Unit Objectives 1understand the evolution of models of

Unit Objectives 1)understand the evolution of models of the atom: Dalton, Thomson, Rutherford, Bohr, the ‘quantum model 2) understand the meaning of mass number, atomic (proton) number. 3) be able to write and work with full atomic symbols 4) be able to calculate relative atomic, molecular and formula mass

Refreshing some Old Terminology A. Bohr Rutherford Diagrams B. Isotopes and ions C. R. A. M. (Relative Atomic Mass)

What is “atom”? The Greek philosopher Democritus

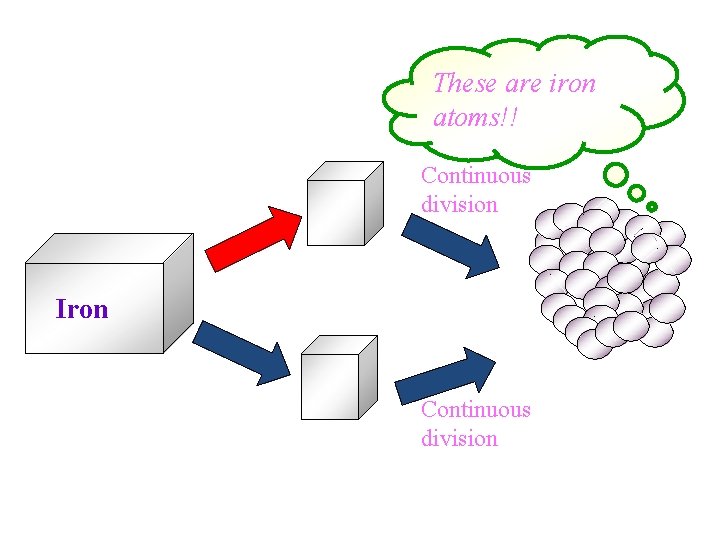
These are iron atoms!! Continuous division Iron Continuous division

Dalton’s atomic theory 1. All elements are made up of atoms. 2. Atoms are not created or destroyed. 3. Atoms of the same element have the same mass and chemical properties. 4. Atoms of different elements are different. They have different masses and chemical properties. 5. Atoms of different elements combine to form a compound. The numbers of various atoms combined bear a simple whole number ratio to each other.
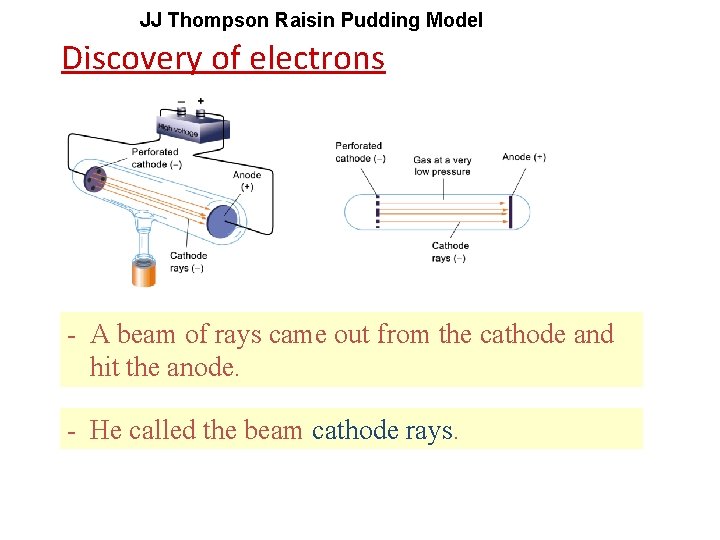
JJ Thompson Raisin Pudding Model Discovery of electrons - A beam of rays came out from the cathode and hit the anode. - He called the beam cathode rays.
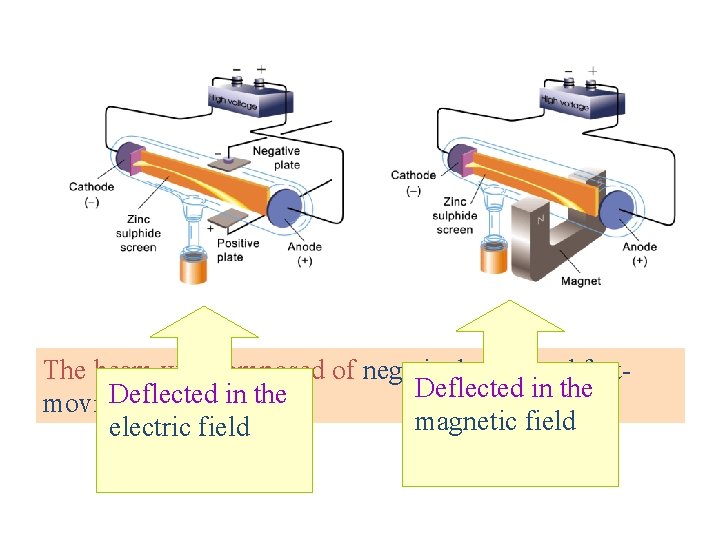
The beam was composed of negatively charged fast. Deflected in the moving particles. magnetic field electric field
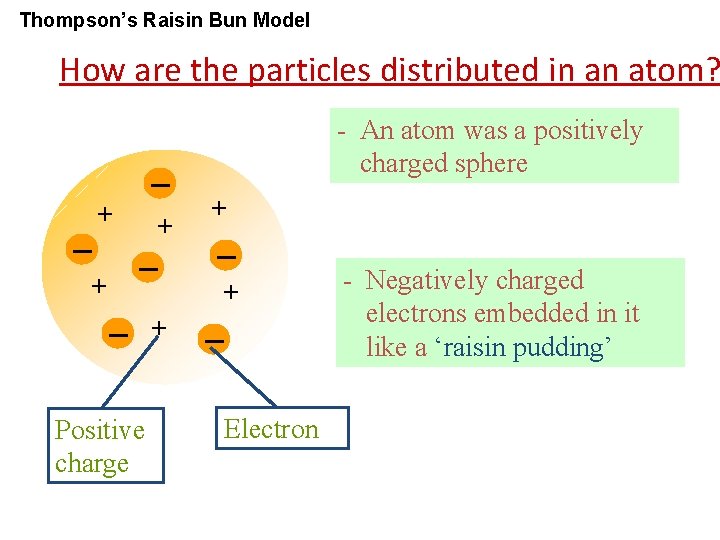
Thompson’s Raisin Bun Model How are the particles distributed in an atom? - An atom was a positively charged sphere + + + Positive charge Electron - Negatively charged electrons embedded in it like a ‘raisin pudding’

Gold foil scattering experiment - performed by Ernest Rutherford

He bombarded a thin gold foil with a beam of fastmoving -particles (+ve charged) Observation: -most -particles passed through the foil without deflection -very few -particles were scattered or rebounded back

Interpretation of the experimental results - The condensed core is called ‘nucleus’. - The positively charged particle is called ‘proton’.

Chadwick’s atomic model Provedthe presence of neutrons Electron Neutron Proton

Interpret the diagram Protons are deflected on a curved path towards the negative plate. Electrons are deflected on a curved path towards the positive plate. The amount of deflection is exactly the same in the electron beam as the proton beam if the energies are the same - but, of course, it is in the opposite direction. Neutrons continue in a straight line.

Practice Fill in the Chart Protons Neutrons Electrons A 19 21 19 B 20 Charge Atomic Number 0 C + 11 23 6 E 92 0 235 F 6 0 13 H 16 Symbol 40 D G 6 Mass Number 0 2 - 16 27 Al 3+

MASS NUMBER AND ATOMIC NUMBER Protons Neutrons Electrons Charge Atomic Number Mass Number Symbol A 19 21 19 0 19 40 40 K B 20 20 20 40 40 Ca C 11 12 10 + 11 23 23 Na+ D 6 6 6 0 6 12 12 C E 92 143 92 0 92 235 U F 6 7 6 0 6 13 13 C G 16 16 18 2 - 16 32 32 S 2 - H 13 14 10 3+ 13 27 27 Al 3+

Review of Models of the Atom (7 min) https: //www. youtube. com/watch? v=k. Bg. IMRV 8 95 w

Bohr's Model of the Atom Niels Bohr (1913): -studied the light produced when atoms was excited by heat or electricity

Bohr's Model of the Atom Niels Bohr (1913): -studied the light produced when atoms were excited by heat or electricity Rutherford's model couldn't explain why unique colours were obtained by atoms of different elements Bohr proposed that electrons are in orbits & when excited jump to a higher orbit. When they fall back to the original they give off light

Bohr's Model of the Atom Bohr's model: -electrons orbit the nucleus like planets orbit the sun -each orbit can hold a specific maximum number of electrons


Atomic Model Development NAME CONTRIBUTION EXPERIMENT John Dalton Postulated that matter is made up of indivisible particles None Democritus Postulated that matter is made up of indivisible particles. None – he was a Greek philosopher. Chemical elements are made of atoms identical in mass. Atoms of different elements have different masses. Atoms only combine in small, whole-number ratios, such as 1 : 1, or 1: 2 J. J. Thomson Discovered the electron Marie Curie Discovered that the atom was not indivisible because radioactive elements emitted particles. Created a glowing stream in a cathode ray tube that woul bend towards a positively charged electric plate. He suggested the stream made up of small particles – pieces of the atoms which carried a negative charge. These particles were later named electrons. Experiments with uranium emitting rays that electrified the air suggested the atom was not indivisible. Ernest Rutherford Discovered that the atom had a very dense positively charged nucleus Bombarded gold foil with -particles. Found that most which made up a very small passed all the way through whilst others ‘ricocheted’. proportion of the volume of the atom. Rutherford noted the disparity between the atomic number of an atom, or number of positive charges, and its mass computed in atomic mass units. The atomic number of an atom is usually about half its atomic mass. James Chadwick Discovered that neutrons were also located in the nucleus of an atoms Bombarded Beryllium with rays from radioactive Polonium, was unable to deflect the and that they contain no charge particle so he said it was neutral.

Characteristics of sub-atomic particles Sub-atomic particle Symbol Proton p or 1 H 1 Neutron n or 1 n 0 Electron e- or 0 e -1 Location in atom Nucleus Surrounding the nucleus Actual charge (C) 1. 6 x 10 -9 0 1. 6 x 10 -9 Relative charge +1 0 -1 Actual mass (g) 1. 7 x 10 -24 9. 1 x 10 -28 1 1 0 Approximate mass (a. m. u. )

Atomic number The atomic number (Z) of an element is the number of protons contained in the nucleus of the atom. Atomic number Number of = protons Reason: Atoms are electrically neutral. Number of = electrons WH Y?

Mass number The mass number (A) of an atom is the sum of the number of protons and neutrons in the nucleus. Mass number Number of = protons Number of + neutrons

Atomic numbers and mass numbers Atom No. of protons No. of electrons No. of neutrons Atomic number Mass number Hydrogen 1 1 0 1 (1 + 0) =1 Oxygen 8 8 9 8 (8 + 9) = 17 Argon 18 18 22 18 (18+22) = 40

Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons. Representation: Mass number Atomic number A Z Symbol of the element X

e. g. the two isotopes of chlorine are written as: 35 Cl 17 37 Cl 17 OR labelled as Cl-35 and Cl-37.

MASS NUMBER AND ATOMIC NUMBER Atomic Number (Z) Number of protons in the nucleus of an atom Mass Number (A) Sum of the protons and neutrons in the nucleus Mass Number (A) PROTONS + NEUTRONS 23 Na 11 Atomic Number (Z) PROTONS

Fuse School Isotopes https: //www. youtube. com/watch? v=o 7 Kpk 3 al_ uo

The Configuration of Electrons
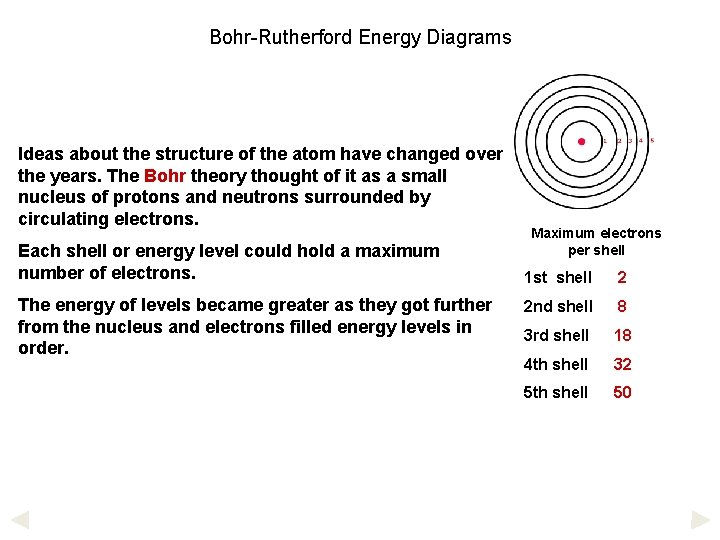
Bohr-Rutherford Energy Diagrams Ideas about the structure of the atom have changed over the years. The Bohr theory thought of it as a small nucleus of protons and neutrons surrounded by circulating electrons. Each shell or energy level could hold a maximum number of electrons. The energy of levels became greater as they got further from the nucleus and electrons filled energy levels in order. Maximum electrons per shell 1 st shell 2 2 nd shell 8 3 rd shell 18 4 th shell 32 5 th shell 50

The arrangement of the electrons The electrons are found in ENERGY LEVELS The first level will only hold 2 electrons, the second holds 8, and the third also 8 electrons. These levels can be thought of as getting progressively further from the nucleus.

What is the electronic arrangement in chlorine ? Atomic number = 17. Therefore there are 17 protons and 17 electrons. The arrangement of the electrons will be 2, 8, 7 (i. e. 2 in the first level, 8 in the second, and 7 in the third). The electronic arrangements of the first 20 elements

Practice Handout Please do the handout on energy level diagrams

ISOTOPES & AVERAGE ATOMIC OR MOLAR MASS

MASS SPECTRA R. A. M. 20 Ne We use these values to determine the average molar mass of an element or Relative atomic mass, called RAM 21 Ne 22 Ne. 90. 92 Abundance / % Consider neon and its 3 isotopes. . . 8. 82 0. 26 19 20 21 22 23 m/z Calculate the average relative atomic mass of neon using the above information. Out of every 100 atoms 90. 92 are Average = 20 Ne , 0. 26 are 21 Ne and 8. 82 are 22 Ne (90. 92 x 20) + (0. 26 x 21) + (8. 82 x 22) = 20. 179 100 Relative atomic mass = 20. 18

Calculate the relative atomic mass of the following – give your answers to 3 significant figures Bromine : 79 Br 50% , 81 Br 50% Copper : 63 Cu 69% , 65 Cu 31% Zirconium : 90 Zr 51. 5% , 91 Zr 11. 2%, 94 Zr 17. 4%, Lead : 204 Pb 1. 5% , 96 63. 6 92 Zr 2. 8% 206 Pb 23. 6%, Zr 17. 1%, 91. 3 207 Pb 22. 6%, 208 Pb 52. 3% 207. Neon : 20 Ne 90. 9% , 21 Ne 0. 2%, 22 Ne 8. 9% 20. 2

Can we calculate the % composition if we know the isotopes ? Naturally occurring potassium consists of potassium-39 and potassium-41. Calculate the percentage of each isotope present if the average is 39. 1. Assume there are x nuclei of 39 K in every 100; so there will be (100 -x) of 41 K so 39 x + 41 (100 -x) 100 = 39. 1 therefore 39 x + 4100 - 41 x = 3910 thus - 2 x = - 190 and ANSWER x = 95 There will be 95% 5% 39 K 41 K and

Right, ok now you try, see the handout
- Slides: 40