Units and the Barometer Mr Shields Regents Chemistry

Units and the Barometer Mr. Shields Regents Chemistry U 05 L 02 1

Pressure Units SI UNITS of Pressure is measured in PASCALS 1 atm = 101. 325 KPa 101, 325 Pascals Other units of pressure are the following: 1 atm = 14. 7 lbs/in 2 1 atm = 760 mm Hg 1 atm = 29. 9 in Hg 1 torr = 1 mm Hg English system ex. tire pressure Know each of these conversions! 2

Pressure Unit Problems 1) How many Pascals in 2 atm? 2 atm = 202, 650 Pascals 2) How many mm Hg in 0. 5 atm? 760 mm Hg/atm x 0. 5 atm = 380 atm 3) How many mm Hg in 25. 33 KPa? (25. 33 KPa/101. 3 KPa) x 760 mm Hg/atm = 190 mm Hg 4) How many Torr in 380 mm Hg? 1 torr = 1 mm Hg so 380 mm Hg = 380 Torr 3

Pressure measurement OK. We now know what units are used in measuring Pressure … So how do we actually MEASURE atmospheric pressure? The instrument is known as a BAROMETER 4

The typical household Barometer Used to forecast A change in weather So how does a barometer Do this? 5

Pressure measurement What is STP: 0 deg C and 1 atm What is STP in SI units? 273 K and 101. 3 KPa But atmospheric Pressure is not always 1 atm; It can vary with weather conditions… Warm fronts (fair weather) have higher pressures Cold Fronts (stormy weather ) have lower pressures And stored gases can vary over any pressure range 6

The Barometer The lower the barometric pressure and the faster it drops the more severe an approaching storm. Hurricanes for example have very low barometric pressures. Most intense storms to ever hit the US Fla Keys Camille Katrina C 4 Andrew C 5 1935 1969 2005 1993 26. 35 in Hg 26. 84 in Hg 27. 11 in Hg 27. 23 in Hg 160 mph 190 mph 140 mph 165 mph Why are these units expressed in inches of Mercury ? 7

The Origin of the Barometer Units of Torr (or mm of Hg) are associated with an Instrument know as a Torricelli Barometer Named after the inventor Evangelista Torricelli (1608 -1647) The first to show that the Atmosphere exerted pressure. 8
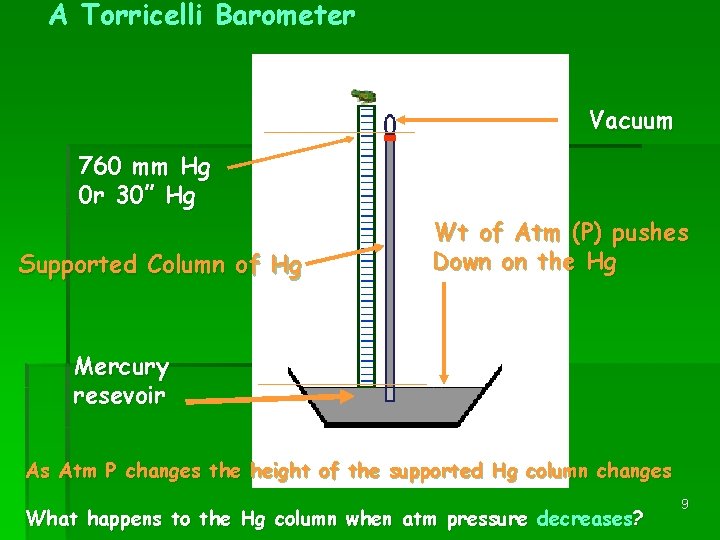
A Torricelli Barometer Vacuum 760 mm Hg 0 r 30” Hg Supported Column of Hg Wt of Atm (P) pushes Down on the Hg Mercury resevoir As Atm P changes the height of the supported Hg column changes What happens to the Hg column when atm pressure decreases? 9
- Slides: 9