Units of Measurement THE SI SYSTEM AND UNITS










- Slides: 14

Units of Measurement THE SI SYSTEM AND UNITS OF MEASUREMENT

MEASUREMENT Measurement is used to measure quantities. Quantity is something that has magnitude, size, or amount (volume). A quantity is not the same as a measurement.

SI SYSTEM OF MEASUREMENT In the late 18 th century, scientists used the metric system. The metric system is a forerunner to the SI System. Scientists all over the world use a single measurement system called Le Systeme International d’Units, abbreviated SI. It was adopted in 1960 and has both base units and derived units.

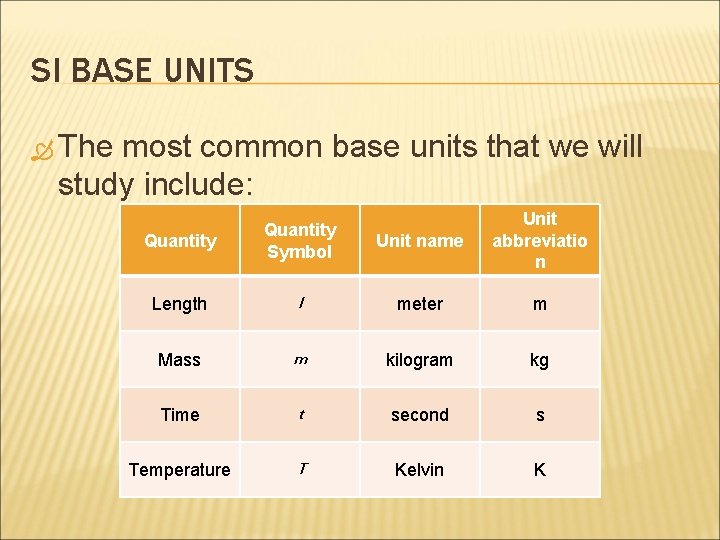
SI BASE UNITS The most common base units that we will study include: Quantity Symbol Unit name Unit abbreviatio n Length l meter m Mass m kilogram kg Time t second s Temperature T Kelvin K

LENGTH The SI standard unit for length is the meter. A distance of 1 m is about the width of an average doorway. To express longer distances, the kilometer (km) is used. One kilometer is equal to 1000 m. To express shorter distances, the centimeter (cm) is used. One centimeter is equal to 1/100 of a meter. Length can be measured using a meter stick or rulers.

MASS Mass is a measure of the quantity of matter. The standard unit for mass is the kilogram (kg). The gram (g), which is 1/1000 of a kg is used for measuring masses of small objects such as flasks and beakers. Mass is typically measured using a balance. Mass is different from weight. Weight is a measure of the gravitational pull on matter. The weight of an object increases as gravity acts on it. The weight of an object on the moon is about 1/6 th of its weight on Earth. So, a human on Earth who weighed 126 lbs. would weight how much on the moon?

TIME The standard unit of measurement for time is the second (s). Time can be measured using stop watches, clocks, count down timers, and other time pieces. Larger amounts of time are measured in minutes and hours. There are 60 seconds in one minute. There are 60 minutes in one hour. Given that there are 24 hours in one day, how many seconds are there in one day?

TEMPERATURE The standard unit of measurement for temperature is degrees Kelvin (K). Temperature can also be measured in degrees Celsius (°C) and degrees Fahrenheit (°F). To convert degrees Celsius (°C) to degrees Fahrenheit (°F) multiply by 1. 8 and then add 32. To convert degrees Fahrenheit to degrees Celsius, subtract 32 and then divide by 1. 8 ° K= °C + 273 Temperature is measured using a thermometer. (measures the degree of heat or coolness)

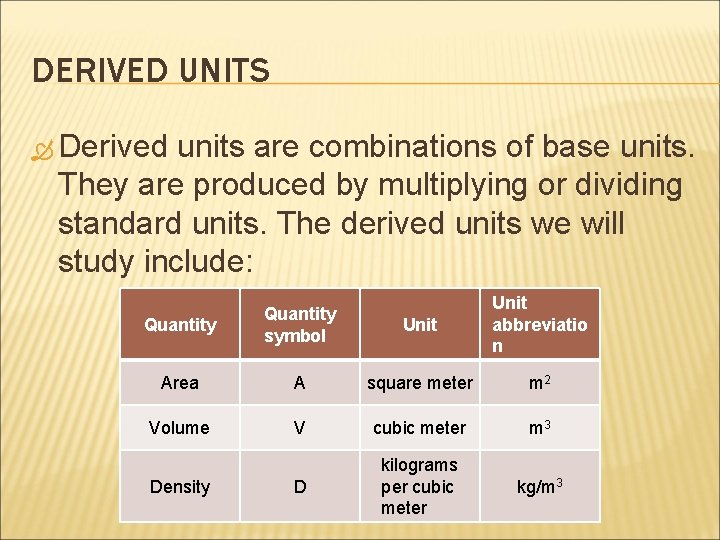
DERIVED UNITS Derived units are combinations of base units. They are produced by multiplying or dividing standard units. The derived units we will study include: Quantity symbol Unit abbreviatio n Area A square meter m 2 Volume V cubic meter m 3 D kilograms per cubic meter kg/m 3 Density

AREA � Area is length times the width. It is expressed as square meters. � Area can also be expressed as cubic centimeters. � What is the area of a rectangle that has an a length of 6 cm and a width of 16 cm? 16 cm � � 1 11111 cmam 6 cm

VOLUME Volume is the amount of space occupied by an object. The derived SI unit for volume is cubic meters. The cubic meter is rather large, so a more common unit of cubic centimeters is more commonly used. Non-SI units are also used to measure volume such as the milliliter (m. L) and the liter (L), which is 1000 cm 3. There are 1000 m. L in 1 L. Beakers, flasks, and graduated cylinders are often used to determine the volume of liquids.

DENSITY Density is the ratio of mass to volume, or mass divided by volume. It can be written: density=mass/volume or D=m/V Density is a characteristic physical property of a substance that does not depend on the size of the sample. As the mass of an object increases, its volume increases. * Question: Are raisins, more or less dense than soda water? Let’s see!

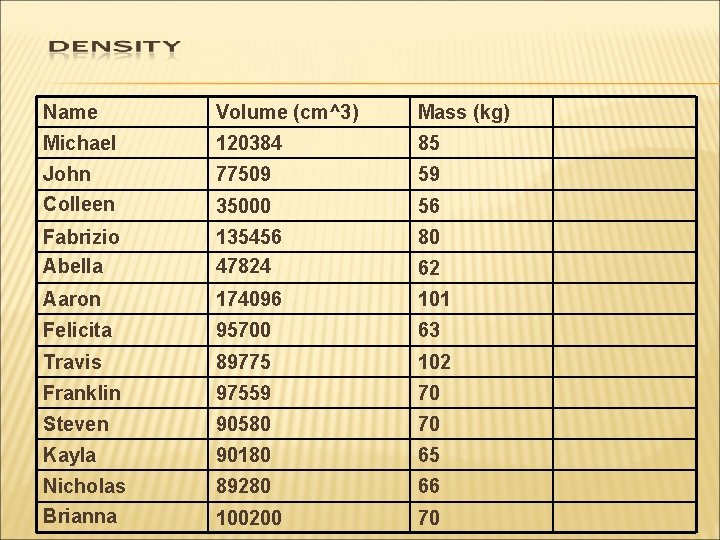
Name Volume (cm^3) Mass (kg) Michael 120384 85 John Colleen 77509 59 35000 56 Fabrizio Abella 135456 47824 80 Aaron 174096 101 Felicita 95700 63 Travis 89775 102 Franklin 97559 70 Steven 90580 70 Kayla 90180 65 Nicholas Brianna 89280 66 100200 70 62

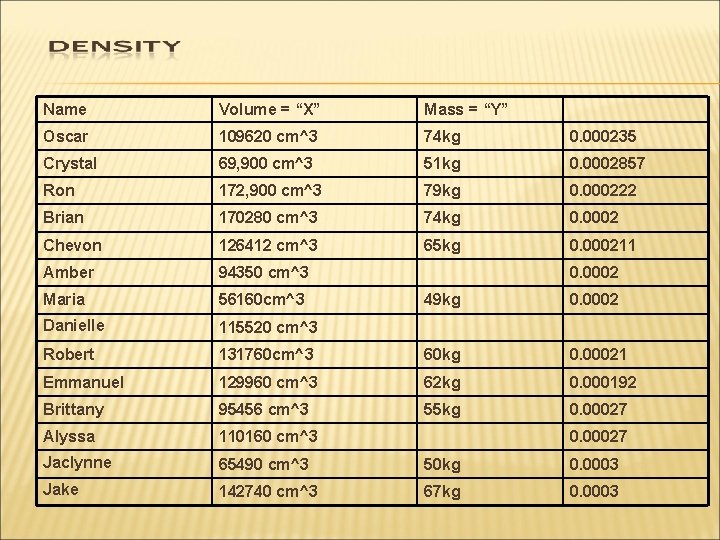
Name Volume = “X” Mass = “Y” Oscar 109620 cm^3 74 kg 0. 000235 Crystal 69, 900 cm^3 51 kg 0. 0002857 Ron 172, 900 cm^3 79 kg 0. 000222 Brian 170280 cm^3 74 kg 0. 0002 Chevon 126412 cm^3 65 kg 0. 000211 Amber 94350 cm^3 Maria 56160 cm^3 Danielle 115520 cm^3 Robert 0. 0002 49 kg 0. 0002 131760 cm^3 60 kg 0. 00021 Emmanuel 129960 cm^3 62 kg 0. 000192 Brittany 95456 cm^3 55 kg 0. 00027 Alyssa 110160 cm^3 Jaclynne 65490 cm^3 50 kg 0. 0003 Jake 142740 cm^3 67 kg 0. 0003 0. 00027