Use of Solar energy Conversion of solar energy







- Slides: 12


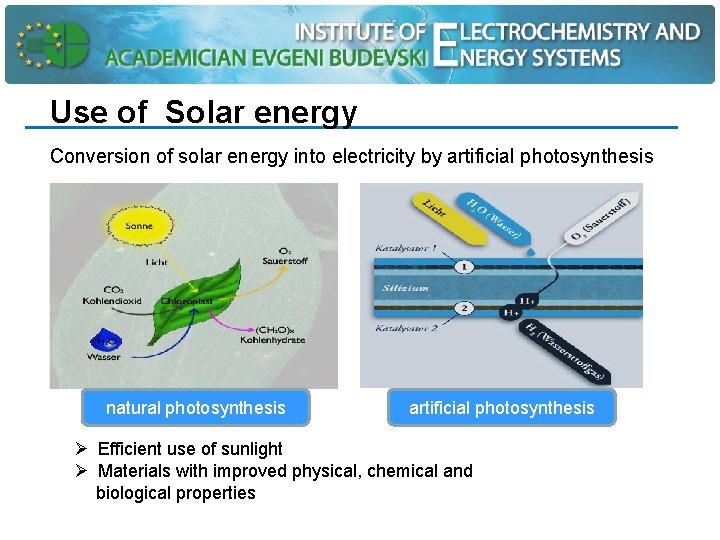
Use of Solar energy Conversion of solar energy into electricity by artificial photosynthesis natural photosynthesis artificial photosynthesis Ø Efficient use of sunlight Ø Materials with improved physical, chemical and biological properties

Materials Diamond Phthalocyanine (Ph. C) Ø Structure and properties of the diamond Ø Organic dyes Ø Photosensitivity ØHigh chemical and thermal stability Ø Nanocrystalline diamond layers (NCD) Ø Diamond layers doped with boron NCD+Ph. C Chlorophyll

Sequence of the experiment 1. Deposition of nanocrystalline layers 2. Modification of diamond surfaces - O 2 -plasma - NH 3 -plasma - UV/O 3 -treatment 3. Immobilization with photosensitive molecules 4. Analyzing the effectiveness of the modification - Measurement of contact angles - X-ray photoelectron spectroscopy - Raman spectroscopy 5. Fotoelektrochemical research

Deposition of diamond layers Ø Substrate preparation Ø Hot-Filament CVD-method Ø Operating parameters (gas Mixture 1% CH 4 + 99% H 2) Substrate’s Temperature Filament’s Temperature Pressure 880… 890 °C > 2000 °C 25 mbar NCD-layer HFCVD-installation Time 180 min

Modification of diamond layers • Unmodified sample • H-Terminierung • UV/O 3 -treatment • Irradiation with UV-light (wave length 175210 nm); time t = 10 min • Modification with O 2 -plasma • time t = 5 min; • power P = 200 W; pressure P = 0, 67 mbar • Modification with NH 3/N 2 -плазма • gas mixture (5% NH 3, 95% N 2) • time t = 5 min; pressure P = 1. 8 x 10 -2 mbar • power P=150 W

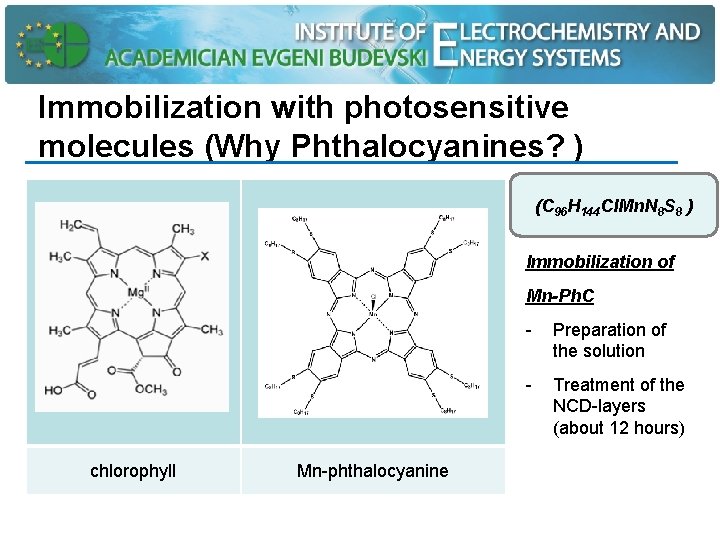
Immobilization with photosensitive molecules (Why Phthalocyanines? ) (C 96 H 144 Cl. Mn. N 8 S 8 ) Immobilization of Mn-Ph. C chlorophyll Mn-phthalocyanine - Preparation of the solution - Treatment of the NCD-layers (about 12 hours)

Fotoelektrochemical characterisation Producing of diamond electrodes Testing • P-doped NCD-layers • Performance of the experiments • Immobilization of phthalocyanine • Three-electrode cell • PBS-solution as electrolyte NCD-electrodes and a using light source Three-electrode cell

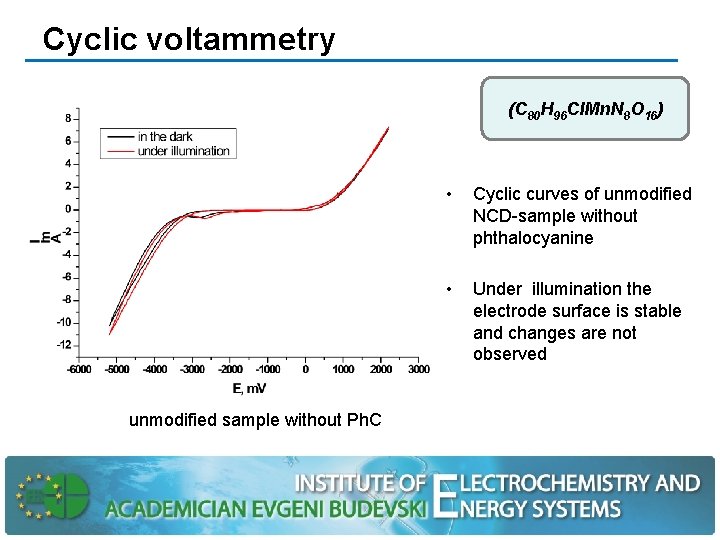
Cyclic voltammetry (C 80 H 96 Cl. Mn. N 8 О 16) unmodified sample without Ph. C • Cyclic curves of unmodified NCD-sample without phthalocyanine • Under illumination the electrode surface is stable and changes are not observed

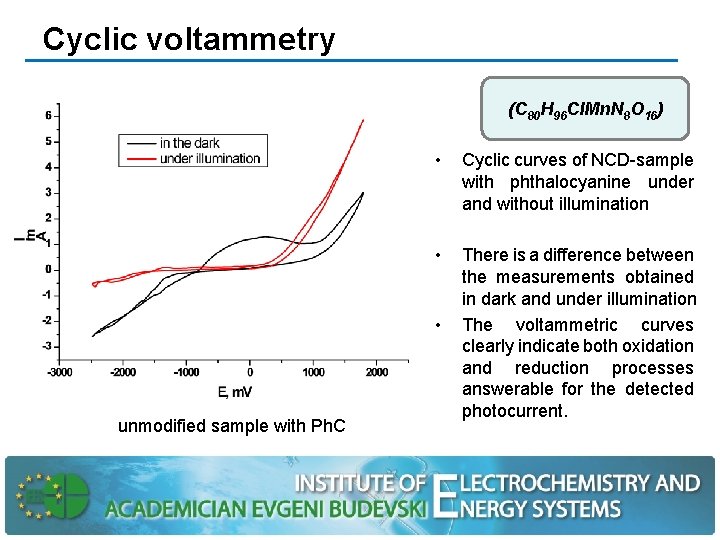
Cyclic voltammetry (C 80 H 96 Cl. Mn. N 8 О 16) • Cyclic curves of NCD-sample with phthalocyanine under and without illumination • There is a difference between the measurements obtained in dark and under illumination The voltammetric curves clearly indicate both oxidation and reduction processes answerable for the detected photocurrent. • unmodified sample with Ph. C

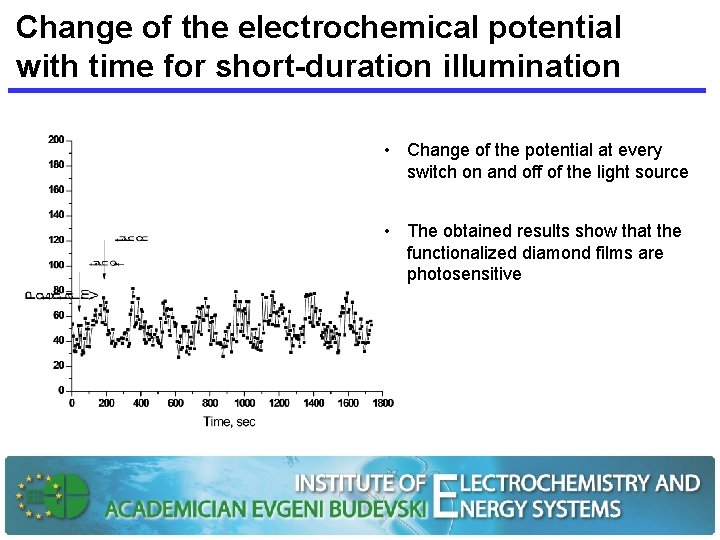
Change of the electrochemical potential with time for short-duration illumination • Change of the potential at every switch on and off of the light source • The obtained results show that the functionalized diamond films are photosensitive
