University of Pittsburgh Senior Design Bio E 11601161

University of Pittsburgh Senior Design – Bio. E 1160/1161 Design of a Wheelchair-Mounted Transfer Assist Device Michael Anderson Andrew Feola Jill Marion Bryan Shelly April 18, 2006 Mentors: Alicia Koontz, Ph. D, RET Jeremy Puhlman, BSE

Background • Wheelchair Patients • Have trouble transferring from chair • Bed, toilet, sofa, etc. • Must carry bulky transfer boards, benches

Current Solutions • Transfer boards, benches • Wall and ceiling-mounted devices • Portability Issues! • Our goal • To design a modified wheelchair armrest such that a transfer assist device is contained within the armrest

Market Analysis- Frost and Sullivan • North American Mobility Aids Market • Manual Wheelchairs • 313, 000 units in 2001(standard) • 77, 000 units in 2001 (lightweight) • 313, 000 units + 77, 000 units = 390, 000 units * $60 /unit= $23, 400, 000 market for our product to be used on manual wheelchairs Frost and Sullivan, 2001

Market Analysis- Frost and Sullivan • Lightweight manual wheelchairs • $102 million/ 77, 000 units in 2001 • $130 million in 2008 • Sunrise Medical Quickie II 30% of market • 0. 3 * 77, 000 = 23, 100 units * $60/unit = $1, 386, 000 market for our product to be used on the Quickie II model Frost and Sullivan, 2001

Market Analysis- Frost and Sullivan • Home Healthcare Market (US) • $2. 03 billion-devices in homecare (2001) • $1. 23 billion- home durable medical equipment (1999) Frost and Sullivan, 1999, 2001

Market Analysis- Frost and Sullivan • Growth rate of home healthcare market- 14% due to: • Increasing number of elderly (baby boomers) • Medicare moving toward covering at home treatments/devices more than in hospital • Improvements in technology • Allows greater number of medical procedures/monitoring to be done at home Frost and Sullivan, 2001

Design Requirements • Armrest converts into a transfer board • Fit in space occupied by standard armrest • Weight < 10 pounds • Provides same comfort as standard armrest • Able to support up to 250 pounds
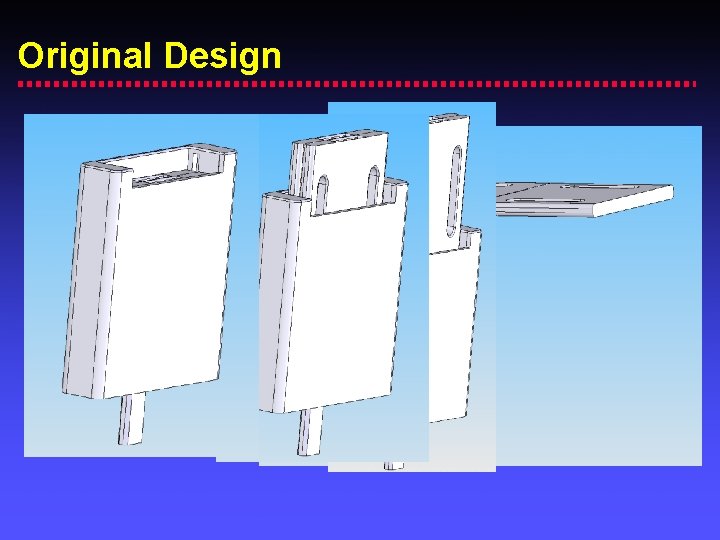
Original Design
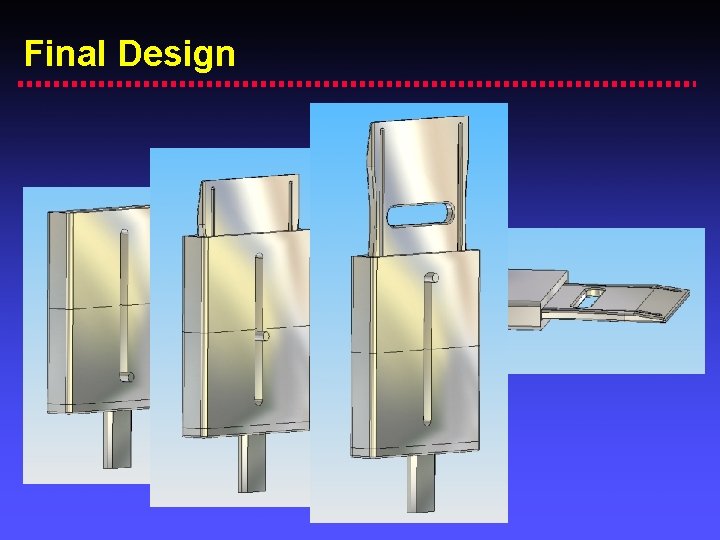
Final Design

Our Prototype

Our Prototype

Prototype Fabrication • Human Engineering Research Laboratory • Wire EDM • Aluminum 6061 Alloy (prototype) • Mass Production • Plastic Injection Molding • High-density polyethylene
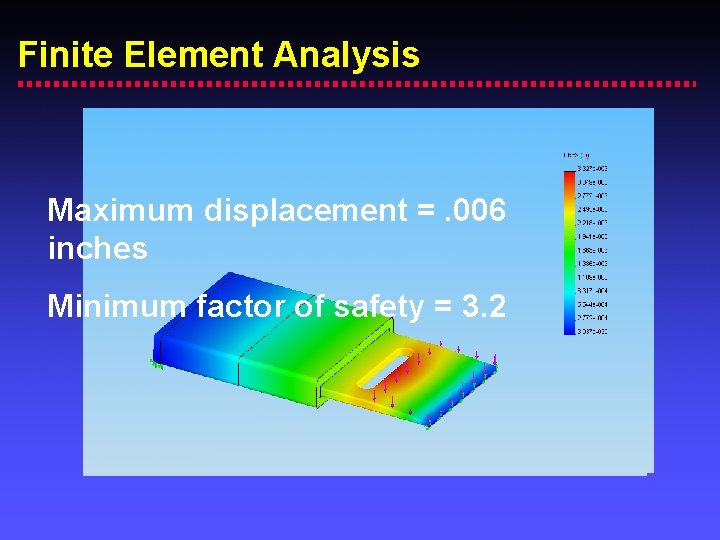
Finite Element Analysis Maximum displacement =. 006 inches Minimum factor of safety = 3. 2
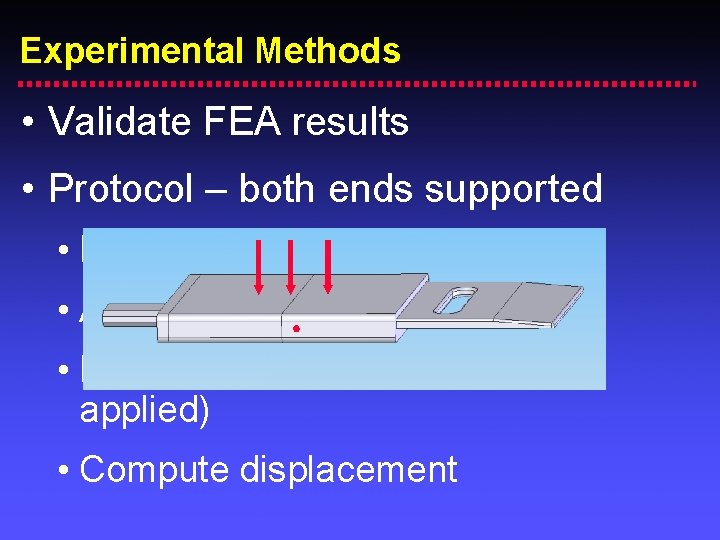
Experimental Methods • Validate FEA results • Protocol – both ends supported • Digitize a point (no load applied) • Apply load • Digitize same point (with load applied) • Compute displacement

Experimental Testing
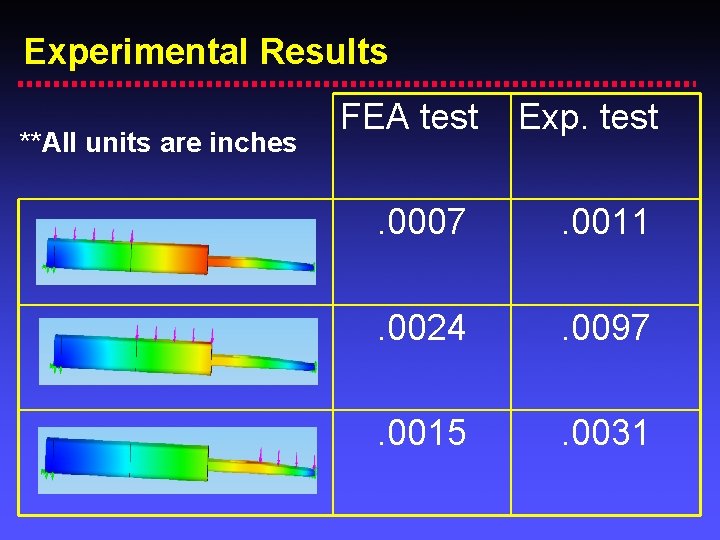
Experimental Results **All units are inches FEA test Exp. test . 0007 . 0011 . 0024 . 0097 . 0015 . 0031
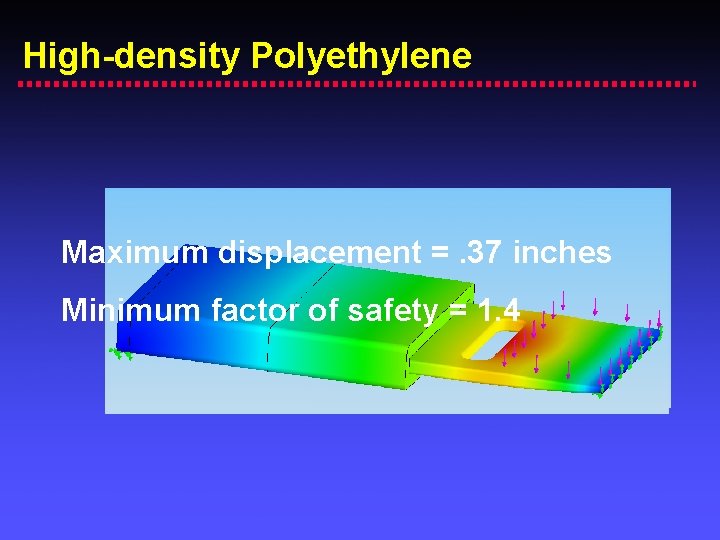
High-density Polyethylene Maximum displacement =. 37 inches Minimum factor of safety = 1. 4

Standard Transfer Board Maximum displacement = 0. 4 inches Minimum factor of safety = 1. 1

Survey Results • Eight (8) volunteers • 7 use transfer boards • 3. 9 / 5 difficulty rating of current boards • 4. 75 / 5 aesthetic rating of our product • All 7 would prefer a transfer board that does not need to be carried

Competitive Analysis Transfer bench • Invacare transfer bench • $120 • Carex transfer bench • $300 Standard transfer board • Allegro Medical wooden board • ~ $37 per unit • Therafin Theraslide transfer board • $100 per unit Our device • Plastic Injection Molding • $50 -60

Competitive Analysis Strengths • Price • Comparable to current transfer boards • Portability • Minimal additional weight Weaknesses • Limited armrest adjustability • One sliding mechanism

Constraints—Testing Human factors • Lack of human subject testing • Falls, etc. Production • Material availability • Testing done on aluminum, not plastic Economic • Cost of prototyping • Only one prototype

Manufacturability Considerations • Simple Design • Ease of injection molding • Standard shapes • Lack of small, irregular pieces • Ease of mass production

Human Factors Considerations • Standard transfer material • Patient can slide easily across board • Cushioning consistent with standard armrests • Handles in transfer board • No sharp edges

FDA Regulation • CDRH website • Sec. 890. 3910 Wheelchair accessories • Includes armrests, transfer boards • Class I device http: //www. accessdata. fda. gov/scripts/cdrh/cfdocs/cf. PCD/classification. cfm

Individual Project Work • Mike Anderson • Andrew Feola • Solid. Works design • Prototype testing • COSMOSWorks analysis • Prototype testing • DHF, SBIR • Jill Marion • Contact companies and patients • Market Analysis • Prototype testing • DHF, SBIR • Bryan Shelly • Material selection • DHF, SBIR • Manufacturing
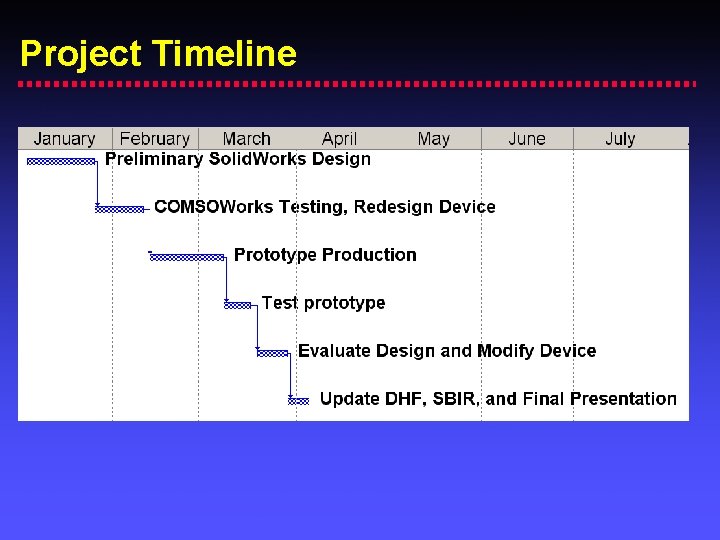
Project Timeline

Future Considerations • Human subject testing • Additional sliding mechanism • Armrest height adjustability

Acknowledgements Alicia Koontz, Ph. D, RET Jeremy Puhlman, BSE Alexis Wickwire, BS Human Engineering Research Labs Pittsburgh Life Sciences Greenhouse University of Pittsburgh Bio. E Dept. A generous gift from Dr. Hal Wrigley and Dr. Linda Baker

Thank You • Questions?
- Slides: 31