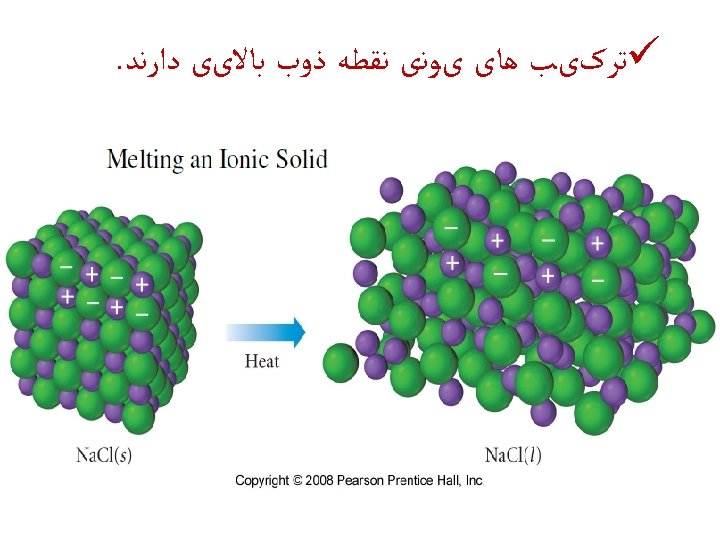
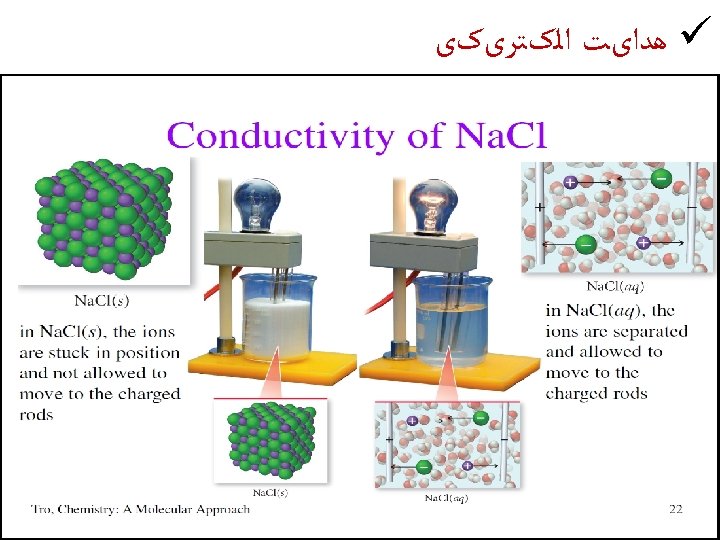
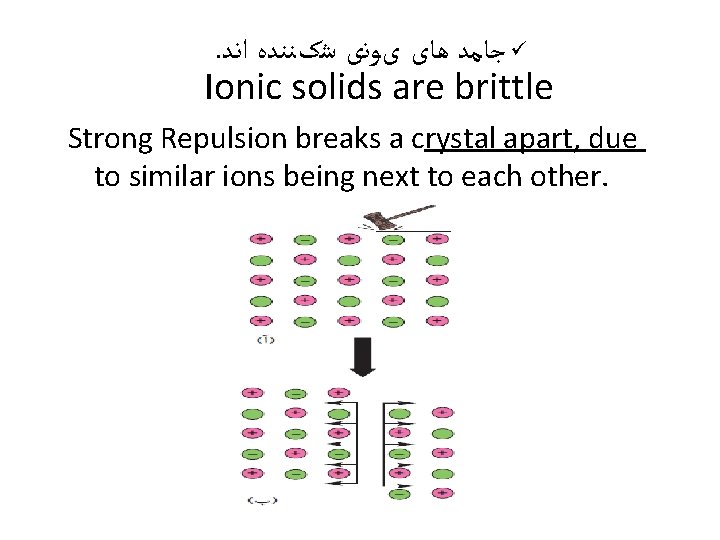
. ﺟﺎﻣﺪ ﻫﺎی یﻮﻧی ﺷکﻨﻨﺪﻩ ﺍﻧﺪ ü Ionic solids are brittle Strong Repulsion breaks a crystal apart, due to similar ions being next to each other.



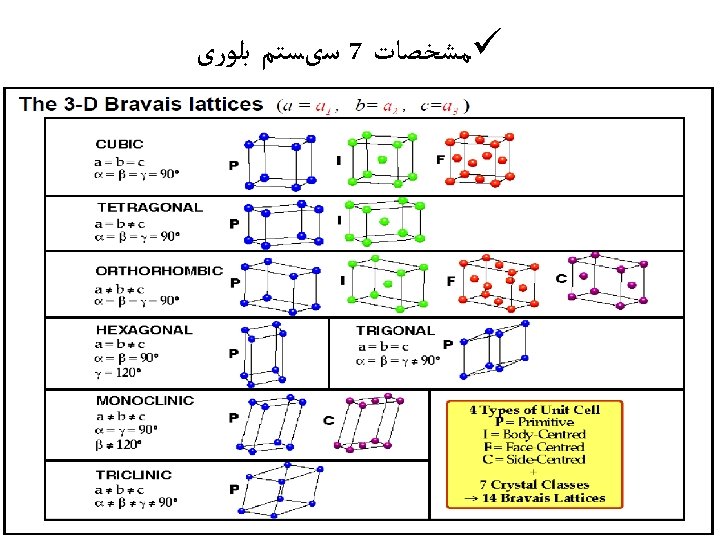
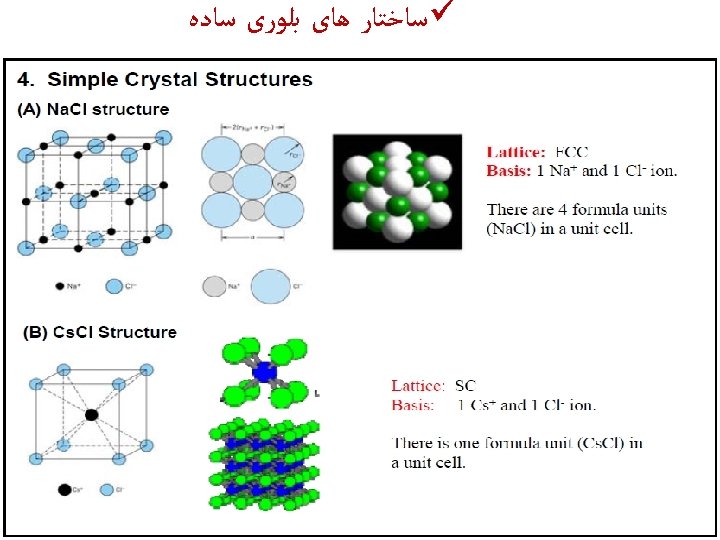
All ionic compounds make a crystal shape. They are held together into a crystal lattice– an arrangement of alternating positive and negative ions held together by opposite charges. Some crystals are held together strongly, some are held together weakly. What are some deter-mining factors about the strength of these crystals? Alum, KAl(SO 4)2 Rutile, Ti. O 2 Aragonite, Ca. CO 3 Beryl, Be. Al 2(Si. O 3)6
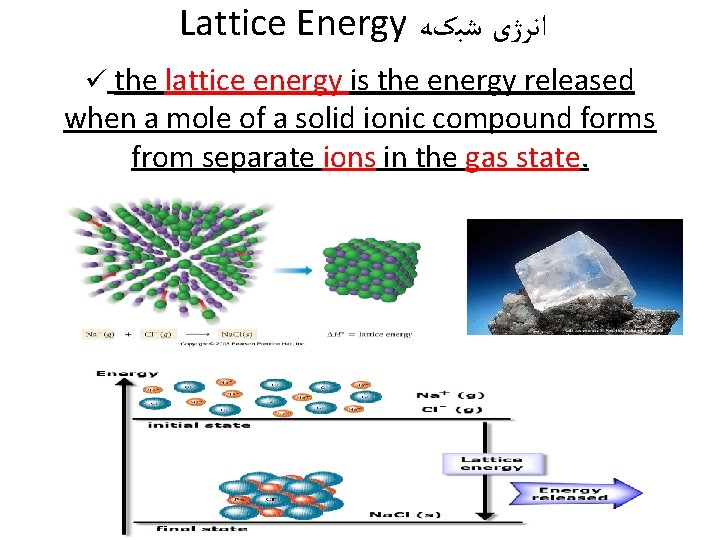
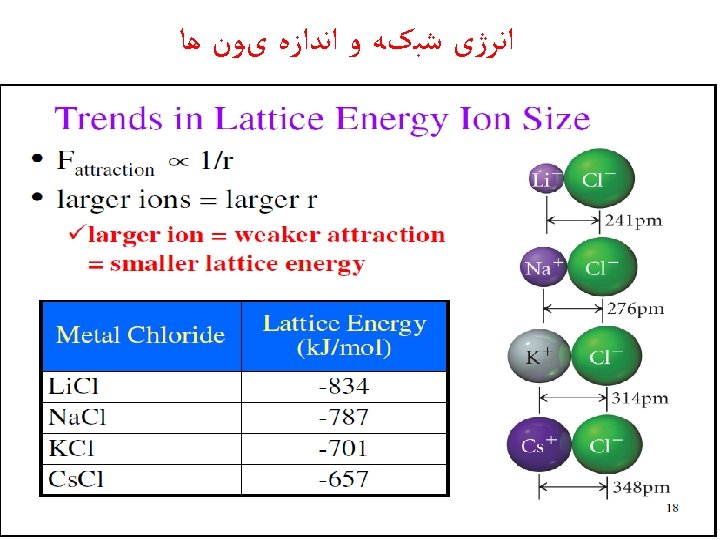
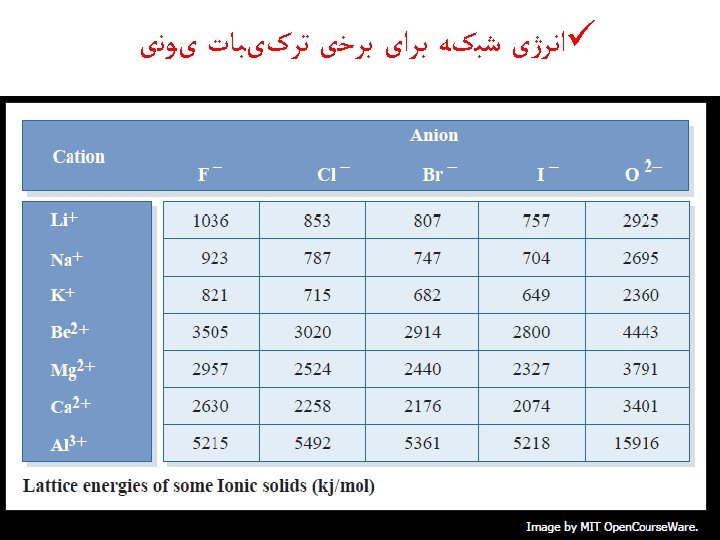
Lattice Energy ﺍﻧﺮژی ﺷﺒکﻪ ü the lattice energy is the energy released when a mole of a solid ionic compound forms from separate ions in the gas state.
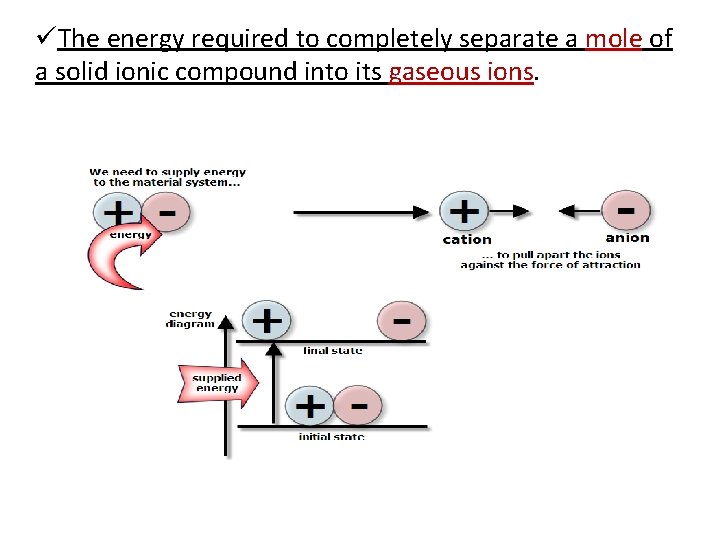
üThe energy required to completely separate a mole of a solid ionic compound into its gaseous ions.


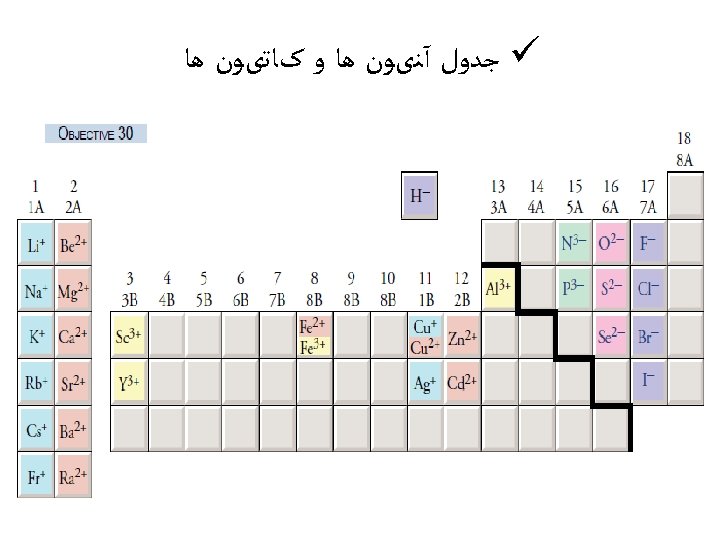
Ionic radius ﺷﻌﺎﻉ یﻮﻧی ü ØThe ionic radius is not a fixed property of a given ion, but varies with coordination number, spin state and other parameters. ØIonic size (for the same ion) also increases with ncreasing coordination number, and an ion in a high-spin state will be larger than the same ion in a low-spin state. In general, ionic radius decreases with increasing positive charge and increases with increasing negative charge.

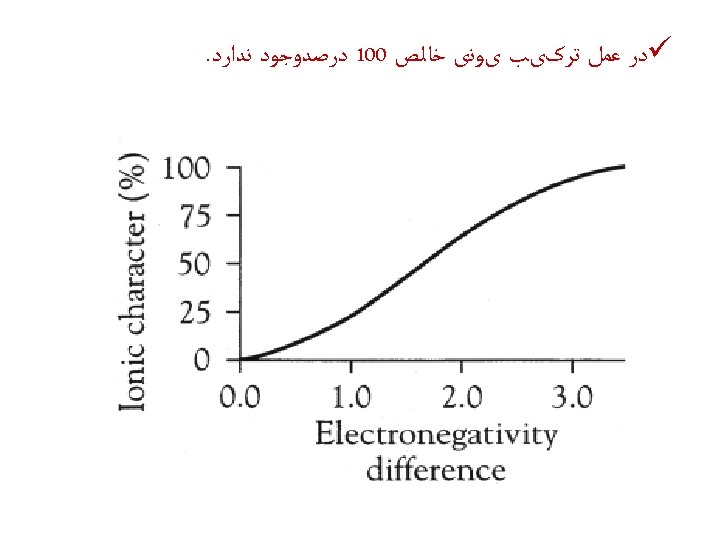
üAn "anomalous" ionic radius in a crystal is often a sign of significant covalent character in the bonding. üOn the basis of the fluorides, one would say that Ag+ is larger than Na+, but on the basis of the chlorides and bromides the opposite appears to be true. ü This is because the greater covalent character of the bonds in Ag. Cl and Ag. Br reduces the bond length and hence the apparent ionic radius of Ag+, an effect which is not present in the halides of the more electropositive sodium, nor in silver fluoride in which the fluoride ion is relatively unpolarizable.
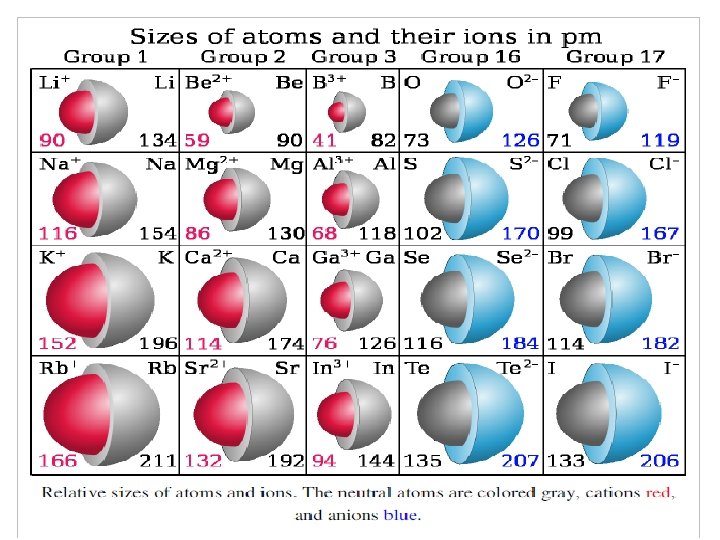

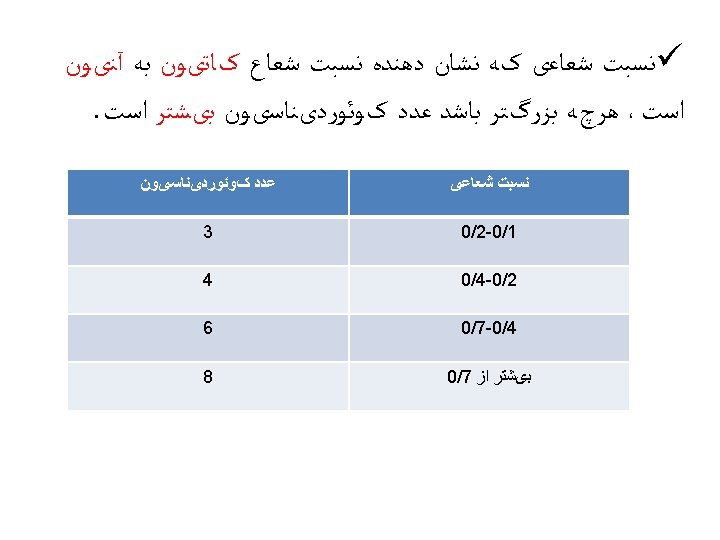
Determination of ionic radii The distance between two ions in an ionic crystal can be determined by X-ray crystallography, which gives the lengths of the sides of the unit cell of a crystal. For example, the length of each edge of the unit cell of sodium chloride is found to be 564. 02 ﺇ pm. Each edge of the unit cell of sodium chloride may be considered to have the atoms arranged as Na+. . . Cl-…. Na+, so the edge is twice the Na-Cl separation. Therefore, the distance between the Na+ and Cl- ions is half of 564. 02 ﺇ pm, which is 282. 01 pm. However, although X-ray crystallography gives the distance between ions, it doesn't indicate where the boundary is between those ions, so it doesn't directly give ionic radii.
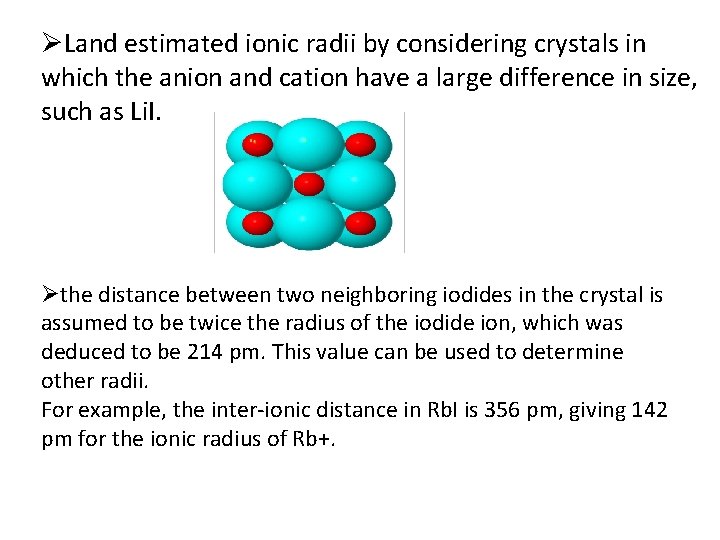
ØLand estimated ionic radii by considering crystals in which the anion and cation have a large difference in size, such as Li. I. Øthe distance between two neighboring iodides in the crystal is assumed to be twice the radius of the iodide ion, which was deduced to be 214 pm. This value can be used to determine other radii. For example, the inter-ionic distance in Rb. I is 356 pm, giving 142 pm for the ionic radius of Rb+.




Total Lattice Energy • The total lattice energy corresponds to the combination of the attracting and repulsing energy terms (N: Avogadro’s number):


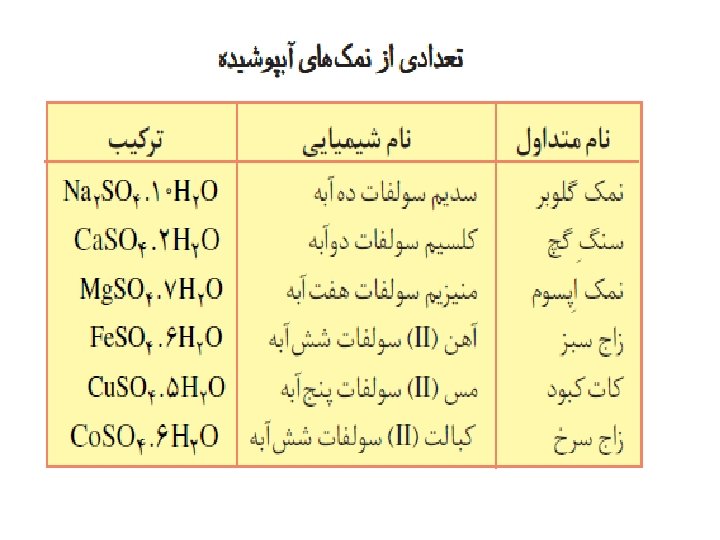
ﻗﻮﺍﻋﺪ ﻧﺎﻣگﺬﺍﺭی ﺗﺮکیﺒﺎﺕ یﻮﻧی آﺒﺪﺍﺭ ü Hydrated ionic compounds (i. e. , hydrates) Rule 1. The ionic compound (without the waters of hydration) is named first by using the rules for naming ionic compounds (e. g. , Ba(OH)2 • 8 H 2 O = "barium hydroxide"). Rule 2. Greek prefixes are attached to the word "hydrate" to indicate the number of water molecules per formula unit for the compound (e. g. , Ba(OH)2 • 8 H 2 O; 8 water molecules = "octahydrate"). Rule 3. When the chemical formula for a hydrated ionic compound is written, the formula for the ionic compound is separated from the waters of hydration by a centered "dot"

REVERSIBILITY OF HYDRATION The waters of hydration can be removed or added by simply heating a hydrated salt or wetting an anhydrous salt. This is called a reversible reaction.
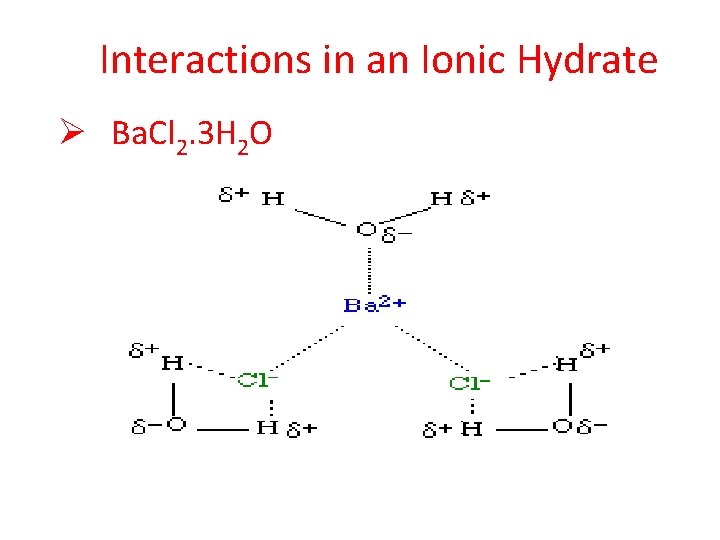
Interactions in an Ionic Hydrate Ø Ba. Cl 2. 3 H 2 O



References D. Shriver, P. Atkins, Inorganic Chemistry, 3 rd Editon, Oxford University Press, 2002; U. Müller, Inorganic Structural Chemistry, 2 nd Edition, John Wiley & Sons Ltd, Chichester, 2007 Bodie E. Douglas, Concepts and Models of Inorganic Chemistry, 2 nd Edition, John Wiley & Sons, Inc, 1983 ﺷیﻤی ﻣﻌﺪﻧی ﻫیﻮﺋی ﺷیﻤی ﻣﻌﺪﻧی ﻣﻼﺭﺩی )پیﺎﻡ ﻧﻮﺭ( ﺩکﺘﺮ ﻣیﺮ ﺷکﺮﺍﺋی 1 ﺷیﻤی ﻋﻤﻮﻣی

ﺑﺮﺍی ﻭﺭﻭﺩ ﺑﻪ ﺷﺒکﻪ آﻤﻮﺯﺷی ﺩﺍﻧﺸﺠﻮیﺎﻥ Title کﻠیک کﻨیﺪ Lorem ipsum dolor sit amet, • consectetuer adipiscing elit. Vivamus et magna. Fusce sed sem sed magna suscipit egestas. Lorem ipsum dolor sit amet, • 808090901000 consectetuer adipiscing elit. Vivamus et magna. Fusce sed sem ﻟﻄﻔﺎ آﺪﺭﺱ ﻣﺎ ﺭﺍ ﺑﻪ ﺧﺎﻃﺮ ﺩﺍﺷﺘﻪ ﺑﺎﺷیﺪ sed magna suscipit egestas. MADSG. COM
- Slides: 51