UNIT II Intermediary Metabolism of monosaccharides and disaccharides

UNIT II: Intermediary Metabolism of monosaccharides and disaccharides
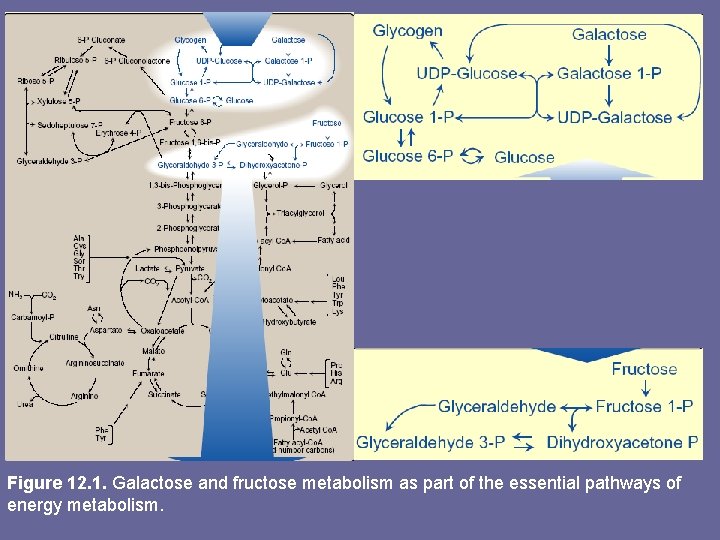
Figure 12. 1. Galactose and fructose metabolism as part of the essential pathways of energy metabolism.

Overview • Although many monosaccharides have been identified in nature, only a few sugars appear as metabolic intermediates or as structural components in mammals. • Gluc is the most common monosacch consumed by humans, & its metabolism has been discussed • However, two other monosacch’s, fructose & galactose, occur in significant amounts in the diet, & make important contributions to energy metabolism • In addition, galactose is an important component of cell structural CHO’s

II. Fructose metabolism - - ~ 10% of the calories contained in Western diet are supplied by fructose (~ 50 g/day). The major source of fructose is the disaccharide sucrose, which, when cleaved in intestine, releases equimolar amounts of fructose & glucose Fructose is also found as a free monosacch in highfructose corn syrup (55% fructose/45% glucose, which is used to sweeten most cola drinks), in many fruits, & in honey. Entry of fructose into cells is not insulin-dependent (unlike that of glucose into certain tissues), & in contrast to glucose, fructose does not promote secretion of insulin

A. Phosphorylation of fructose - For fructose to enter pathways of intermediary metabolism, it must 1 st be phosphorylated. This can be accomplished by either hexokinase or fructokinase (a. k. a ketohexokinase) - Hexokinase phosphorylates gluc in all cells of the body, & several additional hexoses can serve as substrates for this enz. However, it has a low affinity (high Km) for fructose. Therefore, unless intracellular conc. of fructose becomes unusually high, the normal presence of saturating conc’s of gluc means that little fructose is converted to F-6 -P by hexokinase - Fructokinase provides the primary mechanism for fructose phospho. It is found in the liver (which processes most of dietary fructose), kidney, & small intestinal mucosa, & converts fructose to F-1 -P, using ATP as the P donor Note: these 3 tissues also contain aldolase B

B. Cleavage of fructose 1 -phosphate - F 1 -P is not converted to F 1, 6 -BP as F-6 -P, but is cleaved by aldolase B (a. k. a fructose 1 phosphate aldolase) to DHAP & glyceraldehyde Note: both aldolase A (found in all tissues) & aldolase B cleave F 1, 6 -BP produced during glycolysis to DHAP & GA-3 P. - DHAP can directly enter glycolysis or gluconeogenesis, whereas glyceraldehyde can be metabolized by a number of pathways

C. Kinetics of fructose metabolism - The rate of fructose metabolism is more rapid than that of gluc because the trioses formed from F-1 -P bypass PFK, the major rate-limiting step in glycolysis Note: loading the liver with fructose, e. g. , by intravenous infusion, can significantly elevate the rate of lipogenesis caused by enhanced production of acetyl Co. A

D. Disorders of fructose metabolism - A deficiency of one of the key enz’s required for the entry of fructose into intermediary metabolic pathways can result in either a benign condition (fructokinase deficiency essential fructosuria), or a severe disturbance of liver & kidney metabolism as a result of aldolase B deficiency (hereditary fructose intolerance, HFI-”fructose poisoning”) which is estimated to occur in 1: 20, 000 live births - The 1 st symptoms appear when a baby is weaned & begins to be fed food containing sucrose or fructose. F-1 -P accumulates, & ATP & Pi levels fall significantly, with adenine being converted to uric acid, causing hyperuricemia

- The decreased availability of hepatic ATP affects gluconeogenesis (causing hypoglycemia with vomiting), & protein synthesis (causing a decrease in blood clotting factors & other essential proteins) - If fructose (& therefore, sucrose) is not removed from diet, liver failure & death can occur - Diagnosis of HFI can be made on basis of fructose in urine, or by a RFLP test
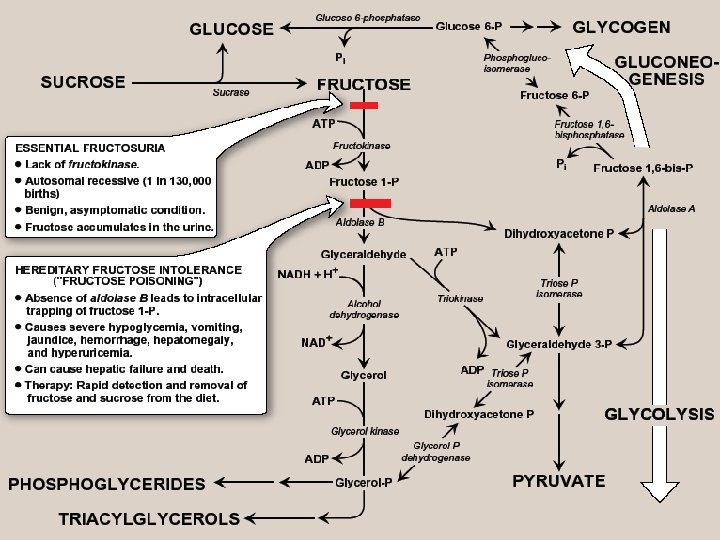

E. Conversion of mannose to fructose 6 -phosphate - Mannose, the C-2 epimer of gluc, is an important component of glycoproteins - Hexokinase phosphorylates mannose 6 -P, which in turn, is (reversibly) isomerized to F-6 -P by phosphomannose isomerase Note: there is little mannose in dietary CHOs. Most intracellular mannose is synthesized from fructose, or is pre-existing mannose produced by degradation of structural CHO’s & salvaged by hexokinase

F. Conversion of glucose to fructose via sorbitol - Most sugars are rapidly phosphorylated following their entry into cells. They are thereby trapped within cells, because organic P’s can’t freely cross memb’s without specific transporters - An alternate mechanism for metabolizing a monosacch is to convert it to a polyol by reduction of an aldehyde group, thereby producing an additional hydoxyl group

1. Synthesis of sorbitol: - Aldose reductase reduces glucose, producing sorbitol (glucitol). This enz is found in many tissues, including the lens, retina, Schwann cells of peripheral nerves, liver, kidney, placenta, RBCs, & cells of the ovaries & seminal vesicles. - In cells of liver, ovaries, sperm & seminal vesicles, there is a 2 nd enz, sorbitol dehydrogenase, that can oxidize sorbitol to produce fructose. - The 2 -reaction pathway from gluc to fruc in seminal vesicles is for the benefit of sperm cells, which use fruc as a major CHO energy source. - The pathway from sorbitol to fruc in liver provides a mechanism by which any available sorbitol is converted into a substrate that can enter glycolysis or gluconeogenesis
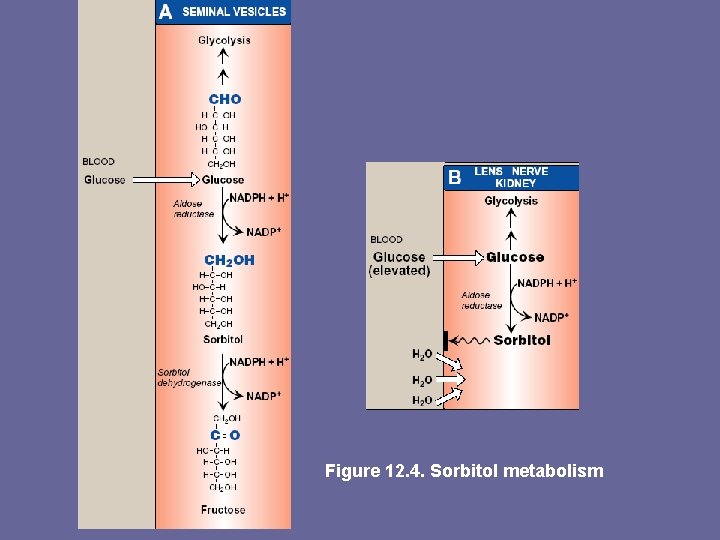
Figure 12. 4. Sorbitol metabolism

2. The effect of hyperglycemia on sorbitol metabolism - Because insulin is not required for entry of gluc into cells listed in previous paragraph, large amounts of gluc may enter these cells during times of hyperglycemia, e. g. , in uncontrolled diabetes. - Elevated intracellular gluc conc’s & an adequate supply of NADPH cause aldose reductase to produce a sufficient increase in the amount of sorbitol, which can’t pass efficiently through CMs &, therefore, remains trapped inside cell. - This is exacerbated when sorbitol dehydrogenase is low or absent, e. g. , in retina, lens, kidney & nerve cells. As a result, sorbitol accumulates in these cells, causing strong osmotic effects &, therefore, cell swelling as a result of water retention - Some of the pathologic alterations associated with diabetes can be attributed, in part, to this phenomenon, including cataract formation, peripheral neuropathy, & vascular problems leading to nephropathy, & retinopathy

III. Galactose metabolism - The major dietary source of galactose is lactose (galactosyl β 1, 4 -glucose) obtained from milk & milk products. Note: digestion of lactose by β-galactosidase (lactase) of the intestinal mucosal CM was discussed earlier - Some galactose can also be obtained by lysosomal degradation of complex CHOs, such as glycoproteins & glycolipids, which are important memb components. - Like fructose, entry of galactose into cells is not insulin dependent A. Phosphorylation of galactose - Like fruc, galactose must be phosphorylated before it can be further metabolized. Most tissues have a specific enz for this purpose, galactokinase, which produces galactose 1 -P. ATP is the P donor.

B. Formation of UDP-galactose • Galactose 1 -P can’t enter glycolytic pathway unless it is 1 st converted to UDP-galactose. This occurs as an exchange reaction, in which UMP is removed from UDPgluc (leaving behind G-1 -P), & is then transferred to the galactose 1 -P, producing UDP-galactose. The enz which catalyzes this reaction is galactose 1 -phosphate uridyl transferase C. Use of UDP-galactose as a carbon source for glycolysis or gluconeogenesis • In order for UDP-galactose to enter the mainstream of gluc metabolism, it must 1 st be converted to its C-4 epimer, UDP-gluc, by UDP-hexose 4 -epimerase • This “new” UDP-gluc (produced from the original UDPgalactose) can then participate in many biosynthetic reactions, as well as being used in uridyl transferase reaction described above, converting another galactose 1 -P into UDP-galactose, & releasing G-1 -P, whose carbons are those of the original galactose
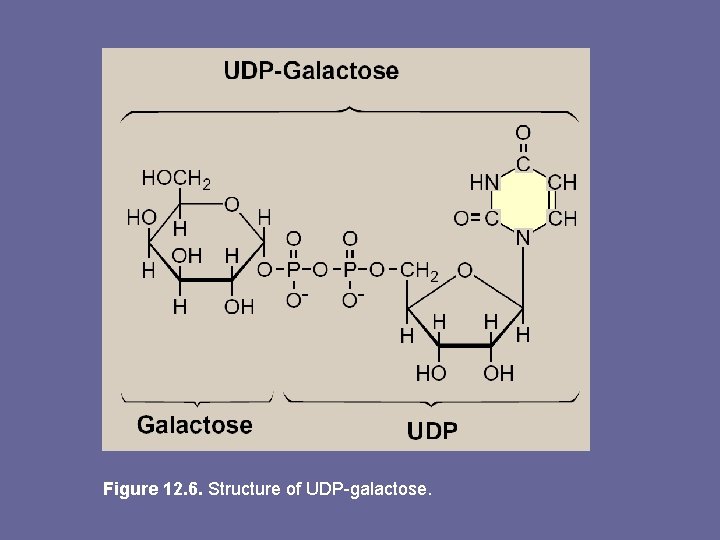
Figure 12. 6. Structure of UDP-galactose.

D. Role of UDP-galactose in biosynthetic reactions - UDP-galactose can serve as the donor of galactose in a number of synthetic pathways, including synthesis of lactose, glycoproteins, glycolipids, & glycosaminoglycans Note: if galactose is not provided by the diet (e. g. , when it can’t be released from lactose as a result of a lack of β-galactosidase in people who are lactose-intolerant), all tissue requirements of UDP-galactose can be met by the action of UDPhexose 4 -epimerase on UDP-glucose, which is efficiently produced from G-1 -P

E. Disorders of galactose metabolism - Galactose 1 -phosphate uridyltransferase is missing in individuals with classic galactosemia. In this disorder, galactose 1 -P &, therefore, galactose, accumulates in cells. - Physiologic consequences are similar to those found in fructose intolerance, but a broader spectrum of tissues is affected. - The accumulated galactose is shunted into side pathways such as that of galactitol production. This reaction is catalyzed by aldose reductase, the same enz that converts gluc to sorbitol - Note: a more benign form of galactosemia is caused by a deficiency of galactokinase
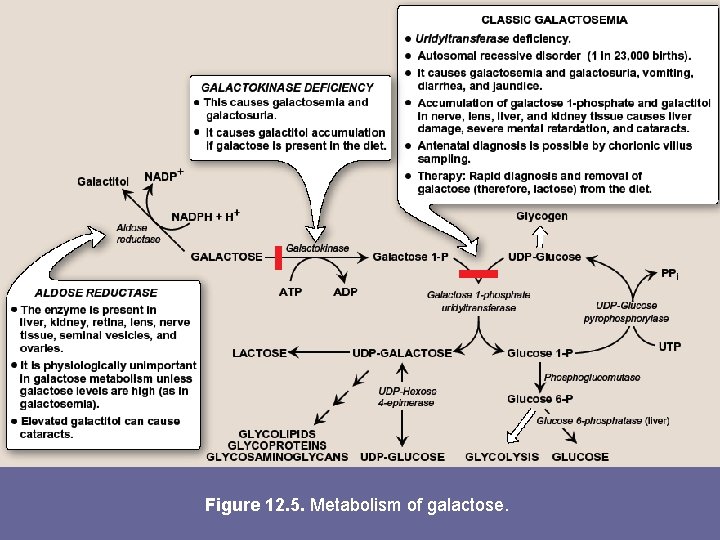
Figure 12. 5. Metabolism of galactose.

IV. Lactose synthesis - Lactose is a disacch that consists of a molecule of β-galactose attached by β(1→ 4) linkage to gluc. Therefore, lactose is galactosyl β(1→ 4)glucose. - Lactose, known as “milk sugar”, is produced by mammary glands of most mammals. Therefore, milk & other dairy products are the dietary sources of lactose. - Lactose is synthesized in the ER by lactose synthase (UDP-galactose: glucose galactosyltransferase), which transfers galactose from UDP-galactose to gluc, releasing UDP.

- This enz is composed of 2 proteins, A & B. protein A is a β-D-galactosyltransferase & is found in a # of body tissues. In tissues other than the lactating mammary gland, this enz transfers galactose from UDP-galactose to N-acetyl-Dglucoasamine, forming the same β(1→ 4) linkage found in lactose, & producing Nacetyllactosamine, a component of the structurally important N-linked glycoproteins - In contrast, protein B is found only in lactating mammary glands. It is α-lactalbumin, & its synthesis is stimulated by the peptide hormone, prolactin. - Protein B forms a complex with the enz, protein A, changing specificity of that transferase so that lactose, rather than N-acetyllactosamine, is produced
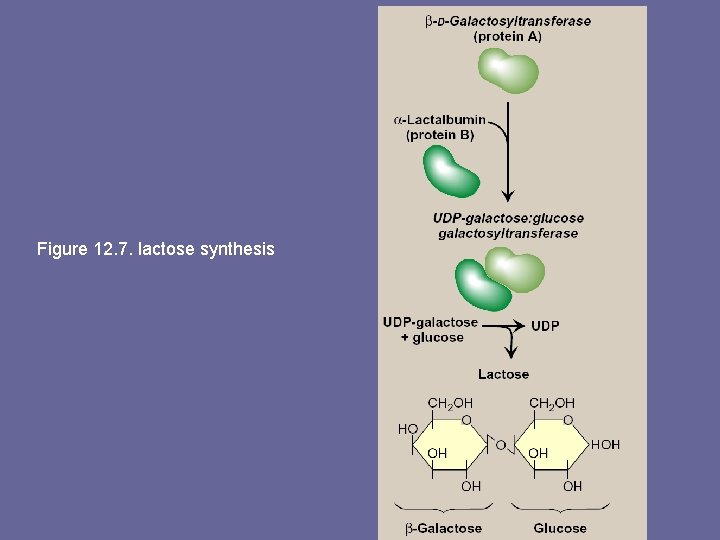
Figure 12. 7. lactose synthesis

Summary • The major source of fructose is sucrose, which when cleaved releases equimolar amounts of fructose & glucose. • Entry of fruc into cells is insulin-independent. Fruc is 1 st phosphorylated to F-1 -P by fructokinase, & then cleaved by aldolase B to DHAP & glyceraldehyde. These enz’s are found in liver, kidney, & small intestinal mucosa. • A deficiency of fructokinase causes a benign condition (fructosuria), but a deficiency of aldolase B causes hereditary fructose intolerance, in which severe hypoglycemia & liver failure lead to death if the amount of fruc (and therefore, sucrose) in the diet is not severely limited.

• Mannose, an important component of glycoproteins, is phosphorylated by hexokinase to mannose-6 -P, which is reversibly isomerized to F-6 -P by phosphomannose isomerase. • Gluc can be reduced to sorbitol (glucitol) by aldose reductase in many tissues, including the lens, retina, Schwann cells, liver, kidney, ovaries, & seminal vesicles. • In cells of liver, ovaries, & seminal vesicles, a 2 nd enz, sorbitol dehydrogenase, can oxidize sorbitol to produce fructose. • Hyperglycemia results in accumulation of sorbitol in those cells lacking sorbitol dehydrogenase. The resulting osmotic events cause cell swelling, & may contribute to the cataract formation, peripheral neuropathy, nephropathy, & retinopathy seen in diabetes

• The major dietary source of galactose is lactose. The entry of galactose into cells is not insulin-dependent. • Galactose is 1 st phosphorylated by galactokinase which produces galactose 1 -P. this cpd is converted to UDPgalactose by galactose 1 -phosphate uridyltransferase, with the nucleotide supplied by UDP-gluc. • A deficiency of this enz causes classic galactosemia. Galactose 1 -P accumulates, & excess galactose is converted to galactitol by aldose reductase. This causes liver damage, severe mental retardation, & cataracts. Treatment requires removal of galactose (& therefore, lactose) from the diet. • In order for UDP-galactose to enter the mainstream of gluc metabolism, it must be converted to UDP-gluc by UDP-hexose-4 -epimerase. This enz can also be used to produce UDP-galactose from UDP-gluc when the former is required for synthesis of structural CHOs

• Lactose is a disacch that consists of galactose & gluc. Milk & other dairy products are the dietary sources of lactose. • Lactose is synthesized by lactose synthase from UDPgalactose & gluc in the lactating mammary gland. The enz has two subunits, protein A (which is a galatosyl transferase) found in most cells where it synthesizes Nacetyllactosamine) & protein B (α-lactalbumin, which is found only in the lactating mammary glands, & whose synthesis is stimulated by the peptide hormone, prolactin). • When both subunits are present, the transferase produces lactose
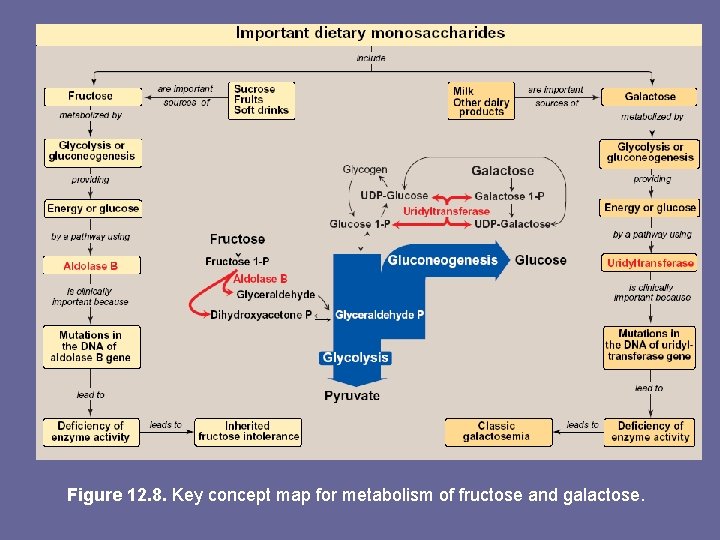
Figure 12. 8. Key concept map for metabolism of fructose and galactose.
- Slides: 29