Units of Measurement SI Units Systme International dUnits

















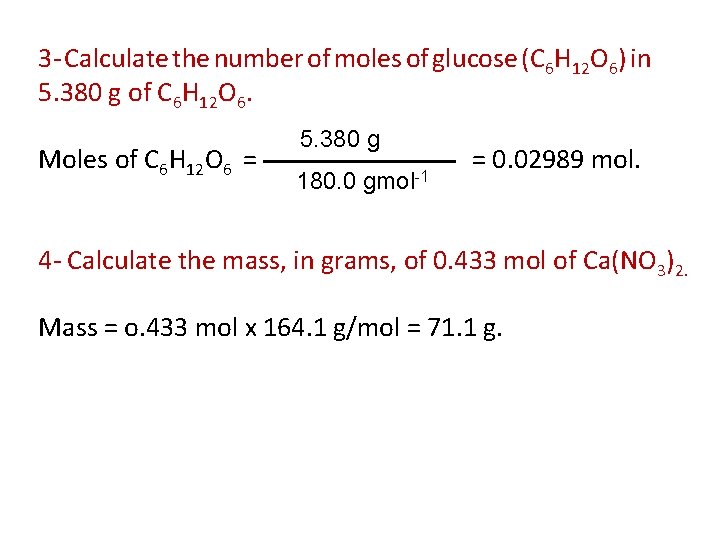
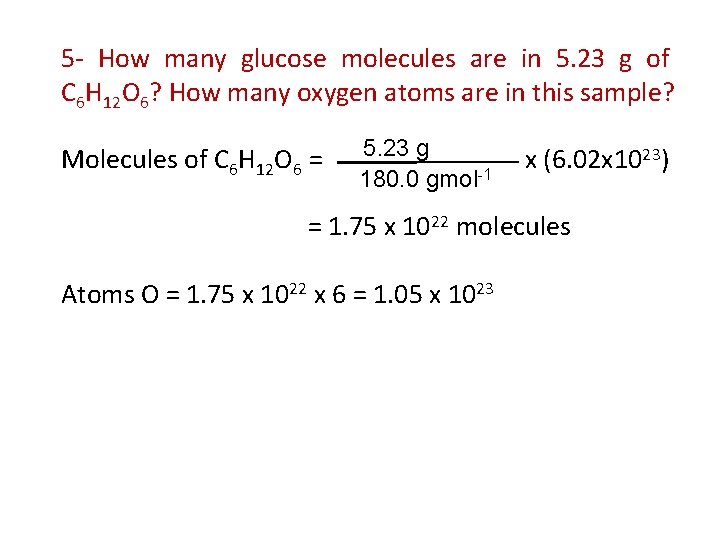



































- Slides: 66


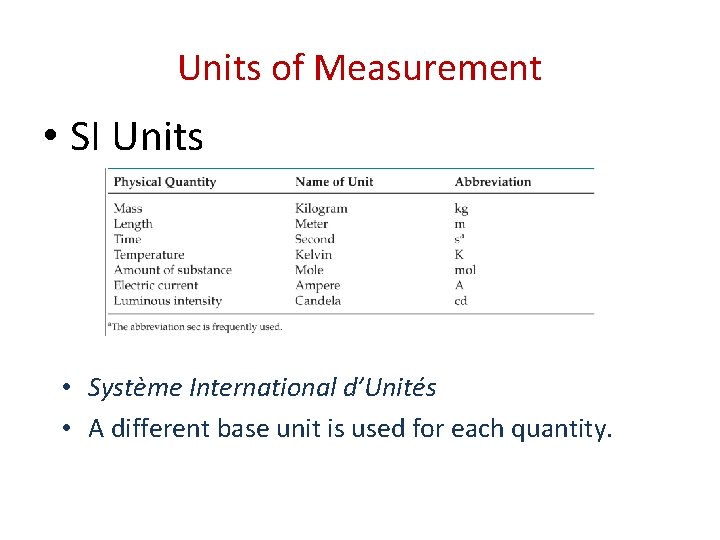
Units of Measurement • SI Units • Système International d’Unités • A different base unit is used for each quantity.

Metric System Prefixes convert the base units into units that are appropriate for the item being measured.

Volume • The most commonly used metric units for volume are the liter (L) and the milliliter (m. L). – A liter is a cube 1 dm long on each side. – A milliliter is a cube 1 cm long on each side.

Density is a physical property of a substance. mass Density = Volume d= m V

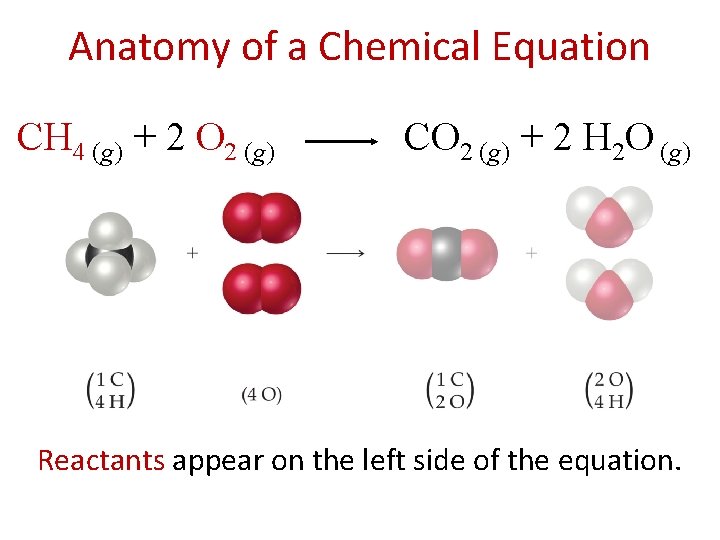
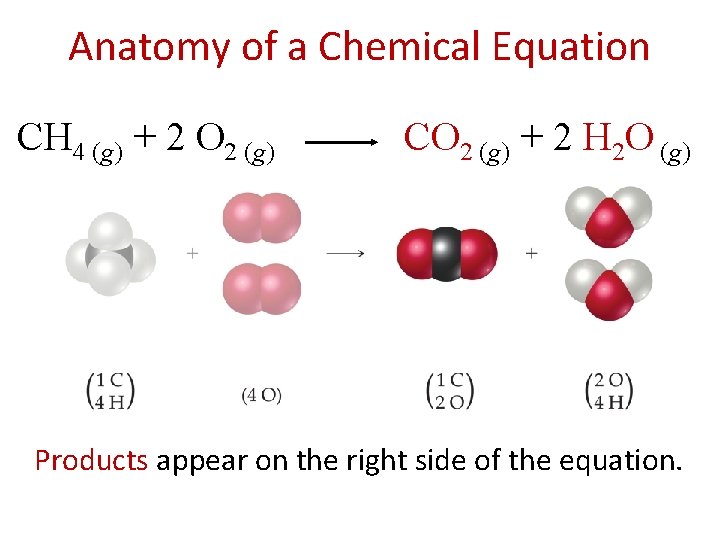
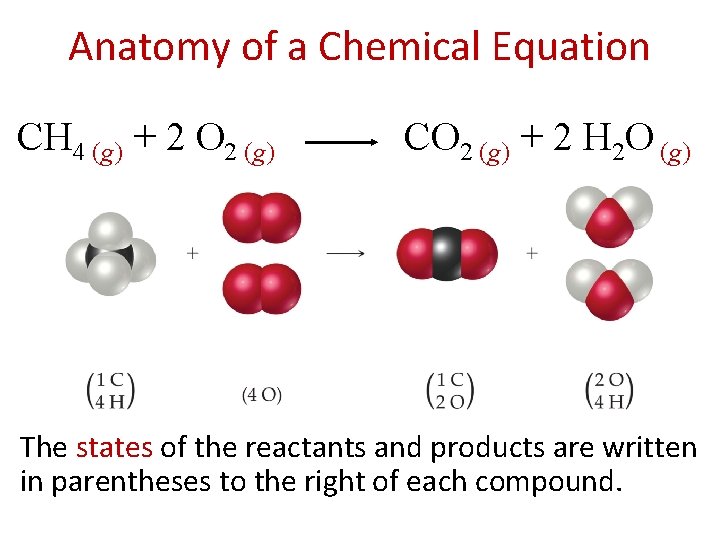
Chemical Equations Chemical equations are concise representations of chemical reactions. CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (g)

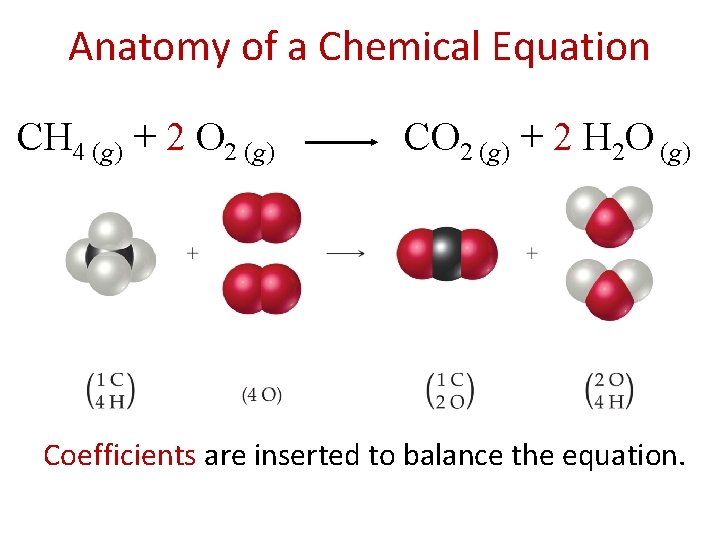
Anatomy of a Chemical Equation CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (g) Reactants appear on the left side of the equation.

Anatomy of a Chemical Equation CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (g) Products appear on the right side of the equation.

Anatomy of a Chemical Equation CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (g) The states of the reactants and products are written in parentheses to the right of each compound.

Anatomy of a Chemical Equation CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (g) Coefficients are inserted to balance the equation.

Subscripts and Coefficients Give Different Information • Subscripts tell the number of atoms of each element in a molecule.

Subscripts and Coefficients Give Different Information • Coefficients tell the number of molecules.

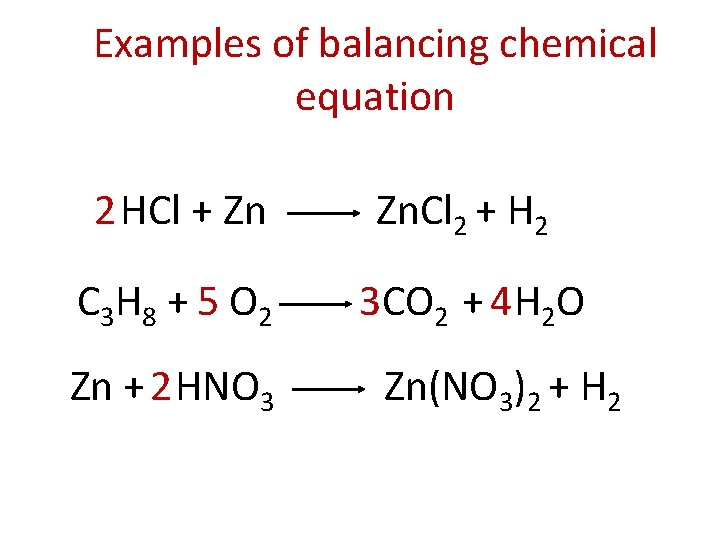
Examples of balancing chemical equation 2 HCl + Zn Zn. Cl 2 + H 2 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O Zn + 2 HNO 3 Zn(NO 3)2 + H 2

Reaction Types Combination & decomposition reactions Combustion in Air

Combination Reactions • Examples: • In this type of reaction two or more substances react to form one product. – 2 Mg (s) + O 2 (g) 2 Mg. O (s) – N 2 (g) + 3 H 2 (g) 2 NH 3 (g) – C 3 H 6 (g) + Br 2 (l) C 3 H 6 Br 2 (l)

Decomposition Reactions • In a decomposition one substance breaks down into two or more substances. • Examples: – Ca. CO 3 (s) Ca. O (s) + CO 2 (g) – 2 KCl. O 3 (s) 2 KCl (s) + O 2 (g) – 2 Na. N 3 (s) 2 Na (s) + 3 N 2 (g)

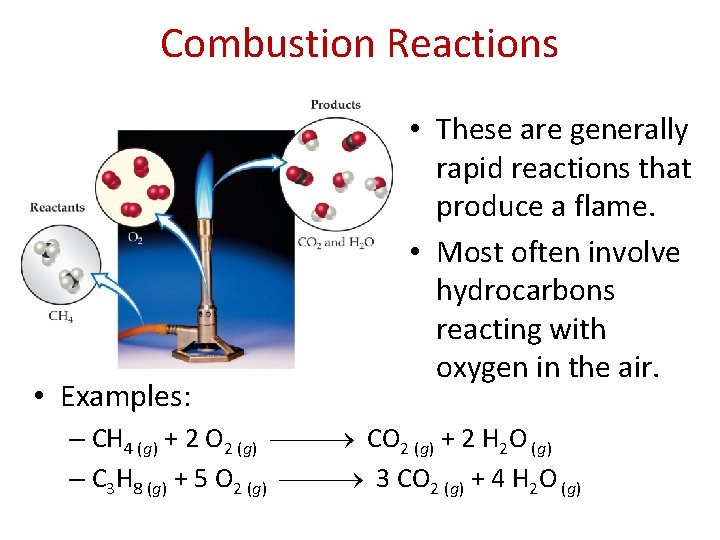
Combustion Reactions • Examples: • These are generally rapid reactions that produce a flame. • Most often involve hydrocarbons reacting with oxygen in the air. – CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (g) – C 3 H 8 (g) + 5 O 2 (g) 3 CO 2 (g) + 4 H 2 O (g)

Formula Weights(FW) • A formula weight is the sum of the atomic weights for the atoms in a chemical formula. • So, the formula weight of calcium chloride, Ca. Cl 2, would be Ca: 1(40. 1 amu*) + Cl: 2(35. 5 amu) 111. 1 amu • Formula weights are generally reported for ionic compounds. *atomic mass unit

Molecular Weight (MW) • A molecular weight is the sum of the atomic weights of the atoms in a molecule. • For the molecule ethane, C 2 H 6, the molecular weight would be C: 2(12. 0 amu) + H: 6(1. 0 amu) 30. 0 amu

Percent Composition One can find the percentage of the mass of a compound that comes from each of the elements in the compound by using this equation: % element = (number of atoms)(atomic weight) (FW of the compound) x 100

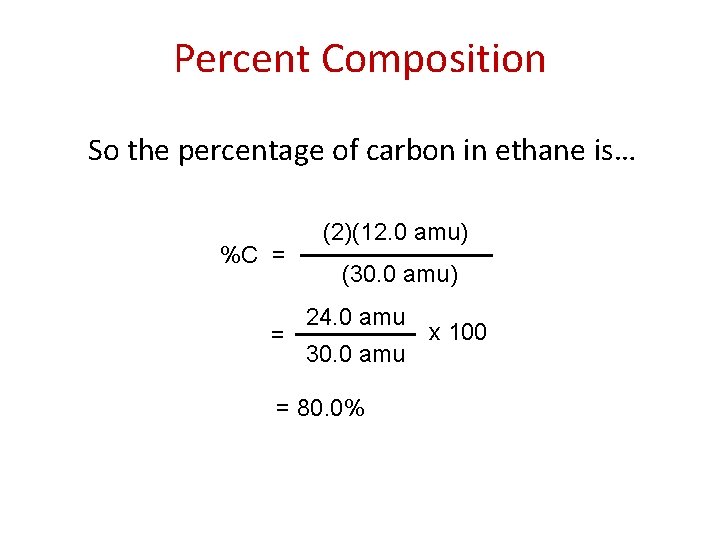
Percent Composition So the percentage of carbon in ethane is… %C = = (2)(12. 0 amu) (30. 0 amu) 24. 0 amu 30. 0 amu = 80. 0% x 100

Avogadro’s Number • 6. 02 x 1023 • 1 mole of 12 C has a mass of 12 g. • 1 mole of H 2 O has a mass of 18 g.

Molar Mass • By definition, a molar mass is the mass of 1 mol of a substance (i. e. , g/mol). – The molar mass of an element is the mass number for the element that we find on the periodic table. – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol).

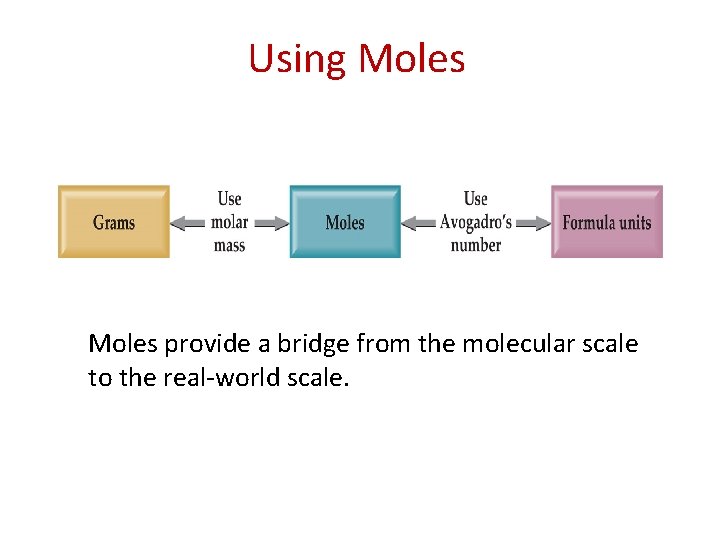
Using Moles provide a bridge from the molecular scale to the real-world scale.

Mole Relationships • One mole of atoms, ions, or molecules contains Avogadro’s number of those particles. • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound.

Examples 1 - Calculate how many atoms there are in 0. 200 moles of copper. The number of atoms in one mole of Cu is equal to the Avogadro number = 6. 02 x 1023. Number of atoms in 0. 200 moles of Cu = (0. 200 mol) x (6. 02 x 1023 mol-1 ) = 1. 20 x 1023. 2 - Calculate how molecules of H 2 O there are in 12. 10 moles of water. Number of water molecules = (12. 10 mol)x(6. 02 x 1023) = 7. 287 x 1024
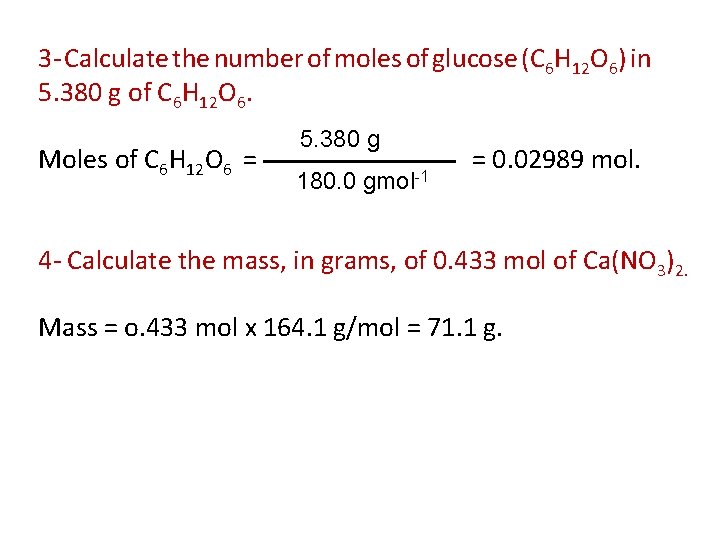
3 - Calculate the number of moles of glucose (C 6 H 12 O 6) in 5. 380 g of C 6 H 12 O 6. Moles of C 6 H 12 O 6 = 5. 380 g 180. 0 gmol-1 = 0. 02989 mol. 4 - Calculate the mass, in grams, of 0. 433 mol of Ca(NO 3)2. Mass = o. 433 mol x 164. 1 g/mol = 71. 1 g.
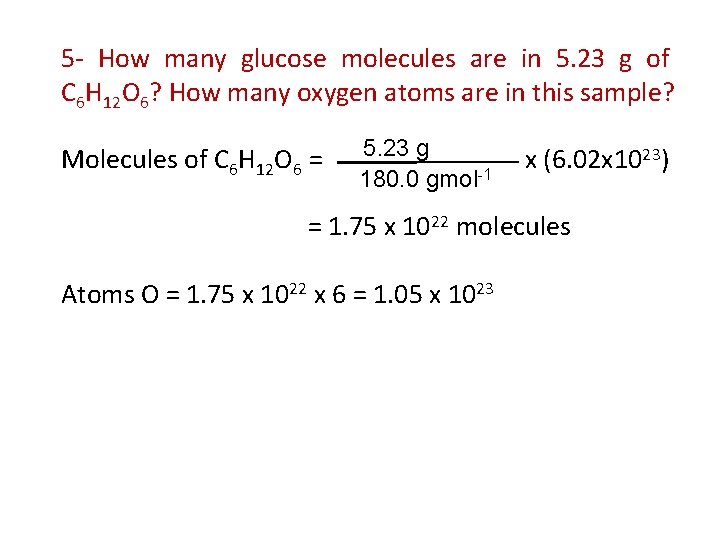
5 - How many glucose molecules are in 5. 23 g of C 6 H 12 O 6? How many oxygen atoms are in this sample? Molecules of C 6 H 12 O 6 = 5. 23 g 180. 0 gmol-1 x (6. 02 x 1023) = 1. 75 x 1022 molecules Atoms O = 1. 75 x 1022 x 6 = 1. 05 x 1023

Finding Empirical Formulas

Calculating Empirical Formulas One can calculate the empirical formula from the percent composition.

Calculating Empirical Formulas The compound para-aminobenzoic acid is composed of carbon (61. 31%), hydrogen (5. 14%), nitrogen (10. 21%), and oxygen (23. 33%). Find the empirical formula of PABA.

Calculating Empirical Formulas Assuming 100. 00 g of para-aminobenzoic acid, C: 61. 31 g x H: 5. 14 g x N: 10. 21 g x O: 23. 33 g x 1 mol 12. 01 g 1 mol 14. 01 g 1 mol 16. 00 g = 5. 105 mol C = 5. 09 mol H = 0. 7288 mol N = 1. 456 mol O

Calculating Empirical Formulas Calculate the mole ratio by dividing by the smallest number of moles: C: 5. 105 mol 0. 7288 mol = 7. 005 7 H: 5. 09 mol 0. 7288 mol = 6. 984 7 N: 0. 7288 mol = 1. 000 O: 1. 458 mol 0. 7288 mol = 2. 001 2

Calculating Empirical Formulas These are the subscripts for the empirical formula: C 7 H 7 NO 2

Combustion Analysis • Compounds containing C, H and O are routinely analyzed through combustion in a chamber like this. – C is determined from the mass of CO 2 produced. – H is determined from the mass of H 2 O produced. – O is determined by difference after the C and H have been determined.

Determining Empirical Formula by Combustion Analysis *Combustion of 0. 255 g of isopropyl alcohol produces 0. 561 g of CO 2 and 0. 306 g of H 2 O. Determine the empirical formula of isopropyl alcohol. 0. 561 g CO 2 Grams of C = x 12 gmol-1 = 0. 153 g -1 44 gmol 0. 306 g H 2 O Grams of H = x 2 x 1 gmol-1 = 0. 0343 g -1 18 gmol Grams of O = mass of sample – (mass of C + mass of H) = 0. 255 g – ( 0. 153 g + 0. 0343 g) = 0. 068 g

0. 153 g C Moles of C = = 0. 0128 mol -1 12 gmol Moles of H = 0. 0343 g H = 0. 0343 mol -1 1 gmol 0. 068 g O Moles of O = = 0. 0043 mol 16 gmol-1 Calculate the mole ratio by dividing by the smallest number of moles: C: H: O 2. 98: 7. 91: 1. 00 C 3 H 8 O

Stoichiometric Calculations The coefficients in the balanced equation give the ratio of moles of reactants and products.

Stoichiometric Calculations Starting with the mass of Substance A you can use the ratio of the coefficients of A and B to calculate the mass of Substance B formed (if it’s a product) or used (if it’s a reactant).

Stoichiometric Calculations C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O Starting with 1. 00 g of C 6 H 12 O 6… we calculate the moles of C 6 H 12 O 6… use the coefficients to find the moles of H 2 O… and then turn the moles of water to grams.

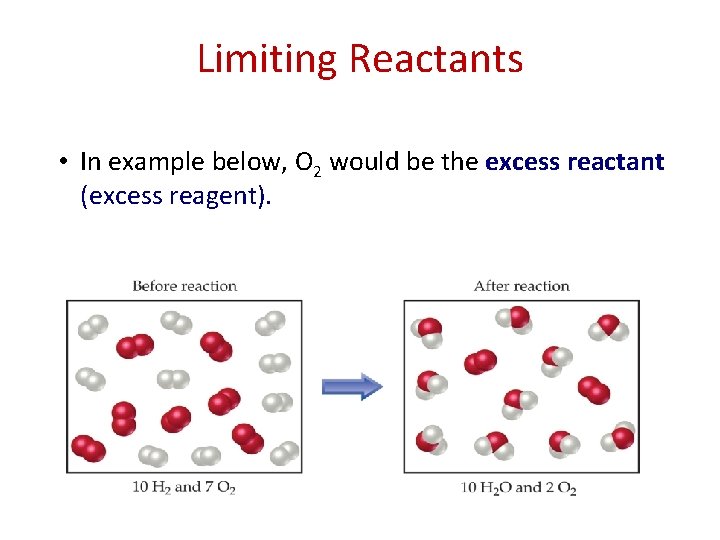
Limiting Reactants • The reactant that is completely consumed in a reaction is called the limiting reactant (limiting reagent). – In other words, it’s the reactant that run out first (in this case, the H 2).

Limiting Reactants • In example below, O 2 would be the excess reactant (excess reagent).

Theoretical Yield • The theoretical yield is the maximum amount of product that can be made. • The amount of product actually obtained in a reaction is called the actual yield.

Theoretical Yield • The actual yield is almost always less than theoretical yield. Why? • Part of the reactants may not react. • Side reaction. • Difficult recovery.

Percent Yield • The percent yield of a reaction relates to the actual yield to theoretical (calculated) yield. Percent Yield = Actual Yield Theoretical Yield x 100

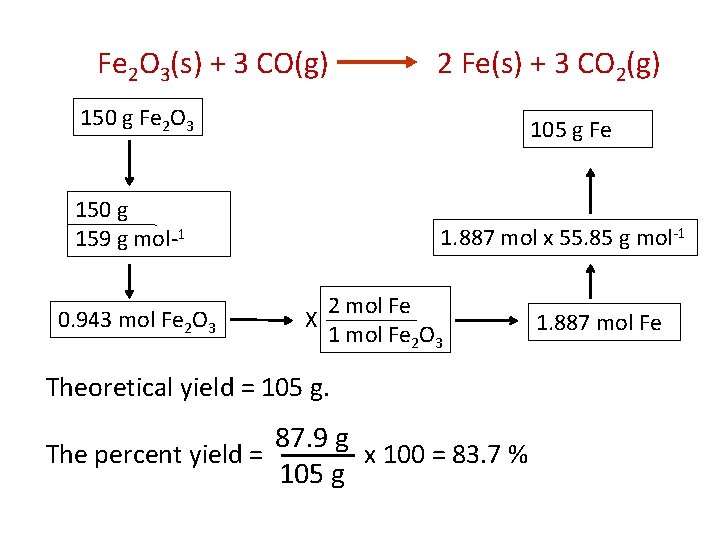
Examples Fe 2 O 3(s) + 3 CO(g) 2 Fe(s) + 3 CO 2(g) If we start with 150 g of Fe 2 O 3 as the limiting reactant, and found actual yield of Fe was 87. 9 g, what is the percent yield? Actual Yield The percent yield = x 100 Theoretical Yield

Fe 2 O 3(s) + 3 CO(g) 2 Fe(s) + 3 CO 2(g) 150 g Fe 2 O 3 105 g Fe 150 g 159 g mol-1 0. 943 mol Fe 2 O 3 1. 887 mol x 55. 85 g mol-1 X 2 mol Fe 1 mol Fe 2 O 3 Theoretical yield = 105 g. 87. 9 g The percent yield = x 100 = 83. 7 % 105 g 1. 887 mol Fe

Solutions • Solutions are defined as homogeneous mixtures of two or more pure substances. • The solvent is present in greatest abundance. • All other substances are solutes.

Molarity • Two solutions can contain the same compounds but be quite different because the proportions of those compounds are different. • Molarity is one way to measure the concentration of a solution. Molarity (M) = moles of solute volume of solution in liters

Mixing a Solution • To create a solution of a known molarity, one weighs out a known mass (and, therefore, number of moles) of the solute. • The solute is added to a volumetric flask, and solvent is added to the line on the neck of the flask.

Examples Calculate the molarity of a solution made by dissolving 0. 750 g of sodium sulfate (Na 2 SO 4) in enough water to form 850 m. L of solution. Moles of Na 2 SO 4 = 0. 750 g = 0. 0053 mol -1 142 g mol 0. 0053 mol Molarity = = 0. 0062 M 0. 850 L

How many moles of KMn. O 4 are present in 250 m. L of a 0. 0475 M solution? mol M= L mol Moles of KMn. O 4 = 0. 0475 x 0. 25 L L = 0. 012 mol

How many milliliters of 11. 6 M HCl solution are needed to obtain 0. 250 mol of HCl? Molarity (M) = moles of solute volume of solution in liters 0. 250 mol 11. 6 M = volume in liters = 0. 022 L = 22 m. L.

What are the molar concentrations of each of the ions present in a 0. 025 M aqueous solution of Ca(NO 3)2? Ca 2+ = 0. 025 M NO 3 - = 0. 025 x 2 = 0. 05 M

Dilution • One can also dilute a more concentrated solution by – Using a pipet to deliver a volume of the solution to a new volumetric flask, and – Adding solvent to the line on the neck of the new flask.

Dilution The molarity of the new solution can be determined from the equation M c Vc = M d Vd where Mc and Md are the molarity of the concentrated and dilute solutions, respectively, and Vc and Vd are the volumes of the two solutions.

Dilution M c Vc = M d Vd Moles solute before dilution = moles solute after dilution

Examples How many milliliters of 3. 0 M H 2 SO 4 are needed to make 450 m. L of 0. 10 M H 2 SO 4 ? Mc Vc = Md VV 3. 0 M x Vc = 0. 10 M x 450 m. L Vc = 15 m. L

Ways of Expressing Concentrations of Solutions • Mass Percentage, ppm, and ppb Ø Mass Percentage mass of A in solution Mass % of A = total mass of solution 100 Example: 36% HCl by mass contains 36 g of HCl for each 100 g of solution (64 g H 2 O)

Ø Parts per Million (ppm) mass of A in solution ppm = total mass of solution 106 ØParts per Billion (ppb) mass of A in solution ppb = total mass of solution 109

Examples Calculate the mass percentage of Na 2 SO 4 in a solution containing 10. 6 g Na 2 SO 4 in 483 g water. 10. 6 g 100 Mass % of Na 2 SO 4 = (483 + 10. 6) g = 2. 15 %

An ore contains 2. 86 g of silver per ton of ore. What is the concentration of silver in ppm? ppm = 2. 86 g 106 g = 2. 86 ppm

• Mole Fraction, Molarity, and Molality Ø Mole Fraction (X) XA = moles of A total moles in solution Ø Molarity (M) = moles of solute volume of solution in liters

üSince volume is temperature-dependent, molarity can change with temperature. Ø Molality (m) moles of solute m= Kilograms of solvent ü Since both moles and mass do not change with temperature, molality (unlike molarity) is not temperature-dependent.

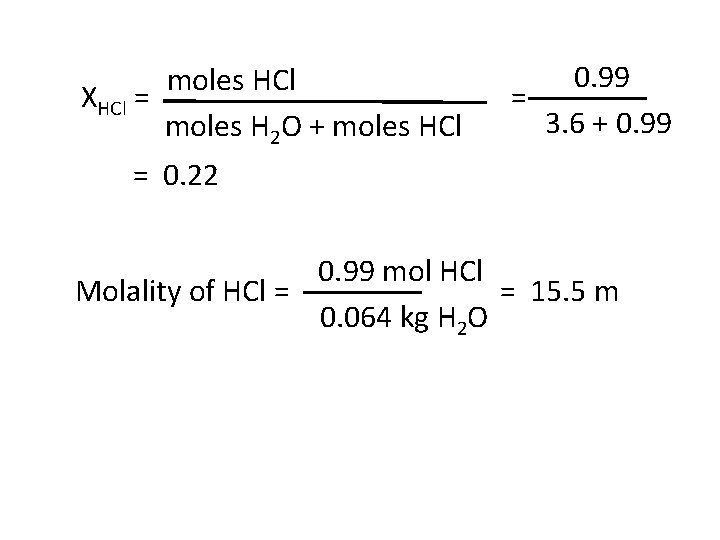
Examples An aqueous solution of hydrochloric acid contains 36 % HCl by mass. (a) Calculate the mole fraction of HCl in the solution. (b) Calculate the molality of HCl in the solution. Moles HCl = 36 g = 0. 99 mol 36. 5 g mol-1 Moles H 2 O = 64 g = 3. 6 mol 18 g mol-1

moles HCl XHCl = moles H 2 O + moles HCl 0. 99 = 3. 6 + 0. 99 = 0. 22 0. 99 mol HCl Molality of HCl = = 15. 5 m 0. 064 kg H 2 O