Update on ABSORB EXTEND Study Preliminary Data from

Update on ABSORB EXTEND Study Preliminary Data from ABSORB EXTEND: A Report of the 6 -month Clinical Outcomes from the First 200 Patients Enrolled Alexandre Abizaid, MD, Ph. D, FACC Antonio L. Bartorelli MD; Robert Whitbourn MD; Lee Clark Ph. D; Bernard Chevalier MD; Karine Miquel-Hebert Ph. D; Xiaolin Li MS; Robert Jan Van Geuns MD, Ph. D; Didier Carrié MD, Ph. D; Pieter C. Smits MD, Ph. D; John A. Ormiston MBCh. B, Ph. D; Robert Mc. Greevy Ph. D; Ashok Seth MD; Patrick W. Serruys MD, Ph. D on behalf of the ABSORB EXTEND Investigators

Alexandre Abizaid, MD, Ph. D § Consulting Fees: § Abbott Vascular § BSC

Conflicts of Interest Alexandre Abizaid; Instituto Dante Pazzanese, Sao Paulo, Brazil Participation in the Advisory Board for Boston Scientific and Abbott Vascular. Antonio L. Bartorelli; Centro Cardiologico Monzino, IRCCS, University of Milan, Italy Participation in the Advisory Board for Abbott Vascular. Robert Whitbourn MD; St. Vincent’s Hospital, Fitzroy, Australia Participation in the Advisory Board for Abbott Vascular. Lee Clark, Xiaolin Li, and Robert Mc. Greevy; Abbott Vascular, Santa Clara, CA Karine Miquel-Hebert; Abbott Vascular, Diegem, Belgium Employees of Abbott Vascular. Bernard Chevalier; Institut Jacques Cartier, Massy, France Consultant for Abbott Vascular, Biotronik, Terumo, and Medtronic. Recipient of research grant from Cordis. Robert Jan Van Geuns; Thoraxcenter, Erasmus Medical Center, Rotterdam, The Netherlands No Conflicts of Interest Didier Carrié; Hôpital Rangueil, Toulouse, France No Conflicts of Interest Pieter C. Smits; Maasstad Ziekenhuis, Rotterdam, The Netherlands Recipient of research grants from Cordis, Boston Scientific and Terumo. Recipient of travel and speaking fees from Abbott Vascular. A consultant for Blue Medical. John A. Ormiston; Mercy Angiography, Mercy Hospital, Auckland, New Zealand Participation in the Advisory Board for Abbott Vascular and Boston Scientific with minor honoraria. Ashok Seth; Escorts Heart Institute and Research Centre, New Delhi, India Participation in the Advisory Board for Abbott Vascular for BVS. Patrick W. Serruys; Thoraxcenter, Erasmus Medical Center, Rotterdam, The Netherlands No Conflicts of Interest
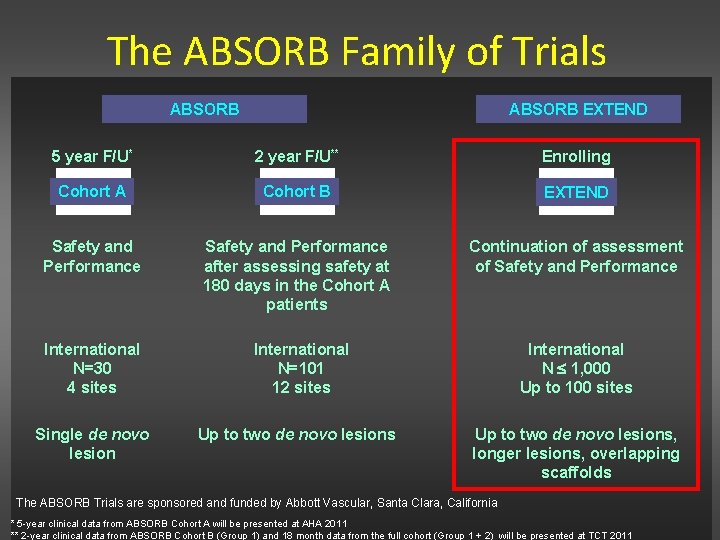
The ABSORB Family of Trials ABSORB EXTEND 5 year F/U* 2 year F/U** Enrolling Cohort A Cohort B EXTEND Safety and Performance after assessing safety at 180 days in the Cohort A patients Continuation of assessment of Safety and Performance International N=30 4 sites International N=101 12 sites International N 1, 000 Up to 100 sites Single de novo lesion Up to two de novo lesions, longer lesions, overlapping scaffolds The ABSORB Trials are sponsored and funded by Abbott Vascular, Santa Clara, California * 5 -year clinical data from ABSORB Cohort A will be presented at AHA 2011 ** 2 -year clinical data from ABSORB Cohort B (Group 1) and 18 month data from the full cohort (Group 1 + 2) will be presented at TCT 2011

Background • Results from ABSORB Cohort A continue to be encouraging, with only 1 MACE and no scaffold thrombosis through 5 years of follow- up*. • The results from ABSORB Cohort B, performed with an ABSORB device with improved mechanical properties, confirm the performance and safety established in the Cohort A investigation: Ø The 12 -month angiographic results from Cohort B (Group 1) demonstrated an in-segment late loss of 0. 16 mm Ø MACE rate of 6. 9% for the full cohort of 101 subjects at 12 months** (3 Non-Q wave MI, 4 ID-TLR) Ø No scaffold thrombosis events out to 12 months * 5 -year clinical data from ABSORB Cohort A will be presented at AHA 2011 ** 2 -year clinical data from ABSORB Cohort B (Group 1) and 18 month data from the full cohort (Group 1 + 2) will be presented at TCT 2011

ABSORB EXTEND Non-Randomized, Single-Arm, Continued Access Trial 1000 subjects Up to 100 International sites Clinical follow-up 30 d 6 mo 12 mo 24 mo 36 mo MSCT follow-up (n=100) OCT follow-up (n=50) Study Objective: Endpoints: Treatment: Continued Access trial. FPI*: Jan 11, 2010 No hypothesis-testing, typical PCI clinical endpoints Up to 2 de novo lesions in different epicardial vessels Planned overlapping allowed in lesions >22 and ≤ 28 mm Device Sizes: Scaffold diameters: 2. 5, 3. 0 mm Scaffold lengths: 18, 28 mm Note: The ABSORB EXTEND Rev 3. 0 protocol is currently pending approval. *FPI: First Patient In
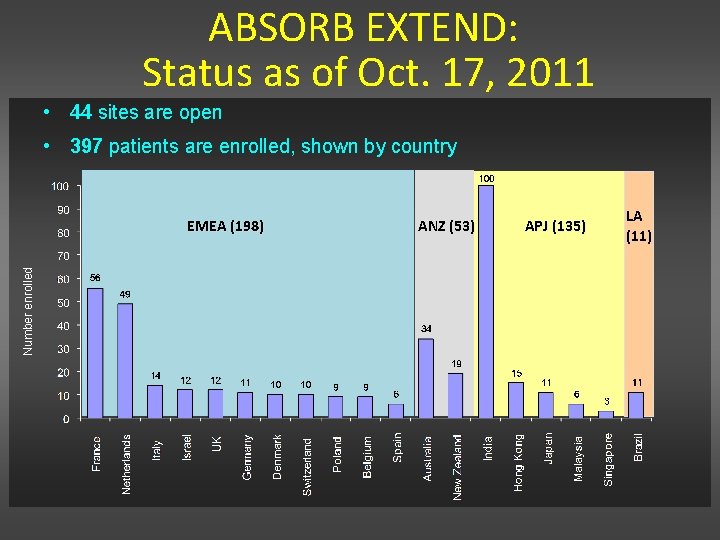
ABSORB EXTEND: Status as of Oct. 17, 2011 • 44 sites are open • 397 patients are enrolled, shown by country Number enrolled EMEA (198) ANZ (53) APJ (135) LA (11)

ABSORB EXTEND Trial Management • Principal Investigator: • Regional Co-Principal Investigators: Alexandre Abizaid, MD, Ph. D; San Paulo, Brazil Antonio Bartorelli, MD; Milano, Italy; Robert Whitbourn, MD; Fitzroy Victoria, Australia; • Chairman: Patrick W. Serruys, Prof, MD, Ph. D; Rotterdam, The Netherlands • Imaging Corelab: Cardialysis B. V. ; Rotterdam, The Netherlands • CEC: Claude Hanet, MD, Ralph Tölg, MD, Angela Hoye, MD, Benno Rensing, MD, Karel Koch, MD, Eugene Mc. Fadden, MD, Vadim A. Kuznetsov, MD • DSMB: Jan G. P. Tijssen, Ph. D, Philip Urban, MD, Marcus Wiemer, MD • Sponsor: Abbott Vascular

Key Inclusion & Exclusion Criteria • Up to two de novo lesions can be treated, each located in a separate native epicardial vessel • Target vessel diameter range is ≥ 2. 0 mm and ≤ 3. 3 mm • Target lesion length is ≤ 28 mm (planned overlapping allowed in lesions >22 and ≤ 28 mm) • Target lesion(s) meeting any of the following criteria are excluded: Ø Ø Ø Left main location; Located within an arterial or saphenous vein graft or distal to a diseased arterial or saphenous vein graft; Involves a bifurcation with a side branch 2 mm in diameter and ostial lesion > 40% stenosed or side branch requiring predilatation; Total occlusion (TIMI flow 0), prior to wire crossing; Excessive tortuosity proximal to or within the lesion; Heavy calcification.
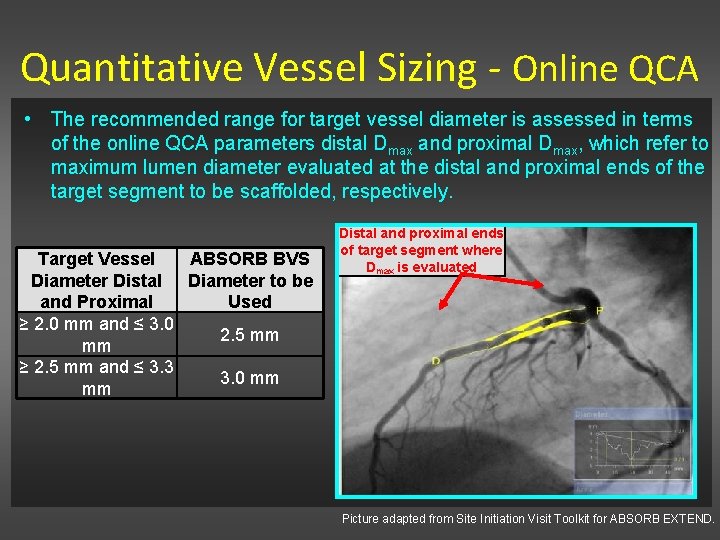
Quantitative Vessel Sizing - Online QCA • The recommended range for target vessel diameter is assessed in terms of the online QCA parameters distal Dmax and proximal Dmax, which refer to maximum lumen diameter evaluated at the distal and proximal ends of the target segment to be scaffolded, respectively. Target Vessel ABSORB BVS Diameter Distal Diameter to be and Proximal Used ≥ 2. 0 mm and ≤ 3. 0 2. 5 mm mm ≥ 2. 5 mm and ≤ 3. 3 3. 0 mm mm Distal and proximal ends of target segment where Dmax is evaluated Picture adapted from Site Initiation Visit Toolkit for ABSORB EXTEND.
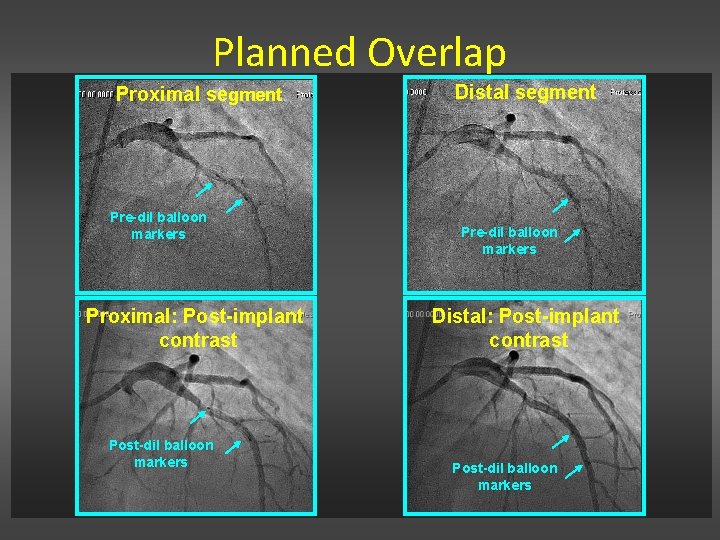
Planned Overlap Proximal segment Pre-dil balloon markers Proximal: Post-implant contrast Post-dil balloon markers Distal segment Pre-dil balloon markers Distal: Post-implant contrast Post-dil balloon markers
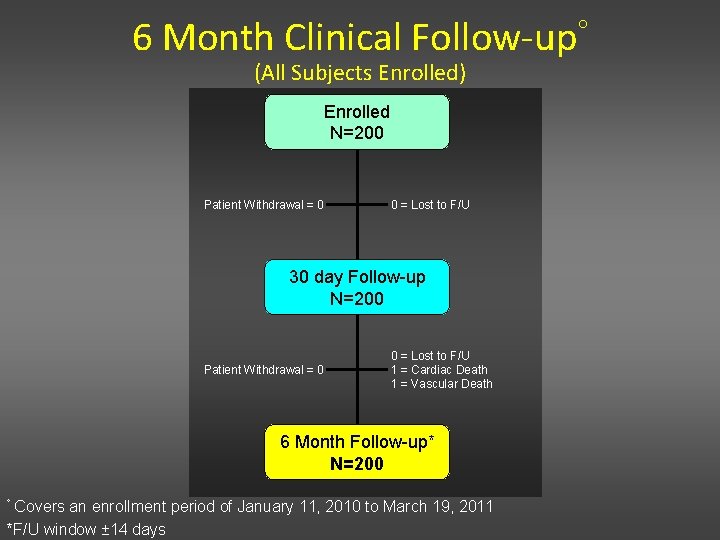
6 Month Clinical Follow-up (All Subjects Enrolled) Enrolled N=200 Patient Withdrawal = 0 0 = Lost to F/U 30 day Follow-up N=200 Patient Withdrawal = 0 0 = Lost to F/U 1 = Cardiac Death 1 = Vascular Death 6 Month Follow-up* N=200 Covers an enrollment period of January 11, 2010 to March 19, 2011 *F/U window ± 14 days
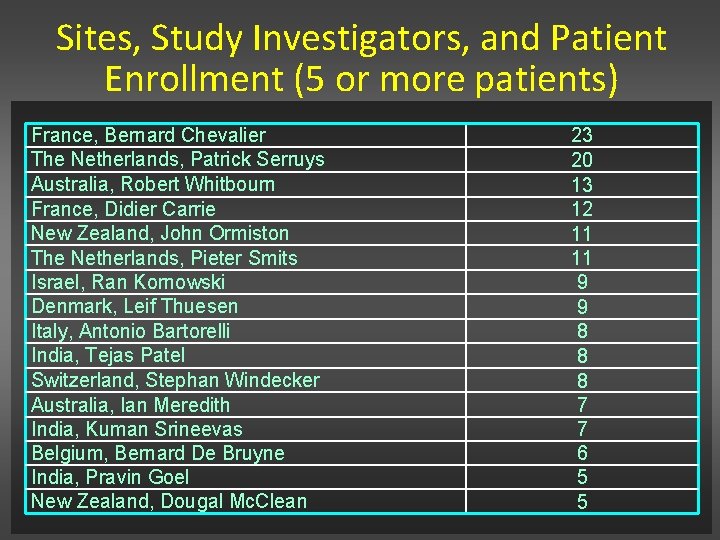
Sites, Study Investigators, and Patient Enrollment (5 or more patients) France, Bernard Chevalier The Netherlands, Patrick Serruys Australia, Robert Whitbourn France, Didier Carrie New Zealand, John Ormiston The Netherlands, Pieter Smits Israel, Ran Kornowski Denmark, Leif Thuesen Italy, Antonio Bartorelli India, Tejas Patel Switzerland, Stephan Windecker Australia, Ian Meredith India, Kuman Srineevas Belgium, Bernard De Bruyne India, Pravin Goel New Zealand, Dougal Mc. Clean 23 20 13 12 11 11 9 9 8 8 8 7 7 6 5 5
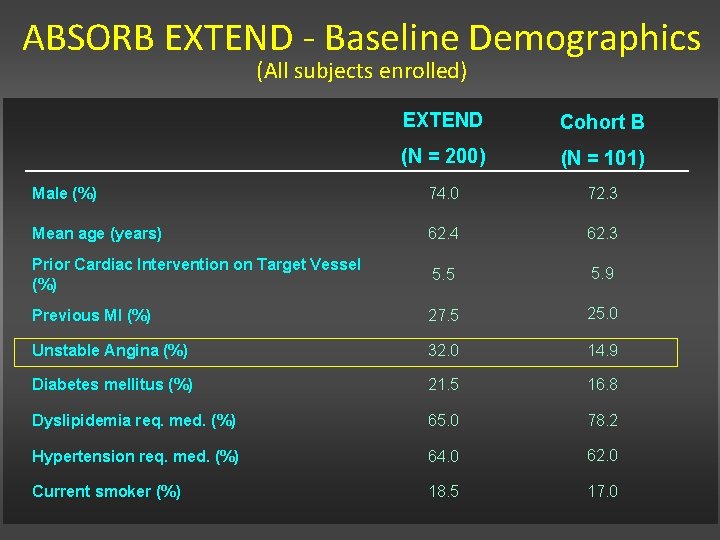
ABSORB EXTEND - Baseline Demographics (All subjects enrolled) EXTEND Cohort B (N = 200) (N = 101) Male (%) 74. 0 72. 3 Mean age (years) 62. 4 62. 3 Prior Cardiac Intervention on Target Vessel (%) 5. 5 5. 9 Previous MI (%) 27. 5 25. 0 Unstable Angina (%) 32. 0 14. 9 Diabetes mellitus (%) 21. 5 16. 8 Dyslipidemia req. med. (%) 65. 0 78. 2 Hypertension req. med. (%) 64. 0 62. 0 Current smoker (%) 18. 5 17. 0
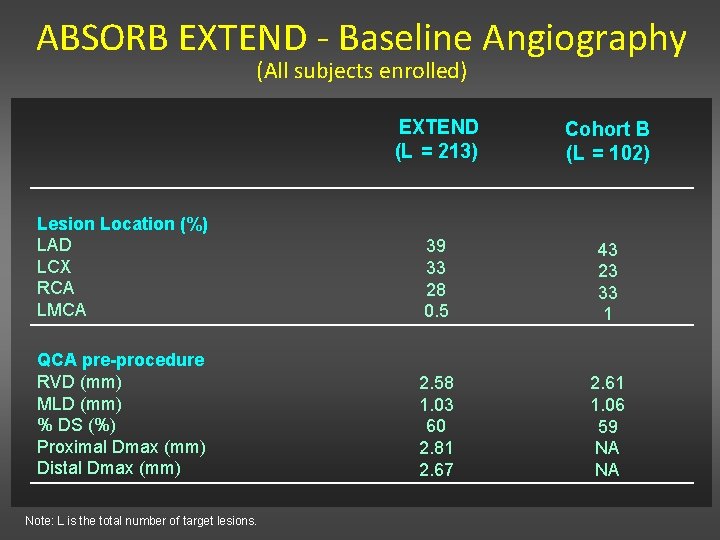
ABSORB EXTEND - Baseline Angiography (All subjects enrolled) EXTEND (L = 213) Cohort B (L = 102) Lesion Location (%) LAD LCX RCA LMCA 39 33 28 0. 5 43 23 33 1 QCA pre-procedure RVD (mm) MLD (mm) % DS (%) Proximal Dmax (mm) Distal Dmax (mm) 2. 58 1. 03 60 2. 81 2. 67 2. 61 1. 06 59 NA NA Note: L is the total number of target lesions.
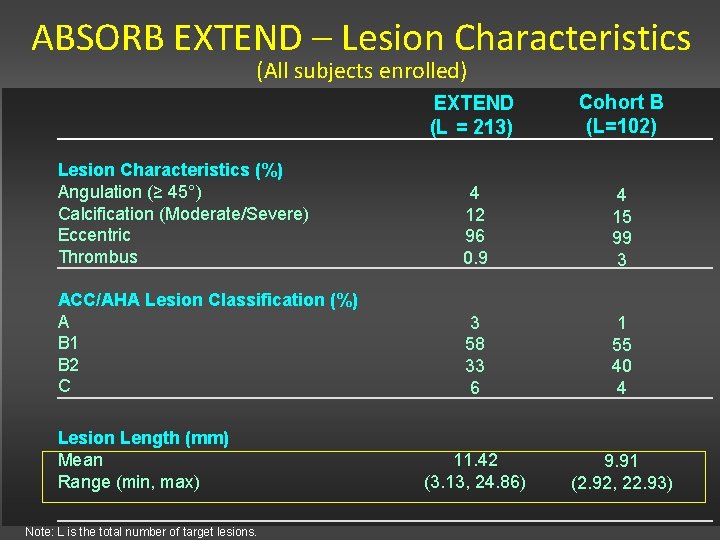
ABSORB EXTEND – Lesion Characteristics (All subjects enrolled) EXTEND (L = 213) Cohort B (L=102) Lesion Characteristics (%) Angulation (≥ 45°) Calcification (Moderate/Severe) Eccentric Thrombus 4 12 96 0. 9 4 15 99 3 ACC/AHA Lesion Classification (%) A B 1 B 2 C 3 58 33 6 1 55 40 4 11. 42 (3. 13, 24. 86) 9. 91 (2. 92, 22. 93) Lesion Length (mm) Mean Range (min, max) Note: L is the total number of target lesions.
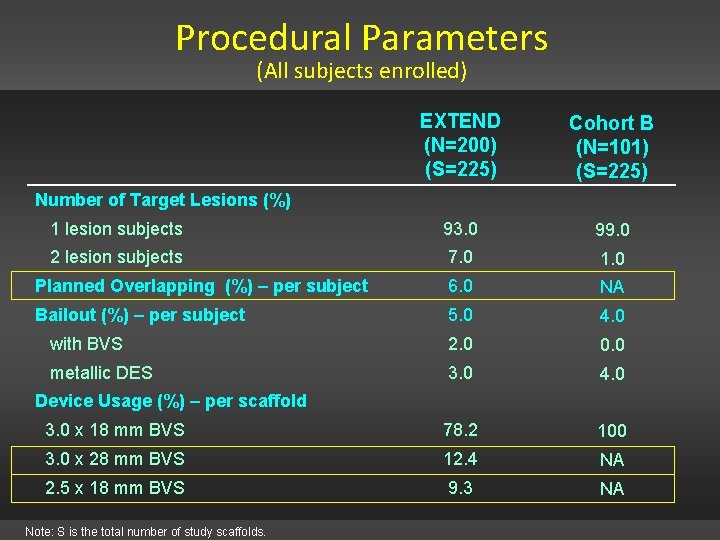
Procedural Parameters (All subjects enrolled) EXTEND (N=200) (S=225) Cohort B (N=101) (S=225) 1 lesion subjects 93. 0 99. 0 2 lesion subjects 7. 0 1. 0 Planned Overlapping (%) – per subject 6. 0 NA Bailout (%) – per subject 5. 0 4. 0 with BVS 2. 0 0. 0 metallic DES 3. 0 4. 0 3. 0 x 18 mm BVS 78. 2 100 3. 0 x 28 mm BVS 12. 4 NA 2. 5 x 18 mm BVS 9. 3 NA Number of Target Lesions (%) Device Usage (%) – per scaffold Note: S is the total number of study scaffolds.
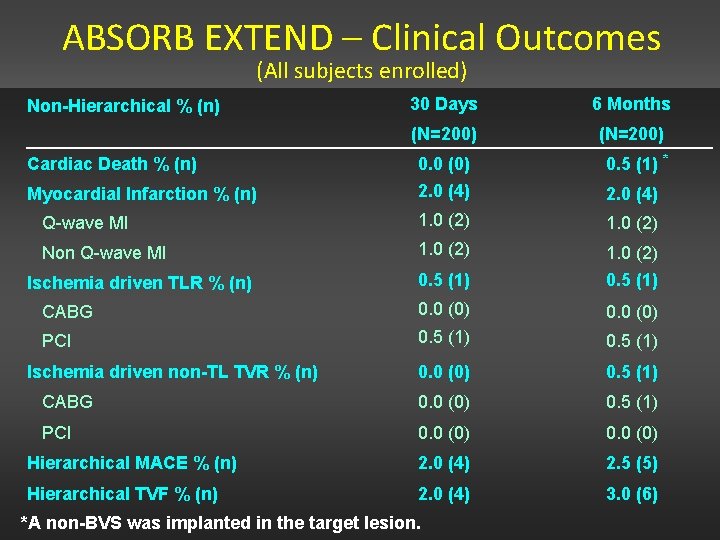
ABSORB EXTEND – Clinical Outcomes (All subjects enrolled) 30 Days 6 Months (N=200) 0. 0 (0) 2. 0 (4) 0. 5 (1) * Q-wave MI 1. 0 (2) Non Q-wave MI 1. 0 (2) 0. 5 (1) CABG 0. 0 (0) PCI 0. 5 (1) 0. 0 (0) 0. 5 (1) CABG 0. 0 (0) 0. 5 (1) PCI 0. 0 (0) Hierarchical MACE % (n) 2. 0 (4) 2. 5 (5) Hierarchical TVF % (n) 2. 0 (4) 3. 0 (6) Non-Hierarchical % (n) Cardiac Death % (n) Myocardial Infarction % (n) Ischemia driven TLR % (n) Ischemia driven non-TL TVR % (n) *A non-BVS was implanted in the target lesion. 2. 0 (4)
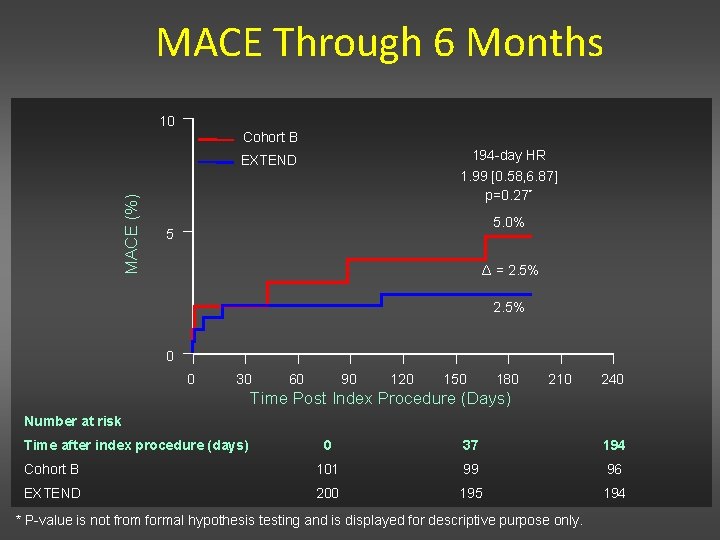
MACE Through 6 Months 10 Cohort B 194 -day HR MACE (%) EXTEND 1. 99 [0. 58, 6. 87] p=0. 27* 5. 0% 5 Δ = 2. 5% 0 0 30 60 90 120 150 180 210 240 Time Post Index Procedure (Days) Number at risk Time after index procedure (days) 0 37 194 Cohort B 101 99 96 EXTEND 200 195 194 * P-value is not from formal hypothesis testing and is displayed for descriptive purpose only.
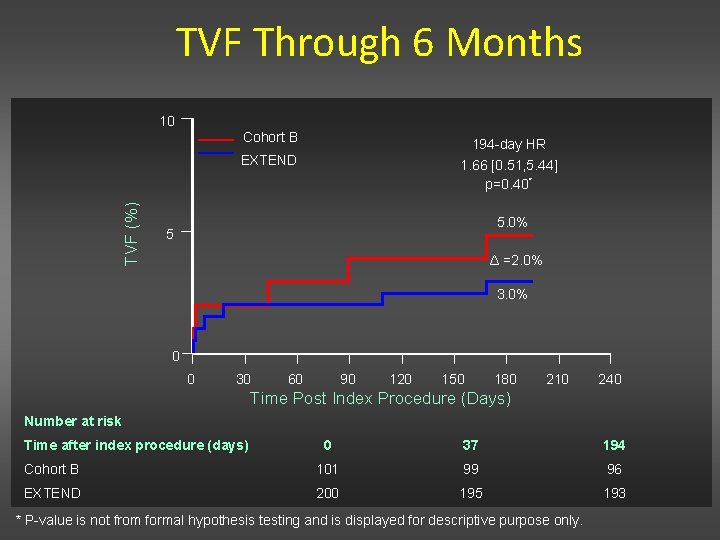
TVF Through 6 Months TVF (%) 10 Cohort B 194 -day HR EXTEND 1. 66 [0. 51, 5. 44] p=0. 40* 5. 0% 5 Δ =2. 0% 3. 0% 0 0 30 60 90 120 150 180 210 240 Time Post Index Procedure (Days) Number at risk Time after index procedure (days) 0 37 194 Cohort B 101 99 96 EXTEND 200 195 193 * P-value is not from formal hypothesis testing and is displayed for descriptive purpose only.
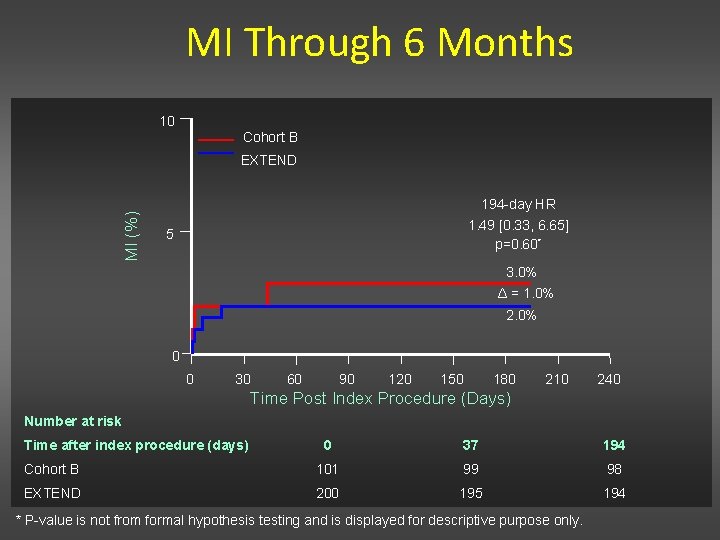
MI Through 6 Months 10 Cohort B MI (%) EXTEND 194 -day HR 1. 49 [0. 33, 6. 65] p=0. 60* 5 3. 0% Δ = 1. 0% 2. 0% 0 0 30 60 90 120 150 180 210 240 Time Post Index Procedure (Days) Number at risk Time after index procedure (days) 0 37 194 Cohort B 101 99 98 EXTEND 200 195 194 * P-value is not from formal hypothesis testing and is displayed for descriptive purpose only.
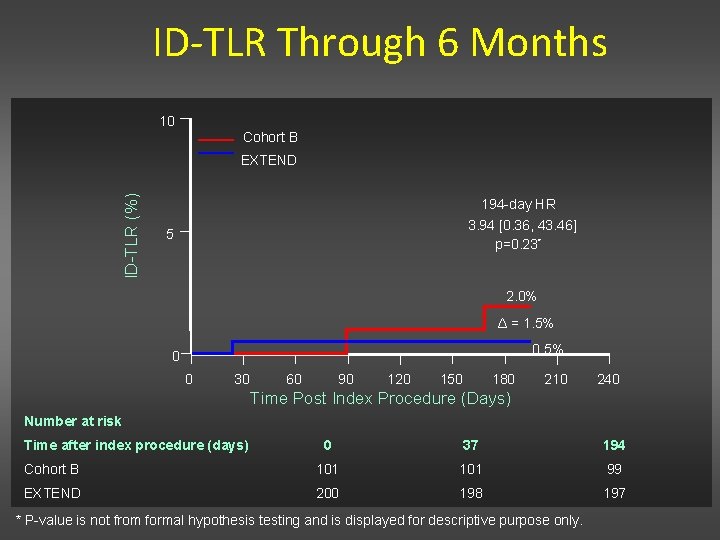
ID-TLR Through 6 Months 10 Cohort B ID-TLR (%) EXTEND 194 -day HR 3. 94 [0. 36, 43. 46] p=0. 23* 5 2. 0% Δ = 1. 5% 0 0 30 60 90 120 150 180 210 240 Time Post Index Procedure (Days) Number at risk Time after index procedure (days) 0 37 194 Cohort B 101 99 EXTEND 200 198 197 * P-value is not from formal hypothesis testing and is displayed for descriptive purpose only.
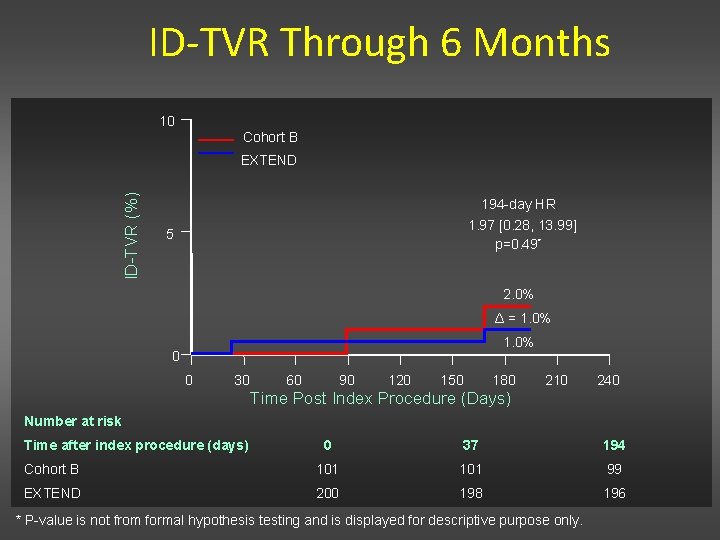
ID-TVR Through 6 Months 10 Cohort B ID-TVR (%) EXTEND 194 -day HR 1. 97 [0. 28, 13. 99] p=0. 49* 5 2. 0% Δ = 1. 0% 0 0 30 60 90 120 150 180 210 240 Time Post Index Procedure (Days) Number at risk Time after index procedure (days) 0 37 194 Cohort B 101 99 EXTEND 200 198 196 * P-value is not from formal hypothesis testing and is displayed for descriptive purpose only.

Scaffold Thrombosis (ARC) through 6 Months (Definite/Probable) ABSORB EXTEND (N = 200) Acute (<1 day) - Definite 0. 0% (0/200) - Probable 0. 0% (0/200) - Definite/probable 0. 0% (0/200) Subacute (1 -30 days) - Definite 0. 0% (0/200) - Probable 0. 5% (1/200) - Definite/probable 0. 5% (1/200) Late (>30 days) - Definite 0. 0% (0/199) - Probable 0. 0% (0/199) - Definite/probable 0. 0% (0/199) The overall definite/probable ST rate out to 194 days is 0. 5%.

Conclusions • This snapshot of 200 subjects from ABSORB EXTEND included follow-up data out to 6 months. • Comparison of the ABSORB EXTEND subject demographics and lesion characteristics showed: Ø Longer mean lesion length than in ABSORB Cohort B Ø Planned overlapping treatment permitted (6. 0% of subjects) Ø Over twice the percent of unstable angina subjects than in Cohort B • Based on 6 months outcomes in 200 subjects, the data showed: Ø Comparable MACE rates to ABSORB Cohort B Ø MACE rate driven primarily by MI events within 30 days postprocedure Ø Low rate of revascularization out to 6 months. • Despite an increase in complexity in ABSORB EXTEND, the data to date demonstrates the consistency in clinical outcomes between ABSORB EXTEND and ABSORB Cohort B.
- Slides: 25