UNIT II Bioenergetics and Carbohydrate Metabolism CHAPTER 13

UNIT II: Bioenergetics and Carbohydrate Metabolism CHAPTER 13: PENTOSE PHOSPHATE PATHWAY AND NICOTINAMIDE ADENINE DINUCLEOTIDE PHOSPHATE

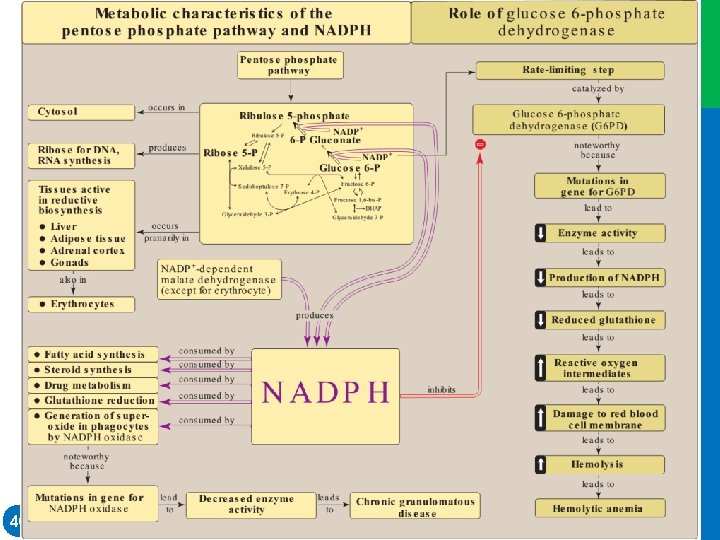
I. OVERVIEW • The pentose phosphate pathway (also called the hexose monophosphate shunt) occurs in the cytosol of the cell. • It includes two irreversible oxidative reactions, followed by a series of reversible sugar–phosphate interconversions (Figure 13. 1). • Carbon 1 of glucose 6 -phosphate is released as CO 2, and two reduced nicotinamide adenine dinucleotide phosphates (NADPHs) are produced for each glucose 6 -phosphate molecule entering the oxidative part of the pathway. 2 • The rate and direction of the reversible reactions of the pentose phosphate pathway are determined by the supply of and demand for intermediates of the cycle. Dr. M. Alzaharna 2016 • No adenosine triphosphate (ATP) is directly consumed or produced in the cycle.
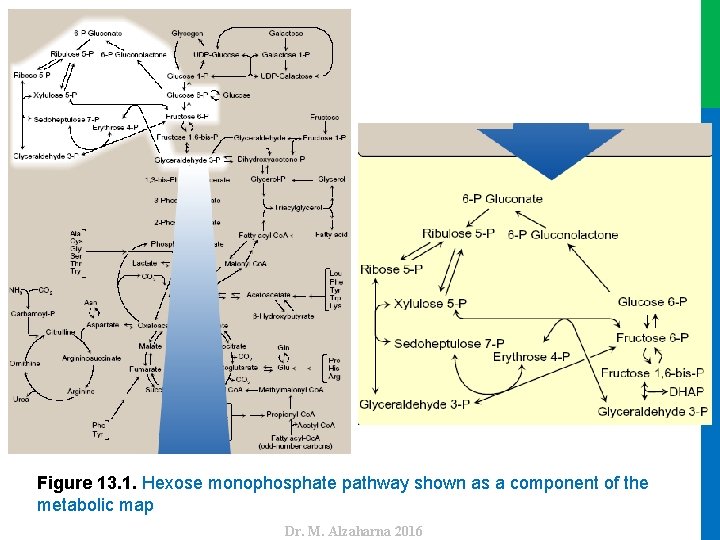
Dr. M. Alzaharna 2016 Figure 13. 1. Hexose monophosphate pathway shown as a component of the metabolic map

I. OVERVIEW • The pathway provides a major portion of the body’s NADPH, which functions as a biochemical reductant. Dr. M. Alzaharna 2016 • It also produces ribose 5 -phosphate, required for the biosynthesis of nucleotides, and provides a mechanism for the metabolic use of five-carbon sugars obtained from the diet or the degradation of structural carbohydrates 4 Dr. M. Alzaharna 2016

II. IRREVERSIBLE OXIDATIVE REACTIONS • The oxidative portion of the pentose phosphate pathway consists of three reactions that lead to the formation of ribulose 5 -phosphate, CO 2, and two molecules of NADPH for each molecule of glucose 6 -phosphate oxidized (Figure 13. 2). • the liver, lactating mammary glands, and adipose tissue, which are active in the NADPH-dependent biosynthesis of fatty acids; • in the testes, ovaries, placenta, and adrenal cortex, which are active in the NADPH-dependent biosynthesis of steroid hormones; • and in red blood cells (RBCs), which require NADPH to keep glutathione reduced 5 Dr. M. Alzaharna 2016 • This portion of the pathway is particularly important in:
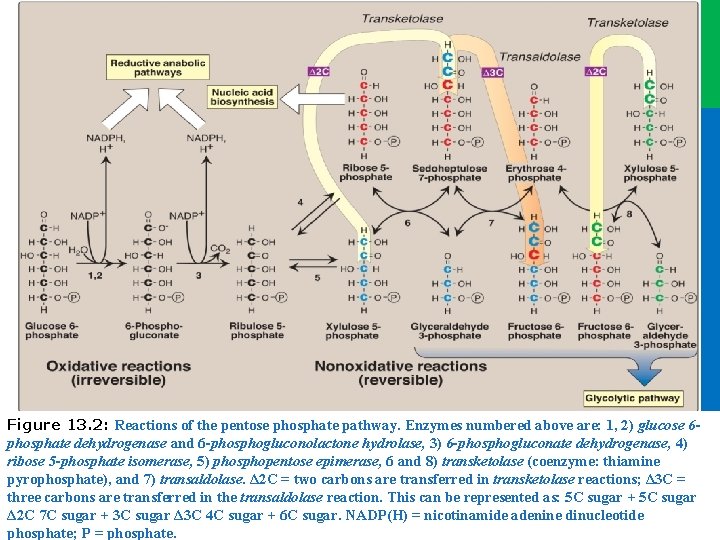
Dr. M. Alzaharna 2016 Figure 13. 2: Reactions of the pentose phosphate pathway. Enzymes numbered above are: 1, 2) glucose 6 phosphate dehydrogenase and 6 -phosphogluconolactone hydrolase, 3) 6 -phosphogluconate dehydrogenase, 4) ribose 5 -phosphate isomerase, 5) phosphopentose epimerase, 6 and 8) transketolase (coenzyme: thiamine pyrophosphate), and 7) transaldolase. Δ 2 C = two carbons are transferred in transketolase reactions; Δ 3 C = three carbons are transferred in the transaldolase reaction. This can be represented as: 5 C sugar + 5 C sugar Δ 2 C 7 C sugar + 3 C sugar Δ 3 C 4 C sugar + 6 C sugar. NADP(H) = nicotinamide adenine dinucleotide Dr. M. Alzaharna 2016 phosphate; P = phosphate.

II. IRREVERSIBLE OXIDATIVE REACTIONS A. Dehydrogenation of glucose 6 phosphate • Glucose 6 -phosphate dehydrogenase (G 6 PD) catalyzes an irreversible oxidation of glucose 6 -phosphate to 6 phosphogluconolactone in a reaction that is specific for oxidized NADP (NADP+) as the coenzyme. • NADPH is a potent competitive inhibitor of the enzyme, and, under most metabolic conditions, the ratio of NADPH/NADP+ is sufficiently high to greatly inhibit enzyme activity. • However, with increased demand for NADPH, the ratio of NADPH/ NADP+ decreases, and flux through the cycle increases in response to the enhanced activity of G 6 PD. • Insulin upregulates expression of the gene for G 6 PD, and flux through the pathway increases in the absorptive state 7 Dr. M. Alzaharna 2016 • The pentose phosphate pathway is regulated primarily at the G 6 PD reaction.

II. IRREVERSIBLE OXIDATIVE REACTIONS B. Formation of ribulose 5 -phosphate • 6 -Phosphogluconolactone is hydrolyzed by 6 phosphogluconolactone hydrolase. • The reaction is irreversible and not rate limiting. • This irreversible reaction produces: • a pentose sugar–phosphate (ribulose 5 -phosphate), • CO 2 (from carbon 1 of glucose), • and a second molecule of NADPH (Figure 13. 2). 8 Dr. M. Alzaharna 2016 • The oxidative decarboxylation of the product, 6 phosphogluconate, is catalyzed by 6 -phosphogluconate dehydrogenase.

III. REVERSIBLE NONOXIDATIVE REACTIONS • The nonoxidative reactions of the pentose phosphate pathway occur in all cell types synthesizing nucleotides and nucleic acids. • These reactions catalyze the interconversion of sugars containing three to seven carbons (Figure 13. 2). • to ribose 5 -phosphate (needed for nucleotide synthesis) • or to intermediates of glycolysis (that is, fructose 6 -phosphate and glyceraldehyde 3 -phosphate). • For example, many cells that carry out reductive biosynthetic reactions have a greater need for NADPH than for ribose 5 phosphate. 9 Dr. M. Alzaharna 2016 • These reversible reactions permit ribulose 5 -phosphate to be converted either:

III. REVERSIBLE NONOXIDATIVE REACTIONS • In this case, • transketolase (which transfers two-carbon units) • and transaldolase (which transfers three –carbon units) • In contrast, when the demand for ribose for nucleotides and nucleic acids is greater than the need for NADPH, the nonoxidative reactions can provide the ribose 5 -phosphate from glyceraldehyde 3 -phosphate and fructose 6 -phosphate in the absence of the oxidative steps (Figure 13. 3). 10 Dr. M. Alzaharna 2016 convert the ribulose 5 -phosphate produced as an end product of the oxidative reactions to glyceraldehyde 3 -phosphate and fructose 6 -phosphate, which are glycolytic intermediates.
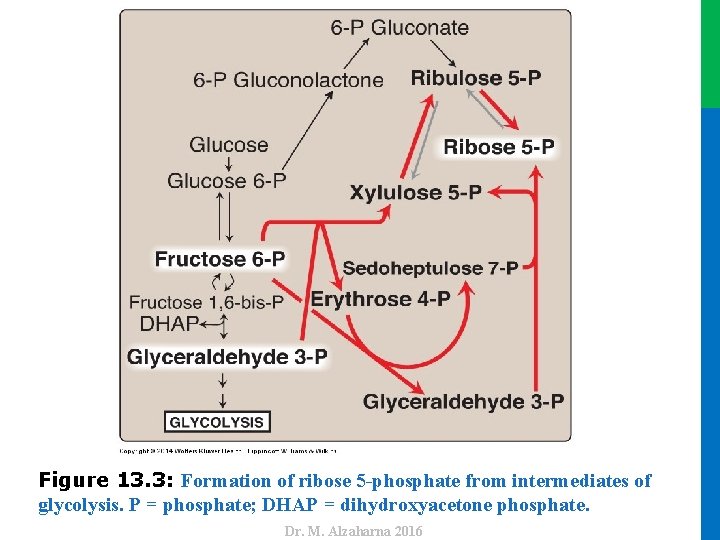
Dr. M. Alzaharna 2016 Figure 13. 3: Formation of ribose 5 -phosphate from intermediates of glycolysis. P = phosphate; DHAP = dihydroxyacetone phosphate.

IV. USES OF NADPH • The coenzyme NADPH differs from nicotinamide adenine dinucleotide (NADH) only by the presence of a phosphate group on one of the ribose units (Figure 13. 4). • This section summarizes some important NADP+ and NADPH specific functions in reductive biosynthesis and detoxification reactions 12 Dr. M. Alzaharna 2016 • This seemingly small change in structure allows NADPH to interact with NADPH-specific enzymes that have unique roles in the cell.
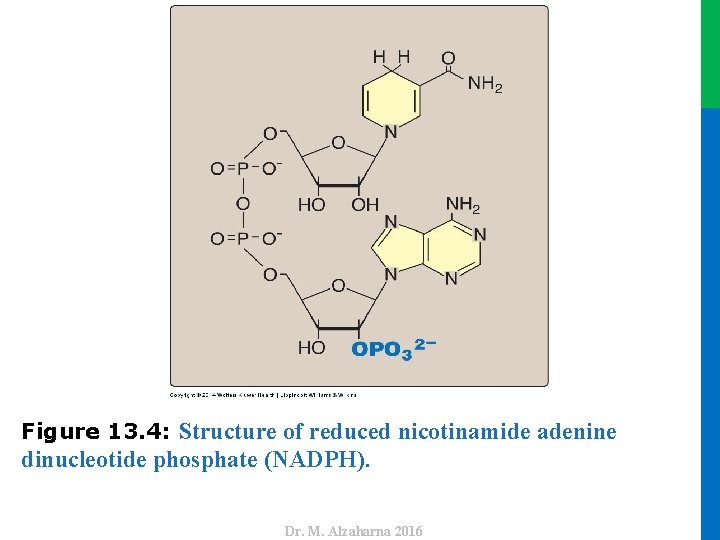
dinucleotide phosphate (NADPH). Dr. M. Alzaharna 2016 Figure 13. 4: Structure of reduced nicotinamide adenine

IV. USES OF NADPH A. Reductive biosynthesis • NADPH can be thought of as a high-energy molecule, much in the same way as NADH. • Thus, in the metabolic transformations of the pentose phosphate pathway, part of the energy of glucose 6 -phosphate is conserved in NADPH, a molecule with a negative reduction potential, that, therefore, can be used in reactions requiring an electron donor, such as fatty acid and steroid synthesis. 14 Dr. M. Alzaharna 2016 • However, the electrons of NADPH are destined for use in reductive biosynthesis, rather than for transfer to oxygen as is the case with NADH.

IV. USES OF NADPH B. Reduction of hydrogen peroxide • Hydrogen peroxide (H 2 O 2) is one of a family of reactive oxygen species (ROS) that are formed from the partial reduction of molecular oxygen (Figure 13. 5 A). • The highly reactive oxygen intermediates can cause serious chemical damage to DNA, proteins, and unsaturated lipids and can lead to cell death. • ROS have been implicated in a number of pathologic processes, including reperfusion injury, cancer, inflammatory disease, and aging. • The cell has several protective mechanisms that minimize the toxic potential of these compounds 15 Dr. M. Alzaharna 2016 • These compounds are formed continuously as byproducts of aerobic metabolism, through reactions with drugs and environmental toxins, or when the level of antioxidants is diminished, all creating the condition of oxidative stress.
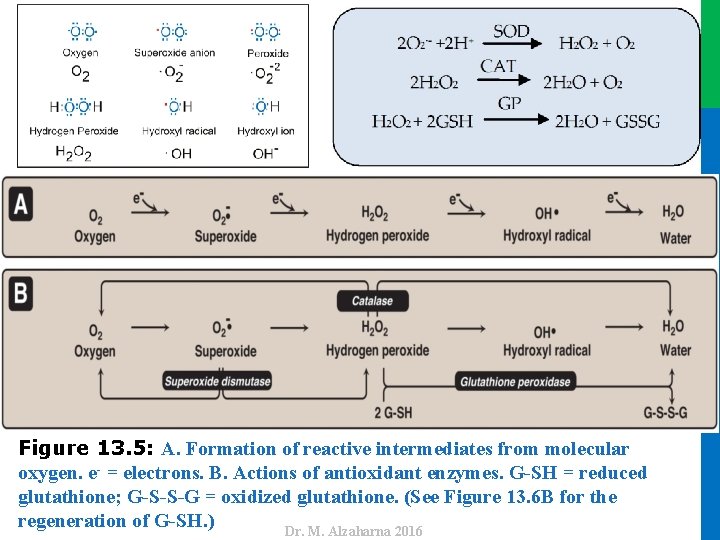
Dr. M. Alzaharna 2016 Figure 13. 5: A. Formation of reactive intermediates from molecular oxygen. e- = electrons. B. Actions of antioxidant enzymes. G-SH = reduced glutathione; G-S-S-G = oxidized glutathione. (See Figure 13. 6 B for the regeneration of G-SH. ) Dr. M. Alzaharna 2016

IV. USES OF NADPH B. Reduction of hydrogen peroxide 17 • Reduced glutathione (G-SH), present in most cells, can chemically detoxify H 2 O 2 (Figure 13. 5 B). • The cell regenerates G-SH in a reaction catalyzed by glutathione reductase, using NADPH as a source of reducing equivalents. • Thus, NADPH indirectly provides electrons for the reduction of H 2 O 2 (Figure 13. 6). • Note: RBCs are totally dependent on the pentose phosphate pathway for their supply of NADPH because, unlike other cell types, RBCs do not have an alternate source for this essential coenzyme. • Additional enzymes, such as superoxide dismutase and catalase, catalyze the conversion of other reactive oxygen intermediates to harmless products (see Figure 13. 5 B). • As a group, these enzymes serve as a defense system to guard against the toxic effects of ROS. Dr. M. Alzaharna 2016 1. Enzymes that catalyze antioxidant reactions:
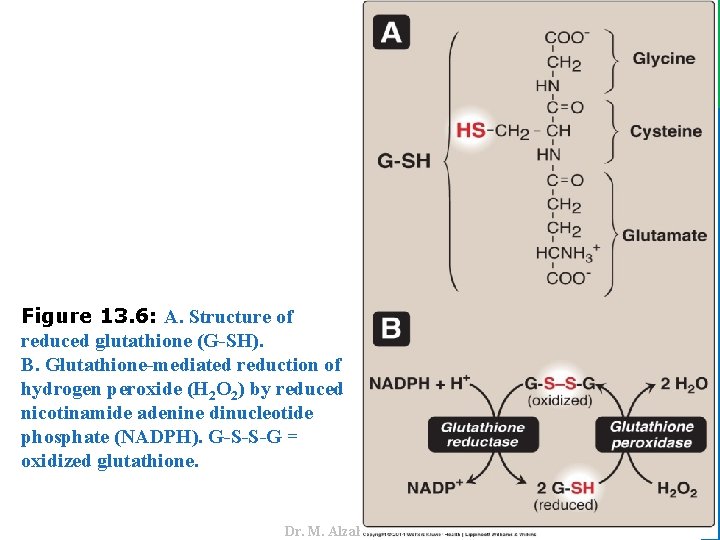
Dr. M. Alzaharna 2016 Figure 13. 6: A. Structure of reduced glutathione (G-SH). B. Glutathione-mediated reduction of hydrogen peroxide (H 2 O 2) by reduced nicotinamide adenine dinucleotide phosphate (NADPH). G-S-S-G = oxidized glutathione.

IV. USES OF NADPH B. Reduction of hydrogen peroxide • A number of intracellular reducing agents, such as ascorbate, vitamin E, and β-carotene, are able to reduce and, thereby, detoxify reactive oxygen intermediates in the laboratory. • Consumption of foods rich in these antioxidant compounds has been correlated with a reduced risk for certain types of cancers as well as decreased frequency of certain other chronic health problems. • However, clinical trials with antioxidants as dietary supplements have failed to show clear beneficial effects. • In the case of dietary supplementation with β-carotene, the rate of lung cancer in smokers increased rather than decreased. • Thus , the health-promoting effects of dietary fruits and vegetables likely reflect a complex interaction among many naturally occurring compounds, 19 Dr. M. Alzaharna 2016 2. Antioxidant chemicals:

IV. USES OF NADPH C. Cytochrome P 450 monooxygenase system • Monooxygenases (mixed-function oxidases) incorporate one atom from molecular oxygen into a substrate (creating a hydroxyl group), with the other atom being reduced to water. • This system performs different functions in two separate locations in cells. The overall reaction catalyzed by a cytochrome P 450 enzyme is: • where R may be a steroid, drug, or other chemical. 20 Dr. M. Alzaharna 2016 • In the cytochrome P 450 monooxygenase system, NADPH provides the reducing equivalents required by this series of reactions (Figure 13. 7).
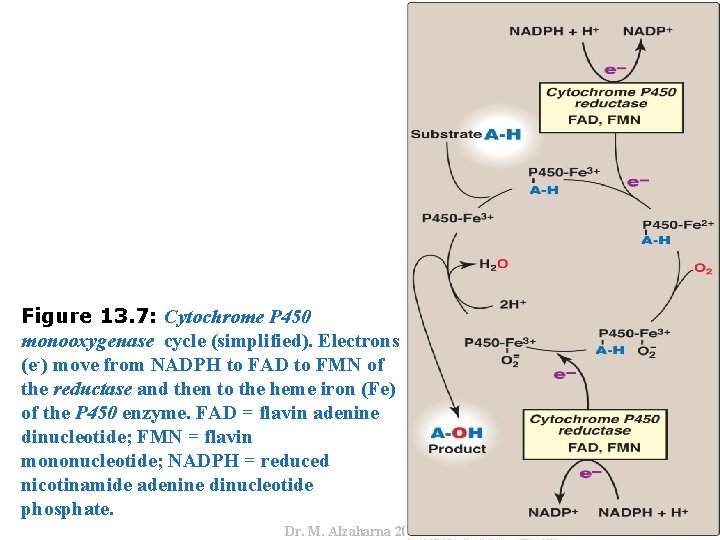
Dr. M. Alzaharna 2016 Figure 13. 7: Cytochrome P 450 monooxygenase cycle (simplified). Electrons (e-) move from NADPH to FAD to FMN of the reductase and then to the heme iron (Fe) of the P 450 enzyme. FAD = flavin adenine dinucleotide; FMN = flavin mononucleotide; NADPH = reduced nicotinamide adenine dinucleotide phosphate.

IV. USES OF NADPH C. Cytochrome P 450 monooxygenase system • An important function of the cytochrome P 450 monooxygenase system found associated with the inner mitochondrial membrane is the biosynthesis of steroid hormones. • The liver uses this same system in bile acid synthesis and the hydroxylation of cholecalciferol to 25 -hydroxycholecalciferol (vitamin D 3), • and the kidney uses it to hydroxylate vitamin D 3 to its biologically active 1, 25 -dihydroxylated form 22 Dr. M. Alzaharna 2016 1. Mitochondrial system:

IV. USES OF NADPH C. Cytochrome P 450 monooxygenase system 2. Microsomal system: • An extremely important function of the microsomal cytochrome P 450 monooxygenase system found associated with the membrane of the smooth endoplasmic reticulum (particularly in the liver) is the detoxification of foreign compounds (xenobiotics). • These include numerous drugs and such varied pollutants as petroleum products and pesticides. • CYP enzymes of the microsomal system (for example, CYP 3 A 4), can be used to hydroxylate these toxins. • The purpose of these modifications is two-fold. • First, it may itself activate or inactivate a drug • and second, make a toxic compound more soluble, thereby facilitating its excretion in the urine or feces. 23 Dr. M. Alzaharna 2016 • A xenobiotic is a foreign chemical substance found within an organism that is not normally naturally produced by or expected to be present within that organism

IV. USES OF NADPH D. Phagocytosis by white blood cells • Phagocytosis is the ingestion by receptor-mediated endocytosis of microorganisms, foreign particles, and cellular debris by cells such as neutrophils and macrophages (monocytes). • Neutrophils and monocytes are armed with both oxygenindependent and oxygen-dependent mechanisms for killing bacteria 1. Oxygen-independent mechanism: • Oxygen-independent mechanisms use p. H changes in phagolysosomes and lysosomal enzymes to destroy pathogens 24 Dr. M. Alzaharna 2016 • It is an important defense mechanism, particularly in bacterial infections.

IV. USES OF NADPH D. Phagocytosis by white blood cells • Oxygen-dependent mechanisms include the enzymes NADPH oxidase and myeloperoxidase (MPO) that work together in killing bacteria (Figure 13. 8). • Overall, the MPO system is the most potent of the bactericidal mechanisms. • An invading bacterium is recognized by the immune system and attacked by antibodies that bind it to a receptor on a phagocytic cell. • After internalization of the microorganism has occurred, NADPH oxidase, located in the leukocyte cell membrane, is activated and reduces O 2 from the surrounding tissue to superoxide , a free radical, as NADPH is oxidized. • The rapid consumption of O 2 that accompanies formation of is referred to as the “respiratory burst. ” 25 Dr. M. Alzaharna 2016 2. Oxygen-dependent system:
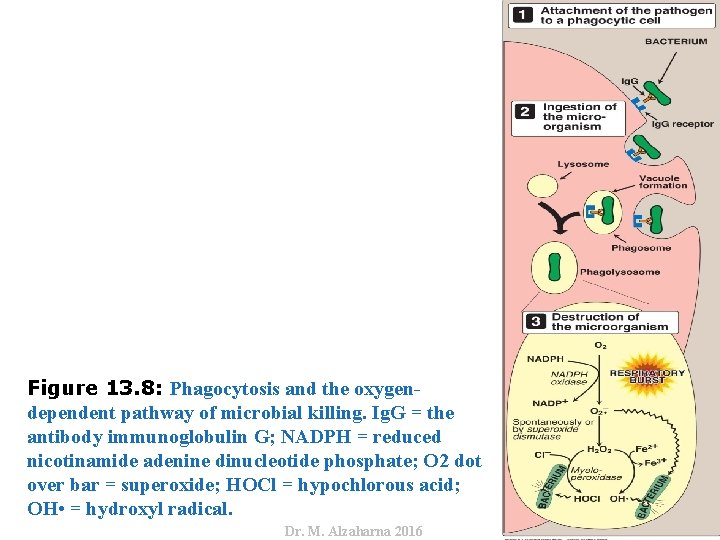
Dr. M. Alzaharna 2016 Figure 13. 8: Phagocytosis and the oxygendependent pathway of microbial killing. Ig. G = the antibody immunoglobulin G; NADPH = reduced nicotinamide adenine dinucleotide phosphate; O 2 dot over bar = superoxide; HOCl = hypochlorous acid; OH • = hydroxyl radical.

IV. USES OF NADPH • Next, is converted to H 2 O 2 (a ROS), either spontaneously or catalyzed by superoxide dismutase. • In the presence of MPO, a heme-containing lysosomal enzyme present within the phagolysosome, peroxide plus chloride ions are converted to hypochlorous acid ([HOCl] the major component of household bleach), which kills the bacteria. • The peroxide can also be partially reduced to the hydroxyl radical (OH • ), a ROS, or be fully reduced to water by catalase or glutathione peroxidase. • Note: Deficiencies in MPO do not confer increased susceptibility to infection because peroxide from NADPH oxidase is bactericidal. 27 Dr. M. Alzaharna 2016 D. Phagocytosis by white blood cells

IV. USES OF NADPH E. Synthesis of nitric oxide • Nitric oxide (NO) is recognized as a mediator in a broad array of biologic systems. • NO is the endothelium-derived relaxing factor, which causes vasodilation by relaxing vascular smooth muscle. • NO has a very short half-life in tissues (3– 10 seconds) because it reacts with oxygen and superoxide and then is converted into nitrates and nitrites including peroxynitrite (O=NOO–), a reactive nitrogen species (RNS). 28 Dr. M. Alzaharna 2016 • NO also acts as a neurotransmitter, prevents platelet aggregation, and plays an essential role in macrophage function.
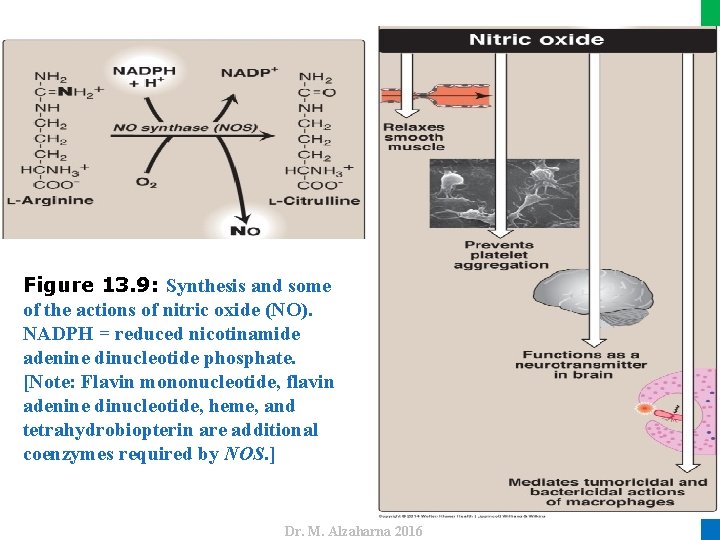
Dr. M. Alzaharna 2016 Figure 13. 9: Synthesis and some of the actions of nitric oxide (NO). NADPH = reduced nicotinamide adenine dinucleotide phosphate. [Note: Flavin mononucleotide, flavin adenine dinucleotide, heme, and tetrahydrobiopterin are additional coenzymes required by NOS. ]

V. GLUCOSE 6 -PHOSPHATE DEHYDROGENASE DEFICIENCY • G 6 PD deficiency is a hereditary disease characterized by hemolytic anemia caused by the inability to detoxify oxidizing agents. • G 6 PD deficiency is the most common disease-producing enzyme abnormality in humans, affecting more than 400 million individuals worldwide. • G 6 PD deficiency is X linked and is, in fact, a family of deficiencies caused by a number of different mutations in the gene coding for G 6 PD. • Only some of the resulting protein variants cause clinical symptoms. 30 Dr. M. Alzaharna 2016 • This deficiency has the highest prevalence in the Middle East, tropical Africa and Asia, and parts of the Mediterranean.

V. GLUCOSE 6 -PHOSPHATE DEHYDROGENASE DEFICIENCY • The jaundice, which may be severe, typically results from increased production of unconjugated bilirubin (see p. 285). • This negative effect of G 6 PD deficiency has been balanced in evolution by an advantage in survival—an increased resistance to Plasmodium falciparum malaria. • [Note: Sickle cell trait and β-thalassemia minor also confer resistance to malaria. ] 31 Dr. M. Alzaharna 2016 • The life span of individuals with a severe form of G 6 PD deficiency may be somewhat shortened as a result of complications arising from chronic hemolysis.

V. GLUCOSE 6 -PHOSPHATE DEHYDROGENASE DEFICIENCY A. Role of glucose 6 -phosphate dehydrogenase in red blood cells • Diminished G 6 PD activity impairs the ability of the cell to form the NADPH that is essential for the maintenance of the G-SH pool. • This results in a decrease in the cellular detoxification of free radicals and peroxides formed within the cell (Figure 13. 10). • G-SH also helps maintain the reduced states of sulfhydryl groups in proteins, including hemoglobin. • Additional oxidation of membrane proteins causes RBCs to be rigid (less deformable), and they are removed from the circulation by macrophages in the spleen and liver. • Although G 6 PD deficiency occurs in all cells of the affected individual, it is most severe in RBCs, where the pentose phosphate pathway provides the only means of generating NADPH. 32 Dr. M. Alzaharna 2016 • Oxidation of those sulfhydryl groups leads to the formation of denatured proteins that form insoluble masses (called Heinz bodies) that attach to RBC membranes (Figure 13. 11).
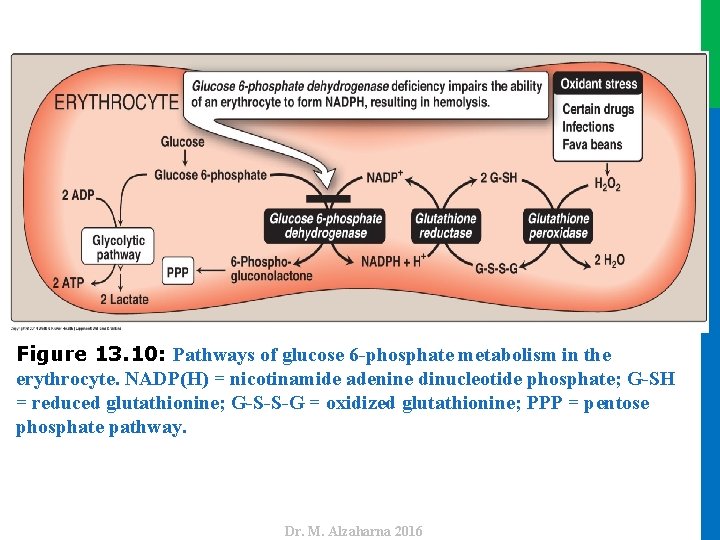
Dr. M. Alzaharna 2016 Figure 13. 10: Pathways of glucose 6 -phosphate metabolism in the erythrocyte. NADP(H) = nicotinamide adenine dinucleotide phosphate; G-SH = reduced glutathionine; G-S-S-G = oxidized glutathionine; PPP = pentose phosphate pathway.
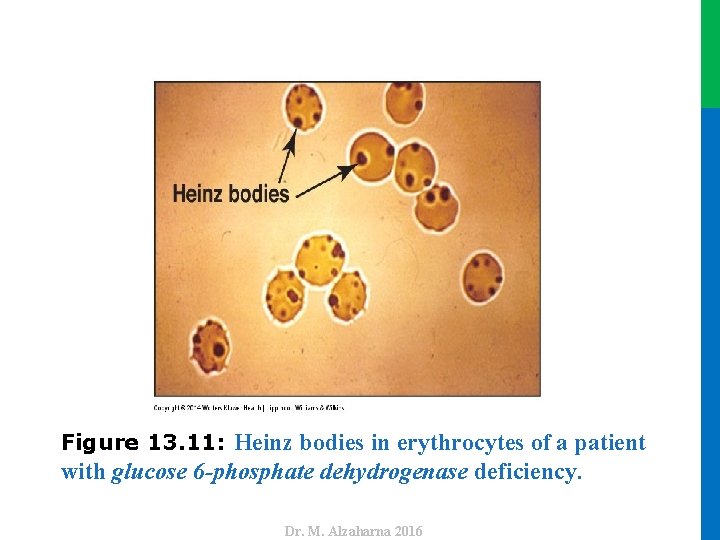
with glucose 6 -phosphate dehydrogenase deficiency. Dr. M. Alzaharna 2016 Figure 13. 11: Heinz bodies in erythrocytes of a patient

V. GLUCOSE 6 -PHOSPHATE DEHYDROGENASE DEFICIENCY A. Role of glucose 6 -phosphate dehydrogenase in red blood cells • Other tissues have alternative sources for NADPH production (such as NADP+-dependent malate dehydrogenase [malic enzyme]; that can keep G-SH reduced. • The RBC has no nucleus or ribosomes and cannot renew its supply of the enzyme. Dr. M. Alzaharna 2016 • Thus, RBCs are particularly vulnerable to enzyme variants with diminished stability 35 Dr. M. Alzaharna 2016

V. GLUCOSE 6 -PHOSPHATE DEHYDROGENASE DEFICIENCY B. Precipitating factors in glucose 6 -phosphate dehydrogenase deficiency • Most individuals who have inherited one of the G 6 PD mutations do not show clinical manifestations (that is, they are asymptomatic). • However, some patients with G 6 PD deficiency develop hemolytic anemia if they are treated with an oxidant drug, ingest fava beans, or contract a severe infection. • Commonly used drugs that produce hemolytic anemia in patients with G 6 PD deficiency are best remembered from the mnemonic AAA: • antibiotics (for example, sulfamethoxazole and chloramphenicol), • antimalarials (for example, primaquine but not chloroquine or quinine ), • and antipyretics (for example, acetanilid but not acetaminophen). 36 Dr. M. Alzaharna 2016 1. Oxidant drugs:

V. GLUCOSE 6 -PHOSPHATE DEHYDROGENASE DEFICIENCY B. Precipitating factors in glucose 6 phosphate dehydrogenase deficiency 2. Favism: • Some forms of G 6 PD deficiency, for example the Mediterranean variant, are particularly susceptible to the hemolytic effect of the fava (broad) bean, a dietary staple in the Mediterranean region. • Favism, the hemolytic effect of ingesting fava beans, is not observed in all individuals with G 6 P D deficiency, but all patients with favism have G 6 PD deficiency. • Infection is the most common precipitating factor of hemolysis in G 6 PD deficiency. • The inflammatory response to infection results in the generation of free radicals in macrophages, which can diffuse into the RBC and cause oxidative damage. 37 Dr. M. Alzaharna 2016 3. Infection:

V. GLUCOSE 6 -PHOSPHATE DEHYDROGENASE DEFICIENCY C. Properties of the variant enzymes • Almost all G 6 PD variants are caused by point mutations in the gene for G 6 PD. • Some mutations do not disrupt the structure of the enzyme’s active site and, therefore, do not affect enzymic activity. • For example, variant enzymes may show decreased catalytic activity, decreased stability, or an alteration of binding affinity for NADP+, NADPH, or glucose 6 -phosphate. • The severity of the disease usually correlates with the amount of residual enzyme activity in the patient’s RBC. • For example, variants can be classified as shown in Figure 13. 12. 38 Dr. M. Alzaharna 2016 • However, many mutant enzymes show altered kinetic properties.

Dr. M. Alzaharna 2016 Figure 13. 12: Classification of glucose 6 -phosphate dehydrogenase (G 6 PD) deficiency variants. Note: Class V variants (not shown in table) result in overproduction of G 6 PD.
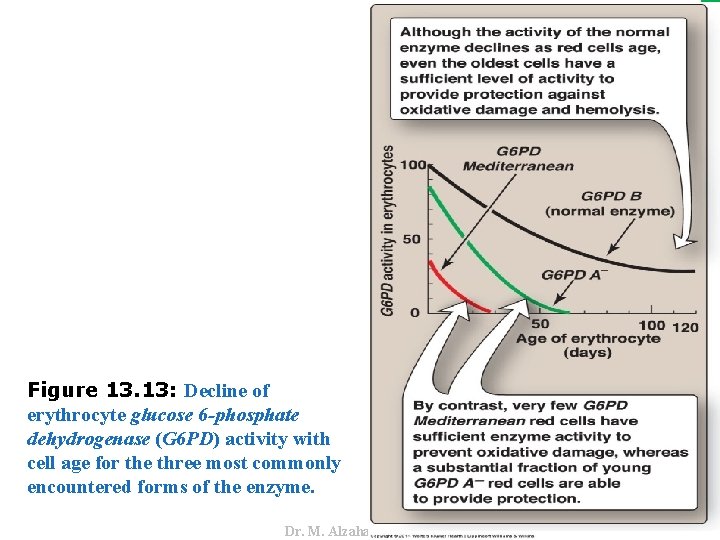
Dr. M. Alzaharna 2016 Figure 13. 13: Decline of erythrocyte glucose 6 -phosphate dehydrogenase (G 6 PD) activity with cell age for the three most commonly encountered forms of the enzyme.

V. GLUCOSE 6 -PHOSPHATE DEHYDROGENASE DEFICIENCY D. Molecular biology of glucose 6 phosphate dehydrogenase • The cloning of the gene for G 6 PD and the sequencing of its DNA have permitted the identification of mutations that cause G 6 PD deficiency. • More than 400 different G 6 PD variants have been identified, a finding that explains the numerous biochemical and clinical phenotypes that have been described. • Both G 6 PD A– and G 6 PD Mediterranean represent mutant enzymes that differ from the respective normal variants by a single amino acid. • Large deletions or frameshift mutations have not been identified, suggesting that complete absence of G 6 PD activity is probably lethal 41 Dr. M. Alzaharna 2016 • Most mutations that result in enzymic deficiency are missense mutations in the coding region.

Summary • The pentose phosphate pathway consists of 2 irreversible oxidative reactions followed by a series of reversible sugar-phosphate interconversions • No ATP is directly consumed or produced in the cycle • The oxidative portion is particularly important in liver & mammary glands, which are active in biosynthesis of fatty acids, in adrenal cortex, which is active in NADPH-dependent synthesis of steroids, & in erythrocytes, which require NADPH to keep glutathione reduced • The regulated step is G 6 PD, which is strongly inhibited by NADPH • Reversible nonoxidative reactions interconvert sugars. This part of pathway is the source of ribose 5 -P required for nt & nucleic acid synthesis • Because reactions are reversible, they can be entered from F-6 -P & GA 3 P (glycolytic intermediates) if ribose is needed & G 6 PD is inhibited Dr. M. Alzaharna 2016 • G-6 -P is irreversibly converted to ribulose-5 -P, & 2 NADPH are produced.

Summary • NADPH is a source of reducing equivalents in reductive biosynthesis, such as production of fatty acids & steroids. It is also required for reduction of hydrogen peroxide, providing the reducing equivalents required by glutathione (GSH). • NADPH provides reducing equivalents for cyt-P 450 monooxygenase system, which is used in hydroxylation of steroids to produce steroid hormones, bile acid synthesis by liver, & activation of vitamin D. the system also detoxify foreign compounds, e. g. , drugs & varied pollutants, including carcinogens, pesticides, & petroleum products Dr. M. Alzaharna 2016 • GSH is used by glutathione peroxidase to reduce peroxide to water. The oxidized glutathione is reduced by glutathione reductase, using NADPH as the source of electrons

Summary • NADPH provides reducing equivalents for phagocytes in the process of eliminating invading microorganisms • NADPH oxidase uses molecular oxygen & NADPH to produce superoxide radicals, which, in turn, can be converted to peroxide, hypochlorous acid, & hydroxyl radicals. Myeloperoxidase is an important enzyme in this pathway • NADPH is required for synthesis of nitric oxide (NO), an important molecule that causes vasodilation by relaxing vascular smooth muscle, acts as a kind of neurotransmitter, prevents platelet aggregation, & helps mediate macrophage bactericidal activity Dr. M. Alzaharna 2016 • A genetic defect in NADPH oxidase causes chronic granulomatosis, a disease characterized by severe, persistent, chronic pyogenic infections

Summary • G 6 PD deficiency is a genetic disease characterized by hemolytic anemia. It impairs ability of cell to form NADPH that is essential for maintenance of reduced glutathione pool. • Cells most affected are RBCs because they do not have additional sources of NADPH • Hemolytic anemia can be caused by production of free radicals & peroxides following the taking of oxidant drugs, ingestion of fava beans, or severe infections • Babies with G 6 PD deficiency may experience neonatal jaundice appearing 1 -4 days after birth • Degree of severity of anemia depends on location of mutation in G 6 PD gene. Class I mutations are the most severe (e. g. , G 6 PD Mediterranean). They are often associated with chronic non-spherocytic anemia. Class III mutations (e. g. , G 6 PD A-) cause a more moderate form of the disease Dr. M. Alzaharna 2016 • Free radicals & peroxides formed within the cells can’t be neutralized, causing denaturation of protein (e. g. , Hb, forming Heinz bodies) & membrane proteins. Cells become rigid, & they are removed by reticuloendothelial system of spleen & liver
Dr. M. Alzaharna 2016 46 Dr. M. Alzaharna 2016
- Slides: 46