Unit2 Lipid Metabolism Protein Metabolism Enzymes 1Lipid Metabolism

Unit-2 �Lipid Metabolism �Protein Metabolism �Enzymes


1)Lipid Metabolism Lipid metabolism refers to all chemical reactions concerned with lipids taking place in the cells. It includes anabolism & catabolism. Anabolism is the synthesis of lipids & catabolism is the breakdown of lipids. Lipids play a major role in the nutrition of man & other animals. Energy value is extremely high, 1 gm of fat release 9. 3 calories energy. at the same time one gram of carbohydrate release only 4. 2 calories. During digestion fats are split into fatty acids and glycerol. From the blood, cells receive them and are used for various purposes


β oxidation of fatty acids Oxidation of glycerol �Glycerol is converted into glycerol phosphate with help of glycrokinase, glycerol phosphate is oxidized to 3 -posphoglyceraldehyde. It enter glycolytic pathway and converted into pyruvic acid. Pyruvic acid is oxidized to CO 2 and H 2 O through Krebs cycle �The net production of energy from one molecule of glycerol is 19 ATP molecules Fat fatty acids + Glycrol glycrokinase Glycerolphosphate 3 -posphoglyceraldehyde Glycolysis Pyruic acid Krebs cycle CO 2+H 2 O+Energy

Fatty Acid Oxidation �Fatty acid are oxidized in various tissues like liver, adipose tissue, muscle, heart, kidney, brain, lungs, testis, etc. �Fatty acids are oxidized to CO 2 and water with the liberation of large amount of energy. �This oxidation occurs in the mitochondria �The mechanism of fatty acid oxidation is β- Oxidation

β -Oxidation � β –Oxidation was first proposed by Knoop in 1905. In β Oxidation, the fatty acid is oxidized at the β carbon atom (2 nd carbon atom in carboxyl group) and is converted into fatty acid having two carbon atom less and acetyl Co. A. �This process is repeated where two carbon atom are removed in each oxidation until a four carbon atom residue is left as butyric acid. The butyric acid is also oxidized in the β -position to form aceto-acetic acid. Both of these compounds undergo final oxidation into CO 2 and water. β α R. CH 2 COOH Higher fatty acid R. CH 2 COCH 2 COOH Keto acid R. CH 2 COOH +CH 2 COOH Lower fatty acid Acetic acid

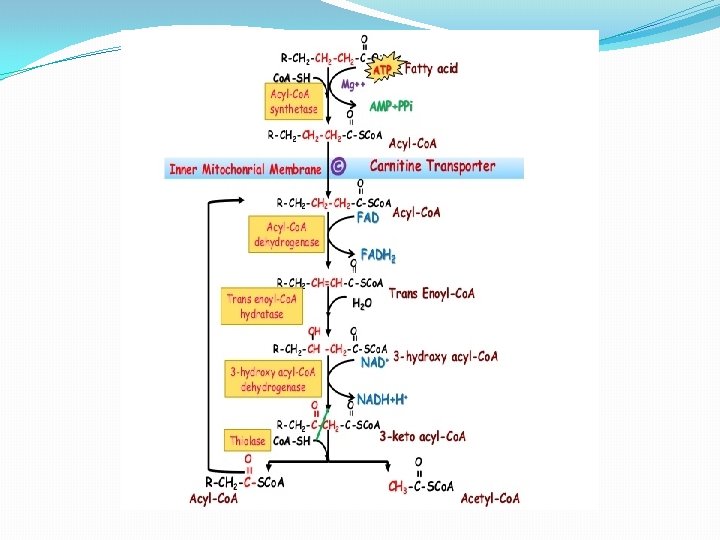
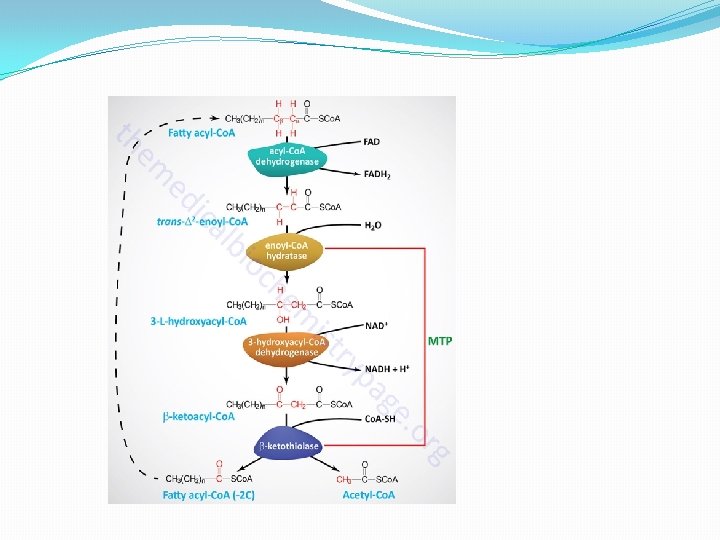
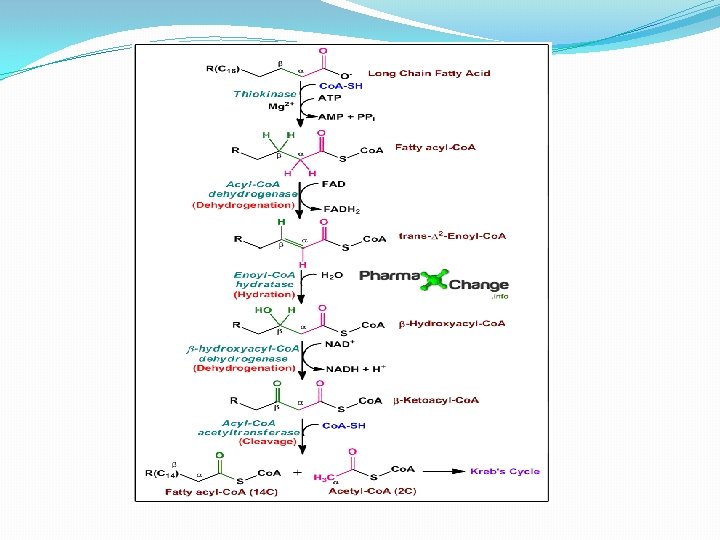
β - oxidation occurs in mitochondria. The acetyl Co. A formed in β - oxidation of fatty acids is oxidized to CO 2 and H 2 O with the release of energy through Krebs cycle. β -oxidation has the following steps: 1 -Activation 2 - Dehydration 3 -Hydration 4 -Dehydrogenation 5 -Thiolytic cleavage �Activation- The fatty acids, as such, are inert chemically. So in the first step, they are activated. The enzyme thiokinase (Acyl Co. A synthetase) in the presence of ATP and Co. A convert free fatty acid. �Dehydration- Acyl Co. A is oxidised by removal of 2 hydrogen atom, one from α- carbon & other from β carbon. � Hydration-
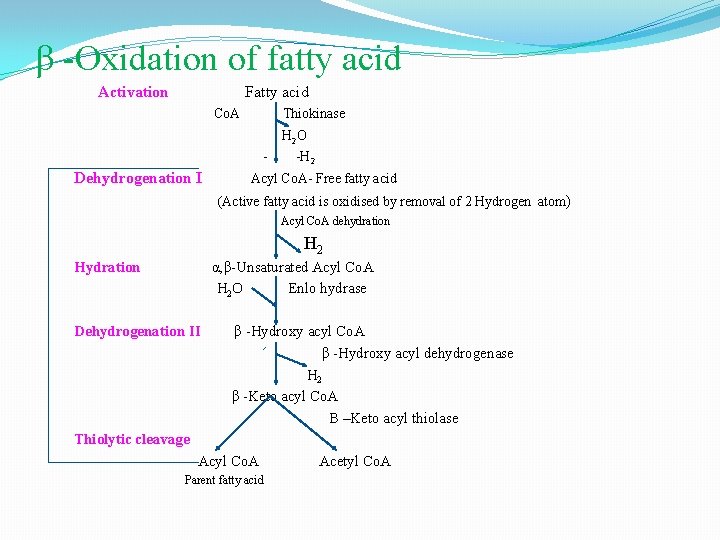
β -Oxidation of fatty acid Activation Fatty acid Co. A Thiokinase - Dehydrogenation I H 2 O -H 2 Acyl Co. A- Free fatty acid (Active fatty acid is oxidised by removal of 2 Hydrogen atom) Acyl Co. A dehydration H 2 Hydration α, β-Unsaturated Acyl Co. A H 2 O Enlo hydrase Dehydrogenation II β -Hydroxy acyl Co. A β -Hydroxy acyl dehydrogenase H 2 β -Keto acyl Co. A B –Keto acyl thiolase Thiolytic cleavage Acyl Co. A Parent fatty acid Acetyl Co. A

2)Protein Metabolism

2) Protein Metabolism All the chemical reactions taking place inside the cells in connection with proteins are called protein metabolism. It is of two types, namely Anabolism & Catabolism. In anabolism, amino acids and proteins are synthesized. In catabolism, amino acids are broken down. Protein are body builders and nitrogen containing compounds. An animal can live without carbohydrates or fats for long time but not without proteins, as they are involved in the formation of new tissue, repair of worn out parts, protect body from attack of foreign toxic elements and much more in the form of enzymes, hormones, antibodies, haemoglobins etc. �Transamination �Deamination �Urea Cycle


1)Transamination It involves the transfer of an amino group from one amino acid to a keto acid under the influence of transeaminase and coenzyme pyridoxol phosphate. In this reaction, the donar amino acid becomes a keto acid and recipient keto acid becomes an amino acid. R 1 R 2 CHNH 2 COOH Amino acid + CO COOH Keto acid CO + COOH Keto acid CHNH 2 COOH Amino acid Transmination is chemical reaction between two molecules. One is amino acid containing amino group(NH 2). Other is keto acid containing keto (CO) group. The amino acid becomes keto acid & keto acid becomes amino acid.

2)Deamination The first stage in the breakdown of amino acids is the removal of their nitrogenous group as ammonia. The removal of amino group from amino acids is called deamination. Deamination convert amino acids into keto acids in liver & kideny. R-CH-NH 2 + 1/2 O 2 | COOH Amino acid R-CO | COOH Keto acid +NH 3 Ammonia

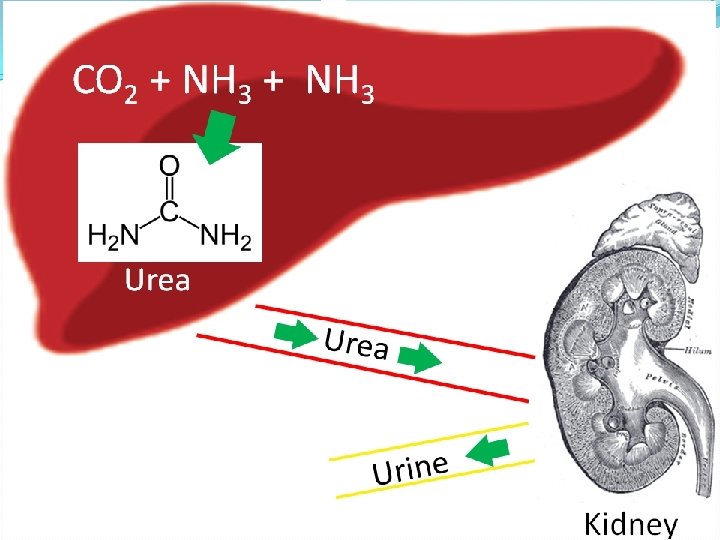
Disposal of Ammonia (NH 3) produced by deamination is toxic & must be deposed off. In aquatic animals NH 3 removal by diffusion, they are called ammonotelic. In snail, insect, and birds ammonia is converted in to amino acids & eliminated. these are called uricotelic. In elasmobranch fishes, amphibians and mammals, ammonia is converted into urea by ornithine cycle and is eliminated, these are called ureotelic.

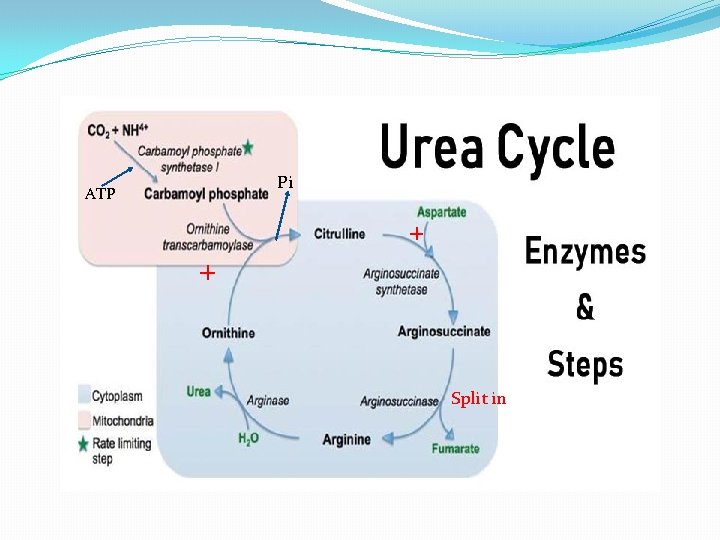
Urea Cycle � The biosynthesis of urea from ammonia is called urea cycle. The conversion of NH 3 into urea is the urea cycle. � The amino acid, ornithine functions as a catalyst in urea cycle. It enters the cycle with ammonia and reappears again after converting ammonia into urea. Hence urea cycle is also called ornithine cycle. � Urea cycle was discovered by Krebs and Henseleit 1932. � It occurs in the liver. It also occur in slow rate in kidney, brain, etc. � The initial two steps occur in cytosol. The remaining steps occur inside mitochondria. � In each cycle 2 molecules of ammonia & 1 molecule of CO 2 are combined to form 1 molecule of urea. 2 ammonia + co 2 Urea � Urea cycles consumes 3 molecules of ATP 2 Ammonia+ Co 2+ 3 ATP Urea+ 3 ADP+ H 2 O

�The urea cycle produces two NADH Molecules. Each NADH produces 2 ATP molecules in electron transport. So 2 NADH produce 4 ATP molecules. �Urea cycle consume 3 ATP molecules; but releases 4 ATP molecule. So net gain of 1 ATP in urea cycle. � 1. Ornithine cycle starts with the combination of CO 2 and NH 3. The resulting compound is called Carbamyl phosphate. This reaction is catalyzed by enzyme Carbamyl phosphate synthetase in the presence of two molecules of ATP & Mg++. Carbamyl phosphate synthetase CO 2 +NH 3 + 2 ATP Carbamyl phosphate + 2 ADP � 2. In the second stage, Carbamyl phosphate reacts with ornithine to form citrulline & inorganic phosphate(Pi) � 3. In the third stage, citrulline condenses with aspartic acid to form an arginosuccinic acid Carbamyl phosphate + Ornithine Citrulline + Aspartic acid Arginsuccinic acid

� 4. In the fourth stage, arginosuccinic acid is cleaved into arginine & fumaric acid in presence of enzyme arginosuccinase Arginosuccinic acid Arginine +Fumaric acid � 5. In the final stage, arginine is cleaved by arginase into ornithine and urea. Ornithine repeats the cycle by accepting another molecule of carbamyl phosphate. Arginase Arginine Ornithine + Urea
Pi ATP + + Split in

3) Enzymes �Introduction �Classification & Nomenclature �Mechanism of action �Enzyme Kinetics �Inhibition & Regulation �Iso-enzymes �Co-enzymes & Co-factors

3)Enzymes �Introduction �They are catalysts of biological systems. Therefore, they called biocatalysts. A catalysts is a substance that accelerates a chemical reaction without affecting the end product of reaction. It regulate various physiological/ biochemical processes of body. Substance on which enzyme acts is called the substrate. �Enzymes are involved in all biological processes such as digestion, respiration, stepwise breaking down, synthesis, interconversion of carbohydrates, proteins, fats and nucleic acids and release and utilization of energy by living cells.

�A)Classification & Nomenclature Enzymes are generally designated by adding the suffix “ase” to the root word of the substance on which the enzyme acts. An enzyme sucrase, acts upon sucrose, lipase upon lipids, protease upon proteins. Some times the suffix “lytic” is used to denote enzymes such as proteolytic ( protein splitting) lipolytic –lipid splitting. In order to have uniformity and unambiguity in identification of enzymes, International Union of Biochemistry (IUB) adopted a nomenclature system based on their reaction specificity. According to this system, enzymes are grouped in six classes.


Enzyme reactions

II) Characteristics of enzymes � Amount or quantity � Chemical nature � Rate of reaction � Reversibility � Denaturation � Activation � Inhibition � Specificity

1. Amount or quantity Enzymes are in small amount & work at extensively small conc. & they are unchanged at the end of reaction so that they can be used over & over. They can catalyze very large quantities of substrates. 2. Chemical nature: - All enzymes are basically protein in nature. Many of them are conjugated proteins. 3. Rate of reaction: - As catalyst, enzymes can only accelerates the rate of reaction by lowering the activation energy. 4. Reversibility: - Their are some enzymes exhibiting reversibility of their reaction. For example unhydrase, succinic dehydrogenase.

5. Denaturation: - Loss of enzyme activity may be seen when the natural conditions are changed. Such denaturation may be brought about by acids, high salt concentration. Heavy metal salts, alkaloidal reagents, UV light treatment, hightemperature. etc. 6. Activation: - Enzymes can be activated by the addition of certain specific agents called activators. In the absence of such activators, enzyme becomes inactive 7. Inhibition: - Enzyme action can be inhibited by certain substances called inhibitors.

8. Specificity: - Specificity is following types a) Stereochemical (optical) specificity: -There are many optical isomers b) c) i) ii) (a) (b) of substrate, but only one isomers acts as a substrate for enzyme activity e. g. succinic dehydrogenase when acts on succinic acid will give only fumaric acid ¬ metallic acid Reaction specificity: - A substrate can undergo many reactions but in a reaction specificity one enzyme can catalyses only one of the various reactions. E. g. oxaloacetic acid can undergo several reactions but each reaction is catalyzed by its own separate enzyme Substrate specificity: - They are of 2 types absolute specificity and relative specificity. Absolute specificity is comparatively rare, e. g. hydrolysis of urea by urease. Relative specificity may be (a) group dependent (b) bond dependent. group dependent- Trypsin hydrolysis residues of only lysine & arginine, chymotrypsin hydrolysis residues of only aromatic amino acids. bond dependent- Found in proteolytic enzymes. E. g. glycosides acts on glycolytic bonds whereas lipase on ester bonds.

B) Mechanism of action �Michaelis and Menten have proposed a hypothesis for enzyme action which is widely accepted. According to this hypothesis, the enzyme molecule (E) first combines with substrate molecule(S) to form an enzyme-substrate complex which further dissociate from complex is free to combine with another molecule of substrate and form product in a similar way. � E+S=ES-Complex �ES= E+ Product
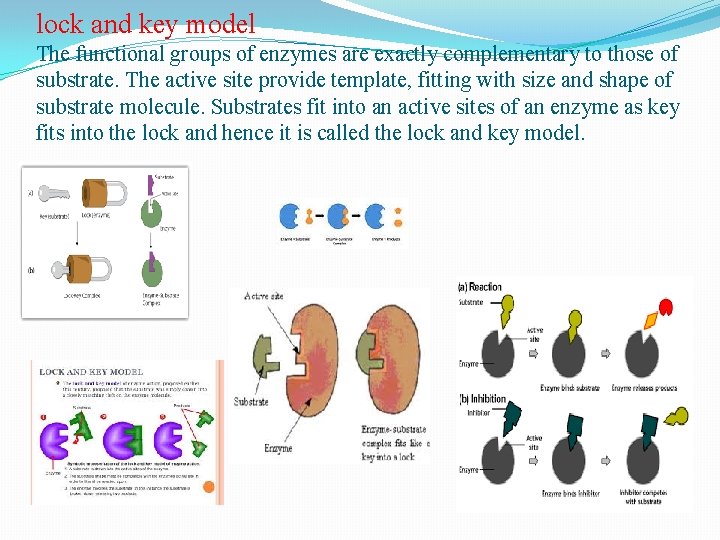
lock and key model The functional groups of enzymes are exactly complementary to those of substrate. The active site provide template, fitting with size and shape of substrate molecule. Substrates fit into an active sites of an enzyme as key fits into the lock and hence it is called the lock and key model.

Induced fit model �According to this model, active site does not posses a rigid structure on enzyme to fit the substrate but are capable of considerable internal movement. This induces conformational changes in the active site to attain the final catalytic shape and form.

�B) Enzyme Kinetics
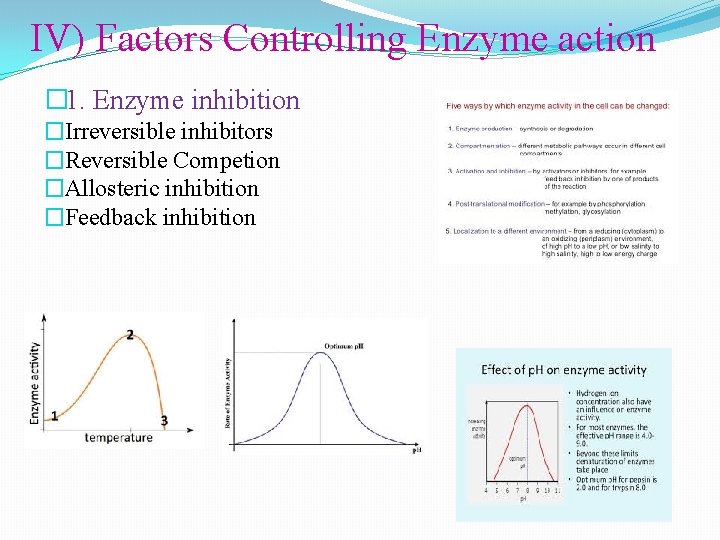
IV) Factors Controlling Enzyme action � 1. Enzyme inhibition �Irreversible inhibitors �Reversible Competion �Allosteric inhibition �Feedback inhibition

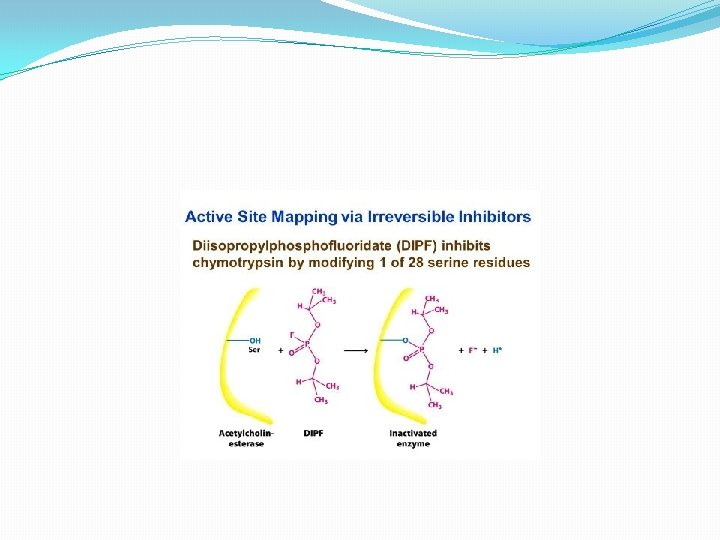
C) Inhibition & Regulation 1. Enzyme inhibition: a) Irreversible inhibitions: - In these types, inhibitors bind very tightly to the enzyme, usually by forming covalent bond to one of its amino acids. All enzymes inactivated. This can be illustrated by acetylcholine- a neurotransmitter responsible for muscle contraction. Its action can be destroyed by an enzyme acetyl cholinesterase. A number of nerve gases such as di-isopropylphosphofluoridate and organophosphate pesticides act as irreversible inhibitors of acetyl cholinesterase
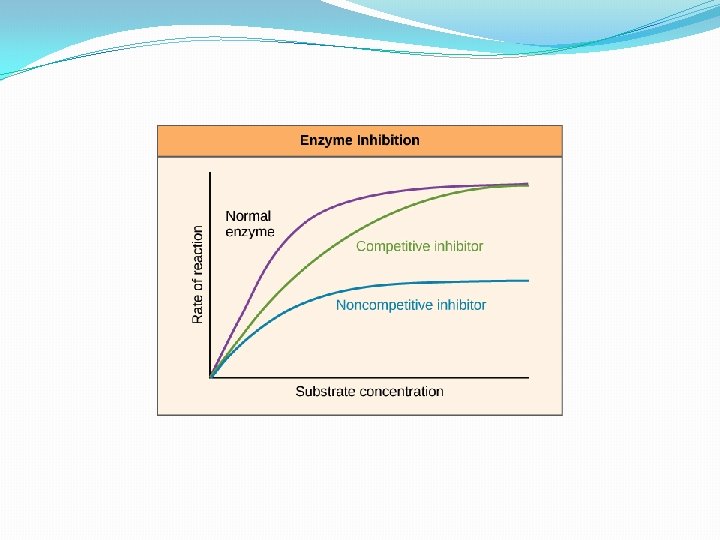
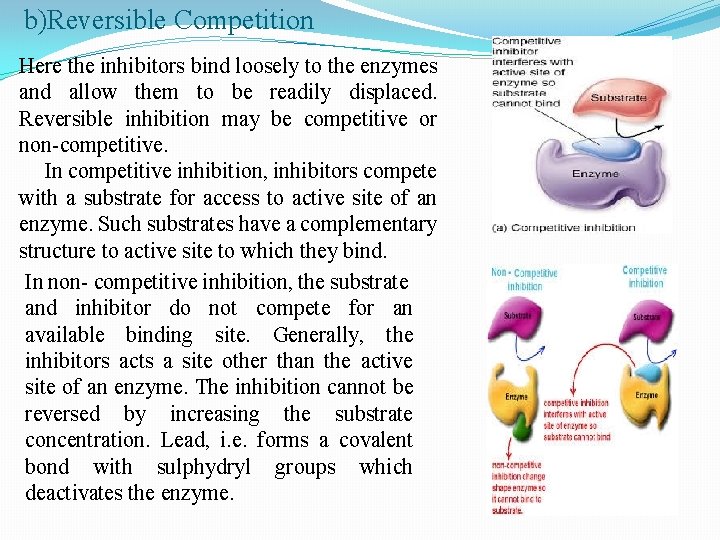
b)Reversible Competition Here the inhibitors bind loosely to the enzymes and allow them to be readily displaced. Reversible inhibition may be competitive or non-competitive. In competitive inhibition, inhibitors compete with a substrate for access to active site of an enzyme. Such substrates have a complementary structure to active site to which they bind. In non- competitive inhibition, the substrate and inhibitor do not compete for an available binding site. Generally, the inhibitors acts a site other than the active site of an enzyme. The inhibition cannot be reversed by increasing the substrate concentration. Lead, i. e. forms a covalent bond with sulphydryl groups which deactivates the enzyme.
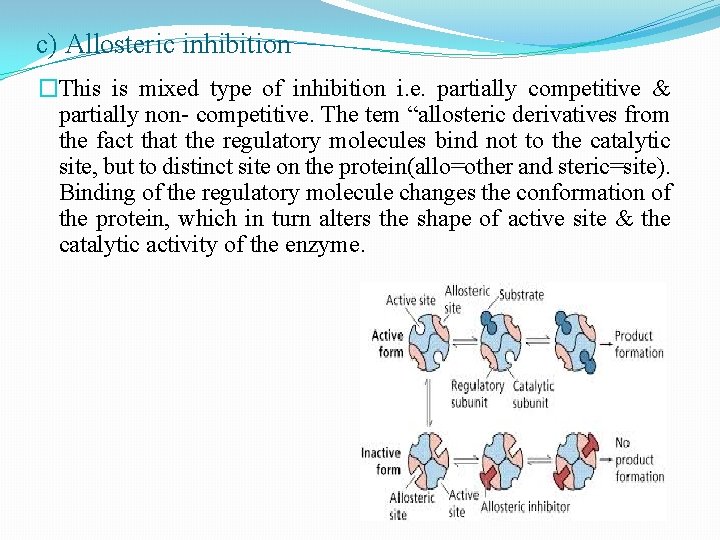
c) Allosteric inhibition �This is mixed type of inhibition i. e. partially competitive & partially non- competitive. The tem “allosteric derivatives from the fact that the regulatory molecules bind not to the catalytic site, but to distinct site on the protein(allo=other and steric=site). Binding of the regulatory molecule changes the conformation of the protein, which in turn alters the shape of active site & the catalytic activity of the enzyme.

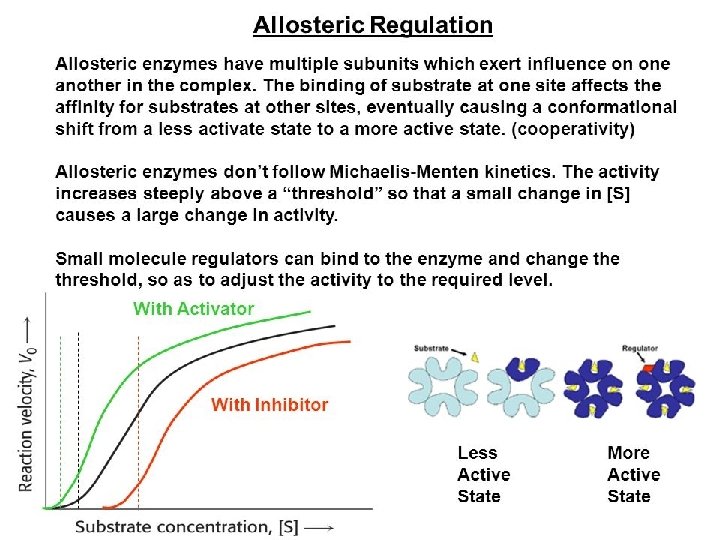


d) Feedback inhibition �It is one the common types of enzyme regulation in which the product of a metallic pathway inhibits the activity of enzyme involved in its synthesis. � E 1 E 2 E 3 � A B C D �In above reaction D is the end product and its high concentration typically inhibits conversion A B. If the concentration of D is decreased, feedback inhibition is relived so that additional D is synthesized.

D) Iso-enzymes V) Isoenzymes, Cactors and Coenzymes Enzymes having similar biological action may be isolated from different sources. Many different molecular species of an enzyme have the same function. Such enzymes which exist in more than one form are called. Isoenzymes are physically distinct forms of the same enzyme but catalyze the same chemical reactions and differ from each other structurally and immunologically. E. g. Lactic dehydrogenase (LDH). There are 5 major isoenzymes of LDH found in tissues. In blood serum these are known as LDH-1, LDH-2, LDH-3, LDH-4 AND LDH-5.

E) Co-enzymes & Co-factors and Coenzymes All enzymes are protein in nature. Few are simple proteins but many of them are conjugated proteins containing non-protein components called co-factors which may be organic or inorganic. The non-protein part is called prosthetic group and the remaining part is called as apoenzymes. The complete structure of apoenzymes and prosthetic group is called holoenzymes.

Certain enzymes require specific, thermostable low molecular weight non-protein organic substance called as co-enzymes. C 0 enzymes may bind covalently or non-covalently to the apoenzyme. The reactions involving oxido-reductions, group transfers isomerization and covalent bond formation require coenzymes. The coenzymes usually acts as an acceptor or donor of an atom or a group, e. g. NAD, NADP and CO 2 are hydrogen carrier, ATP transfers the phosphate group and Co A the acetyl group. Many coenzymes are closely related to vitamins. The B complex vitamin ( except biotin and lipoic acid) functions as parts of some coenzymes. e. g. Co A is derivative of pantothenic acid. Other examples are cobamide, tetrahydropolate- folic acid, niacin, ribflavin, pyrodoxine, thiamine etc.

The activity of many enzymes depends on the presence of certain metal ions such as Na+ , K+, Mg++, Ca++, Co++, Mn++, Cd++, Cr+++, AI+++. These substances acts as activators.

- Slides: 52