UNIT II Bioenergetics and Carbohydrate Metabolism CHAPTER 9

UNIT II: Bioenergetics and Carbohydrate Metabolism CHAPTER 9: TRICARBOXYLIC ACID CYCLE AND PYRUVATE DEHYDROGENASE COMPLEX

• The tricarboxylic acid cycle (TCA cycle), also called the citric acid cycle or the Krebs cycle, plays several roles in metabolism • It is the final pathway where the oxidative catabolism of carbohydrates, amino acids, and fatty acids convert them to CO 2 • This oxidation provides energy for the production of the majority of ATP in most animals, including humans. • Occurs totally in the mitochondria and is, therefore, in close proximity to the reactions of ETC which oxidize the reduced coenzymes (NADH and FADH 2) produced by the cycle • It is an aerobic pathway, because O 2 is required as the final electron acceptor • Supplies intermediates for a number of important synthetic reactions. • formation of glucose from the carbon skeletons of some amino acids, • and it provides building blocks for the synthesis of some amino acids 2 Dr. M. Alzaharna 2016 I. OVERVIEW
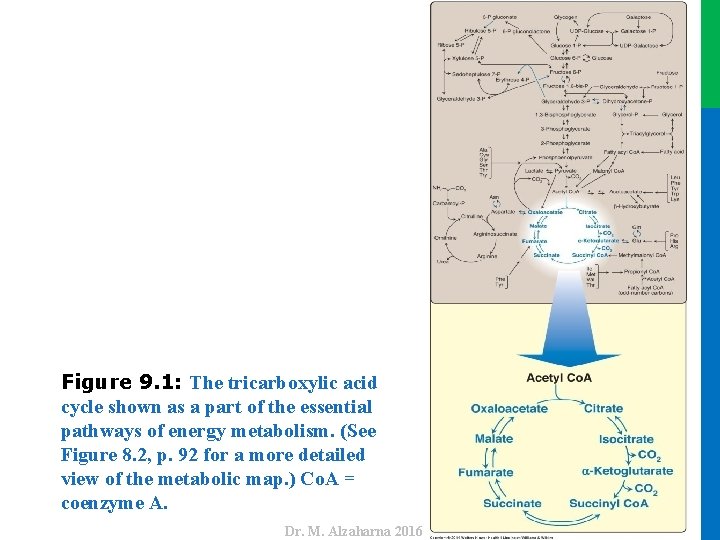
Dr. M. Alzaharna 2016 Figure 9. 1: The tricarboxylic acid cycle shown as a part of the essential pathways of energy metabolism. (See Figure 8. 2, p. 92 for a more detailed view of the metabolic map. ) Co. A = coenzyme A.

II. REACTIONS OF THE CYCLE • In the TCA cycle, oxaloacetate is first condensed with an acetyl group from acetyl coenzyme A (Co. A) and then is regenerated as the cycle is completed (Figure 9. 1). • Note: Two carbons entering the cycle as acetyl Co. A are balanced by two CO 2 exiting. 4 Dr. M. Alzaharna 2016 • Therefore, the entry of one acetyl Co. A into one round of the TCA cycle does not lead to the net production or consumption of intermediates.

II. REACTIONS OF THE CYCLE A. Oxidative decarboxylation of pyruvate • Pyruvate, must be transported from the cytosol into the mitochondrion by a specific transporter • Once in the mitochondrial matrix, pyruvate is converted to acetyl Co. A by the pyruvate dehydrogenase complex (PDH complex), which is a multienzyme complex. • Note: Strictly speaking, the PDH complex is not part of the TCA cycle, but it supplies substrate for the cycle. • The PDH complex is a protein aggregate of three enzymes, each catalyzes a part of the overall reaction (Figure 9. 2) • E 1: pyruvate carboxylase (sometimes called pyruvate dehydrogenase), • E 2: dihydrolipoyl transacetylase, • E 3: and dihydrolipoyl dehydrogenase • Their physical association links the reactions in proper sequence without the release of intermediates. 5 Dr. M. Alzaharna 2016 1. Component enzymes:
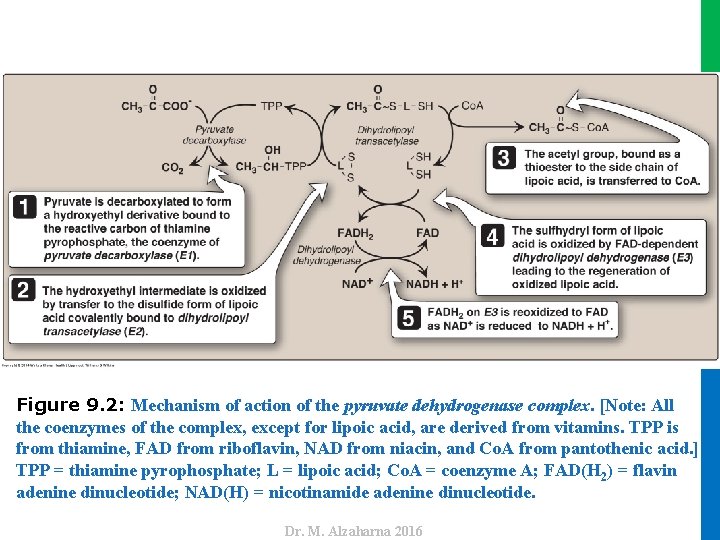
Dr. M. Alzaharna 2016 Figure 9. 2: Mechanism of action of the pyruvate dehydrogenase complex. [Note: All the coenzymes of the complex, except for lipoic acid, are derived from vitamins. TPP is from thiamine, FAD from riboflavin, NAD from niacin, and Co. A from pantothenic acid. ] TPP = thiamine pyrophosphate; L = lipoic acid; Co. A = coenzyme A; FAD(H 2) = flavin adenine dinucleotide; NAD(H) = nicotinamide adenine dinucleotide.

II. REACTIONS OF THE CYCLE A. Oxidative decarboxylation of pyruvate • In addition to the enzymes participating in the conversion of pyruvate to acetyl Co. A, the complex also contains two tightly bound regulatory enzymes, pyruvate dehydrogenase kinase (PDH kinase) and pyruvate dehydrogenase phosphatase (PDH phosphatase ). • The PDH complex contains five coenzymes that act as carriers or oxidants for the intermediates of the reactions • All the coenzymes of the complex, except for lipoic acid, are derived from vitamins. 7 Dr. M. Alzaharna 2016 2. Coenzymes:

II. REACTIONS OF THE CYCLE A. Oxidative decarboxylation of pyruvate • Covalent modifications by the two regulatory enzymes that are part of the complex alternately activate and inactivate E 1. • The cyclic AMP–independent PDH kinase phosphorylates and, thereby, inactivates E 1, whereas PDH phosphatase dephosphorylates and activates E 1 (Figure 9. 3). • The kinase itself is allosterically activated by ATP, acetyl Co. A, and NADH. • Therefore, in the presence of these high energy signals, the PDH complex is turned off. • Note: It is actually the rise in the ATP/ADP, NADH/NAD+, or acetyl Co. A/Co. A ratios that affects enzymatic activity. • Pyruvate is a potent inhibitor of PDH kinase. 8 Dr. M. Alzaharna 2016 3. Regulation of the pyruvate dehydrogenase complex:
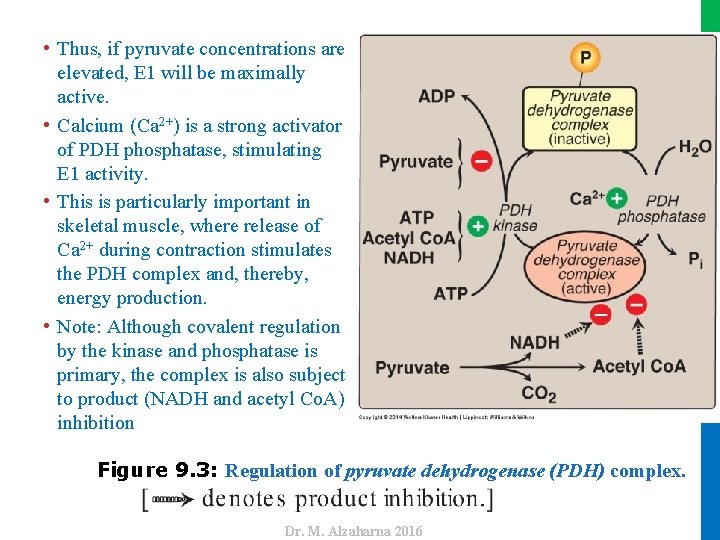
Figure 9. 3: Regulation of pyruvate dehydrogenase (PDH) complex. Dr. M. Alzaharna 2016 • Thus, if pyruvate concentrations are elevated, E 1 will be maximally active. • Calcium (Ca 2+) is a strong activator of PDH phosphatase, stimulating E 1 activity. • This is particularly important in skeletal muscle, where release of Ca 2+ during contraction stimulates the PDH complex and, thereby, energy production. • Note: Although covalent regulation by the kinase and phosphatase is primary, the complex is also subject to product (NADH and acetyl Co. A) inhibition

II. REACTIONS OF THE CYCLE A. Oxidative decarboxylation of pyruvate 4. Pyruvate dehydrogenase complex deficiency: Dr. M. Alzaharna 2016 • A deficiency in the activity of E 1 component of the PDH complex, although rare, is the most common biochemical cause of congenital lactic acidosis. • This enzyme deficiency results in an inability to convert pyruvate to acetyl Co. A, causing pyruvate to be shunted to lactate via lactate dehydrogenase. • This creates particular problems for the brain, which relies on the TCA cycle for most of its energy and is particularly sensitive to acidosis. 10 Dr. M. Alzaharna 2016

II. REACTIONS OF THE CYCLE B. Synthesis of citrate from acetyl coenzyme A and oxaloacetate • The condensation of acetyl Co. A and oxaloacetate (OAA) to form citrate (a tricarboxylic acid) is catalyzed by citrate synthase (Figure 9. 4) • In humans, citrate synthase is not an allosteric enzyme. It is inhibited by its product, citrate. • The binding of OAA causes a conformational change in the enzyme that generates a binding site for acetyl Co. A. 11 Dr. M. Alzaharna 2016 • Substrate availability is another means of regulation for citrate synthase.
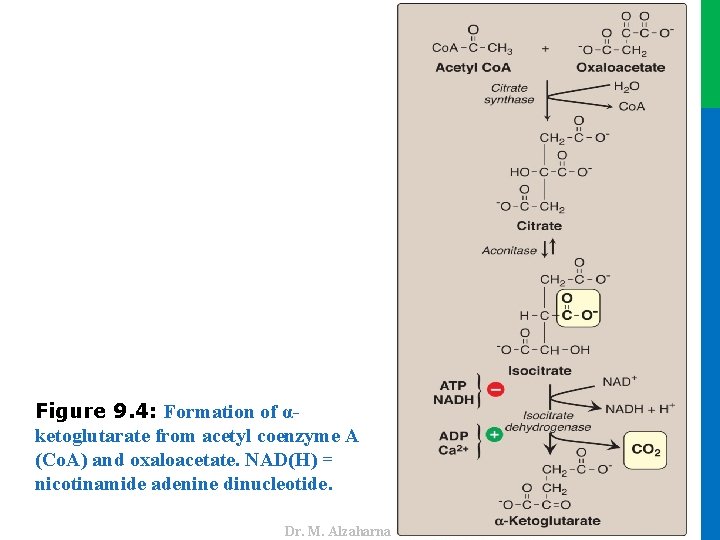
Dr. M. Alzaharna 2016 Figure 9. 4: Formation of αketoglutarate from acetyl coenzyme A (Co. A) and oxaloacetate. NAD(H) = nicotinamide adenine dinucleotide.

II. REACTIONS OF THE CYCLE C. Isomerization of citrate • Citrate is isomerized to isocitrate by aconitase (aconitate hydratase), an Fe-S protein (see Figure 9. 4) • Aconitase is inhibited by fluoroacetate, a plant toxin that is used as a pesticide Dr. M. Alzaharna 2016 • Fluoroacetate is converted to fluoroacetyl Co. A, which condenses with OAA to form fluorocitrate (a potent inhibitor of aconitase), resulting in citrate accumulation 13 Dr. M. Alzaharna 2016

II. REACTIONS OF THE CYCLE D. Oxidative decarboxylation of isocitrate • Isocitrate dehydrogenase catalyzes the irreversible oxidative decarboxylation of isocitrate giving α-ketoglutarate, • This yields the first of three NADH molecules produced by the cycle and the first release of CO 2 (see Figure 9. 4) • The enzyme is allosterically activated by ADP (a low-energy signal) and Ca 2+ and is inhibited by ATP and NADH, levels of which are elevated when the cell has abundant energy stores 14 Dr. M. Alzaharna 2016 • This is one of the rate-limiting steps of the TCA cycle

II. REACTIONS OF THE CYCLE E. Oxidative decarboxylation of αketoglutarate • The conversion of α-ketoglutarate to succinyl Co. A is catalyzed by the α-ketoglutarate dehydrogenase complex, a protein aggregate of three enzymes (Figure 9. 5) • The reaction releases the second CO 2 and produces the second NADH of the cycle • α-ketoglutarate dehydrogenase complex is inhibited by its products, NADH and succinyl Co. A, and activated by Ca 2+ 15 Dr. M. Alzaharna 2016 • The mechanism of this oxidative decarboxylation is very similar to that used for the conversion of pyruvate to acetyl Co. A by the PDH complex
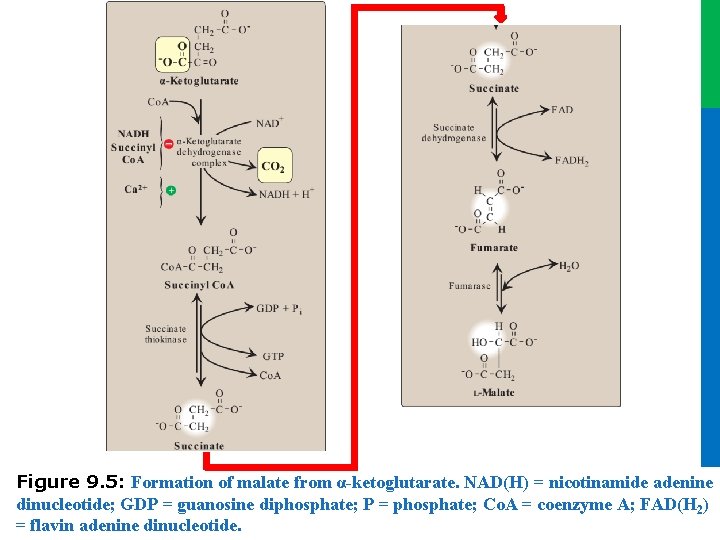
Dr. M. Alzaharna 2016 Figure 9. 5: Formation of malate from α-ketoglutarate. NAD(H) = nicotinamide adenine dinucleotide; GDP = guanosine diphosphate; P = phosphate; Co. A = coenzyme A; FAD(H 2) = flavin adenine dinucleotide. Dr. M. Alzaharna 2016

II. REACTIONS OF THE CYCLE F. Cleavage of succinyl coenzyme A • Succinate thiokinase (also called succinyl Co. A synthetase, named for the reverse reaction) cleaves the high-energy thioester bond of succinyl Co. A (see Figure 9. 5) • This reaction is coupled to phosphorylation of guanosine diphosphate (GDP) to guanosine triphosphate (GTP) • The generation of GTP by succinate thiokinase is another example of substrate-level phosphorylation 17 Dr. M. Alzaharna 2016 • GTP and ATP are energetically interconvertible by the nucleoside diphosphate kinase reaction

II. REACTIONS OF THE CYCLE G. Oxidation of succinate • Succinate is oxidized to fumarate by succinate dehydrogenase, as FAD (its coenzyme) is reduced to FADH 2 (see Figure 9. 5) • Succinate dehydrogenase is the only enzyme of the TCA cycle that is embedded in the inner mitochondrial membrane • Note: FAD, rather than NAD+, is the electron acceptor because the reducing power of succinate is not sufficient to reduce NAD+ 18 Dr. M. Alzaharna 2016 • As such, it functions as Complex II of the electron transport chain

II. REACTIONS OF THE CYCLE H. Hydration of fumarate • Fumarate is hydrated to malate in a freely reversible reaction catalyzed by fumarase (fumarate hydratase; see Figure 9. 5) I. Oxidation of malate • This reaction produces the third and final NADH of the cycle • The standard free energy change (∆Go) of the reaction is positive, but the reaction is driven in the direction of OAA by the highly exergonic citrate synthase reaction 19 Dr. M. Alzaharna 2016 • Malate is oxidized to oxaloacetate by malate dehydrogenase (Figure 9. 6)
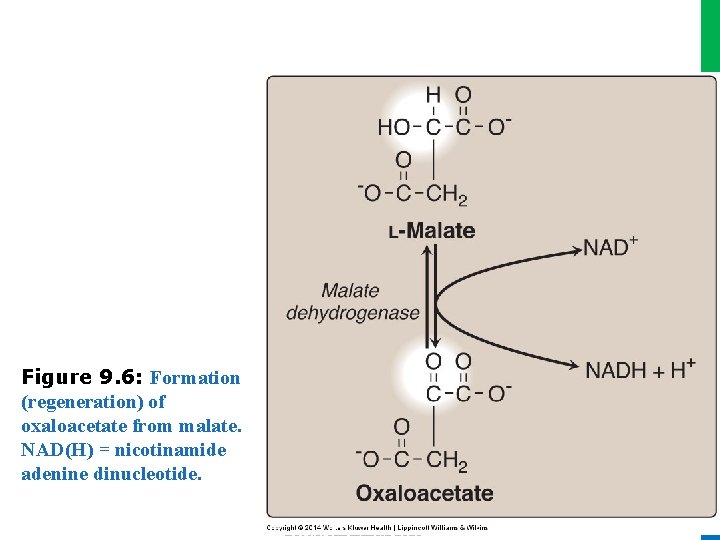
Dr. M. Alzaharna 2016 Figure 9. 6: Formation (regeneration) of oxaloacetate from malate. NAD(H) = nicotinamide adenine dinucleotide. Dr. M. Alzaharna 2016

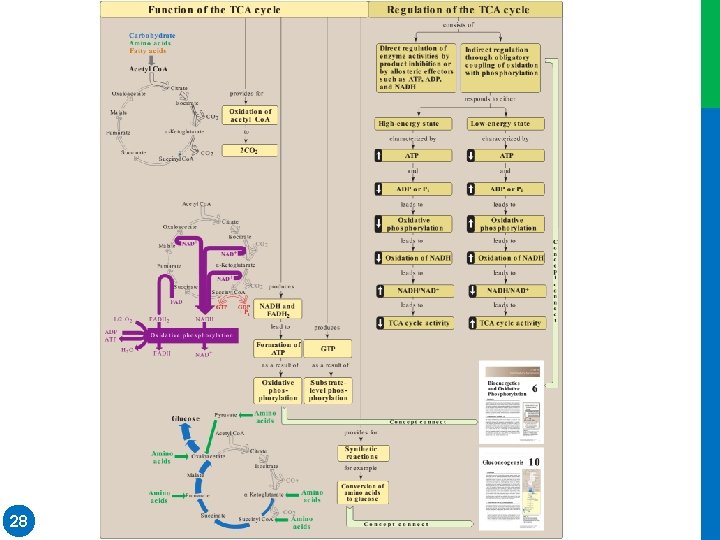
III. ENERGY PRODUCED BY THE CYCLE • Two carbon atoms enter the cycle as acetyl Co. A and leave as CO 2. • The cycle does not involve net consumption or production of OAA or of any other intermediate • Four pairs of electrons are transferred during one turn of the cycle: • Oxidation of one NADH by the electron transport chain leads to formation of three ATP, whereas oxidation of FADH 2 yields two ATP • The total yield of ATP from the oxidation of one acetyl Co. A is shown in Figure 9. 7 • Figure 9. 8 summarizes the reactions of the TCA cycle 21 Dr. M. Alzaharna 2016 • three pairs of electrons reducing three NAD+ to NADH and one pair reducing FAD to FADH 2

Dr. M. Alzaharna 2016 • Figure 9. 7: Number of ATP molecules produced from the oxidation of one molecule of acetyl coenzyme A (Co. A) using both substrate-level and oxidative phosphorylation.
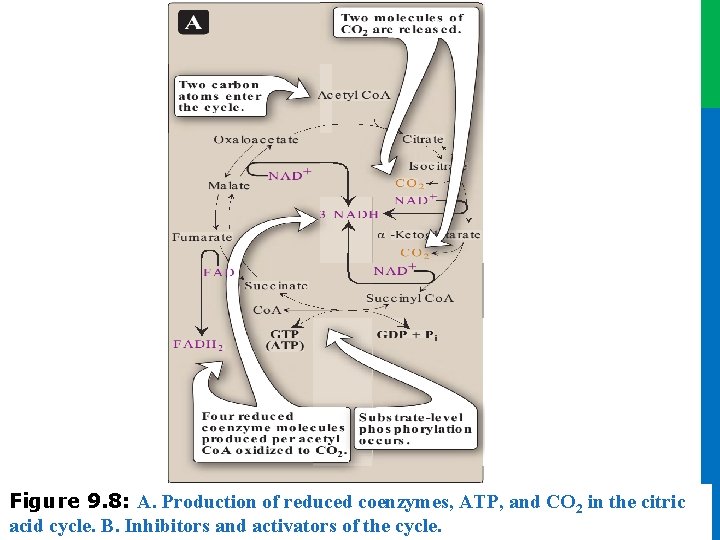
Dr. M. Alzaharna 2016 Figure 9. 8: A. Production of reduced coenzymes, ATP, and CO 2 in the citric acid cycle. B. Inhibitors and activators of the 2016 cycle. Dr. M. Alzaharna

IV. REGULATION OF THE CYCLE • In contrast to glycolysis, which is regulated primarily by PFK 1, the TCA cycle is controlled by the regulation of several enzymes Figure 9. 8) • citrate synthase, • isocitrate dehydrogenase, • and α-ketoglutarate dehydrogenase complex • Reducing equivalents needed for oxidative phosphorylation are generated by the PDH complex and the TCA cycle, and both processes are upregulated in response to a decrease in the ratio of ATP to ADP 24 Dr. M. Alzaharna 2016 • The most important of these regulated enzymes are those that catalyze reactions with highly negative ∆G 0:
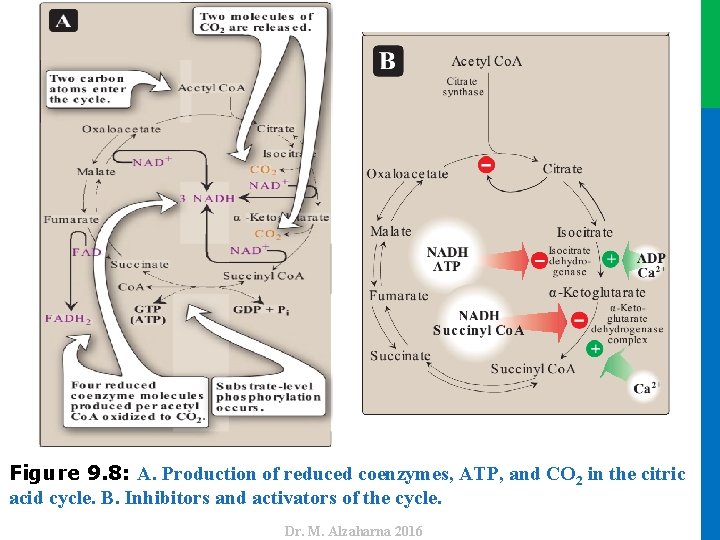
Dr. M. Alzaharna 2016 Figure 9. 8: A. Production of reduced coenzymes, ATP, and CO 2 in the citric acid cycle. B. Inhibitors and activators of the cycle.

• Pyruvate is oxidatively decarboxylated by pyruvate dehydrogenase (P DH) complex, producing acetyl coenzyme A (Co. A), which is the major fuel for the tricarboxylic acid cycle ([TCA cycle] Figure 9. 9). PDH complex is regulated by covalent modification of E 1 (pyruvate decarboxylase ) by PDH kinase a nd P DH phosphatase: phosphorylation inhibits E 1. PDH kinase is allosterically activated by ATP, acetyl Co. A, and NADH and inhibited by pyruvate. The phosphatase is activated by Ca 2+. PDH complex deficiency is the most common biochemical cause of congenital lactic acidosis. Citrate is synthesized from oxaloacetate and acetyl Co. A by citrate synthase. This enzyme is subject to product inhibition by citrate. Citrate is isomerized to is ocitrate by aconitase (aconitate hydratase). Isocitrate is oxidatively decarboxylated by isocitrate dehydrogenase to α-ketoglutarate, producing CO 2 and NADH. The enzyme is inhibited by ATP and NADH, and activated by ADP and Ca 2+. 26 Dr. M. Alzaharna 2016 V. CHAPTER SUMMARY

• α-Ketoglutarate is oxidatively decarboxylated to succinyl Co. A by the αKetoglutarate dehydrogenase complex, producing CO 2 and NADH. The enzyme is very similar to the PDH complex and uses the same coenzymes. α-ketoglutarate dehydrogenase complex is activated by Ca+2 and inhibited by NADH and succinyl Co. A but is not covalently regulated. Succinyl Co. A is cleaved by succinate thiokinase (also called succinyl Co. A synthetase), producing succinate and GTP. This is an example of substrate-level phosphorylation. Succinate is oxidized to fumarate by succinate dehydrogenase , producing FADH 2. Fumarate is hydrated to malate by fumarase (fumarate hydratas e ), and malate is oxidized to oxaloacetate by malate dehydrogenase , producing NADH. Three NADH, one FADH 2, and one GTP (whose terminal phosphate can be transferred to ADP by nucleoside diphosphate kinase, producing ATP) are produced by one round of the TCA cycle. The generation of acetyl Co. A by the oxidation of pyruvate via the PDH complex also produces an NADH. Oxidation of the NADH and FADH 2 by the electron transport chain yields 14 ATP. An additional ATP (GTP) comes from substrate level phosphorylation in the TCA cycle. Therefore, a total of 15 ATP are produced from the complete mitochondrial oxidation of pyruvate to CO 2. 27 Dr. M. Alzaharna 2016 V. CHAPTER SUMMARY
Dr. M. Alzaharna 2016 28 Dr. M. Alzaharna 2016
- Slides: 28