US Early Feasibility Studies for Transcatheter Mitral Therapy

US Early Feasibility Studies for Transcatheter Mitral Therapy Changfu Wu, Ph. D Food and Drug Administration Center for Devices and Radiological Health Office of Device Evaluation Division of Cardiovascular Devices

I have no relevant financial relationships.
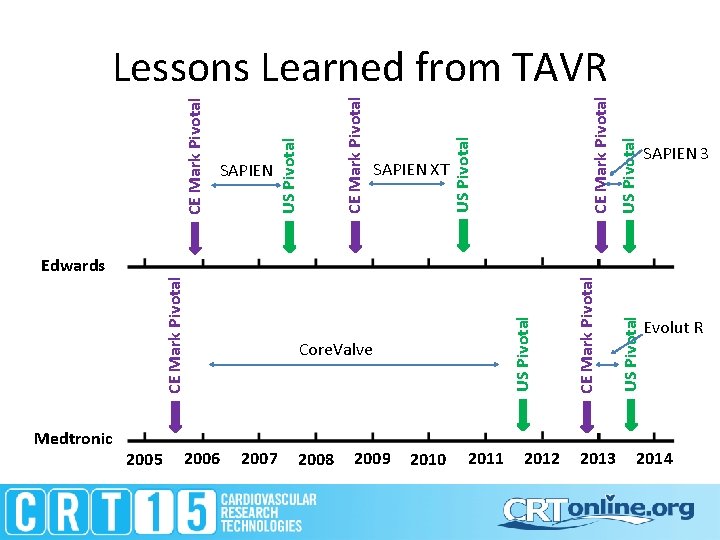
Edwards Medtronic CE Mark Pivotal 2005 2006 2007 2008 2009 2010 Core. Valve 2011 2012 2013 US Pivotal SAPIEN 3 US Pivotal CE Mark Pivotal US Pivotal SAPIEN XT CE Mark Pivotal US Pivotal SAPIEN US Pivotal CE Mark Pivotal Lessons Learned from TAVR Evolut R 2014

CDRH Early Feasibility Study Initiative CDRH 2014 -2015 Strategic Priorities:

Preclinical Testing Requirements for EFS • FDA HV Team has adopted a staged (tiered) preclinical testing strategy for EFS, pivotal IDE, and PMA • For EFS – Less test items – Smaller sample size – Shorter test duration – Allowing more leveraging in lieu of direct testing

When Should You Talk to FDA? • When you think you have a “final” design, and plan to start the “formal” design verification studies • Too late when you are ready to start or have started an OUS study • The earlier, the better; better late than never

What Should You Tell FDA? • Your plan for – Bench testing – Animal testing – Biocompatibility testing – Sterility validation – MRI compatibility testing – Clinical investigation plan • Plan includes: what have been completed, what are planned, test items, test conditions, sample size, test duration, what will be leveraged from what source, timeline for the EFS, etc.

Common Holdups • Design Evaluation Strategy (DES) table – A concept that has some overlap with the FMEA – FDA can provide hands-on training – Recommended, but not required • Can be substituted with the FMEA table • Animal testing – GLP is recommended, but not required – Sample size and follow-up duration are negotiable – It’s not the end of the world if anatomical limitations in animal cannot be overcome

What Happens after You Talk to FDA? • We will meet with you, if needed, and provide our feedback on your plan – A live device demo is extremely helpful • You complete the preclinical testing that’s agreed upon by FDA and prepare the EFS IDE package • Lots of interactions in-between about details of the preclinical testing plan or any snags encountered during execution of the plan • Some statistics based on past TMVR device EFS IDE approvals – 2 -7 months from first contact with FDA to EFS IDE approval – 30 days on average from IDE submission to approval

Call of Action • CDRH needs your help in fulfilling the strategic priority plan • If you’re a device developer, come talk to FDA • If you’re a clinician working with a device developer, urge them to go talk to FDA

Contact Info Changfu Wu Email: Changfu. Wu@fda. hhs. gov Tel: (301) 796 -6086 OR Nicole Ibrahim, Branch Chief Email: Nicole. Ibrahim@fda. hhs. gov Tel: (301) 796 -5171
- Slides: 11