Upstream Process Fermentation Batch Culture Part II WarilaCCRI

Upstream Process Fermentation Batch Culture Part II Warila-CCRI

Mammalian Cell Scale-Up Translating the small quantities of cell culture used in the laboratory to a large production setting Often involves intermediate “stepping stone” volumes to work up to the final production quantities mammalian cells scale-up is different than for bacteria Not all products are successful at scaling-up

Cell Bank - LN 2 Cryogenic Storage Initiating a new working cell line begins by rapidly thawing a vial stored in liquid nitrogen (-160 to -174⁰C )

Inoculum Train If this little vial of cells is placed into a 15, 000 L culture vessel filled with media – would they thrive? Cells need buddies too! Cells like to be in contact with other cells and their metabolism conditions the media – gets them out of lag phase!

Inoculum Train initial cell density may be only 1 x 106 cells/ml or often less. Gradual expansion into larger and larger culture vessels can lead to high cell densities 4 x 106 cells/ml or more! That’s 4, 000 cells/ml! or 4 billion cells/L or 4 trillion cells/1000 L or 40 trillion cells/10, 000 L

Spinner Flask Bacteria may simply be grown in shake flasks but mammalian cells (such as CHO cells) can’t withstand the shear force of agitation and need gentle mixing with an agitator.

Alternatives Tissue culture flasks (T-flasks) or roller bottles are common in cells that are adherent (stick to surfaces)

Scale-Up Spinners of increasing size will be used over a period of several days until volume and density is sufficient to be transferred to a seed bioreactor (about 40 L).

Scale-Up Once the cells reach a target density of 4 x 106 cells/ml in 2 to 3 days, they will be used to inoculate a larger bioreactor of approximately 600 -800 L. The final production bioreactors are large typically 10, 000 to 15, 000 L in size.

Scale-Up

Production Once the cells are scaled up to production volume the cells need to be subcultured in the growth (continuous culture) vessels every few days… Why? Need to add fresh media, remove waste, reduce cell density!

Continuous Culture Depending on the cell density a calculated amount of culure (thousands of litres) is literally sent down the drain or to a second tank for production. Fresh media will be added to compensate for the volume removed. Typical target cell density is 1 x 106 cells/ml after subculture (a. k. a. splitting or passaging)

Perfusion Bioreactors There also perfusion bioreactors that trap the cells inside of hollow fibers. There is a continuous removal of old media and equal additions of fresh media. Advantages are there is no “subculture shock” thus no lag phase. Disadvantages are the complexities of the system.

Wave Bioreactors Wave bioreactors are becoming more common as they are disposable Less worry of maintenance and contamination, may actually be cheaper in long run. Can’t scale up to thousand liter volumes though…

Wave Bioreactors

Production To make a production batch, the growth vessels are subcultured (some not all of culture) is transferred to a production vessel by pressure. The production vessel then has a different type of media (production) added that doesn’t stimulate cell growth but makes the cell focus on protein production. Chemicals are sometimes added to stimulate protein production (sodium butyrate) and limit growth.

Harvest After a few days of production the protein is ready to be recovered or “harvested” Harvest activities often include filtration (microfiltration, diafiltration, and TFF –tangential flow filtration) Centrifugation is also an important part of harvesting product.

Harvest The advantages of using mammalian cells to produce protein: Bacteria do a poor job of proper folding of the protein produced – Mammalian cells (CHO in particular) do a great job with complex proteins Unlike bacteria, mammalian cells secrete the protein outside the cell making protein purification much easier allowing higher yields of product.

Harvest Disadvantages: Mammalian cells not as hardy, slow growing, prone to contamination, and may harbor viruses dangerous to humans.

Harvest Since Mammalian cells produce extracellular proteins our job here is simple… Separate the cells from the media. What do we keep the cells or the media?

Harvest The point of centrifugation skids are separate the cells from the media. Cells go down the drain and the media is recovered as it contains the protein. Production centrifuges are large, loud, but can go through CIP, SIP processes.

Filtration follows centrifugation and helps concentrate the product and reduce the volume of media from thousands of liters to hundreds or less. This is done by recirculating the media through (microfiltration) or across a filter (TFF) trapping proteins (retentate) but letting supernatant (fluid) though to waste
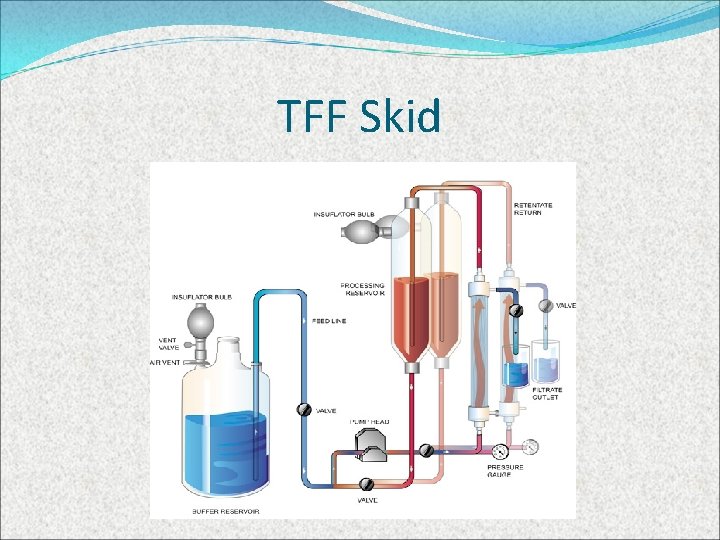
TFF Skid

TFF Skid

Centrifuges • A centrifuge is an instrument that rotates samples around a central axis • This centripetal acceleration separates substances in the sample according to density

Lab vs. Production Centrifuge

Centrifuges • The heavier or more dense the substance is the greater its sedimentation rate • The more dense the substance is the closer it will be to the bottom of the centrifuge tube.


Centrifuges • The force the centrifuge exerts on the sample is measured in g’s or the # of times greater than the force of the earth’s gravity • The speed of the centrifuge can also be monitored in RPM’s or revolutions per minute.

Centrifuges • Generally the greater the force exerted by the centrifuge and the longer the cycle is run the greater the separation • You need to be careful with delicate samples that they are not crushed or sheared apart • So… use only as much force as is necessary for separation

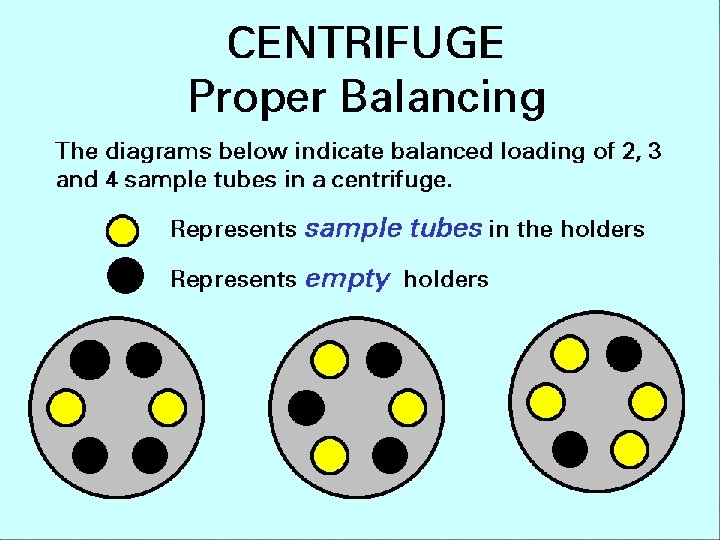
Centrifuges • The samples are loaded into numbered wells in the rotor of the centrifuge • The rotor needs to be balanced so it operates smoothly • Unbalanced centrifuges are dangerous, breaking samples, spilling chemicals and hurling pieces in addition to damaging the centrifuge
- Slides: 32