Upper respiratory tract infections acute bronchitis and communityacquired

Upper respiratory tract infections, acute bronchitis and community-acquired pneumonia in outpatient setting

Upper respiratory tract infections
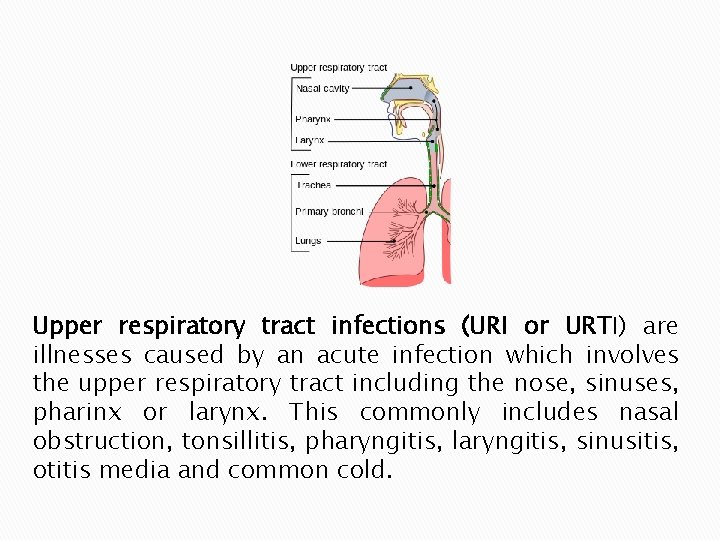
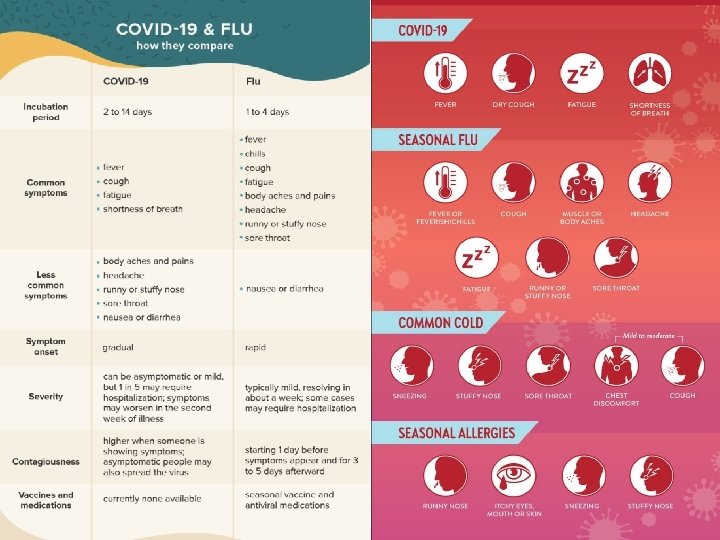
Upper respiratory tract infections (URI or URTI) are illnesses caused by an acute infection which involves the upper respiratory tract including the nose, sinuses, pharinx or larynx. This commonly includes nasal obstruction, tonsillitis, pharyngitis, laryngitis, sinusitis, otitis media and common cold.

Acute respiratory disease (ARD) or viral respiratory infections (VRI) are viral infectious disease of the upper respiratory tract that primarily affects the nose. The throat, sinuses, and voice box may also be affected. Without a doubt, ARD is the most well known upper respiratory tract infections (URI). Other types of URIs include sinusitis, pharyngitis, epiglottitis. Influenza, on the other hand, is not an upper respiratory infection because it is a systemic illness.
Viral respiratory infection

Causes of acute upper respiratory tract infection � Viruses ◦ rhinovirus (33%) ◦ adenovirus (20%) ◦ respiratory syncytial virus (15%) ◦ human coronavirus ◦ coxsackie virus ◦ parainfluenza virus ◦ bocavirus � Bacteria ◦ group A beta-hemolytic streptococcus (GABHS) ◦ chlamydia pneumonia ◦ streptococcus pneumonia ◦ haemophilus influenzae ◦ mycoplasma pneumonia

Rhinovirus � Epidemiology: wide spread (more in the North) � Pathway: droplet, contact � Incubation period: 1 -3 days � Symptoms: indisposition, low-grade fever, runny nose, pharyngitis, conjunctivitis, submaxillary lymphatic nodes enlargement � Diagnosis: polymerase chain reaction

Adenovirus � Epidemiology: world over � Pathway: droplet, contact � Incubation period: 6 days � Clinical forms: ◦ Rhinitis +pharyngitis + tonsillatis ◦ Acute bronchitis ◦ Pharyngitis + conjunctivitis ◦ Keratitis + conjunctivitis ◦ Viral pneumonia � Diagnosis: ◦ enzyme-linked immunosorbent assay (ELISA) ◦ polymerase chain reaction (PCR)

Coronavirus � Epidemiology: world over � Pathway: droplet, contact � Incubation period: 2 -14 days � Symptoms: chill, fever, headache, myalgias, fatigue, cough, dyspnea, bubbling rails, nausea, vomiting, diarrhea, acute respiratory distress syndrome � Diagnosis: polymerase chain reaction

Clinical features of ARD � Typical ◦ ◦ ◦ symptoms: rhinorrhea nasal congestion sneezing coughing sore throat � Accompany ◦ ◦ ◦ symptoms low-grade fever (3 days) headache myalgias fatigue loss of appetite
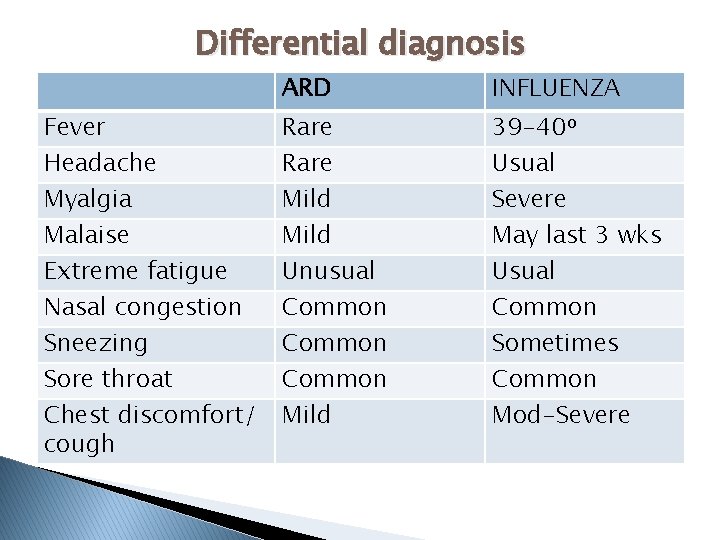
Differential diagnosis Fever Headache Myalgia Malaise Extreme fatigue Nasal congestion Sneezing Sore throat Chest discomfort/ cough ARD INFLUENZA Rare Mild Unusual Common Mild 39 -40 o Usual Severe May last 3 wks Usual Common Sometimes Common Mod-Severe

Diagnosis of ARD � Symptoms and a physical examination are all the doctor needs to diagnose the common cold � Usually, no blood tests or X-ray are necessary � During the physical examination, the doctor will pay careful attention to the head, neck, and chest � Examine the eyes, ears, throat, and chest to help determine if a bacterial source is causing the illness

Acute respiratory disease also known as a common cold is the third most common primary diagnosis in office visits. ARD is self-limited, usually lasting up to 10 days; therefore, management is directed at symptom relief rather than treating the infection. Multiple remedies, including complementary and alternative medicine products, over-thecounter (OTC) products, and prescription drugs, have been used to prevent and treat ARD symptoms.

Complementary and alternative medicine products

Therapies not effective for ARD � Antibiotics � Antihistamine monotherapy (sedating and nonsedating) � Codeine � Intranasal corticosteroids � Nasal irrigation with hypertonic or normal saline � Vitamin C

Therapies effective for ARD � Decongestants ◦ Ephedrine, pseudoephedrine ◦ Phenylephrin, naphazoline, oxymetazoline, tramazoline, xylometazoline � Antihistamines ◦ Diphenhydramine (dimedrol) ◦ Chloropyramine (suprastin) � Anticholinergics ◦ Ipratropium bromide orally inhaled or nasal form � Nonsteroid anti-inflammatory drugs ◦ Naproxen (naprosyn) ◦ Paracetomol � Guaifenesin (mucolitic) � Humidified air and fluid intake

Complementary and alternative medicine products for the common cold � Andrographis paniculata (Maha-tikta, India echinacea) ◦ 200 mg daily five days � Ocimum tenuiflorum (Holy basil, Tulasi, Tulsi) � Echinacea purpurea (solution of pressed juice of aerial parts and alcohol) ◦ 4 m. L twice daily eight weeks ◦ 20 drops every two hours on day 1, then 20 drops three times daily 10 days � Pelargonium Coldcare) sidoides (geranium) extract (Umcka ◦ 30 drops three times daily, alcohol root extract 10 days � Zinc acetate or gluconate ◦ Variable (lozenges contain between 4. 5 and 23. 7 mg of zinc). As long as symptoms persist

Complementary and alternative medicine products Pelargonium sidoides Andrographis paniculata

Complementary and alternative medicine products

Prophylaxis of ARD � Garlic ◦ Supplement with 180 mg of allicin 12 weeks � Vitamin C (dog rose, bell pepper) ◦ 0. 25 to 2 g daily 40 days to 28 weeks (generally around three months) � Frequent hand washing � Face mask
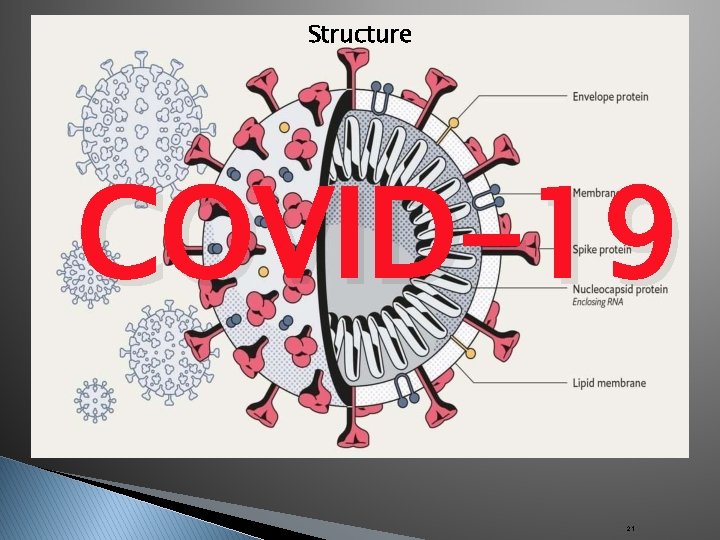
Structure COVID-19 21

Description ▪ Coronaviruses are positive-stranded RNA viruses with a crown-like appearance under an electron microscope (coronam is the Latin term for crown) due to the presence of spike glycoproteins on the envelope. ▪ SARS-Co. V-2 has round or elliptic and often pleomorphic form, and a diameter of approximately 60– 140 nm. Like other Coronaviruses, it is sensitive to ultraviolet rays and heat. ▪ Its single-stranded RNA genome contains 29891 nucleotides, encoding for 9860 amino acids. ▪ It has 89% nucleotide identity with bat SARS-like. Co. VZXC 21 and 82% with that of human SARS-Co. V. 22
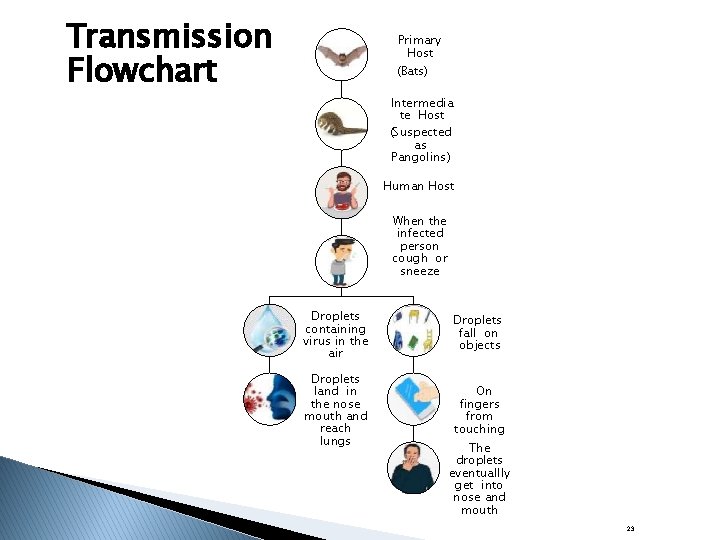
Transmission Flowchart Primary Host (Bats) Intermedia te Host (Suspected as Pangolins) Human Host When the infected person cough or sneeze Droplets containing virus in the air Droplets fall on objects Droplets land in the nose mouth and reach lungs On fingers from touching The droplets eventuallly get into nose and mouth 23

At highest risk for COVID-19 severe clinical course are people with: � Hypertension � Cardiovascular � Diabetes � Chronic � Cancer � Renal disease respiratory disease � Obesity � > 65 years � On immunosuppressive therapy

Suspect/Probable infected person Suspect - a person with acute respiratory illness (fever > 37, 50 C and at least one sign/symptom of respiratory disease (eg. cough, shortness of breath) AND � A history of travel to or residence in a country/area or territory reporting local transmission of COVID-19 disease during the 14 days prior to symptom onset OR � Probable - a person with any acute respiratory illness AND having being in contact with a confirmed COVID-19 case in the last 14 days prior to onset of symptoms OR � Probable - a person with severe acute respiratory infection {fever and at least one sign/symptom of respiratory disease (eg. , Cough, shortness of breath)} AND requiring hospitalisation AND with no other etiology that fully explains the clinical presentation OR � A case for whom testing for COVID-19 is inconclusive. � � Probable - a person with any acute respiratory illness AND typical changes in the lung (X-ray, CT) independent of testing for COVID-19

Who is a contact? �A CONTACT IS A PERSON WHO IS INVOLVED IN ANY OF THE FOLLOWING: ◦ PROVIDING DIRECT CARE WITHOUT PROPER PERSONAL PROTECTIVE EQUIPMENT (PPE) FOR COVID-19 PATIENTS ◦ STAYING IN THE SAME CLOSE ENVIRONMENT OF A COVID-19 PATIENT (INCLUDING WORKPLACE, CLASSROOM, HOUSEHOLD, GATHERINGS). ◦ TRAVELING TOGETHER IN CLOSE PROXIMITY (LESS THAN 1 M) WITH A SYMPTOMATIC PERSON WHO LATER TESTED POSITIVE FOR COVID-19.

High risk contacts: ▪ TOUCHED BODY FLUIDS OF THE PATIENT (RESPIRATORY TRACT SECRETIONS, BLOOD, VOMIT, SALIVA, URINE, FEACES) ▪ HAD DIRECT PHYSICAL CONTACT WITH THE BODY OF THE PATIENT, SHOOK HANDS, HUGGED OR TOOK CARE OF. ▪ TOUCHED OR CLEANED THE LINEN, CLOTHES, OR DISHES OF THE PATIENT. ▪ LIVED IN THE SAME HOUSEHOLD AS THE PATIENT. ▪ ANYONE IN CLOSE PROXIMITY (LESS THAN ONE METER) OF THE CONFIRMED CASE WITHOUT PRECAUTIONS. ▪ PASSENGER TRAVELING IN CLOSE PROXIMITY (LESS THAN ONE METER) FOR MORE THAN 6 HOURS WITH A SYMPTOMATIC PERSON WHO LATER TESTED POSITIVE FORCOVID-19.

Low risk contacts: • SHARED THE SAME SPACE (SAME CLASS FOR SCHOOL/WORKED IN SAME ROOM/SIMILAR AND NOT HAVING A HIGH RISK EXPOSURE TO CONFIRMED OR SUSPECT CASE OFCOVID-19). • TRAVELLED IN SAME ENVIRONMENT (BUS/TRAIN/FLIGHT/ANY MODE OF TRANSIT) BUT NOT HAVING A HIGH-RISK EXPOSURE.

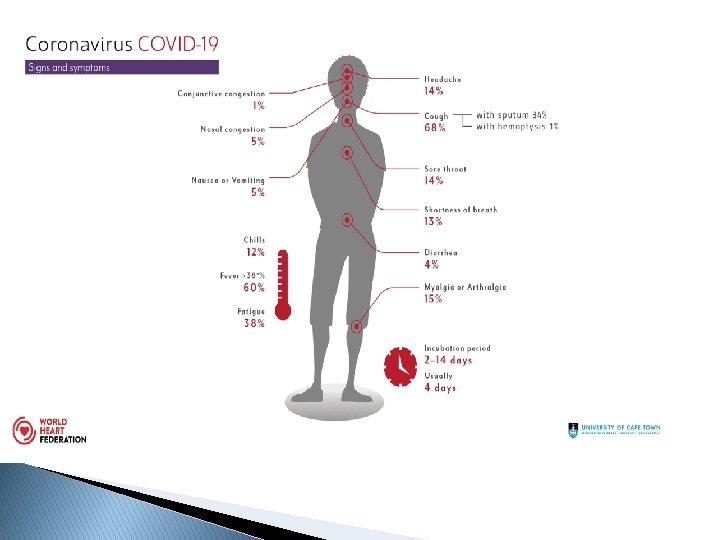
SYMPTOMS � Common symptoms include fever, cough, fatigue, shortness of breath and loss of smell � While the majority of cases result in mild symptoms, some progress to viral pneumonia, multi-organ failure or cytokine storm. � Concerned symptoms include difficulty breathing, persistent chest pain, confusion, difficulty waking and bluish skin.

Effects in other organs ▪ The virus can also enter the bloodstream, and may be able to infect the gastrointestinal system, causing symptoms like diarrhea and indigestion. ▪ The infection can also directly damage organs including the heart, kidneys, and liver, and cause bone marrow to become inflamed. Small blood vessels may also be vulnerable to inflammation. ▪ The body’s own immune response to the infection cause inflammation and organ malfunction. It is still unclear if the brain is affected. 30

Classification (Russian) � Mild � Moderate � � ◦ T < 380 C, cough, fatigue, sore throat ◦ Absents of moderate/severe criteria ◦ T > 380 C ◦ ◦ ◦ RR > 22 per min Exertion dyspnea Pneumonia (CT scan) Sp. O 2 < 95% C-reactive protein > 10 vg/l ◦ ◦ ◦ RR > 30 per min Sp. O 2 < 93% Pa. O 2 /Fi. O 2 ≤ 300 мм рт. ст. Pneumonia progression (x-ray, CT scan) Loss of consciousness BP < 90/60 mm. Hg Severe (15%) Very severe (5% - China) ◦ Acute respiratory failure (artificial ventilation) ◦ Septic shock ◦ polyorganic insufficiency

Risk factors and prognostic determinants Higher body temperature is associated with more severe disease and higher fatality. � Lymphopenia: appeared to be a good predictor of severe disease and worse outcomes, a lymphocyte count of less than 2000/cu. mm was associated with poorer outcomes. � Neutrophil/lymphocyte ratio greater than 2 was associated with higher mortality � Lactate dehydrogenase (LDH)>245 U/L � HS-Cardiac Troponin>28 ng/ml � Prothrombin time>16 s � Serum ferritin>300 μg/L � ↑D-Dimer � CRP>100 mg/dl �

Acute respiratory distress syndrome � ARDS is a type of respiratory failure characterized by rapid onset of widespread inflammation in the lungs. Symptoms include shortness of breath, and bluish skin. � The underlying mechanism involves diffuse injury of cells which form alveolar barrier, surfactant dysfunction, activation of the immune system, and dysfunction of the body's regulation of blood clotting. � Diagnosis is based on a Pa. O 2/Fi. O 2 ratio of less than 300.

Management �Mild cases – treated at home �Moderate/severe cases – hospitalization to isolation ward �Very severe cases - hospitalization to intensive care unit (require artificial ventilation)

Diagnosis � WHO recommends collecting specimens from both the upper respiratory tract (naso and oropharyngeal samples) and lower respiratory tract such as expectorated sputum, endotracheal aspirate or broncho-alveolar lavage. � Amplification of the genetic material extracted from the saliva or mucus sample is through a reverse polymerase chain reaction (RT-PCR), which involves the synthesis of a double-stranded DNA molecule from an RNA

Priority system for diagnostic testing for COVID-19 (USA): � Priority 1: Hospitalized patients and symptomatic health care workers (to reduce the risk of nosocomial infections and maintain the health care system). � Priority 2: Individuals with symptoms who live in long -term care facilities, who are aged ≥ 65 years, or who have underlying conditions, and symptomatic first responders (to ensure those at highest risk of complications of infection are rapidly identified and triaged). � Priority 3: In communities experiencing high COVID 19 hospitalizations, critical infrastructure workers and other individuals with symptoms, health care workers and first responders, and individuals with mild symptoms (to decrease community spread and ensure the health of essential workers).

Treatment � There is no specific antiviral treatment recommended for COVID-19, and no vaccine is currently available. � The treatment is symptomatic, and oxygen therapy represents the major treatment intervention for patients with severe infection. � Mechanical ventilation may be necessary in cases of respiratory failure refractory to oxygen therapy, whereas hemodynamic support is essential for managing septic shock

Drugs under research (WHO) ▪ While studies are on, and different countries try various combinations to treat its patients, the WHO launched a multicountry clinical trial to test four drug combinations that have yielded results – an experimental antiviral drug called remdesivir, the antimalarial drug chloroquine (or the related hydroxychloroquine + azitromycin), a combination of two HIV drugs (lopinavir/ritonavir), and those same two HIV drugs along with the anti-inflammatory interferon beta. 40

Drugs (USA: COVID-19 treatment guidelines) There are insufficient clinical data to recommend either for or against using chloroquine or hydroxychloroquine for the treatment of COVID-19. � Antiviral agent remdesivir is recommended for the treatment of COVID-19 in hospitalized patients with severe disease, defined as Sp. O 2 ≤ 94% on ambient air (at sea level), requiring supplemental oxygen, mechanical ventilation, or extracorporeal membrane oxygenation. � Remdesivir does not recommended for the treatment of mild or moderate COVID-19 outside the setting of a clinical trial. � Except in the context of a clinical trial, the Panel recommends against the use of the following drugs for the COVID-19 treatment : ◦ The combination of hydroxychloroquine plus azithromycin because of the potential for toxicities. ◦ Lopinavir/ritonavir or other HIV protease inhibitors because of unfavorable pharmacodynamics and negative clinical trial data. �

Drugs (USA: COVID-19 treatment guidelines) � There are insufficient clinical data to recommend either for or against the use of convalescent plasma or hyperimmune immunoglobulin for the treatment of COVID-19. � There are insufficient clinical data to recommend either for or against the use of the following agents for the treatment of COVID-19: ◦ Interleukin-6 inhibitors (e. g. , sarilumab, siltuximab, tocilizumab) ◦ Interleukin-1 inhibitors (e. g. , anakinra) � Except in the context of a clinical trial, the Panel recommends against the use of other immunomodulators, such as: ◦ Interferons, because of lack of efficacy in treatment of severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS) and toxicity. ◦ Janus kinase inhibitors (e. g. , baricitinib), because of their broad immunosuppressive effect.

Drugs (Covid-19 temporary recommendations. Russia) � Low molecular weight heparins are indicated for all in-patients with COVID-19 for normalization of microcirculation and inflammation reduction. ◦ Low doses for prophylactic ◦ Full doses for thrombosis treatment ◦ Anticoagulants treatment is controlled by D-dimer, fibrinogen, prothrombin, platelet count. ◦ USA guidelines recommend the use of antithrombotic agents for the prevention of venous thromboembolic events in hospitalized patients with COVID-19. � Rehydration (oral, i/v- slowly) therapy should not exceed 1, 5 -2 l/day ◦ May be in combination with furosemide for prophylaxis of brain and pulmonary edema. � Antibiotics in case of concomitant bacterial infection

Drugs (USA: COVID-19 treatment guidelines) � The Panel recommends against the routine use of systemic corticosteroids for the treatment of mechanically ventilated patients with COVID-19 without acute respiratory distress syndrome (ARDS). � For mechanically ventilated patients with ARDS, there is insufficient evidence to recommend for or against the use of systemic corticosteroids. � For adults with COVID-19 and refractory shock, the Panel recommends using low-dose corticosteroid therapy (i. e. , shock reversal) over no corticosteroids.

Drugs (USA: COVID-19 treatment guidelines) � Oral or inhaled corticosteroid therapy used prior to COVID-19 diagnosis for another underlying condition (e. g. , primary or secondary adrenal insufficiency, rheumatological diseases) should not be discontinued. � Persons with COVID-19 who are taking NSAIDs for a co-morbid condition should continue therapy as previously directed by their physician. � The Panel recommends that there be no difference in the use of antipyretic strategies (e. g. , with acetaminophen or NSAIDs) between patients with or without COVID-19.

Drugs � Persons (USA: COVID-19 treatment guidelines) with COVID-19 who are prescribed ACE inhibitors or ARBs for cardiovascular disease (or other indications) should continue these medications. � The COVID-19 Treatment Guidelines Panel (the Panel) recommends against the use of ACE inhibitors or ARBs for the treatment of COVID-19 outside of the setting of a clinical trial. � Persons with COVID-19 who are prescribed statin therapy for the treatment or prevention of cardiovascular disease should continue these medications. � The Panel recommends against the use of statins for the treatment of COVID-19 outside of the setting of a clinical trial.

Acute bronchitis

Lower respiratory tract infection is an acute illness (present for 21 days or less), usually with cough as the main symptom, and with at least 1 other lower respiratory tract symptom (such as fever, sputum production, breathlessness, wheeze or chest discomfort or pain) and no alternative explanation (such as sinusitis or asthma). Pneumonia, acute bronchitis and exacerbation of chronic obstructive airways disease are included in this definition.

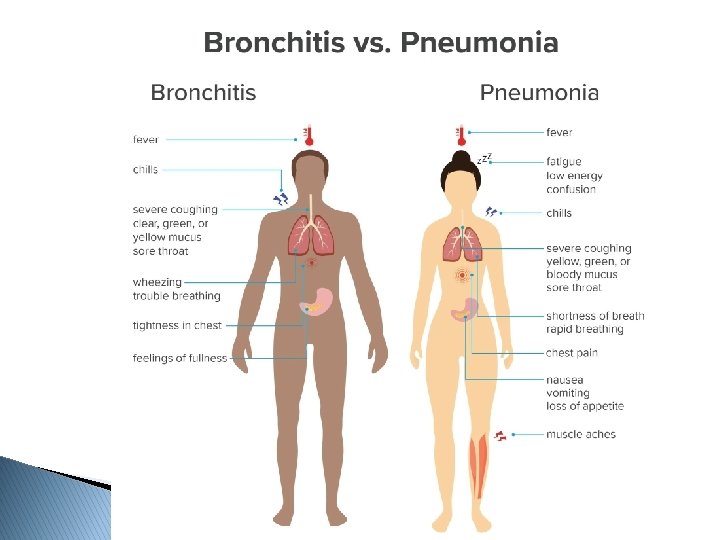
Acute bronchitis, also known as a chest cold, is short-term inflammation of the bronchi (large and medium-sized airways) of the lungs. The most common symptom is a cough. Other symptoms include coughing up mucus, wheezing, shortness of breath, fever, and chest discomfort.
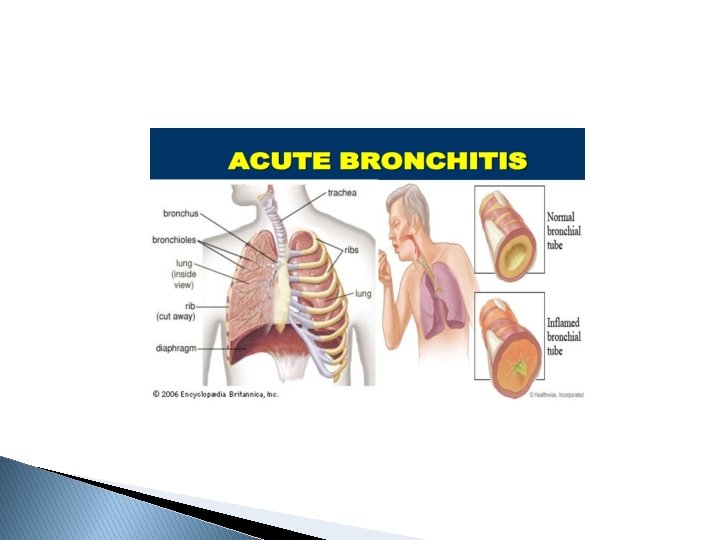

Cough is the most common illness-related reason for ambulatory care visits. Acute bronchitis is a clinical diagnosis characterized by cough due to acute inflammation of the trachea and large airways without evidence of pneumonia. Pneumonia should be suspected in patients with tachypnea, tachycardia, dyspnea, or lung findings suggestive of pneumonia, and radiography is warranted. The cough associated with acute bronchitis typically lasts about two to three weeks. Acute bronchitis is usually caused by viruses, and antibiotics are not indicated in patients without chronic lung disease or complications.

Clinical features of acute bronchitis � Typical ◦ ◦ symptoms: expectorating cough wheezing sore throat runny nose � Accompany ◦ ◦ symptoms fever (5 -7 days) chest pain fatigue malaise

Most common differential diagnosis of acute cough � Acute bronchitis � Pneumonia � Allergic rhinitis � Asthma � Chronic obstructive pulmonary disease exacerbation � ARD � Congestive heart failure exacerbation � Gastroesophageal reflux disease � Malignancy

Diagnosis of acute bronchitis � Obligatory ◦ ◦ ◦ anamnesis and complaints objective examination thermometry common blood count fluorography ◦ ◦ ◦ lung X-ray (fever > 3 days) sputum analysis spirometry C-reactive protein consultation by phthisiologist (tuberculosis specialist) � Additional

Key recommendations for acute bronchitis � Avoid prescribing antibiotics for uncomplicated acute bronchitis � Over-the-counter cough medications containing antihistamines and antitussives should not be used in children younger than four years because of the high potential for harm � Consider using dextromethorphan, guaifenesin, or honey to manage acute bronchitis symptoms � Avoid using beta 2 agonists for the routine treatment of acute bronchitis unless wheezing is present � Employ strategies to reduce antibiotic use, such as asking patients to hold an antibiotic prescription for a set amount of time

Expectorants Guaifenesin

Antitussives Dextromethorphan

Complementary and alternative medicine products for the acute bronchitis � Echinacea � Pelargonium � Dark honey in children

Complementary and alternative medicine products
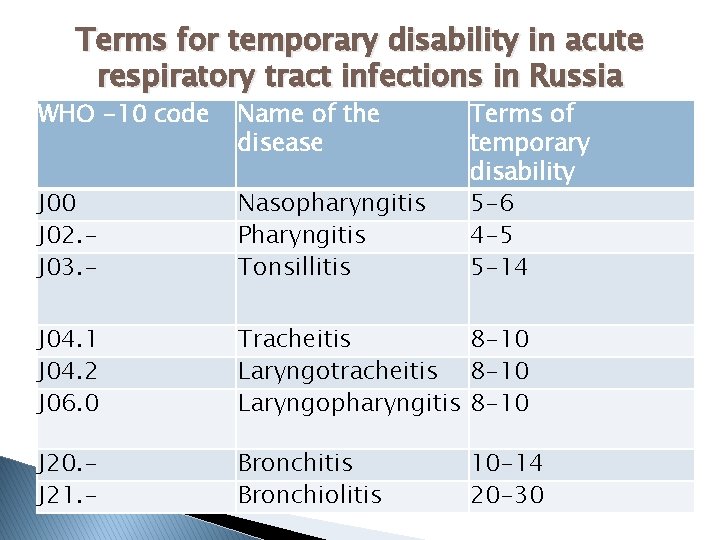
Terms for temporary disability in acute respiratory tract infections in Russia WHO -10 code Name of the disease J 00 J 02. - J 03. - Nasopharyngitis Pharyngitis Tonsillitis J 04. 1 J 04. 2 J 06. 0 Tracheitis Laryngotracheitis Laryngopharyngitis Bronchitis Bronchiolitis J 20. - J 21. - Terms of temporary disability 5 -6 4 -5 5 -14 8 -10 10 -14 20 -30

Community-acquired pneumonia (CAP)

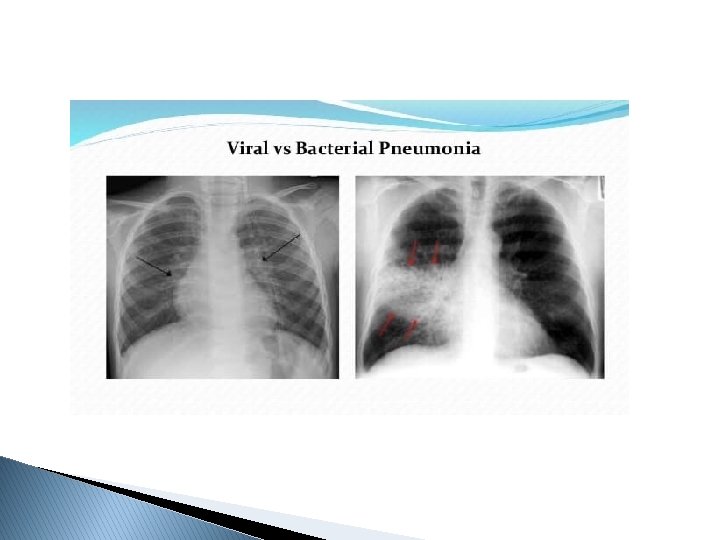
Community-acquired pneumonia (CAP) is pneumonia that is acquired outside hospital. Pneumonia that develops in a nursing home resident is included in this definition. When managed in hospital the diagnosis is usually confirmed by chest X‑ray.

COVID – 19 Pneumonia � Type ◦ ◦ Low Low � Type ◦ ◦ L is characterized by elastance (i. e. , high compliance) ventilation to perfusion ratio, lung weight recruitability H is characterized by High elastance right to left shunt lung weight recruitability

Diagnostic tests (1) � Physical exam: ◦ Crackles or rales, bronchial breath sounds, hypoxemia, tachypnic ◦ Signs/symptoms of cough, fever, sputum production, pleuritic chest pain � Chest radiograph: ◦ Observe apparent lobar or bilateral infiltrates with or without microbiological evidence ◦ Hospitalized for suspected pneumonia but negative chest radiograph: may receive empiric antibiotics with repeat chest radiograph 24− 48 hrs later � CT scan � Pulse oximetry
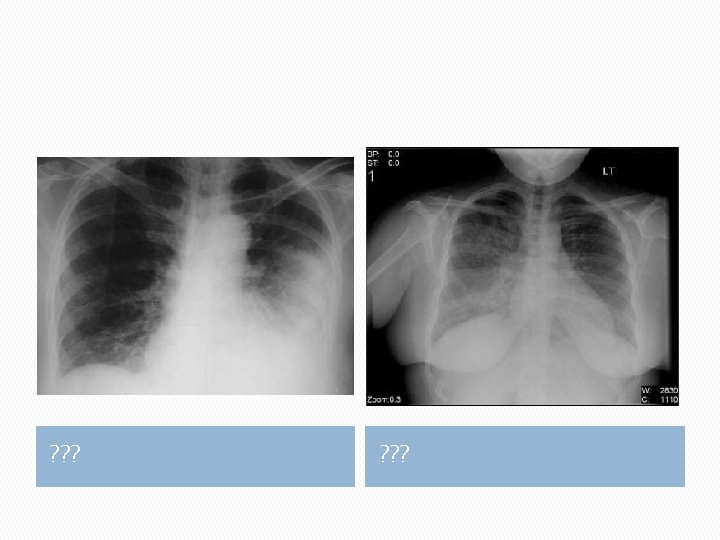

Abnormalities seen in computed tomography (CT) in COVID-19: �Bilateral peripheral ground-glass opacities with the development of areas of consolidation later in the clinical course. �Imaging may be normal early in infection and can be abnormal in the absence of symptoms
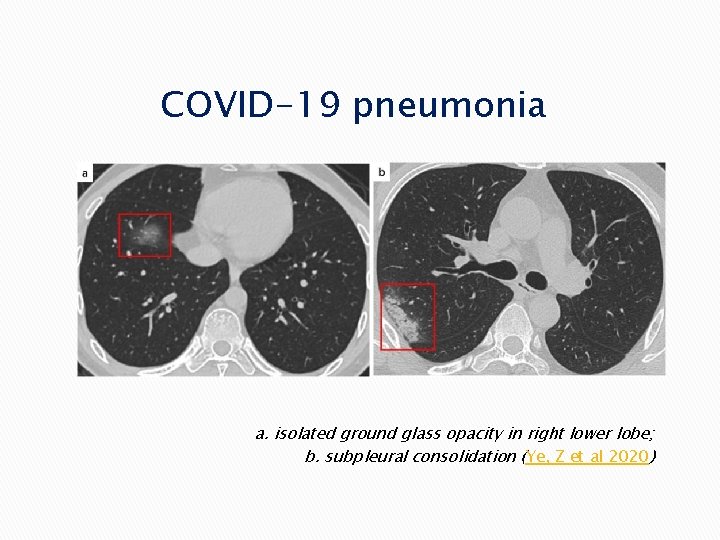
COVID-19 pneumonia a. isolated ground glass opacity in right lower lobe; b. subpleural consolidation (Ye, Z et al 2020)
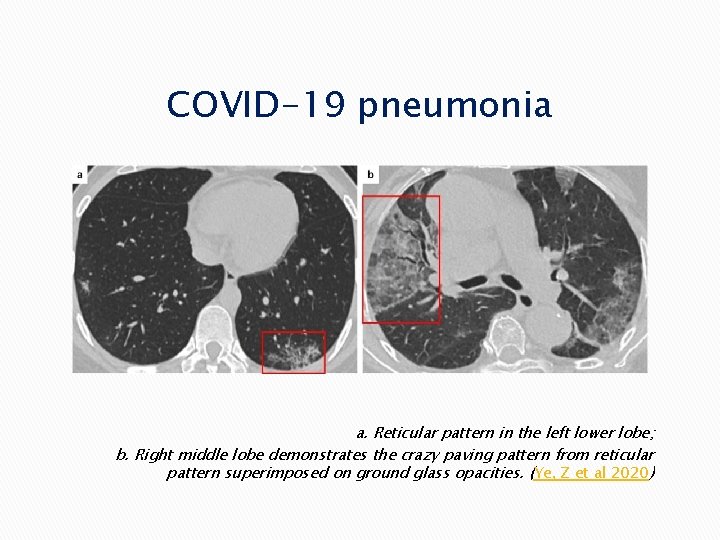
COVID-19 pneumonia a. Reticular pattern in the left lower lobe; b. Right middle lobe demonstrates the crazy paving pattern from reticular pattern superimposed on ground glass opacities. (Ye, Z et al 2020)

Diagnostic tests (2) � We recommend not obtaining sputum Gram stain and culture and blood cultures routinely in adults with CAP managed in the outpatient setting � We recommend obtaining pretreatment Gram stain and culture of respiratory secretions and blood cultures in adults with CAP managed in the hospital setting who: ◦ are classified as severe CAP, especially if they are intubated ◦ are being empirically treated for MRSA or P. aeruginosa ◦ were previously infected with MRSA or P. aeruginosa, especially those with prior respiratory tract infection ◦ were hospitalized and received parenteral antibiotics, whether during the hospitalization event or not, in the last 90 days

Diagnostic tests (3) suggest not routinely testing urine for Pneumococcal antigen in adults with CAP, except in adults with severe CAP. � We suggest not routinely testing urine for Legionella antigen in adults with CAP, except � We ◦ in cases where indicated by epidemiological factors, such as association with a Legionella outbreak or recent travel ◦ in adults with severe CAP. � When influenza viruses are circulating in the community, we recommend testing for influenza with a rapid influenza molecular assay (i. e. , influenza nucleic acid amplification test), which is preferred over a rapid influenza diagnostic test (i. e. , antigen test). � We recommend that empiric antibiotic therapy should be initiated in adults with clinically suspected and radiographically confirmed CAP regardless of initial serum procalcitonin level

Indications for hospitalization � In addition to clinical judgment, we recommend that clinicians use a validated clinical prediction rule for prognosis, preferentially the Pneumonia Severity Index (PSI) over the CURB-65 to determine the need for hospitalization in adults diagnosed with CAP. � We recommend direct admission to an ICU for patients with hypotension requiring vasopressors or respiratory failure requiring mechanical ventilation. � For patients not requiring vasopressors or mechanical ventilator support, we suggest using the IDSA/ATS 2007 minor severity criteria together with clinical judgment to guide the need for higher levels of treatment intensity.

Pneumonia. CURB 65 score � CURB 65 Score is calculated by giving 1 Point for each of the following prognostic features: ◦ Confusion (abbreviated Mental Test score 8 or less, or new disorientation in person, place or time) ◦ Raised Blood Urea Nitrogen (over 7 mmol/litre) ◦ Raised Respiratory Rate (30 breaths per minute or more) ◦ Low Blood Pressure (Diastolic 60 mm. Hg or less, or Systolic less than 90 mm. Hg) ◦ Age 65 years or more

CURB 65 � Use Clinical Judgment in conjunction with the CURB 65 Score to Guide Management of Community Acquired Pneumonia as follows: ◦ Consider Home-Based Care for patients with a CURB 65 Score of 0 -1 ◦ Consider Hospital-Based Care for patients with a CURB 65 Score of 2 or more ◦ Consider Intensive Care assessment for patients with a CURB 65 Score of 3 or more

PSI (Pneumonia Severity Index) algorithm Presence of: Over 50 years of age Yes/No Altered mental status Yes/No Pulse ≥ 125/minute Yes/No Respiratory rate >30/minute Yes/No History of: Systolic blood pressure <90 mm Hg Temperature <35 °C or ≥ 40 °C Yes/No Neoplastic disease Yes/No Congestive heart failure Yes/No Cerebrovascular disease Renal disease Liver disease Yes/No If any "Yes", then proceed to Step 2 If all "No" then assign to Risk Class I
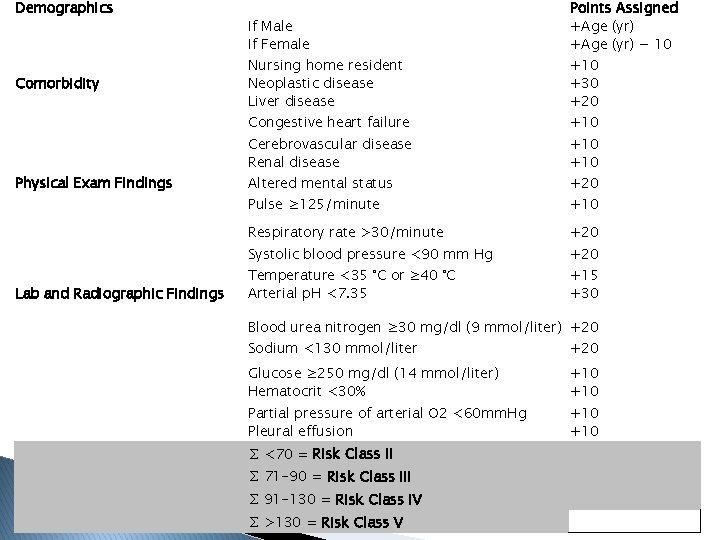
Demographics Comorbidity Physical Exam Findings Lab and Radiographic Findings If Male If Female Points Assigned +Age (yr) − 10 Congestive heart failure +10 Nursing home resident Neoplastic disease Liver disease Cerebrovascular disease Renal disease Altered mental status Pulse ≥ 125/minute +10 +30 +20 +10 Respiratory rate >30/minute +20 Systolic blood pressure <90 mm Hg +20 Temperature <35 °C or ≥ 40 °C Arterial p. H <7. 35 +15 +30 Blood urea nitrogen ≥ 30 mg/dl (9 mmol/liter) +20 Sodium <130 mmol/liter +20 Glucose ≥ 250 mg/dl (14 mmol/liter) Hematocrit <30% +10 Partial pressure of arterial O 2 <60 mm. Hg Pleural effusion +10 ∑ <70 = Risk Class II ∑ 71 -90 = Risk Class III ∑ 91 -130 = Risk Class IV ∑ >130 = Risk Class V

PSI (Pneumonia Severity Index) �A Risk Class I or Risk Class II pneumonia patient can be sent home on oral antibiotics. � A Risk Class III patient, after evaluation of other factors including home environment and followup, may either: � be sent home with oral antibiotics � be admitted for a short hospital stay with antibiotics and monitoring. � Patients with Risk Class IV-V pneumonia patient should be hospitalized for treatment.

Criteria for Defining Severe Community-acquired Pneumonia Validated definition includes either one major criterion or three or more minor criteria Minor criteria Respiratory rate ≥ 30 breaths/min Pa. O 2/Fi. O 2 ratio ≤ 250 Multilobar infiltrates Confusion/disorientation Uremia (blood urea nitrogen level ≥ 20 mg/dl) Leukopenia* (white blood cell count < 4, 000 cells/μl) Thrombocytopenia (platelet count < 100, 000/μl) Hypothermia (core temperature < 36°C) Hypotension requiring aggressive fluid resuscitation Major criteria Septic shock with need for vasopressors Respiratory failure requiring mechanical ventilation

Community-acquired pneumonia: treatment in outpatient setting � For healthy outpatient adults without comorbidities listed below or risk factors for antibiotic resistant pathogens, we recommend: � amoxicillin 1 g three times daily, or � doxycycline 100 mg twice daily, or � macrolide (azithromycin 500 mg on first day then 250 mg daily or clarithromycin 500 mg twice daily or clarithromycin extended release 1, 000 mg daily) only in areas with pneumococcal resistance to macrolides <25%

Community-acquired pneumonia: treatment in outpatient setting outpatient adults with comorbidities such as chronic heart, lung, liver, or renal disease; diabetes mellitus; alcoholism; malignancy; or asplenia we recommend: � Combination therapy: � For ◦ amoxicillin/clavulanate 500 mg/125 mg three times daily, or amoxicillin/clavulanate 875 mg/125 mg twice daily, or 2, 000 mg/125 mg twice daily, or a cephalosporin (cefpodoxime 200 mg twice daily or cefuroxime 500 mg twice daily); AND ◦ macrolide (azithromycin 500 mg on first day then 250 mg daily, clarithromycin [500 mg twice daily or extended release 1, 000 mg once daily]), or doxycycline 100 mg twice daily; OR � Monotherapy: ◦ respiratory fluoroquinolone (levofloxacin 750 mg daily, moxifloxacin 400 mg daily, or gemifloxacin 320 mg daily).

Community-acquired pneumonia: treatment in outpatient setting suggest that antiinfluenza treatment be prescribed for adults with CAP who test positive for influenza in the outpatient setting, independent of duration of illness before diagnosis � We recommend that standard antibacterial treatment be initially prescribed for adults with clinical and radiographic evidence of CAP who test positive for influenza in the inpatient and outpatient settings � We
Treatment duration and follow-up � We recommend that the duration of antibiotic therapy should be guided by a validated measure of clinical stability (resolution of vital sign abnormalities [heart rate, respiratory rate, blood pressure, oxygen saturation, and temperature], ability to eat, and normal mentation), and antibiotic therapy should be continued until the patient achieves stability and for no less than a total of 5 days � In adults with CAP whose symptoms have resolved within 5 to 7 days, we suggest not routinely obtaining follow-up chest imaging
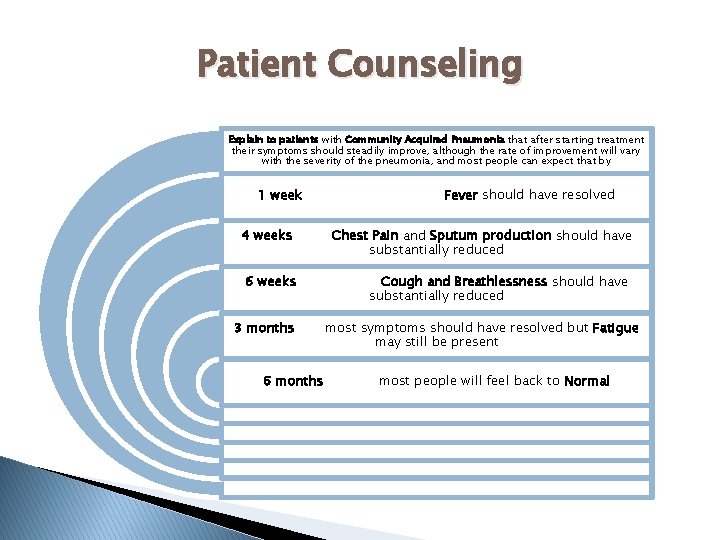
Patient Counseling Explain to patients with Community Acquired Pneumonia that after starting treatment their symptoms should steadily improve, although the rate of improvement will vary with the severity of the pneumonia, and most people can expect that by 1 week Fever should have resolved 4 weeks Chest Pain and Sputum production should have substantially reduced 6 weeks Cough and Breathlessness should have substantially reduced 3 months 6 months most symptoms should have resolved but Fatigue may still be present most people will feel back to Normal

Prevention � Vaccination against Streptococcus pneumonia and Influenza Virus �Vaccination status should be assessed at the time of admission �Vaccination should be performed either at Discharge or during Out Patient follow up if needed � Risk Factor Modifications �Smoking and Alcohol abuse Cessation

COVID-19 protection for each of us


HOW TO USE A MASK ● ● ● Before wearing a mask, wash your hands with an alcohol-based disinfectant or with soap and water. Cover your mouth and nose with the mask and make sure the mask is firmly pressed against your face. Do not touch the mask while you are wearing it; if you do, wash your hands with an alcohol-based disinfectant or with soap and water afterward. Replace the mask as soon as it gets wet and do not reuse disposable masks. Remove the mask from behind (do not touch its front side); throw it away in a closed container and then wash your hands with an alcohol-based disinfectant or with soap and water.

Take care!
- Slides: 91