Unit II Atomic Structure Obj 1Daltons Atomic Theory

Unit II Atomic Structure

Obj. 1…Dalton’s Atomic Theory • Four postulates (1808). . . proton, neutron and electron * * isotopes exist 1. All elements are composed of tiny, indivisible particles called ‘atoms’. 2. Atoms of the same element are identical. . . each element is unique 3. Atoms of different elements can physically mix or chemically combine (compounds). 4. Chemical rxns. occur when atoms are separated, joined, or rearranged. • Much of this theory is still accepted, with 2 exceptions

Obj. 2…Atomic Scientists • J. J. Thomson discovered electrons (e-) in 1897. • He passed an electric current through a glass tube filled with gas. • He discovered that a beam of negative charges traveled from the cathode (-) to the anode (+).

Obj. 2 cont. . . • E. Goldstein discovered positive particles in atoms • He observed rays traveling in the opposite direction of cathode rays. • These particles were termed ‘ protons’ (p+) by Ernest Rutherford in 1920. • James Chaddwick discovered neutrons (n 0) in 1932. • His discovery was based on the fact that different atoms’ atomic mass and atomic # (# of protons) were not adding up. in 1886.

Obj. 2 cont. . . • Robert Millikan furthered Thomson’s work by describing the electron more in depth. • all e- carry exactly one unit of negative charge. • mass of an e- is 1/1840 the mass of a hydrogen atom (p+).

Obj. 3 and 4…The Bohr-Rutherford Model • Ernest Rutherford conducted the famous ‘gold foil experiment’ (1911) which concluded that: 1. atoms are mostly space. (football arena) 2. atoms have a solid nucleus at the center which contains most of the mass. • This overturned the accepted ‘plum pudding model’ of the time.
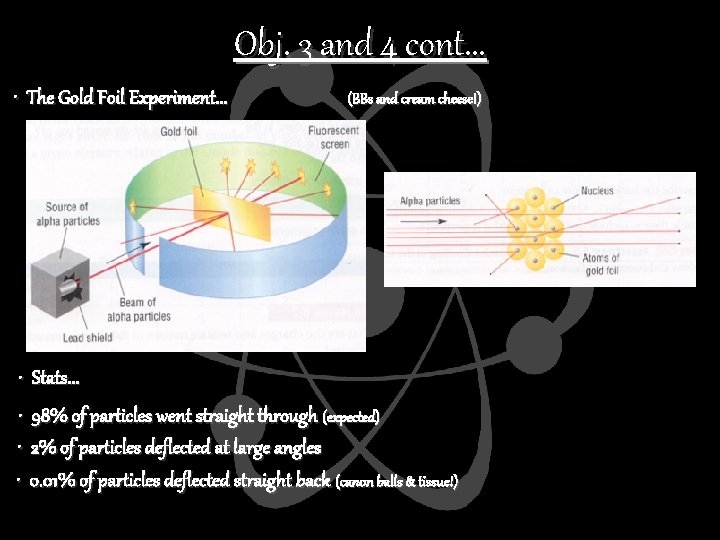
Obj. 3 and 4 cont. . . • The Gold Foil Experiment. . . (BBs and cream cheese!) • Stats. . . • 98% of particles went straight through (expected) • 2% of particles deflected at large angles • 0. 01% of particles deflected straight back (canon balls & tissue!)
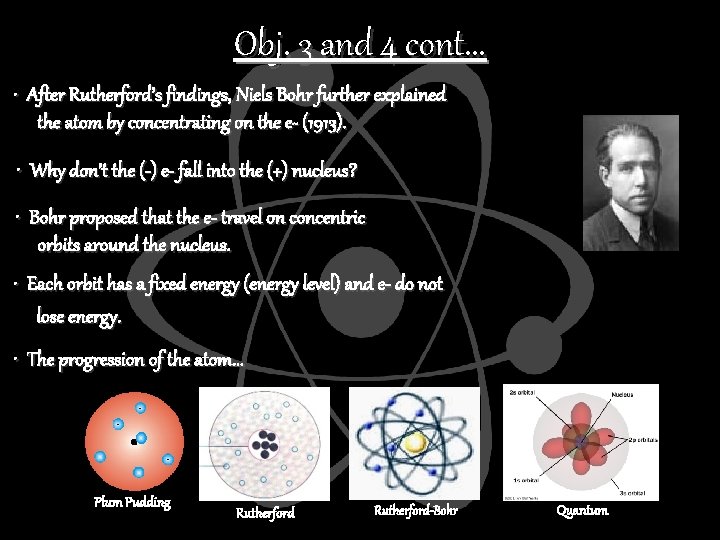
Obj. 3 and 4 cont. . . • After Rutherford’s findings, Niels Bohr further explained the atom by concentrating on the e- (1913). • Why don’t the (-) e- fall into the (+) nucleus? • Bohr proposed that the e- travel on concentric orbits around the nucleus. • Each orbit has a fixed energy (energy level) and e- do not lose energy. • The progression of the atom. . . Plum Pudding Rutherford-Bohr Quantum

Obj. 5…Subatomic Particles PROPERTIES OF SUBATOMIC PARTICLES SYMBOL ELECTRICAL CHARGE RELATIVE MASS ACTUAL MASS (g) Electron e- -1 1/1840 9. 11 x 10 -28 Proton p+ +1 1 1. 67 x 10 -24 Neutron 0 0 1 1. 67 x 10 -24 PARTICLE n
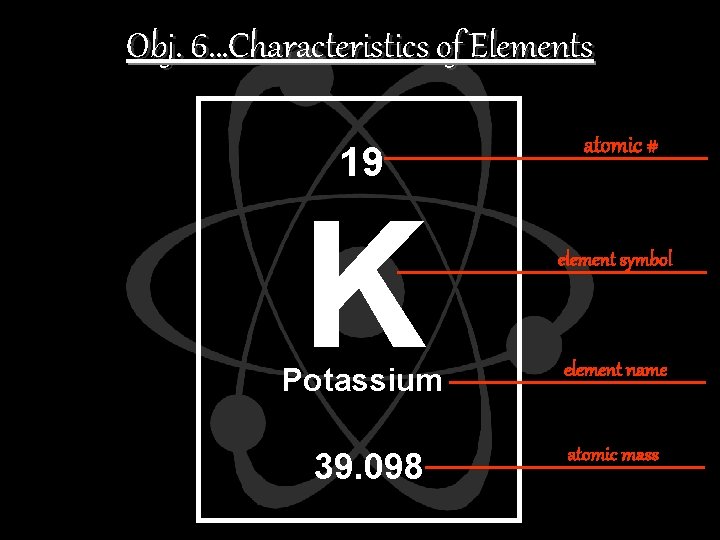
Obj. 6…Characteristics of Elements 19 K atomic # element symbol Potassium element name 39. 098 atomic mass
Obj. 6 cont. . . • Atomic #: E # of p+ in an atom E identifies element (change atomic # = change of element). E # p+ = # e- in neutral atom ** (+) charge = less e- than p+ ** (-) charge = more e- than p+ • Atomic mass: (a. k. a. mass #) E mass of the nucleus E p+ + n 0 E units are a. m. u. (atomic mass unit)
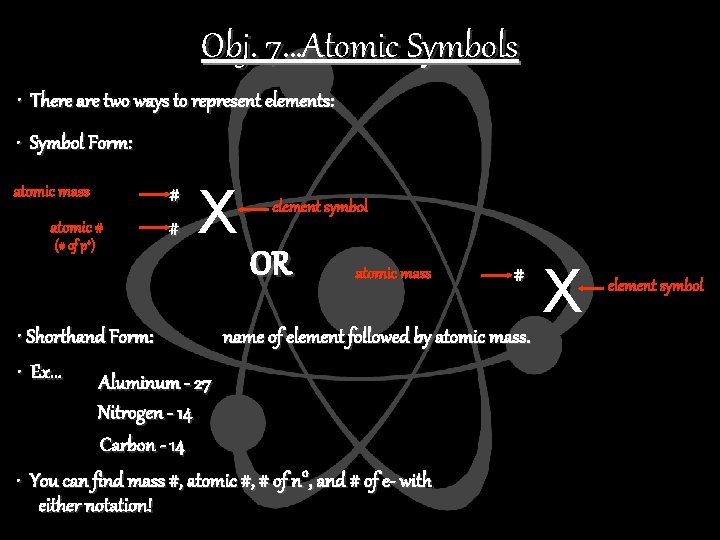
Obj. 7…Atomic Symbols • There are two ways to represent elements: • Symbol Form: atomic mass atomic # (# of p+) # # X element symbol OR atomic mass # • Shorthand Form: name of element followed by atomic mass. • Ex. . . Aluminum - 27 Nitrogen - 14 Carbon - 14 • You can find mass #, atomic #, # of n 0, and # of e- with either notation! X element symbol

Obj. 8…Atomic Calculations • All mass of the atom is in the nucleus. - only p+ and n 0 are in the nucleus. - if you know the mass of any atom, you can find the # of n 0. - if you know the # of n 0 and the # of p+, you can find the mass. • Ex. . . electrical charge on atom 31 15 +1 P Manganese - 55 element = ________ Phosphorus atomic mass = ______ atomic # = _______ # of p+ = ________ # of n 0 = ________ # of e- = ________ 31 a. m. u. s 15 15 16 14 symbol = _______ atomic mass = _____ atomic # = ______ # of p+ = _______ # of n 0 = _______ # of e- = _______ Mn 55 a. m. u. s 25 25 30 25

Obj. 9…Isotopes • Isotopes: atoms of the same element with different atomic masses. - different # of n 0 !!! • Three isotopes of Carbon: element of life (6 p+, 6 n 0) Carbon - 12 extremely rare (6 p+, 7 n 0) Carbon - 13 (6 p+, 8 n 0) Carbon - 14 radioactive…carbon dating • Note: atomic # will NEVER change in isotopes… only mass and # of n 0 do! • Which of the following are isotopes of the same element? 22 12 +3 X 22 10 X 25 10 -1 X Neon - 20 Fluorine - 20 Neon - 22

Obj. 10…Average Mass of Isotopes • Isotopes are naturally occurring. • The mass # of an element (periodic table) is the weighted avg. of all isotopes that exist in nature. - abundance of isotope is just as important as mass! • Ex. . . Natural copper (Cu) consists of 2 isotopes. . . Copper - 63 (mass = 62. 930 g/mole) Copper - 65 (mass = 64. 930 g/mole) • To calculate avg. mass. . . mass x abundance for each isotope Step 1 : add the two values from step 1 together Step 2 : 43. 42 62. 93 x. 69 = 43. 42 + 20. 13 64. 93 x. 31 = 20. 13 63. 55 g/mole 69% 31%
Obj. 10 cont. . . • The average mass of an element is closest to the isotope that is most plentiful in nature. • Ex. . . Three isotopes of Oxygen: Oxygen - 16 Oxygen - 17 Oxygen - 18 99. 759% 0. 037% 0. 204% • The avg. mass (from P. T. ) is closest to 16, therefore, Oxygen-16 is the isotope that is most abundant in nature.
- Slides: 16