Unit No 5 Chemical Kinetics Kote S B

Unit No. 5 Chemical Kinetics Kote S. B.

Introduction • Studies the rate at which a chemical process occurs. • Besides information about the speed at which reactions occur, kinetics also sheds light on the reaction mechanism (exactly how the reaction occurs). • Thermodynamics – does a reaction take place? • Kinetics – how fast does a reaction proceed?

Reaction rate is the change in the concentration of a reactant or a product per unit time (M/s) Average Rate-The change in conc. Of a reactant or product divided by time interval over which the change occurs. Average Rate = DC/ Dt A B D[A] rate = Dt D[A] = change in concentration of A over time period Dt D[B] rate = Dt D[B] = change in concentration of B over time period Dt Because [A] decreases with time, D[A] is negative. 13. 1


Rate of Reaction is expressed in unit Mol L-1 t-1 e. g. N 2(g) + 3 H 2(g) OR M t-1 M- molarity of substanc 2 NH 3(g) Average rate of consumption of N 2= - D[N 2]/Dt Average rate of consumption of H 2= - D[H 2]/Dt Average rate of formation of NH 3= + D[NH 3]/Dt Average rate of overall reaction = -D[N 2]/Dt = -1/3 D[H 2]/Dt = +1/2 D[NH 3]/Dt

Instantaneous rate = The rate of reaction at a specific instant e. g. R P
![Reaction Rates a. A + b. B Products . 1 D [A] rate = Reaction Rates a. A + b. B Products . 1 D [A] rate =](http://slidetodoc.com/presentation_image_h2/9360029ca410bdee863a52e009160fc4/image-7.jpg)
Reaction Rates a. A + b. B Products . 1 D [A] rate = a Dt a. A + b. B = K[A]X [B]y c. C + d. D 1 D[A] 1 D[C] 1 D[D] 1 D[B] = = rate = =a Dt b Dt c Dt d Dt 13. 1

ORDER OF REACTION The order of a chemical reaction with respect to each reactant is defined as the exponent to which the concentration term of that reactant in the rate law is raised. The overall order of the reaction is defined as the sum of the exponent to which the concentration terns in the rate law are raised. A Product The rate law is rate = k [A]. Such a reaction is a first order reaction.

e. g. : 1) The decomposition of H 2 O 2, according to the reaction. 2 H 2 O 2(g) 2 H 2 O(l) + O 2(g). Rate = k[H 2 O 2] The is first order. 2) H 2(g)+ I 2(g) 2 HI(g) Rate = k[H 2] [I 2] The reaction is first order in H 2 and first order in I 2. Hence the overall order of the reaction is 1+1 = 2 3) a. A + b. B c. C + d. D Rate = k[A]x [B]y The overall order of the reaction is (x + y). 4) The order can also be zero

The Rate Law The rate law expresses the relationship of the rate of a reaction to the rate constant and the concentrations of the reactants raised to some powers. a. A + b. B c. C + d. D Rate = k [A]x[B]y reaction is xth order in A reaction is yth order in B reaction is (x +y)th order overall 13. 2

Integrated rate laws The equation which are obtained by integrating the differential rate laws & which give a direct relationship between the conc. of the reactants & time are called Integrated rate laws

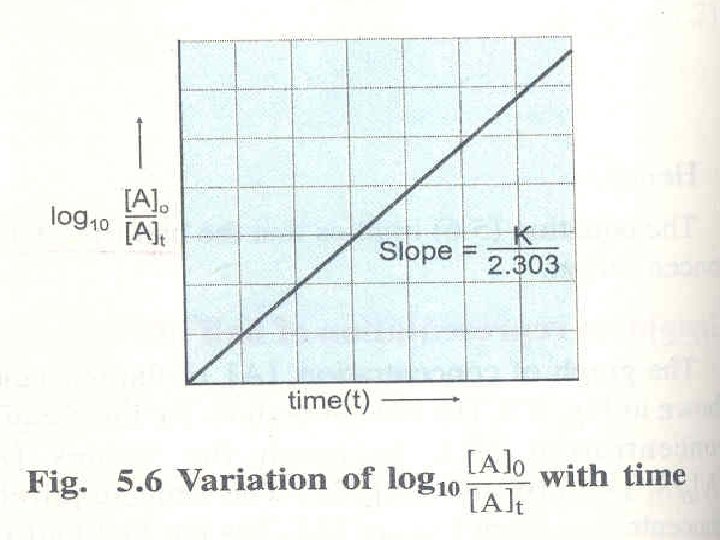
Integrated Rate Laws Consider a simple 1 st order : A Product Differential form: How much A is left after time t? Integrate: K = 2. 303/t log 10 [A]0/[A]t

Integrated Rate Laws The integrated form of first order rate law: Can be rearranged to give: [A]0 is the initial concentration of A (t=0). [A]t is the concentration of A at some time, t, during the course of the reaction.

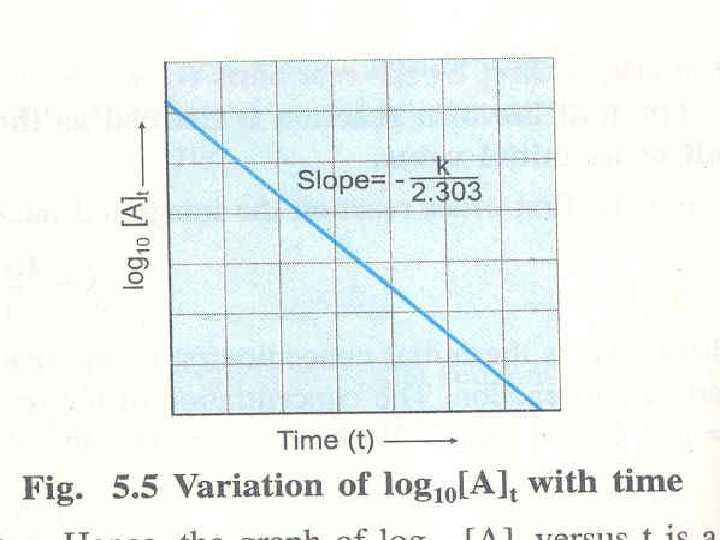
Integrated Rate Laws Manipulating this equation produces… …which is in the form y = mx + b The Unit of k is reciprocal of time s-1
![First-Order Processes If a reaction is first-order, a plot of ln [A]t vs. t First-Order Processes If a reaction is first-order, a plot of ln [A]t vs. t](http://slidetodoc.com/presentation_image_h2/9360029ca410bdee863a52e009160fc4/image-15.jpg)
First-Order Processes If a reaction is first-order, a plot of ln [A]t vs. t will yield a straight line with a slope of -k.

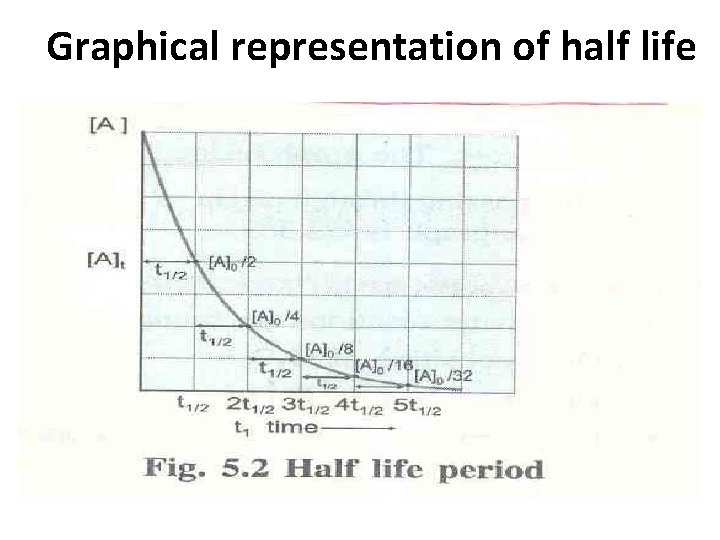
Graphical representation of half life
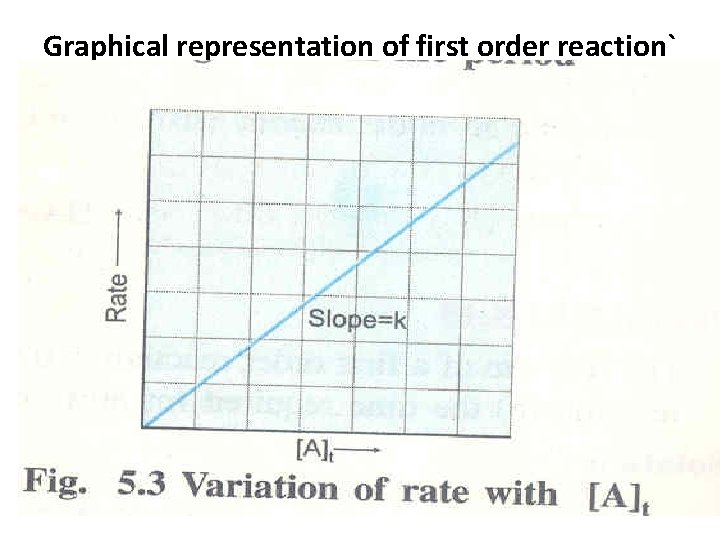
Graphical representation of first order reaction`
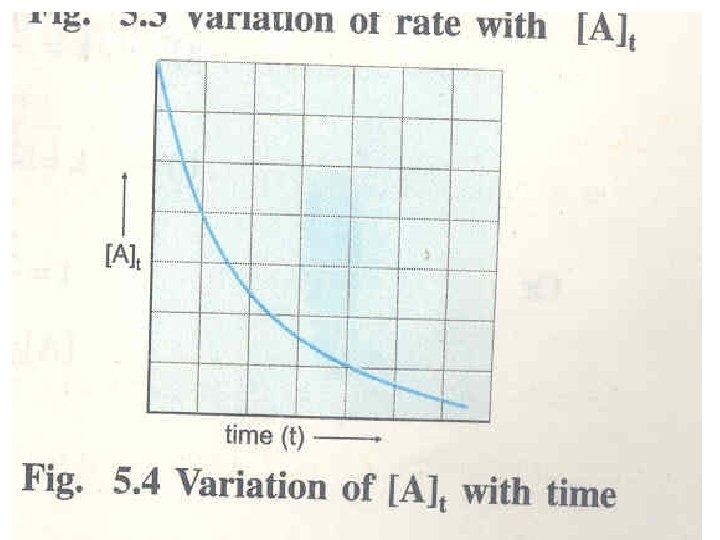

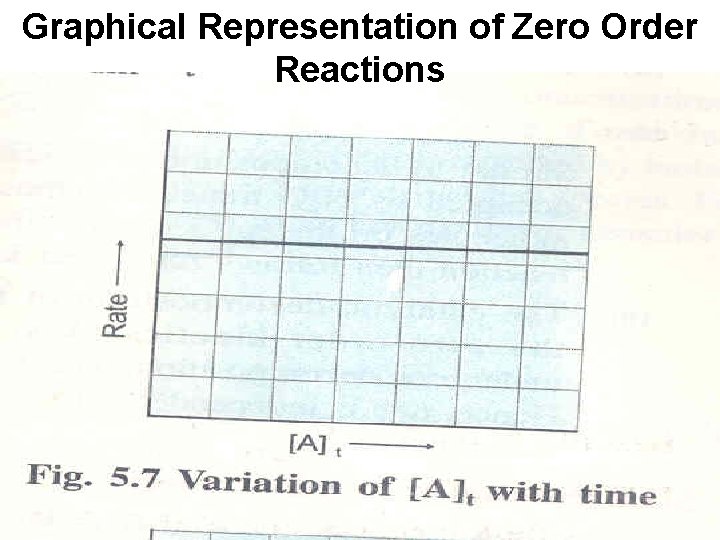
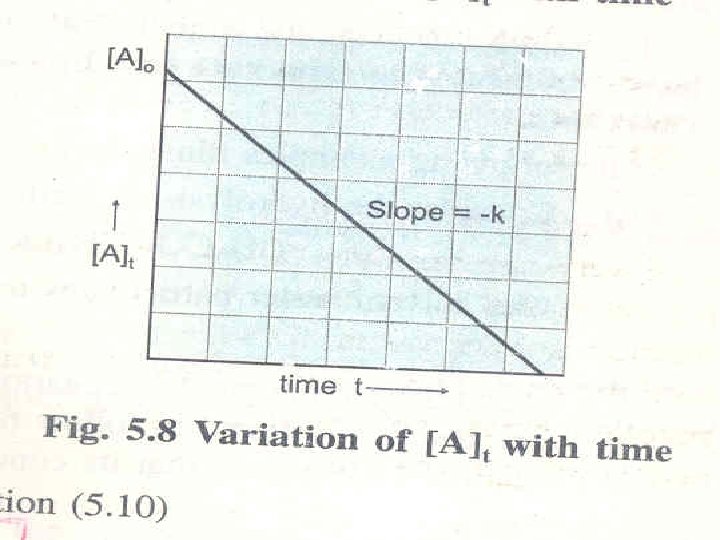
Zero Order Reaction The zero order reaction is defined as the reaction whose rate is independent of the reactant concentration and remains constant through out course of the reaction A → Product Rate = - d[A]/dt = k[A]o= k The unit of k zero order reaction is = Mt-1=mol dm-3 t-1
Graphical Representation of Zero Order Reactions
![Zero-Order Reactions A product D[A] rate = Dt D[A] =k Dt rate = M/s Zero-Order Reactions A product D[A] rate = Dt D[A] =k Dt rate = M/s](http://slidetodoc.com/presentation_image_h2/9360029ca410bdee863a52e009160fc4/image-25.jpg)
Zero-Order Reactions A product D[A] rate = Dt D[A] =k Dt rate = M/s k= 0 [A] = [A]0 - kt rate = k [A]0 = k [A] is the concentration of A at any time t [A]0 is the concentration of A at time t=0 t½ = t when [A] = [A]0/2 [A]0 t½ = 2 k 13. 3

Summary of the Kinetics of Zero-Order, First-Order and Second-Order Reactions Order 0 Rate Law rate = k 1 rate = k [A] 2 [A]2 rate = k Concentration-Time Equation [A] = [A]0 - kt Half-Life t½ = [A]0 2 k ln[A] = ln[A]0 - kt t½ = ln 2 k 1 1 = + kt [A]0 1 t½ = k[A]0 13. 3

Pseudo first Order Reaction Such reaction that having higher order true rate law but are found behave as first order are pseudo first order reaction. 1)Consider the hydrolysis of methyl acetate CH 3 COOCH 3(aq)+ H 2 O(l) → CH 3 COOH(aq) + CH 3 OH(aq) Rate =K’ [CH 3 COOCH 3] [H 2 O] The reaction is expected to have second order kinetics. [H 2 O] = constant = k’’ Thus rate = k’ k’’ [CH 3 COOCH 3] = k [CH 3 COOCH 3] k = k’ k’’

Experimental determination of rate laws and order • Isolation method • Method of initial rates • Use of integrated rate law
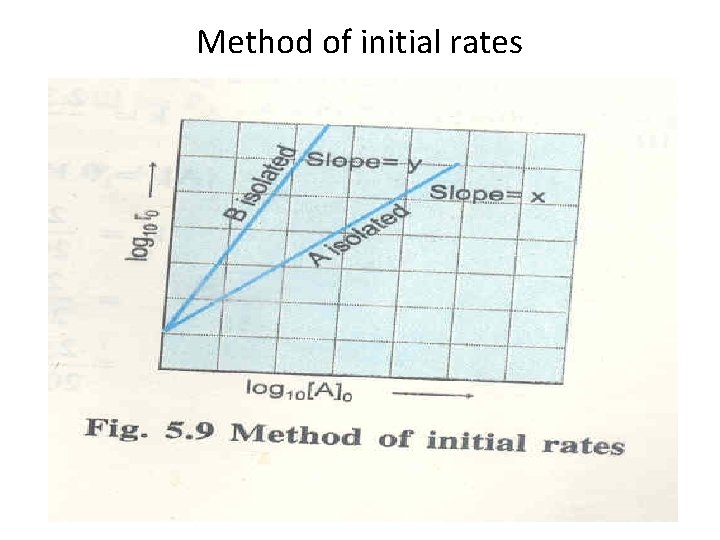
Method of initial rates
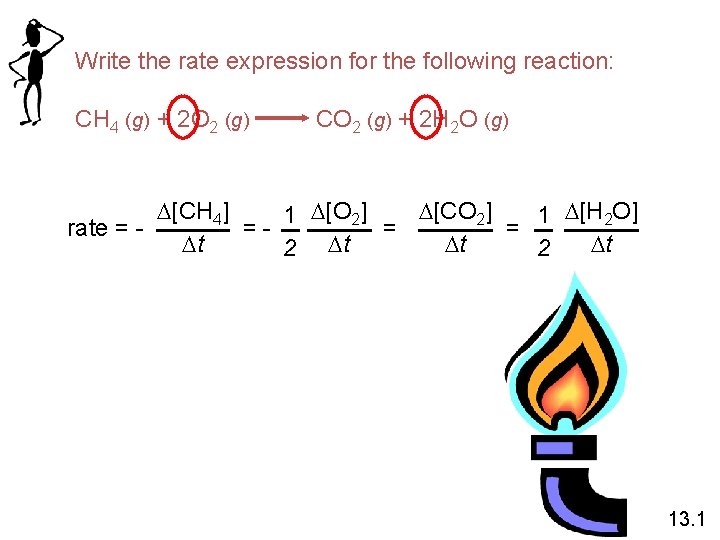
Write the rate expression for the following reaction: CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (g) D[CH 4] D[CO 2] 1 D[H 2 O] rate = = == Dt Dt Dt 2 13. 1

Rate Laws • Rate laws are always determined experimentally. • Reaction order is always defined in terms of reactant (not product) concentrations. • The order of a reactant is not related to the stoichiometric coefficient of the reactant in the balanced chemical equation. F 2 (g) + 2 Cl. O 2 (g) 2 FCl. O 2 (g) rate = k [F 2][Cl. O 2] 1 13. 2

Determine the rate law and calculate the rate constant for the following reaction from the following data: S 2 O 82 - (aq) + 3 I- (aq) 2 SO 42 - (aq) + I 3 - (aq) Experiment [S 2 O 82 -] [I-] Initial Rate (M/s) 1 0. 08 0. 034 2. 2 x 10 -4 2 0. 08 0. 017 1. 1 x 10 -4 3 0. 16 0. 017 2. 2 x 10 -4 rate = k [S 2 O 82 -]x[I-]y y=1 x=1 rate = k [S 2 O 82 -][I-] Double [I-], rate doubles (experiment 1 & 2) Double [S 2 O 82 -], rate doubles (experiment 2 & 3) 2. 2 x 10 -4 M/s rate k= = = 0. 08/M • s 2[S 2 O 8 ][I ] (0. 08 M)(0. 034 M) 13. 2

Molecularity of Elementary Reactions An elementary reaction is defined as a reaction that takes place in A singal step & cannot be broken down further into simpler chemica Reaction. The molecularity of an elementary reaction is defined as As the number of reactant molecules taking part in the reaction. O 3(g) + O O 2(g) + O(g) 2 O 2(g) + O(g) ( Unimolecular) ( Bimolecular) Order & molecularity of elementary reactions
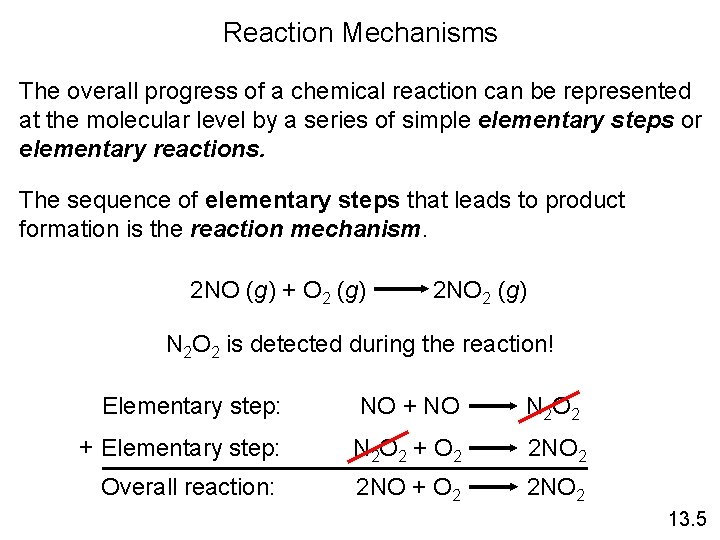
Reaction Mechanisms The overall progress of a chemical reaction can be represented at the molecular level by a series of simple elementary steps or elementary reactions. The sequence of elementary steps that leads to product formation is the reaction mechanism. 2 NO (g) + O 2 (g) 2 NO 2 (g) N 2 O 2 is detected during the reaction! Elementary step: NO + NO N 2 O 2 + Elementary step: N 2 O 2 + O 2 2 NO 2 Overall reaction: 2 NO + O 2 2 NO 2 13. 5
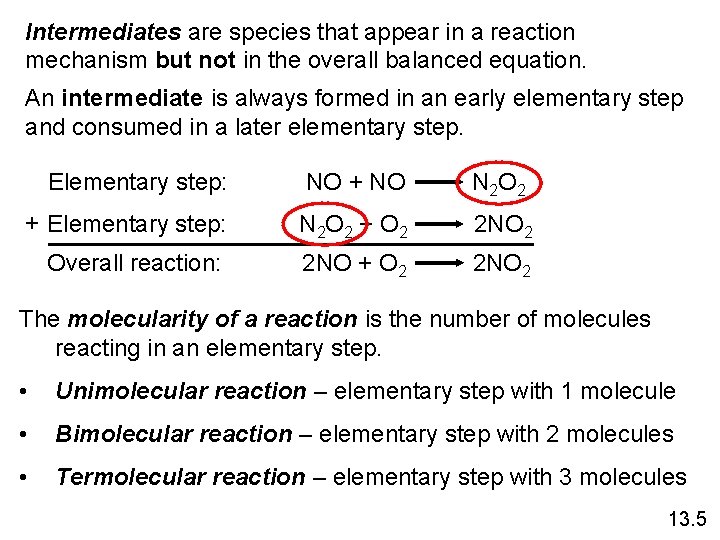
Intermediates are species that appear in a reaction mechanism but not in the overall balanced equation. An intermediate is always formed in an early elementary step and consumed in a later elementary step. Elementary step: NO + NO N 2 O 2 + Elementary step: N 2 O 2 + O 2 2 NO 2 Overall reaction: 2 NO + O 2 2 NO 2 The molecularity of a reaction is the number of molecules reacting in an elementary step. • Unimolecular reaction – elementary step with 1 molecule • Bimolecular reaction – elementary step with 2 molecules • Termolecular reaction – elementary step with 3 molecules 13. 5
![Rate Laws and Elementary Steps Unimolecular reaction A products rate = k [A] Bimolecular Rate Laws and Elementary Steps Unimolecular reaction A products rate = k [A] Bimolecular](http://slidetodoc.com/presentation_image_h2/9360029ca410bdee863a52e009160fc4/image-36.jpg)
Rate Laws and Elementary Steps Unimolecular reaction A products rate = k [A] Bimolecular reaction A+B products rate = k [A][B] Bimolecular reaction A+A products rate = k [A]2 Writing plausible reaction mechanisms: • The sum of the elementary steps must give the overall balanced equation for the reaction. • The rate-determining step should predict the same rate law that is determined experimentally. The rate-determining step is the slowest step in the sequence of steps leading to product formation. 13. 5

The experimental rate law for the reaction between NO 2 and CO to produce NO and CO 2 is rate = k[NO 2]2. The reaction is believed to occur via two steps: Step 1: NO 2 + NO 2 NO + NO 3 Step 2: NO 3 + CO NO 2 + CO 2 What is the equation for the overall reaction? NO 2+ CO NO + CO 2 What is the intermediate? NO 3 What can you say about the relative rates of steps 1 and 2? rate = k[NO 2]2 is the rate law for step 1 so step 1 must be slower than step 2 13. 5

COLLISION THEORY & ACTIVATION ENERGY (i) Collision between reactant molecule (ii) Energy requirment (activation energy) (iii) Orientation of reactant molecule (iv) Potential energy barrier
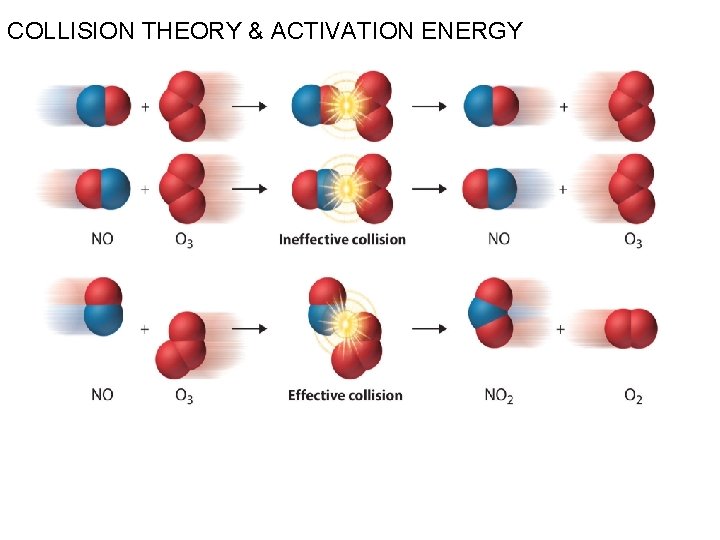
COLLISION THEORY & ACTIVATION ENERGY

Collision Theory & Activation Energy
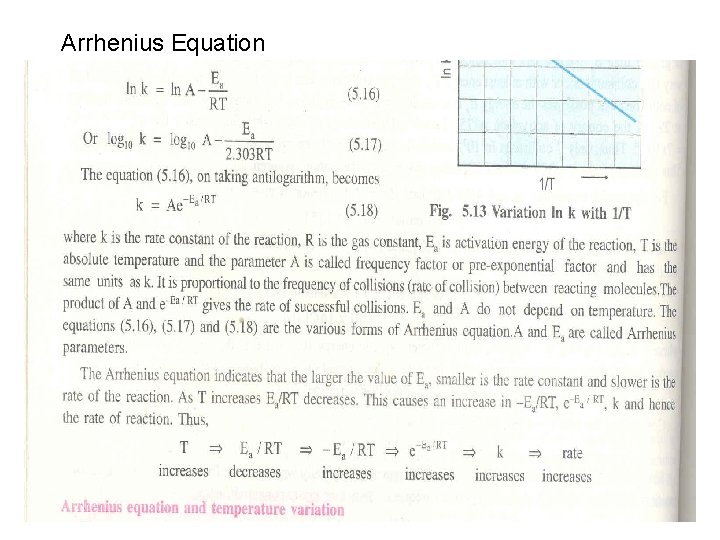
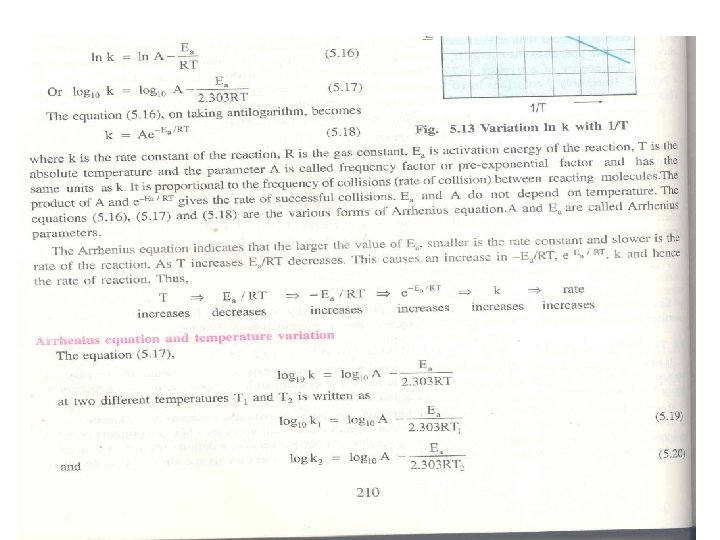
Arrhenius Equation


Effect of Catalyst on rates of reactions 2 KCl. O 3(s) Mn. O 2 2 KCl(s) + 3 O 2(g)
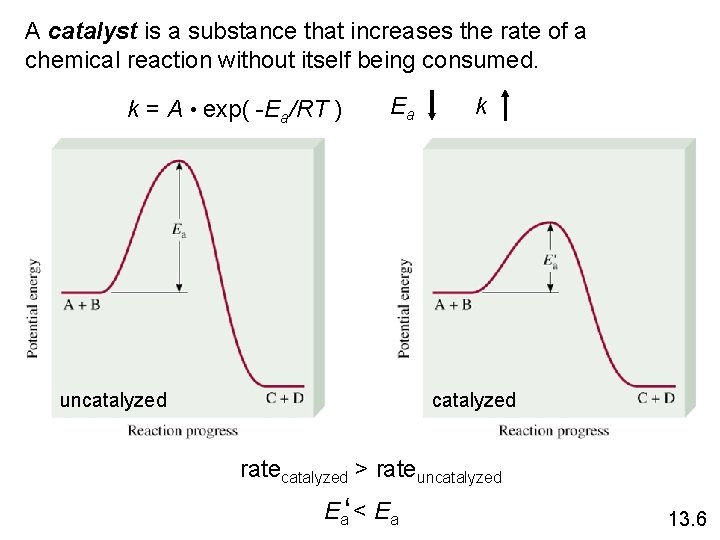
A catalyst is a substance that increases the rate of a chemical reaction without itself being consumed. k = A • exp( -Ea/RT ) Ea uncatalyzed k catalyzed ratecatalyzed > rateuncatalyzed Ea‘ < Ea 13. 6
- Slides: 45