UPADACITINIB A SUCCESSOR TO ADALIMUMAB Miranda Ochs Pharm

UPADACITINIB: A SUCCESSOR TO ADALIMUMAB? Miranda Ochs, Pharm. D PGY-1 Pharmacy Resident Millcreek Community Hospital October 26, 2019

OBJECTIVES • Following the presentation, pharmacists will be able to • Identify appropriate dosing and monitoring for patients taking upadacitinib • Explain upadacitinib's place in the treatment of rheumatoid arthritis • Following the presentation, pharmacy technicians will be able to • Describe appropriate storage requirements for upadacitinib • Recognize available preparations of upadacitinib

UPADACITINIB MONOGRAPH OVERVIEW

UPADACITINIB GENERAL INFORMATION • Brand name: RINVOQ™ • Indication • Moderate-severe rheumatoid arthritis in patients that have had an inadequate response or intolerance to methotrexate • Monotherapy or in combination with nonbiologic agent • Pharmacologic category: Disease modifying antirheumatic drug (DMARD) • Janus Associated Kinase (JAK) inhibitor • Mechanism of action • Inhibits JAK enzymes involved with hematopoiesis and immune cell function • Decreased immune cell production decreased inflammatory response improved symptoms RINVOQ™ (upadacitinib) Treatment for Rheumatoid Arthritis. Rinvoq. https: //www. rinvoq. com/. Accessed October

UPADACITINIB DOSING • Dosage • 15 mg tablet by mouth once daily • Dosage adjustments • No renal dosing recommendations; not studied in e. GFR <15 ml/min/1. 73 m 2 • Not recommended in severe liver impairment (Child-Pugh class C) • Formulations • Extended release tablet A 15 Pill Images (Purple / Capsule-shape). Drugs. com. https: //www. drugs. com/imprints/a 15 -29542. html. Accessed October 11, 2019.

UPADACITINIB PATIENT MANAGEMENT SIDE EFFECTS MONITORING AND SCREENING • Upper respiratory tract infection • Lymphocyte, neutrophil counts • Nausea • Liver function tests (LFTs) at baseline and as clinically indicated • Cough • Pyrexia • Lipids 12 weeks after initiation and periodically thereafter
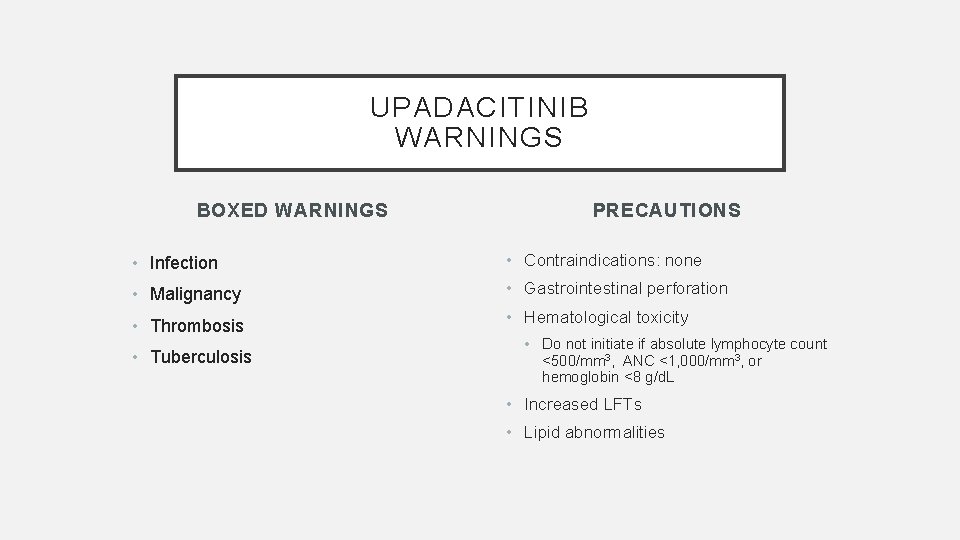
UPADACITINIB WARNINGS BOXED WARNINGS PRECAUTIONS • Infection • Contraindications: none • Malignancy • Gastrointestinal perforation • Thrombosis • Hematological toxicity • Tuberculosis • Do not initiate if absolute lymphocyte count <500/mm 3, ANC <1, 000/mm 3, or hemoglobin <8 g/d. L • Increased LFTs • Lipid abnormalities

UPADACITINIB STORAGE AND ADMINISTRATION • Swallowed whole with or without food • Do not crush, split, or chew • Storage • Room temperature in original bottle • Not on the 2016 National Institution for Occupational Safety and Health (NIOSH) list of hazardous drugs • Qualifying criteria: carcinogenicity, teratogenicity • Use appropriate precautions during handling • Assessment of risk • Dispense in original bottle

USE IN PRACTICE CLINICAL IMPLICATIONS
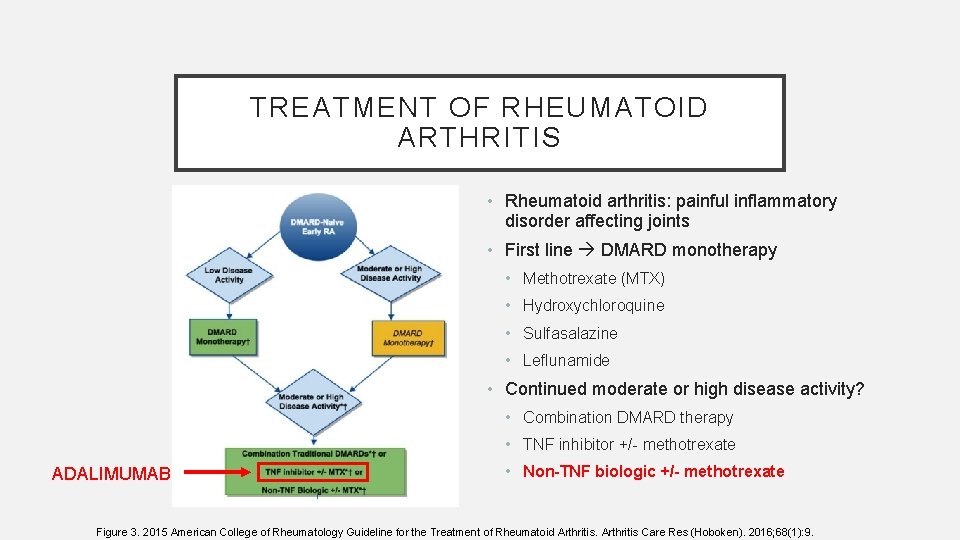
TREATMENT OF RHEUMATOID ARTHRITIS • Rheumatoid arthritis: painful inflammatory disorder affecting joints • First line DMARD monotherapy • Methotrexate (MTX) • Hydroxychloroquine • Sulfasalazine • Leflunamide • Continued moderate or high disease activity? • Combination DMARD therapy • TNF inhibitor +/- methotrexate ADALIMUMAB • Non-TNF biologic +/- methotrexate Figure 3. 2015 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis Care Res (Hoboken). 2016; 68(1): 9.

A SUCCESSOR TO ADALIMUMAB? • Upadacitinib and adalimumab share placement in the algorithm for treatment of rheumatoid arthritis • Adalimumab: weekly or biweekly self-injection • Upadacitinib: once daily pill • Upadacitinib shown to be superior to adalimumab with regards to American College of Rheumatology (ACR) 20/50/70 achievement • Achievement of ACR 20/50/70 correlates with a 20%/50%/70% improvement in symptoms (i. e. swollen joints, pain)
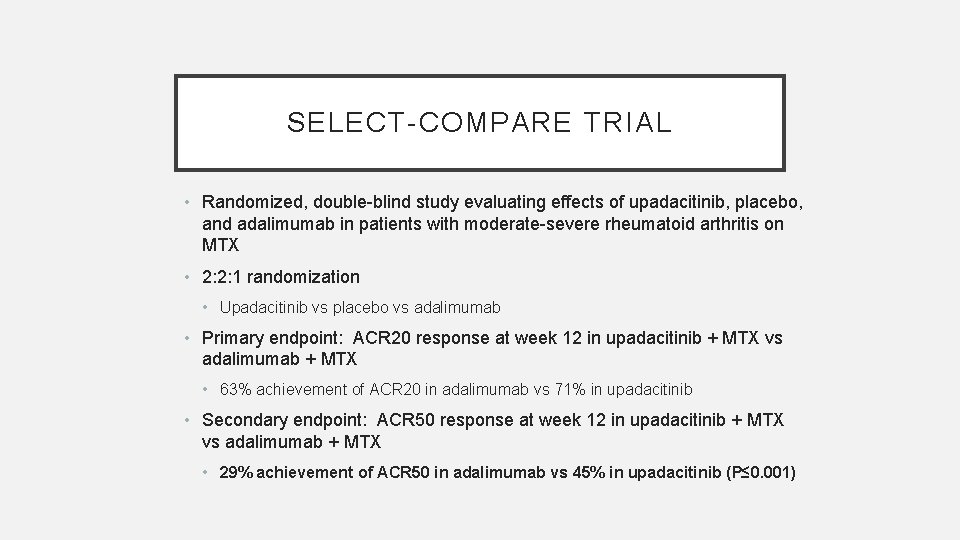
SELECT-COMPARE TRIAL • Randomized, double-blind study evaluating effects of upadacitinib, placebo, and adalimumab in patients with moderate-severe rheumatoid arthritis on MTX • 2: 2: 1 randomization • Upadacitinib vs placebo vs adalimumab • Primary endpoint: ACR 20 response at week 12 in upadacitinib + MTX vs adalimumab + MTX • 63% achievement of ACR 20 in adalimumab vs 71% in upadacitinib • Secondary endpoint: ACR 50 response at week 12 in upadacitinib + MTX vs adalimumab + MTX • 29% achievement of ACR 50 in adalimumab vs 45% in upadacitinib (P≤ 0. 001)

QUESTION #1 • A 45 year old female brings in four new prescriptions. After reviewing them, it is determined the physician needs to be contacted. What is the problem with the following regimen? • Upadacitinib 15 mg by mouth once daily for initiation of therapy • Certolizumab 200 mg subcutaneously every two weeks for continuation of therapy • Methotrexate 15 mg by mouth once weekly for continuation of therapy • Folic acid 1 mg by mouth daily A. Inappropriate dosing: the methotrexate should be 20 mg daily B. Drug interaction: folic acid can’t be taken with methotrexate C. Upadacitinib can’t be taken with certolizumab D. The prescription for upadacitinib needs to be sent to a specialty pharmacy for compounding

QUESTION #1 • A 45 year old female brings in four new prescriptions. After reviewing them, it is determined the physician needs to be contacted. What is the problem with the following regimen? • Upadacitinib 15 mg by mouth once daily for initiation of therapy • Certolizumab 200 mg subcutaneously every two weeks for continuation of therapy • Methotrexate 15 mg by mouth once weekly for continuation of therapy • Folic acid 1 mg by mouth daily A. Inappropriate dosing: the methotrexate should be 20 mg daily B. Drug interaction: folic acid can’t be taken with methotrexate C. Upadacitinib can’t be taken with certolizumab D. The prescription for upadacitinib needs to be sent to a specialty pharmacy for compounding

QUESTION #2 What consideration must be taken when dispensing upadacitinib? A. Must be stored in the refrigerator until dispensed B. Must be mixed before dispensing to patient C. Pennsylvania Drug Monitoring Program (PDMP) needs to be checked before dispensing D. Must be dispensed in the original container

QUESTION #2 What consideration must be taken when dispensing upadacitinib? A. Must be stored in the refrigerator until dispensed B. Must be mixed before dispensing to patient C. Pennsylvania Drug Monitoring Program (PDMP) needs to be checked before dispensing D. Must be dispensed in the original container

CONCLUSION • Upadacitinib is a new biologic DMARD indicated for the treatment of rheumatoid arthritis in patients who have had an inadequate response to or cannot tolerate methotrexate • Upadacitinib can be used as monotherapy or combination therapy • Administered as a single pill once day • Upadacitinib may require handling precautions in accordance with USP <800> • Clinical data is proving upadacitinib to be superior to adalimumab in the treatment of rheumatoid arthritis and may become a preferred agent.

UPADACITINIB: A SUCCESSOR TO ADALIMUMAB? Miranda Ochs, Pharm. D PGY-1 Pharmacy Resident Millcreek Community Hospital October 26, 2019

REFERENCES • <800> Hazardous Drugs—Handling in Healthcare Settings. United States Pharmacopeia and National Formulary (USP 40 -NF 35). Rockville, MD: United States Pharmacopeia Convention; 2018: 84 -103. • Figure 3. 2015 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis Care Res (Hoboken). 2016; 68(1): 9. • Fleischmann R, Pangan AL, Mysler E, Bessette L, Peterfy C, Durez P, Ostor A, Li Y, Zhou Y, Othman AA, Song IH, Genovese MC. A Phase 3, Randomized, Double-Blind Study Comparing Upadacitinib to Placebo and to Adalimumab, in Patients with Active Rheumatoid Arthritis with Inadequate Response to Methotrexate [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 10). https: //acrabstracts. org/abstract/a-phase-3 -randomized-double-blind-study-comparingupadacitinib-to-placebo-and-to-adalimumab-in-patients-with-active-rheumatoid-arthritis-with-inadequate-response-tomethotrexate/. Accessed October 5, 2019. • Humira (adalimumab) [product monograph]. St-Laurent, Quebec, Canada: Abb. Vie Corporation; June 2019. • Rinvoq (upadacitinib) [prescribing information]. North Chicago, IL: Abb. Vie Inc; August 2019. • RINVOQ™ (upadacitinib) Treatment for Rheumatoid Arthritis. Rinvoq. https: //www. rinvoq. com/. Accessed October 5, 2019. • Singh JA, Saag KG, Bridges SL, et al. 2015 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis Care Res (Hoboken). 2016; 68(1): 1 -25. • Taylor P. Abb. Vie prices Rinvoq close to Humira after arthritis approval. pharmaphorum. https: //pharmaphorum. com/news/abbvie-prices-upadacitinib-close-to-humira-after-arthritis-approval/. Published August 19, 2019. Accessed October 5, 2019. • US Department of Health and Human Services; Centers for Disease Control and Prevention; National Institute for Occupational Safety and Health. NIOSH list of antineoplastic and other hazardous drugs in healthcare settings 2016. http: //www. cdc. gov/niosh/topics/antineoplastic/pdf/hazardous-drugs-list_2016 -161. pdf. Updated September 2016. Accessed October 6, 2019.
- Slides: 19