Update Measles Mumps Rubella and Varicella MMRV Vaccine

Update: Measles, Mumps, Rubella, and Varicella (MMRV) Vaccine Safety and Recommendations National Immunization Conference April 21, 2010, Atlanta, GA Karen Broder, MD, Immunization Safety Office Mona Marin, MD, Division of Viral Diseases Centers for Disease Control and Prevention 1

Outline • Risk for Fever and Febrile Seizures after MMRV Vaccine • MMRV Vaccine Recommendations and Framework for Development • Additional Resources 2

MMRV Vaccine • 2005: The Food and Drug Administration (FDA) licensed combined Measles, Mumps, Rubella and Varicella Virus Vaccine Live (MMRV: Pro. Quad®) for use in children 12 months to 12 years of age • 2006: The Advisory Committee on Immunization Practices (ACIP) recommended use of MMRV vaccine. MMRV preferred over separate administration of MMR and varicella vaccines as part of general preference for combination vaccines Pro. Quad® Measles, Mumps, Rubella and Varicella Virus Vaccine Live DESCRIPTION Pro. Quad* is a combined attenuated live virus vaccine containing measles, mumps, rubella, and varicella viruses. Pro. Quad is a sterile lyophilized preparation of (1) the components of M-M-R*II (Measles, Mumps and Rubella Virus Vaccine Live): Measles Virus Vaccine Live, a more attenuated line of measles virus, derived from Enders' attenuated Edmonston strain and propagated in chick embryo cell 3 culture; Mumps Virus Vaccine Live, the Jeryl Lynn™

Prelicensure Studies: Vaccine-related Fever and Systemic Rash during Days 0– 42 in Children Aged 12– 23 Months Administered Dose 1 MMRV or MMR and Varicella Vaccine Dose 1 MMRV N=4, 497 MMR+V N=2, 038 Days 0– 42 Fever > 102°F or abnormal 21. 5%* 14. 9% Measles-like rash 3. 0%* 2. 1% Varicella-like rash 2. 1% 2. 2% * Rate significantly higher in MMRV group • • Among subjects who had fever after vaccination, the proportion who had fever during the 5– 12 days was 45% for MMRV and 36% for MMR + V Among subjects who had measles-like rash after vaccination, the proportion who had this rash during the 5– 12 days was 82% for MMRV and 81% for MMR + V Source: Package insert 2 -2008 and unpublished data from Merck on 10 -20 -08

Febrile Seizures • Seizures that occur in febrile children who do not have an intracranial infection, metabolic disturbance, or history of afebrile seizures 1, 2 – Usually occur at ages 6– 60 months – Peak age 14– 18 months • Affects 2%– 5% of young children in the United States 1 • Generally excellent prognosis 1, 2 – >90% of children who have a febrile seizure will not develop epilepsy 3 • One third of children with a first febrile seizure will have recurrent febrile seizures 3 5 1. AAP. Pediatrics. 2008. 2. Johnston M. Nelson Textbook of Pediatrics. 2007. 3. Baulac. The Lancet Neurology. 2004.

Mechanisms Leading to Febrile Seizures • Age-related increased susceptibility to seizures induced by fever • Peak temperature is a major determining factor 1 • Certain infections (e. g. rosela [HHV 6], salmonella, shigella) have higher likelihood of febrile seizure • DTP and MMR vaccines are transiently associated with increased risk for febrile seizures 2 1. Berg AT, et al. Epilepsia 36: 334, 1995 2. Davis. Pediatric Drugs. 2003. 6

MMRV Postlicensure Safety Studies • Two postlicensure studies conducted to assess risk for febrile seizures after MMRV – Vaccine Safety Datalink (VSD): Collaboration between CDC and 8 managed care organizations – Merck-sponsored study • February 2008 ACIP meeting: * – Preliminary information from both postlicensure studies suggested an increased risk for febrile seizures during 1 st 2 nd week after 1 st dose MMRV vaccine among children aged 12 -23 months – ACIP removed the preference for use of MMRV over separate administration of MMR and varicella vaccines 7 *CDC. MMWR. March 14, 2008 / 57(10); 258 -260
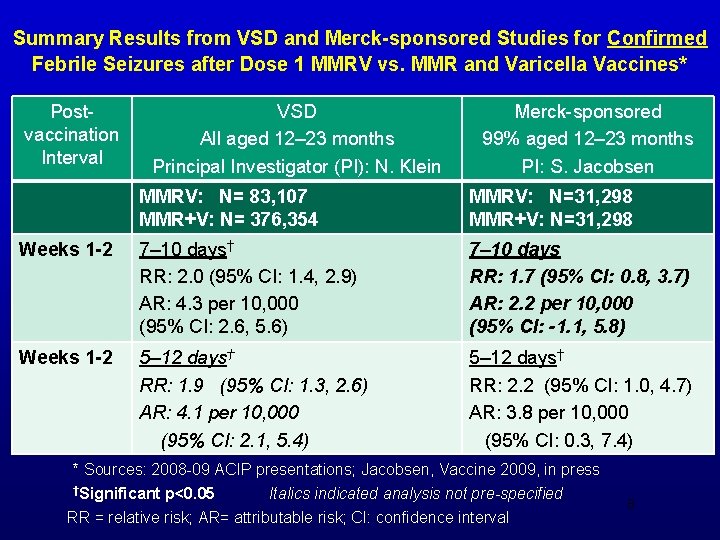
Summary Results from VSD and Merck-sponsored Studies for Confirmed Febrile Seizures after Dose 1 MMRV vs. MMR and Varicella Vaccines* Postvaccination Interval VSD All aged 12– 23 months Principal Investigator (PI): N. Klein Merck-sponsored 99% aged 12– 23 months PI: S. Jacobsen MMRV: N= 83, 107 MMR+V: N= 376, 354 MMRV: N=31, 298 MMR+V: N=31, 298 Weeks 1 -2 7– 10 days† RR: 2. 0 (95% CI: 1. 4, 2. 9) AR: 4. 3 per 10, 000 (95% CI: 2. 6, 5. 6) 7– 10 days RR: 1. 7 (95% CI: 0. 8, 3. 7) AR: 2. 2 per 10, 000 (95% CI: -1. 1, 5. 8) Weeks 1 -2 5– 12 days† RR: 1. 9 (95% CI: 1. 3, 2. 6) AR: 4. 1 per 10, 000 (95% CI: 2. 1, 5. 4) 5– 12 days† RR: 2. 2 (95% CI: 1. 0, 4. 7) AR: 3. 8 per 10, 000 (95% CI: 0. 3, 7. 4) * Sources: 2008 -09 ACIP presentations; Jacobsen, Vaccine 2009, in press †Significant p<0. 05 Italics indicated analysis not pre-specified RR = relative risk; AR= attributable risk; CI: confidence interval 8

Vaccine Safety: Febrile Seizure Risk after Dose 1 MMRV Vaccination - Conclusion • Among children aged 12 -23 months: – 1 additional febrile seizure is expected to occur during 5 -12 days after vaccination per ~2, 300 to 2, 600 children vaccinated with dose 1 MMRV, compared with dose 1 with MMR vaccine and varicella vaccine (MMR+V) 9

Vaccine Safety: Febrile Seizure Risk after Dose 2 MMRV Vaccination - Conclusion • Among children aged 4 -6 years: – Those who receive dose 2 MMRV vaccine do not appear to have an increased risk for febrile seizures after vaccination compared with those who receive MMR vaccine and varicella vaccine at the same visit (MMR+V) – Less information is available regarding the risk of febrile seizures after dose 2 than for dose 1 MMRV vaccine 10

Recommendations for Use of MMRV Vaccine 11

Outline • Recommendations for use of MMRV vaccine • Framework considered formulating policy recommendations • Assessment of the elements of the framework used to arrive at recommendations (dose 1 and dose 2) • Additional resources 12

Recommendations for Use of MMRV Vaccine - Approved by ACIP in June 2009 - 13

Recommendations for Use of MMRV Vaccine for Dose 1 at Age 12 -47* Months • For the first dose of measles, mumps, rubella, and varicella vaccines at ages 12 -47 months, either MMR vaccine and varicella or MMRV vaccine may be used. Providers who are considering administering MMRV vaccine should discuss the benefits and risks of both vaccination options with the parents or caregivers. Unless the parent or caregiver expresses a preference for MMRV vaccine, for the first dose in this age group, MMR vaccine and varicella vaccine as separate injections should be administered. • The discussion with parents should be centered on helping them understand the risk-benefit tradeoff *The 47 -month cut-off was chosen on the bases of the epidemiology of febrile seizures. First febrile seizures are uncommon at age >4 years, and approximately 94% of febrile seizures occur in children aged <4 years. 14

Recommendations for Use of MMRV Vaccine for Dose 1 at Age 12 -47 Months • Discussion with parents – Compared with use of MMR vaccine and varicella vaccine at the same visit, use of MMRV vaccine results in one fewer injection but is associated with a higher risk for fever and febrile seizures 5 -12 days after the first dose among children aged 12 -23 months (approximately one extra febrile seizure for every 2, 300 -2, 600 MMRV vaccine doses). Use of MMR vaccine and varicella vaccine avoids this increased risk for fever and febrile seizures following MMRV vaccine 15

Recommendations for Use of MMRV Vaccine for Dose 2 and Dose 1 at Age >48 Months • For the second dose of measles, mumps, rubella, and varicella vaccines at any age (15 months-12 years) and for the first dose at age >48 months, use of MMRV vaccine generally is preferred over separate injections of its equivalent component vaccines (i. e. , MMR vaccine and varicella vaccine). Considerations should include provider assessment, patient preference, and the potential for adverse events – This recommendation is consistent with the new ACIP general recommendations on combination vaccines (June 2009) • The routinely recommended ages for measles, mumps, rubella and varicella vaccination continue to be 12 -15 months for dose 1 and 4 -6 age for dose 2 16

Framework Used for Policy Discussions 1. Vaccine safety 2. Efficacy/effectiveness and immunogenicity 3. Burden of disease to prevent (measles, mumps, rubella, and varicella) 4. Program implementation 5. Equity in access to vaccines and use of public funds 6. Social context 17

Assessment of the Elements of the Framework - Dose 1 18

Vaccine Safety: Febrile Seizure Risk after Dose 1 MMRV Vaccination - Conclusion • Among children aged 12 -23 months – 1 additional febrile seizure is expected to occur during 5 -12 days after vaccination per ~2, 300 to 2, 600 children vaccinated with dose 1 MMRV, compared with dose 1 with MMR vaccine and varicella vaccine (MMR+V) 19

Dose 1: Efficacy/Effectiveness and Immunogenicity Evidence • MMRV licensed on the basis of non-inferior immunogenicity of the antigenic components compared with simultaneous administration of MMR vaccine and varicella vaccine* – Efficacy inferred on the basis of non-inferior immunogenicity Assessment • Equal immunogenicity • Assumed equal effectiveness • Both vaccination options considered to provide the same protection against the four diseases *FDA, Pro. Quad clinical review, available at http: //www. fda. gov/cber/review/proquad. R. pdf 20

Dose 1: Burden of Disease to Prevent Evidence • Current burden of disease in the US – Measles, rubella, mumps: eliminated/very low 1 -3 – Varicella: low 4 • Vaccine coverage levels: high – Measles, mumps, rubella (MMR) vaccine § Among 19 to 35 month-olds: 90%-93% since 19965 – Varicella vaccine § Among 19 to 35 month-olds: 91% in 20085 • Disease reduction and coverage levels achieved in the absence of MMRV vaccine 1. Katz SL and Hinman AR. J Infect Dis, 2004 4. Guris D et al. J Infect Dis, 2008 2. Reef SE and Cochi SL. J Infect Dis, 2006 5. NIS, www. cdc. gov/vaccines/stats-surv/default. htm 3. Barskey A et al. IDSA abstract, 2008 21

Dose 1: Burden of Disease to Prevent Assessment • MMRV as a first dose likely has similar impact to that of MMR+V for prevention of measles, mumps, rubella and varicella – MMRV may lead to more children vaccinated against varicella at age 12 months rather than 15 to 18 months 22

Dose 1: Program Implementation - Evidence • Storage conditions – MMR stored in the refrigerator – Varicella and MMRV must be stored frozen • Number of injections – 9 -10 vaccinations recommended either routinely at age 12 -15 months or for ages that overlap age 12 -15 months 1 – Parent and provider preference for fewer injections at a visit 2 -4 • Current experience with vaccine administration, 2007 -085 – The majority of children receive dose 1 MMR vaccine and varicella vaccine at age 12 months • Potential impact of combination vaccines – Combination vaccines may offer benefit in terms of overall coverage or timeliness but the literature is limited to quantitate the benefit 1. http: //www. cdc. gov/vaccines/recs/schedules/child-schedule. htm 2. Meyerhoff A. Clinical Pediatrics, 2004 3. Lieu TA et al. Vaccine, 2000 4. Freed GL et at. Pediatrics, 2006 5. Source: Immunization information Systems, MI and OR sentinel sites 23

Dose 1: Program Implementation - Evidence • Information from focus groups with mothers in 2 cities 1 – Questions and concerns related to safety of vaccines – If MMRV were recommended by their physician, 41% would accept and 31% would refuse • Physician use and opinions of MMRV vaccine – Studies found good uptake when MMRV was available – Physicians interviewed reported no differences in experience using MMRV compared with MMR 1 – Intended use of MMRV given postlicensure safety data § 9% FP and 21% Peds would definitely give MMRV to a healthy 12 -15 month-old; 20%-24% would let parents decide 2 § 51% of prior users and 22% of non-prior users expect their patients aged 12 -15 months to receive dose 1 as MMRV 3 1. A. Janssen, CDC. ACIP meeting June 25, 2009 2. Dr. A. Kempe, Univ of Colorado. ACIP meeting February 26, 2009 3. J. Kramer, Merck & Co. Presentation to the WG, May, 2009 24

Dose 1: Program Implementation Assessment • Insufficient evidence to express a benefit for one vaccination option − MMRV potentially can have a favorable impact on the on-time administration of other vaccines recommended for age 12 -15 months − For some parents and providers the benefit of the reduced number of injections is outweighed by the increase in febrile seizure rate after MMRV 25

Dose 1: Equity in Access to Vaccines and Use of Public Funds Evidence • Price of the vaccines per dose is similar* • MMRV and varicella vaccines stored frozen – no difference in availability due to storage requirements • ACIP recommendations for MMR, MMRV, and Varicella vaccine use • Vaccines for Children (VFC) resolutions Assessment • No difference in access and use of public funds * Available at http: //www. cdc. gov/vaccines/programs/vfc/cdc-vac-price-list. htm 26

Dose 1: Social Context Ethics perspective • Does the increased protection for the population offset any increased risk for the individual? • Consider risk/benefit analyses, parental and provider preferences, and justice and fairness considerations Assessment – Ethicists recommended an approach that makes the option with the fewer adverse events the default, therefore favoring a preference for MMR vaccine and varicella vaccine for dose 1 – Choice to use MMRV vaccine should be made available if the physician and parent decide it is the best option – If future data show that a preference for separate vaccines has a larger than expected impact on coverage, then the option should be reconsidered CDC Public Health ethics activity and academic medical ethicists, 2009 27

Assessment of the Elements of the Framework - Dose 2 - Elements that are different from Dose 1 - 28

Vaccine Safety: Febrile Seizure Risk after Dose 2 MMRV Vaccination - Conclusion • Among children aged 4 -6 years – Those who receive dose 2 MMRV vaccine do not appear to have an increased risk for febrile seizures after vaccination compared with those who receive MMR vaccine and varicella vaccine at the same visit (MMR+V) – Less information is available regarding the risk of febrile seizures after dose 2 than for dose 1 MMRV vaccine 29

Dose 2: Burden of Disease to Prevent Evidence • Burden of disease to prevent considered similar to that for dose 1 at age 12 -15 months, with several differences in coverage for varicella – 2 nd dose recommended in 2006 compared with 1989 for MMR – In 2009, 22 states had school entry requirements for 2 doses of varicella vaccine* compared with all states for measles-containing vaccine Assessment • MMRV as a second dose potentially more beneficial for varicella than MMR+V – Through increasing 2 nd dose varicella coverage until states implement school entry requirements *CDC, unpublished data (courtesy of Jessica Leung, MPH) 30

Dose 2: Program Implementation - Evidence • 5 vaccines routinely recommended at ages 4 -6 years 1 – Anecdotal reports that it is more difficult to administer multiple injections at one visit to children aged 4 -6 years • Physician opinions on use of MMRV – 20% FP and 38% Peds would definitely give MMRV to a healthy 4 -6 year-old and 21%-25% would let parents decide 2 – 67% of prior users and 36% of non-prior users expect their patients aged 4 -6 years to receive dose 2 as MMRV 3 1. http: //www. cdc. gov/vaccines/recs/schedules/child-schedule. htm 2. Dr. A. Kempe, Univ of Colorado. ACIP meeting February 26, 2009 3. J. Kramer, Merck & Co. Presentation to the WG, May, 2009 31

Dose 2: Program Implementation Assessment • It is likely that vaccination against measles, mumps, rubella and varicella at age 4 -6 years is preferred as one injection (MMRV) instead of two (MMR+V) by both parents and providers provided safety profile similar • A potential benefit for MMRV is a more rapid increase of coverage for dose 2 varicella vaccine and improved coverage for other vaccines recommended at this age • MMRV more beneficial than MMR+V 32

Dose 2 at Ages 4 -6 Years – Summary To protect against measles, mumps, rubella and varicella, two vaccine options are available: MMRV vaccine, or MMR vaccine and varicella vaccine administered as separate vaccines • The two options are considered equivalent in terms of disease protection • Use of MMRV vaccine results in one fewer injection compared with the use of MMR vaccine and varicella vaccine and has the potential to improve the coverage for dose 2 varicella vaccine as well as for other vaccines recommended at this age • Available data do not suggest an increase in risk for fever or febrile seizure after the second dose MMRV vaccine at ages 4 -6 years compared with second dose administered as MMR vaccine and varicella vaccine at the same visit 33

Additional Resources • CDC has developed materials to help with the implementation of the new recommendations for use of MMRV vaccine: www. cdc. gov/vaccines/vpd-vac/combovaccines/mmrv/vacopt. htm – Will be updated by early May 2010 • A new Vaccine Information Statement (VIS) was developed for MMRV vaccine – In CDC clearance • MMWR ACIP statement on use of MMRV vaccine – Scheduled for electronic release by early May and print in mid-May 2010 34

Disclaimer • The findings and conclusions in this presentation are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention (CDC) • Recommendations from the Advisory Committee on Immunization Practices are provisional until they are published by CDC in the Morbidity and Mortality Weekly Report (MMWR) 35

MMRV Vaccine Safety Working Group Members CDC ACIP Jon Temte, Chair H. Cody Meissner Mark Sawyer FDA Sang Ahnn* Judy Beeler Hector Izurieta Karen Broder (Co-Lead) Mona Marin (Co-Lead) Elizabeth Skillen* (Coordinator) Aisha Jumaan* Preeta Kutty Jim Sejvar Jane Seward Dixie Snider Eric Weintraub HRSA Rosemary Johann-Liang NVPO Daniel Salmon *Service on Working Group from June – October 2008 36

MMRV Vaccine Safety Working Group Members Elizabeth Andrews Lawrence Brown RTI International Children’s Hospital of Philadelphia Bruce Fireman Kaiser Permanente Northern California (VSD) Kathleen Gensheimer Anne Gershon Sam Katz David Kimberlin Nicola Klein Thomas Koinis Martin Kulldorff Tracy Lieu Georges Peter Lin Watson Katherine Yih Maine State Health Department, CSTE Columbia University Duke University (Emeritus) (IDSA) University of Alabama (AAP) Kaiser Permanente Northern California (VSD) Duke Health (AAFP) Harvard Medical School (VSD) Brown University (Emeritus) Washington State Department of Health/CDC Harvard Medical School (VSD) 37

MMRV Vaccine Safety Working Group Acknowledgments Academia Robert Baumann Saad Omer Joe Bocchini Combination Vaccine WG Andrew Kroger Ciro Sumaya Program Diana Bartlett Zhen Zhao A. Osborn, MB Kurilo – MI IIS R. Porter & B. Salada – OR IIS Jack Sims Barbara Watson Sandra-Jo Hammer Ethics Drue Barett Bernard Lo Rick Zimmerman Division of Viral Diseases Stephanie Bialek Greg Armstrong Susan Goldstein Charley Le. Baron Scott Schmid Communication Kris Sheedy Alan Janssen Merck Barbara Kuter Patricia Saddier 38

Back-up Slides 39
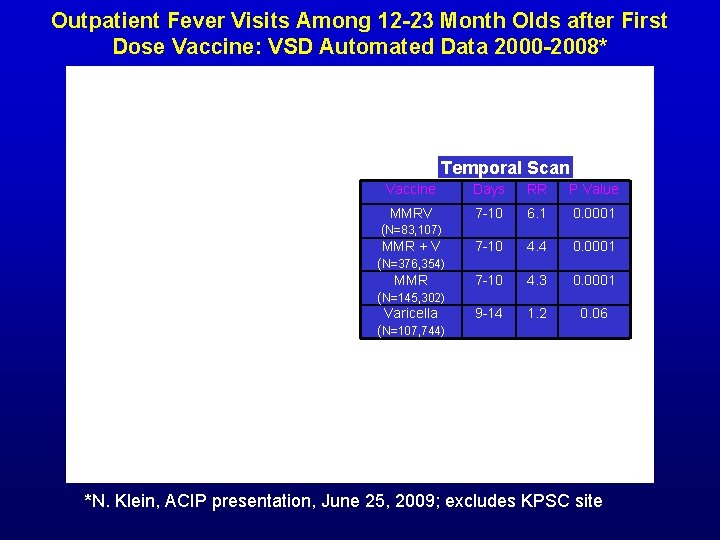
Outpatient Fever Visits Among 12 -23 Month Olds after First Dose Vaccine: VSD Automated Data 2000 -2008* Temporal Scan Vaccine Days RR P Value MMRV 7 -10 6. 1 0. 0001 7 -10 4. 4 0. 0001 7 -10 4. 3 0. 0001 9 -14 1. 2 0. 06 (N=83, 107) MMR + V (N=376, 354) MMR (N=145, 302) Varicella (N=107, 744) *N. Klein, ACIP presentation, June 25, 2009; excludes KPSC site

Biological Plausibility Assessment: Increased Risk for Fever and Febrile Seizures in Children Receiving MMRV vs. MMR and Varicella (V) Vaccines • During the 8– 14 days after vaccination, about one additional febrile seizure occurs among every 3, 000– 4, 000 children who receive MMR vaccine, compared with children who are not recently vaccinated 1 • MMRV (Proquad®) has ~7 times more varicella component than varicella vaccine (Varivax®) and the same amount of measles component as MMR • Immune response to MMRV suggests measles virus replication may be higher after MMRV vs. MMR+V vaccines • Higher rates of fever reported after MMRV vs. MMR+V • Febrile seizures occur in a setting a fever 1. Barlow, Davis et al. NEJM, 2001. 41

Dose 1 at Age 12 -15 Months – Summary To protect against measles, mumps, rubella and varicella, two vaccine options are available: MMRV vaccine, or MMR and varicella vaccines • The two options are considered equivalent in terms of disease protection • Use of MMRV vaccine results in one fewer injection compared with use of MMR vaccine and varicella vaccine and may improve the on-time coverage for the vaccines recommended at ages 12 -15 months • Use of MMRV vaccine results in a higher risk for fever and febrile seizures 5 -12 days after the first dose when administered during the second year of life compared with use of MMR vaccine and varicella vaccine at the same visit (about one extra febrile seizure for every 2, 3002, 600 MMRV doses) • The risk for febrile seizures after MMRV can be reduced by administering separate injections of MMR vaccine and varicella vaccine • Children who have febrile seizures generally have an excellent prognosis; however, febrile seizures often require a medical visit to the emergency room for the child and are distressing to parents/caregivers 42

Other MMRV Vaccine Related Guidance 43

New Precaution for MMRV Vaccine Use • A personal or family (i. e. , sibling or parent) history of seizures of any etiology is a precaution for MMRV vaccination. − These children generally should be vaccinated with MMR vaccine and varicella vaccine because the risks for using MMRV vaccine generally outweigh the benefits “A precaution is a condition in a recipient that might increase the risk for a serious adverse reaction or that might compromise the ability of the vaccine to produce immunity (e. g. , administering measles vaccine to a person with passive immunity to measles from a blood transfusion). A person might experience a more severe reaction to the vaccine than would have otherwise been expected; however, the risk for this happening is less than expected with a contraindication. In general, vaccinations should be deferred when a precaution is present. However, a vaccination might be indicated in the presence of a precaution because the benefit of protection from the vaccine outweighs the risk for an adverse reaction. ” ACIP General Recommendations on Immunization. MMWR 2006; 55(No. RR-15) 44

MMRV Vaccine for Dose 1 and Dose 2 at Ages Other Than Routinely Recommended - Summary • Dose 1 MMRV among children aged >23 months – No safety data available – Febrile seizure usually occur at ages 6 to 60 mos; peak age 14 to 18 mos – MMR experience: decreased risk for febrile seizures with increasing age at vaccination within the age group 15 to 23 months 1 • Dose 2 MMRV at age <4 years (data for dose 2 at ages 15 to 26 months) – Lower incidence of fever, measles- and varicella-like rash than post dose 1 2, 3 – Among ~2, 700 subjects 2 febrile seizures (1 in setting of roseola) post dose 2 administered 3 and 6 to 9 months after dose 14 – 95% of measles-containing vaccine recipients have antibodies after dose 1 – Immunogenicity not inferior when 2 nd dose administered at ages <4 years 2, 3 • Dose 1 or dose 2 MMRV at ages 7 to 12 years – No safety or immunogenicity data available – Febrile seizures occur almost exclusively among children aged 6 to 60 mos 1. Vestergaard M et al. JAMA, 2004 2. Shinefield H et al. Pediatr Infect Dis J, 2005 3. Shinefield H et al. Pediatr Infect Dis J, 2005 4. Merck & Co. Presentation to the WG, December, 2008 45

Include Among Precautions for MMRV Vaccine Use Children with Personal and Family History of Seizure • Studies suggest that children who have a personal or family history of febrile seizures or family history of epilepsy are at increased risk for febrile seizures compared with children who do not have such histories Characteristic Personal history of febrile seizure Family history of epilepsy Risk difference within 14 days after MMR vaccine vs. nonvaccinated, children age 15 -17 mos (95% CI), per 1, 0001 Matched Odds Ratio Cases: children with first febrile seizure Controls: febrile, no history of febrile seizure 2 19. 5 (16. 1 -23. 6) 6. 5 (2. 2, 19. 6) 4. 0 (2. 9 -5. 4) 3. 6 (1. 4, 9. 3) 3. 4 (1. 9 -5. 9) 2. 6 (0. 5, 14. 3) 1. Vestergaard M et at. JAMA, 2004 2. Berg AT et al. Epilepsia, 1995 • The MMRV (Pro. Quad®) package insert lists individual or family history of convulsions as a warning 46

Dose 1: Social Context Working Group Discussion • Need to communicate the risks and benefits of vaccination strategies in a clear and transparent manner • Make evidence-based policy recommendations considering the best information available and recognizing the legitimacy of questions and concerns 47

MMRV Dose 1 at Ages Older Than 23 Months • • No published data on safety or immunogenicity for MMRV vaccine administered as dose 1 to children aged >23 months MMR experience* – – No data on MMR administered as dose 1 to children aged >2 years (some papers lumped together 1 yr-4/6 yrs, no age breakdown of subjects) Risk difference of febrile seizures during 2 weeks after MMR vaccination compared with children who did not receive MMR, per 1000 children, by age at vaccination (~537, 000 children, 82% vaccinated)** • Age 15 -17 mo. : 1. 56 (95% CI, 1. 44 -1. 68) • Age 18 -20 mo. : 1. 46 (95% CI, 1. 10 -1. 91) • Age 21 -23 mo. : 0. 64 (95% CI, 0. 22 -1. 40) ** M. Vestergaard et al. MMR vaccination and febrile seizures: evaluation of susceptible subgroups and long-term prognosis. JAMA. 2004 * Thanks to Dr. Preeta Kutty, MMR team, who did the literature review including data from clinical trials for M, MR, MMR 48
- Slides: 48