Unit One Matter Part One Matter Objective To

Unit One: Matter

Part One: Matter Objective: To learn what matter is

Part One: What is Matter • Matter is anything that has mass and takes up space

Part Two: The Atomic Nature of Matter • Objective: to understand that matter is composed of atoms

• Matter is made up of atoms • Scanning Tunneling Microscope gives us the chance to see atoms • Atoms are like sand on a beach

Part Three: Elements and Compounds • Atom- tiny particle that makes up matter • Contains: • Negative Electrons • Positive Protons • Neutral Neutrons (in nucleus)

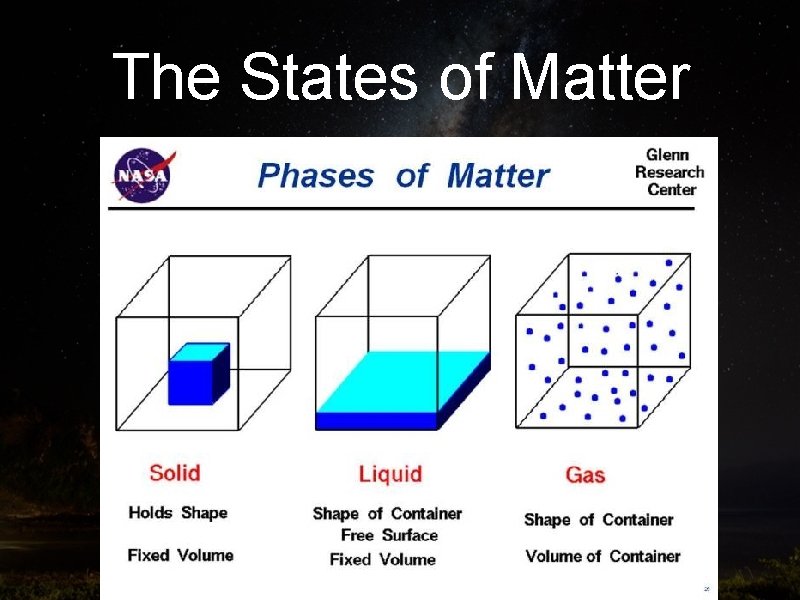
Part Three: The States of Matter Objective: To define three states of matter

The States of Matter • A SOLID is rigid and has a fixed shape and volume

The States of Matter • A LIQUID is fluid, has a definite volume, but takes up the shape/space of its container

The States of Matter • A GAS has no fixed volume or shape. It uniformly fills any container
The States of Matter

What state am I? • I am matter that loves to take the space of whatever I’m in. I’m often invisible and very full of energy.

What state am I? • Some people call me a moderate. I am flexible enough to take the shape of my container, though my volume never changes

What state am I? • I am never changing, I keep my shape no matter what I’m stuck in. Some people say I’m cold.

What is Jello? • Is Jello a liquid or a solid? • Write your opinion down and EXPLAIN YOUR ANSWER

Part 4: Kinetic Theory of Matter Objective: To understand that molecules are always moving and how we measure temperature

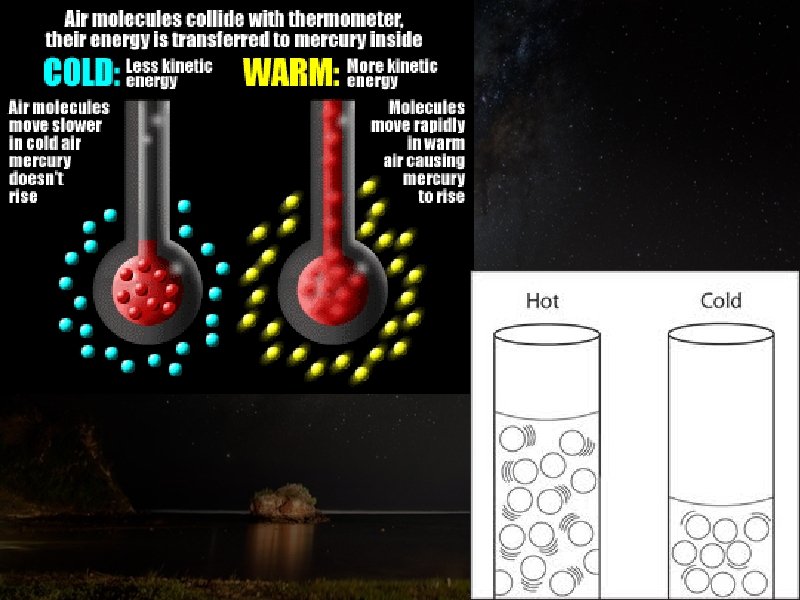
Kinetic Theory of Matter • Molecules are always moving. This is the Kinetic theory of matter. • Movement of molecules uses energy. • The more energy something has, the faster it moves.

Temperature • Heat is the energy that flows between objects of different temperatures. • We measure kinetic energy with a thermometer and call it temperature. • The warmer it is, the faster air molecules are moving.

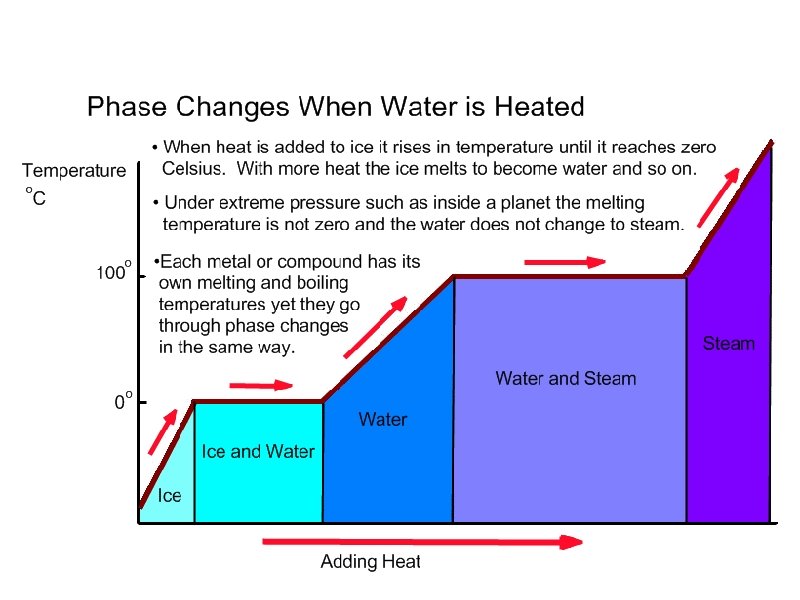
Part Five: Phase Changes of Matter Objectives: Understand how energy changes the state of matter, understand how to graph and read phase change graphs

Energy and phase changes • As energy is added or released from molecules, the substance changes phases
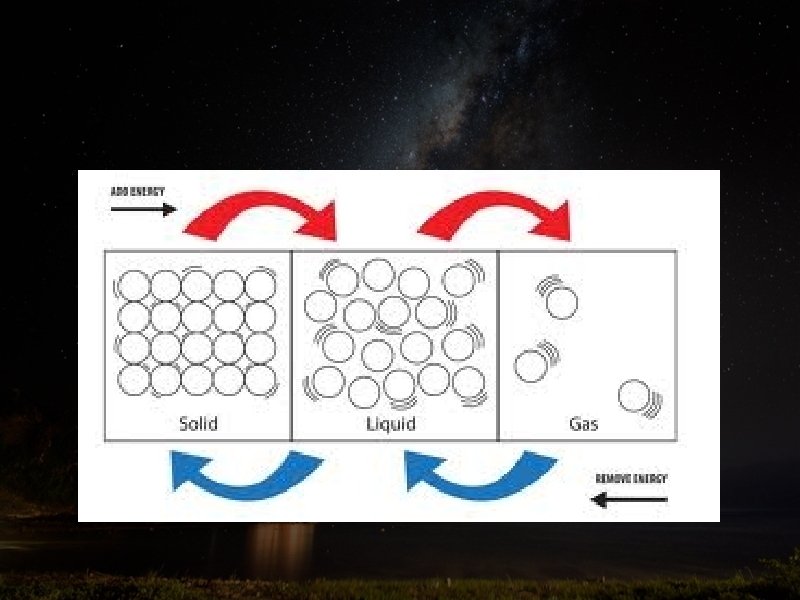

Vocabulary Terms • When Energy is added: • Solids MELT into liquid • Liquids EVAPORATE into gas • Solids SUBLIME into gas

Vocabulary Terms • When Energy is lost: • Gas CONDENSATES into liquid • Liquid FREEZES into solid • Gas DEPOSITES into solid
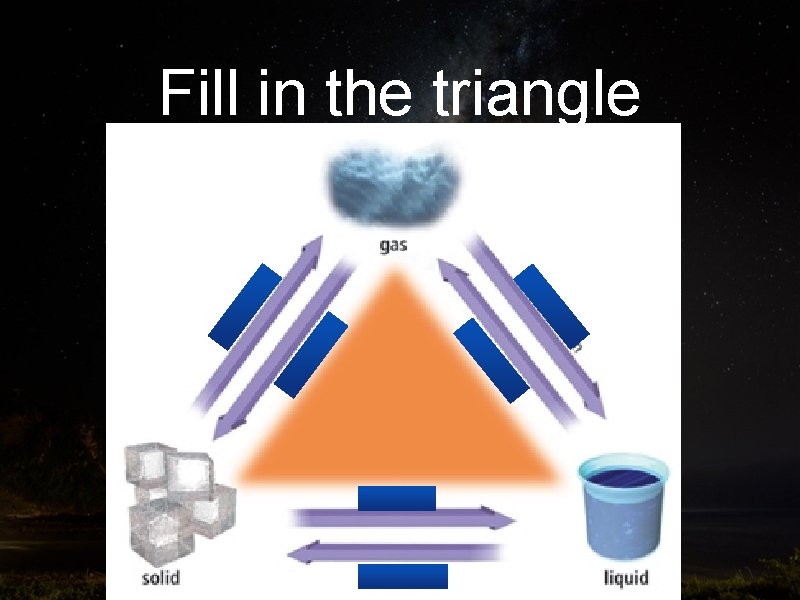
Fill in the triangle
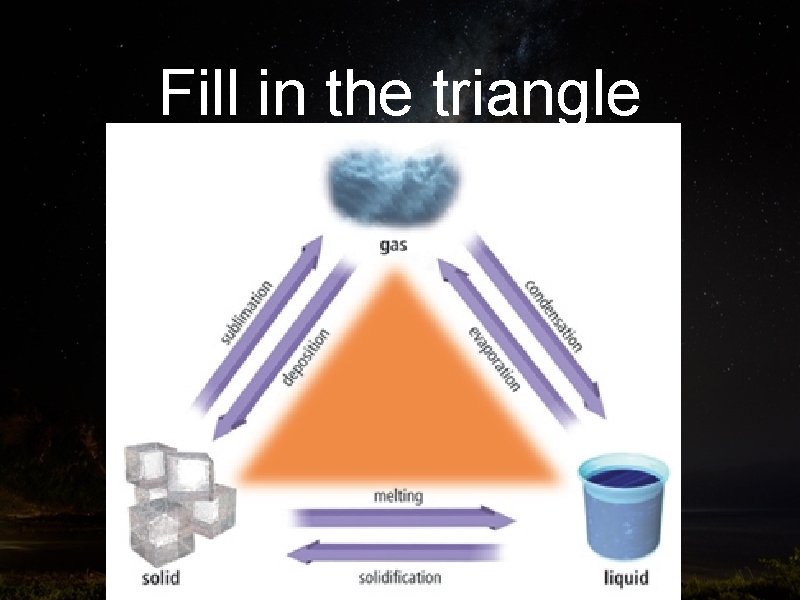
Fill in the triangle
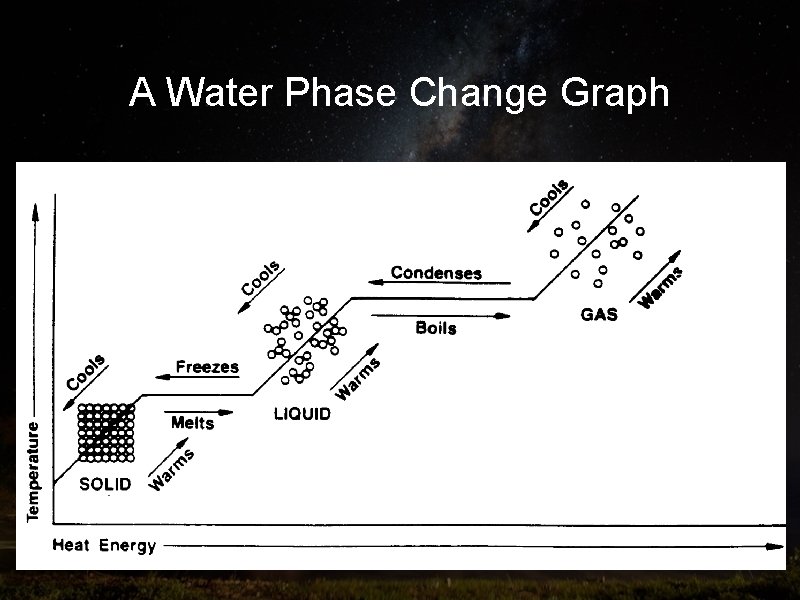
A Water Phase Change Graph
- Slides: 28