Uranium Radioactive Decay 238 234 230 Mass number
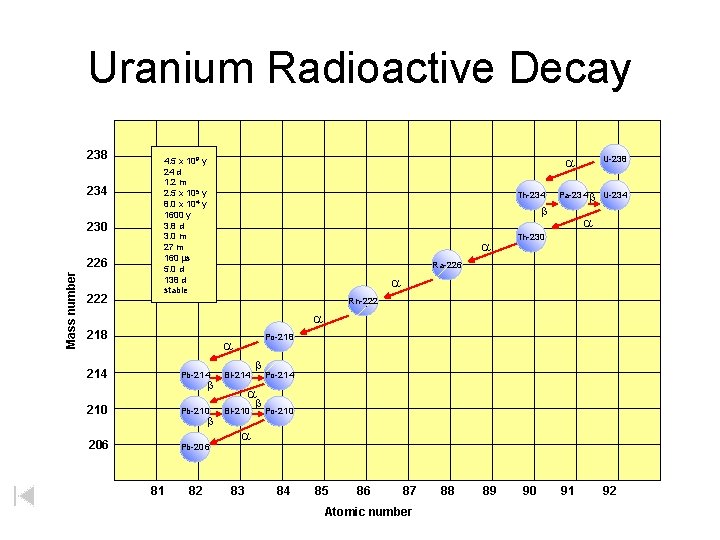
Uranium Radioactive Decay 238 234 230 Mass number 226 222 a 4. 5 x 109 y 24 d 1. 2 m 2. 5 x 105 y 8. 0 x 104 y 1600 y 3. 8 d 3. 0 m 27 m 160 ms 5. 0 d 138 d stable Th-234 b a U-238 Pa-234 b U-234 a Th-230 Ra-226 a Rn-222 a 218 Po-218 a 214 Pb-214 b 210 Pb-210 206 Pb-206 b 81 82 Bi-214 b Po-214 a Bi-210 b Po-210 a 83 84 85 86 87 Atomic number 88 89 90 91 92
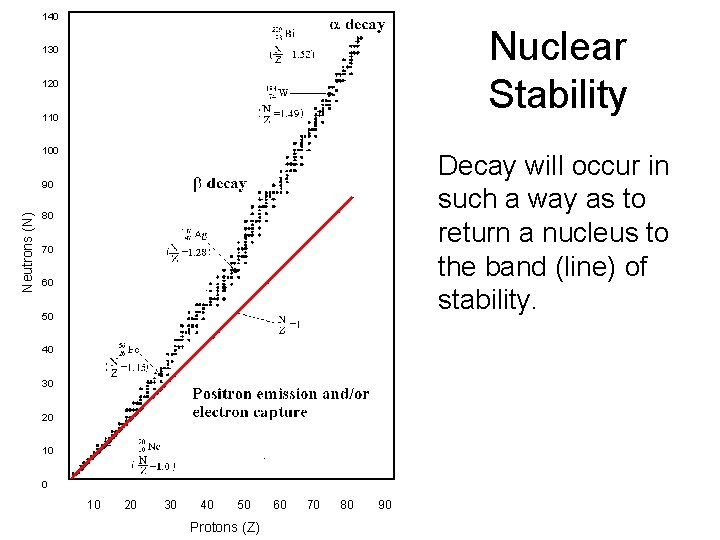
140 Nuclear Stability 130 120 110 100 Decay will occur in such a way as to return a nucleus to the band (line) of stability. Neutrons (N) 90 80 70 60 50 40 30 20 10 20 30 40 50 Protons (Z) 60 70 80 90
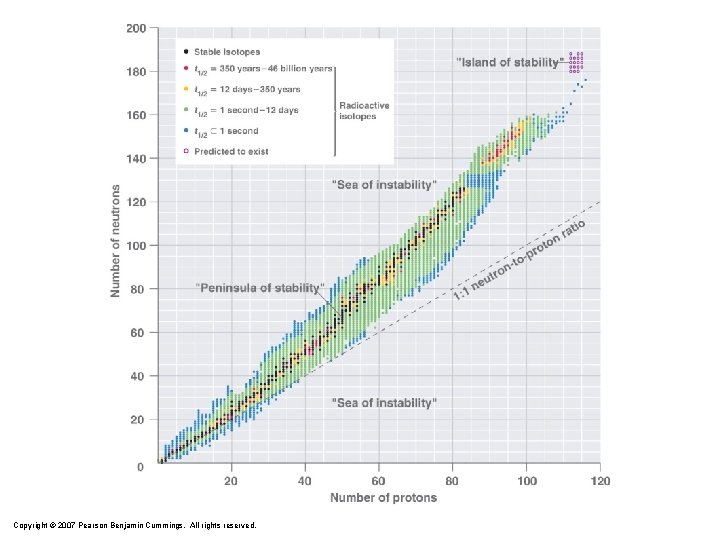
Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.
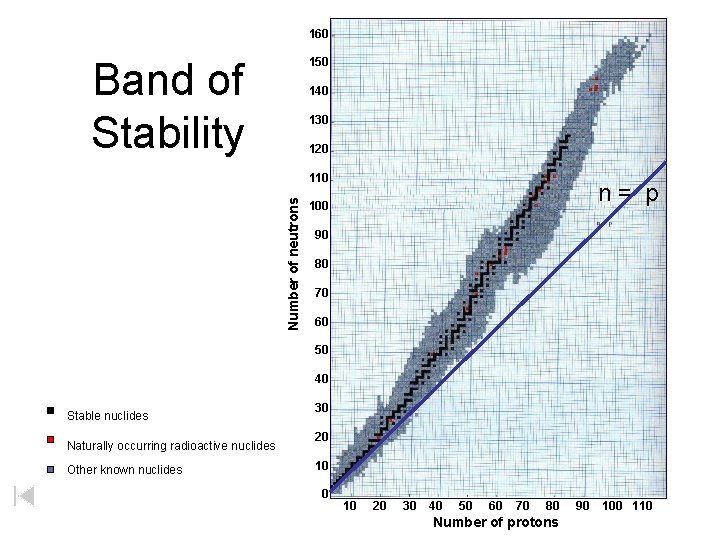
160 Band of Stability 150 140 130 120 Number of neutrons 110 n= p 100 90 80 70 60 50 40 Stable nuclides Naturally occurring radioactive nuclides Other known nuclides 30 20 10 20 30 40 50 60 70 80 Number of protons 90 100 110
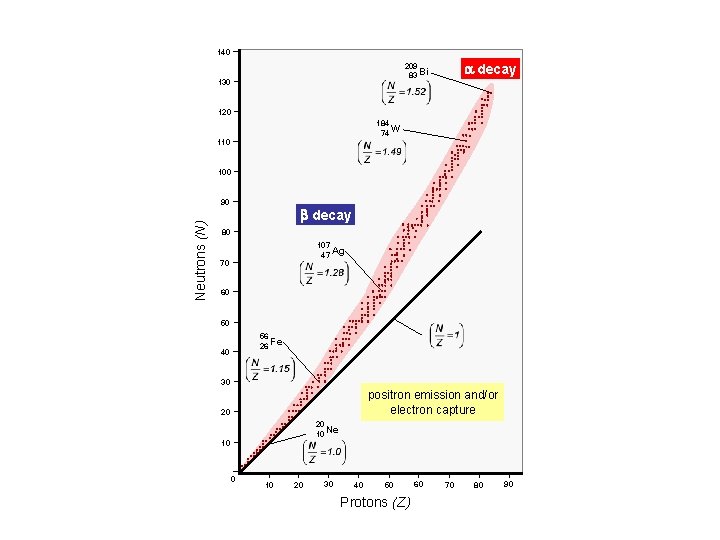
140 a decay 209 83 Bi 130 120 184 74 W 110 100 Neutrons (N) 90 b decay 80 107 47 Ag 70 60 50 56 26 Fe 40 30 positron emission and/or electron capture 20 20 10 Ne 10 0 10 20 30 40 50 Protons (Z) 60 70 80 90
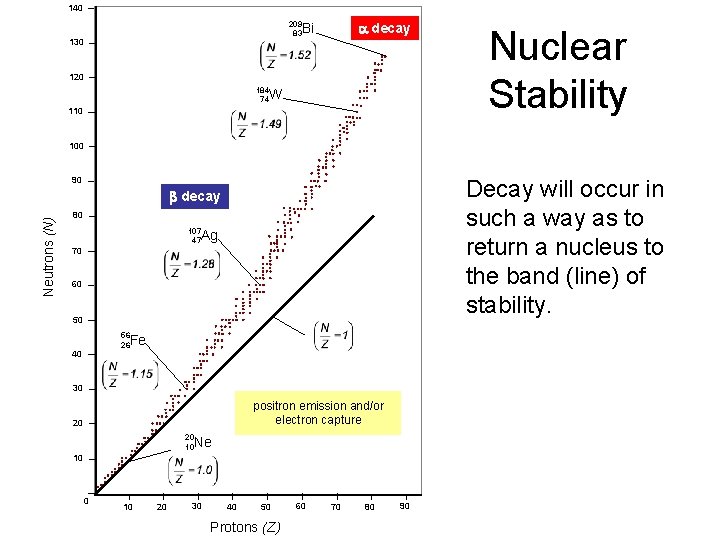
140 a decay 209 83 Bi 130 120 184 74 W 110 Nuclear Stability 100 90 Decay will occur in such a way as to return a nucleus to the band (line) of stability. Neutrons (N) b decay 80 107 47 Ag 70 60 50 56 26 Fe 40 30 positron emission and/or electron capture 20 20 10 Ne 10 0 10 20 30 40 50 Protons (Z) 60 70 80 90
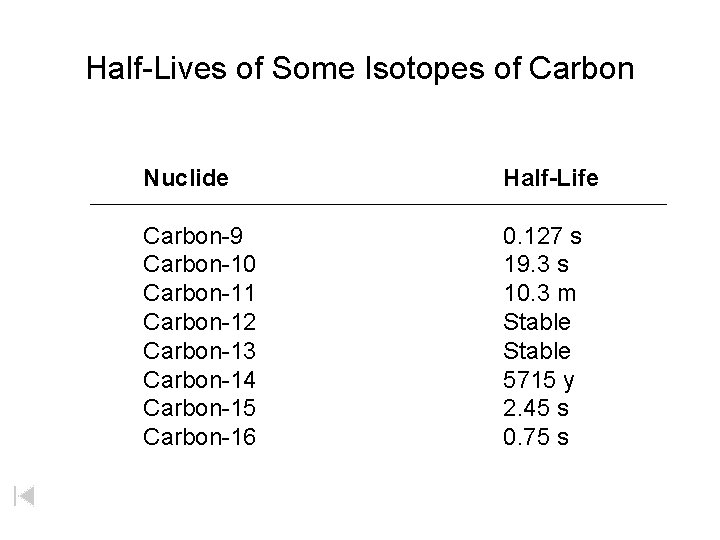
Half-Lives of Some Isotopes of Carbon Nuclide Half-Life Carbon-9 Carbon-10 Carbon-11 Carbon-12 Carbon-13 Carbon-14 Carbon-15 Carbon-16 0. 127 s 19. 3 s 10. 3 m Stable 5715 y 2. 45 s 0. 75 s
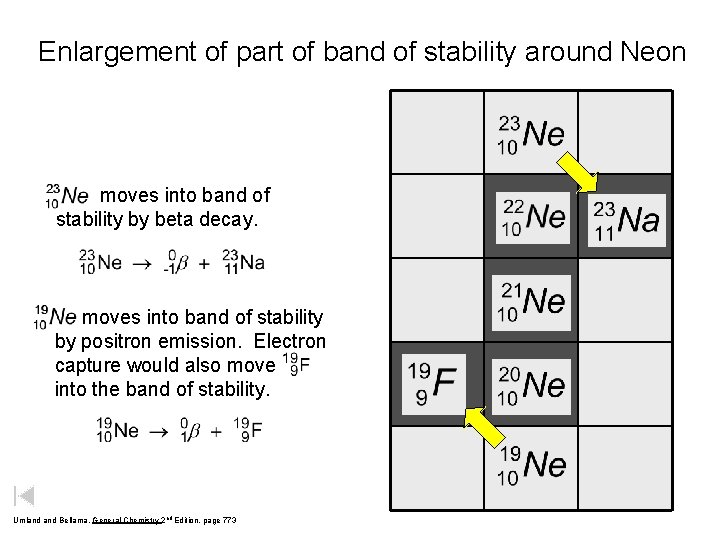
Enlargement of part of band of stability around Neon moves into band of stability by beta decay. moves into band of stability by positron emission. Electron capture would also move into the band of stability. Umland Bellama, General Chemistry 2 nd Edition, page 773
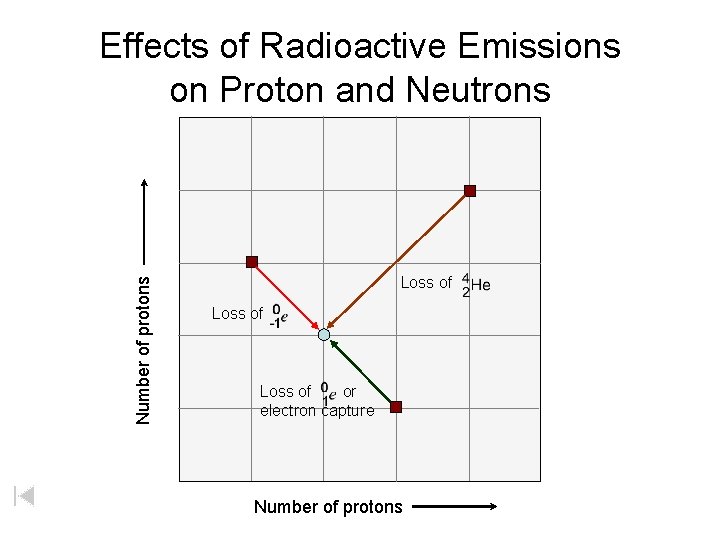
Number of protons Effects of Radioactive Emissions on Proton and Neutrons Loss of or electron capture Number of protons

Nuclear Decay “absorption”, “bombardment” vs. “production”, “emission” 223 88 4 2 a 2+ 87 37 4 2 Ra 14 7 + a 2+ 17 8 N 0 -1 b Rb 219 86 + O + 87 38 + Alpha 4 2 a 1 1 Sr Beta 2+ 0 -1 b neutron 1 0 n 2 1 Rn H H 2 1 + H 14 6 + 3 1 2 1 4 2 H 0 -1 b C Positron 0 +1 + He 17 7 N Gamma 0 0 b proton 1 1 H 1+ g 1 0 n

Units Used in Measurement of Radioactivity Units Measurements Curie (C) radioactive decay Becquerel (Bq) radioactive decay Roentgens (R) exposure to ionizing radiation Rad (rad) energy absorption caused by ionizing radiation Rem (rem) biological effect of the absorbed dose in humans

Effects of Instantaneous Whole-Body Radiation Doses on People Dose, Sv (rem) Effect Alexander Litvinenko >10 (1000) Death within 24 h from destruction of the neurological system. 7. 5 (750) Death within 4 -30 d from gastrointestinal bleeding. 1. 5 – 7. 5 (150 – 750) Intensive hospital care required for survival. At the higher end of range, death through infection resulting from destruction of white-blood cell-forming organs usually takes place 4 – 8 weeks after accident. Those surviving this period usually recover. < 0. 5 (50) Only proven effect is decrease in white blood cell count.

The intensity of radiation is proportional to 1/d 2, where d is the distance from the source.

Alpha, Beta, Positron Emission Examples of Nuclear Decay Processes a emission (alpha) b- emission (beta) b+ emission (positron) Although beta emission involves electrons, those electrons come from the nucleus. Within the nucleus, a neutron decays into a proton and an electron. The electron is emitted, leaving behind a proton to replace the neutron, thus transforming the element. (A neutrino is also produced and emitted in the process. ) Herron, Frank, Sarquis, Schrader, Kulka, Chemistry, Heath Publishing, 1996, page 275

Nuclear Reactions First recognized natural transmutation of an element (Rutherford and Soddy, 1902) First artificial transmutation of an element (Rutherford, 1919) ? Discovery of the neutron (Chadwick, 1932) ? Discovery of nuclear fission (Otto Hahn and Fritz Strassman, 1939) Bailar, Chemistry, pg 361
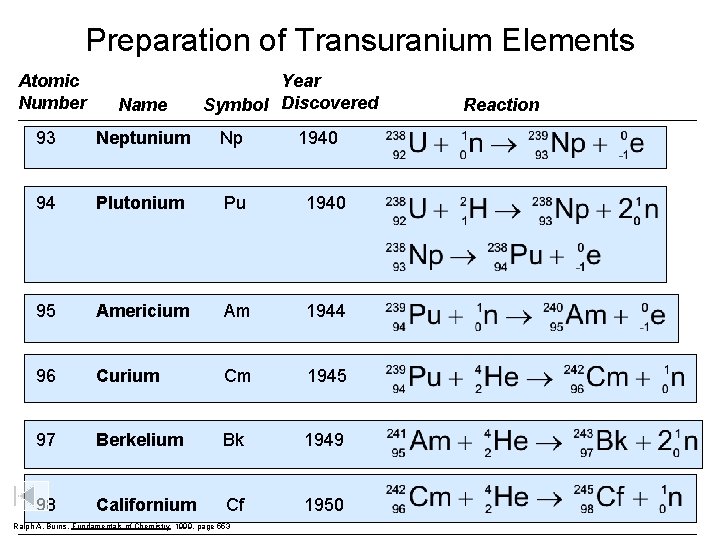
Preparation of Transuranium Elements Atomic Number Name Year Symbol Discovered 93 Neptunium Np 94 Plutonium Pu 1940 95 Americium Am 1944 96 Curium Cm 1945 97 Berkelium Bk 1949 98 Californium Cf 1950 Ralph A. Burns, Fundamentals of Chemistry 1999, page 553 1940 Reaction
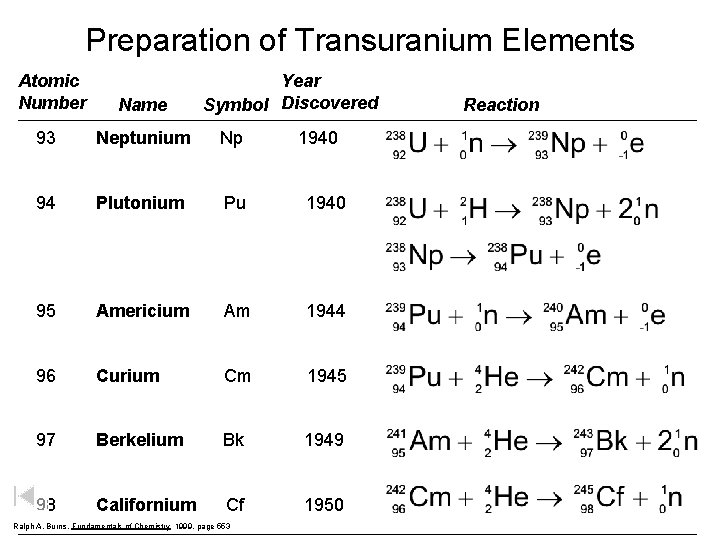
Preparation of Transuranium Elements Atomic Number Name Year Symbol Discovered 93 Neptunium Np 94 Plutonium Pu 1940 95 Americium Am 1944 96 Curium Cm 1945 97 Berkelium Bk 1949 98 Californium Cf 1950 Ralph A. Burns, Fundamentals of Chemistry 1999, page 553 1940 Reaction

Additional Transuranium Elements 99 100 101 102 103 104 105 106 107 108 109 110 111 112 114 116 118 Einsteinium Fermium Mendelevium Nobelium Lawrencium Rutherfordium Dubnium Seaborgium Bohrium Hassium Meitnerium Darmstadtium Unununium Ununbium Es Fm Md Nb Lr Rf Db Sg Bh Hs Mt Ds Uun Uub Uuq 1952 1955 1958 1961 1964 1970 1974 1981 1984 1988 1994 1996 1999 2002 2006 (Russia)
- Slides: 18