Units of Measure Conversions Number vs Quantity Quantity

Units of Measure & Conversions

Number vs. Quantity Ø Quantity - number + unit UNITS MATTER!!

SI Units Quantity Symb Base Unit Abbre ol v. l Length meter m Mass m kilogram kg Time t second s Temp T kelvin K Amount n mole mol

SI Units Prefix mega- Symbol M Factor 106 kilo- k 103 BASE UNIT --- 100 deci- d 10 -1 centi- c 10 -2 milli- m 10 -3

SI Prefix Conversions 1. Find the difference between the exponents of the two prefixes. 2. Move the decimal that many places. To the left or right?

SI Prefix Conversions 532 m NUMBER UNIT 0. 532 km = _______ = NUMBER UNIT

SI Prefix Conversions move right move left Prefix mega- Symbol M Factor 106 kilo- k 103 BASE UNIT --- 100 deci- d 10 -1 centi- c 10 -2 milli- m 10 -3 micro- 10 -6 nano- n 10 -9 pico- p 10 -12

SI Prefix Conversions 1) 20 cm = 0. 2 _______ m 32 2) 0. 032 L = _______ m. L 3) 45 m = 45, 000 _______ nm 0. 0805 4) 805 dm = _______ km

Dimensional Analysis Ø The “Factor-Label” Method w Units, or “labels” are canceled, or “factored” out

Dimensional Analysis Ø Steps: 1. Identify starting & ending units. 2. Line up conversion factors so units cancel. 3. Multiply all top numbers & divide by each bottom number. 4. Check units & answer.

Dimensional Analysis 5) Your European hairdresser wants to cut your hair 8. 0 cm shorter. How many inches will he be cutting off? cm in 8. 0 cm 1 in 2. 54 cm = 3. 2 in

Dimensional Analysis 6) Lake Taylor football needs 550 cm for a 1 st down. How many yards is this? cm 550 cm yd 1 in 1 ft 1 yd 2. 54 cm 12 in 3 ft = 6. 0 yd

Dimensional Analysis 7) A piece of wire is 1. 3 m long. How many 1. 5 -cm pieces can be cut from this wire? cm pieces 1. 3 m 100 cm 1 m 1 piece 1. 5 cm = 86 pieces

Warm-Up: Density Ø What is the density of an object that has a mass of 8. 76 g and a volume of 3. 07 cm 3? Ø Find the density of a granite if a rectangular piece, 5. 00 cm by 10. 0 cm by 23. 0 cm, has a mass of 3220 g. Ø Iron has a density of 7. 9 g/m. L. What volume would 10. 0 g of iron occupy? Ø Determine the density of an object that has a mass of 50. 50 g and when placed in a graduated cylinder containing 50. 0 m. L of water causes the new volume to be 63. 2 m. L.

Ø In your book read section 1. 3 p 22 -26 Ø Make notes on the different types of graphs and when we use them Ø Do practice problem #1 -3 on p 24

Scientific Notation 65, 000 kg 6. 5 × 104 kg Ø Converting into Scientific Notation: w Move decimal until there’s 1 digit to its left. Places moved = exponent. w Large # (>1) positive exponent Small # (<1) negative exponent w Only include sig figs.
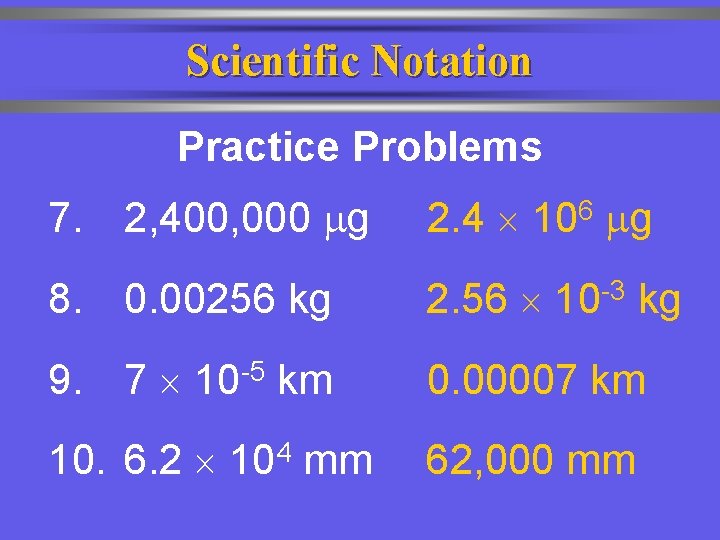
Scientific Notation Practice Problems 7. 2, 400, 000 g 2. 4 8. 0. 00256 kg 2. 56 9. 7 10 -5 km 0. 00007 km 10. 6. 2 104 mm 62, 000 mm 6 10 g -3 10 kg

Scientific Notation Ø Calculating with Scientific Notation (5. 44 × 107 g) ÷ (8. 1 × 104 mol) = Type on your calculator: 5. 44 2 nd EE 7 ÷ 8. 1 2 nd EE 4 ENTER = 671. 6049383 = 670 g/mol = 6. 7 × 102 g/mol


Accuracy vs. Precision Ø Accuracy - how close a measurement is to the accepted value Ø Precision - how close a series of measurements are to each other ACCURATE = CORRECT PRECISE = CONSISTENT

Percent Error Ø Indicates accuracy of a measurement your value / actual accepted value / theoretical

Percent Error Ø A student determines the density of a substance to be 1. 40 g/m. L. Find the % error if the accepted value of the density is 1. 36 g/m. L. % error = 2. 9 %
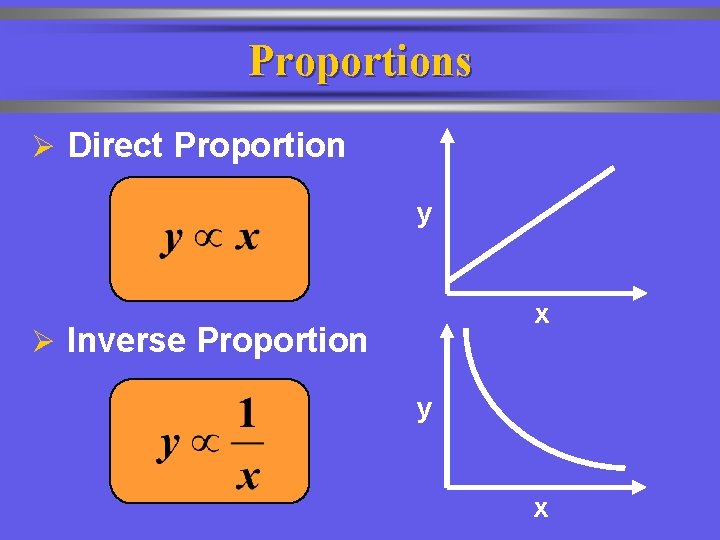
Proportions Ø Direct Proportion y x Ø Inverse Proportion y x

Derived Units Ø Combination of base units. 1 cm 3 = 1 m. L w length 1 dm 3 = 1 L Ø Volume (m 3 or cm 3) Ø Density (kg/m 3 or g/cm 3) w mass per volume M D= V
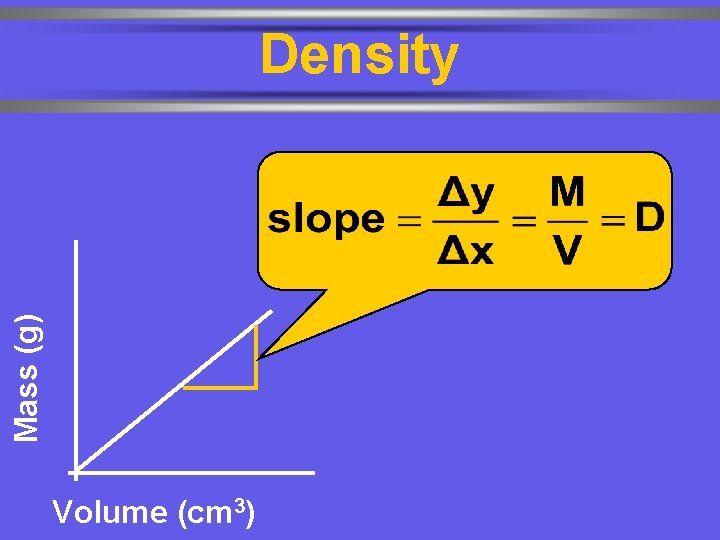
Mass (g) Density Volume (cm 3)

Density Ø An object has a volume of 825 cm 3 and a density of 13. 6 g/cm 3. Find its mass. GIVEN: WORK: V = 825 cm 3 D = 13. 6 g/cm 3 M=? M = DV M = (13. 6 g/cm 3)(825 cm 3) M = 11, 200 g

Density Ø A liquid has a density of 0. 87 g/m. L. What volume is occupied by 25 g of the liquid? GIVEN: WORK: D = 0. 87 g/m. L V=? M = 25 g V=M D V= 25 g 0. 87 g/m. L V = 29 m. L


Significant Figures Ø Indicate precision of a measurement. Ø Recording Sig Figs w Sig figs in a measurement include the known digits plus a final estimated digit 2. 35 cm

Significant Figures Ø Counting Sig Figs (pg. 57) w Count all numbers EXCEPT: ² Leading ² Trailing zeros -- 0. 0025 zeros without a decimal point -- 2, 500

Significant Figures Counting Sig Fig Examples 1. 23. 50 4 sig figs 2. 402 3 sig figs 3. 5, 280 3 sig figs 4. 0. 080 2 sig figs

Significant Figures Ø Calculating with Sig Figs w Multiply/Divide - The # with the fewest sig figs determines the # of sig figs in the answer. (13. 91 g/cm 3)(23. 3 cm 3) = 324. 103 g 4 SF 324 g

Significant Figures Ø Calculating with Sig Figs (con’t) w Add/Subtract - The # with the lowest decimal value determines the place of the last sig fig in the answer. 3. 75 m. L + 4. 1 m. L 7. 85 m. L 7. 9 m. L 224 g + 130 g 354 g Since no decimals, the number stays as

Significant Figures Ø Calculating with Sig Figs (con’t) w Exact Numbers do not limit the # of sig figs in the answer. ² Counting ² Exact ² “ 1” numbers: 12 students conversions: 1 m = 100 cm in any conversion: 1 in = 2. 54 cm

Significant Figures Practice Problems 5. (15. 30 g) ÷ (6. 4 m. L) 4 SF 2 SF = 2. 390625 g/m. L 2. 4 g/m. L 2 SF 6. 18. 9 g - 0. 84 g 18. 06 g 18. 1 g
- Slides: 35