UNIT I Material science STRUCTURE OF Chapter 1






































- Slides: 84

UNIT I Material science STRUCTURE OF Chapter 1 CRYSTALLINE SOLIDS

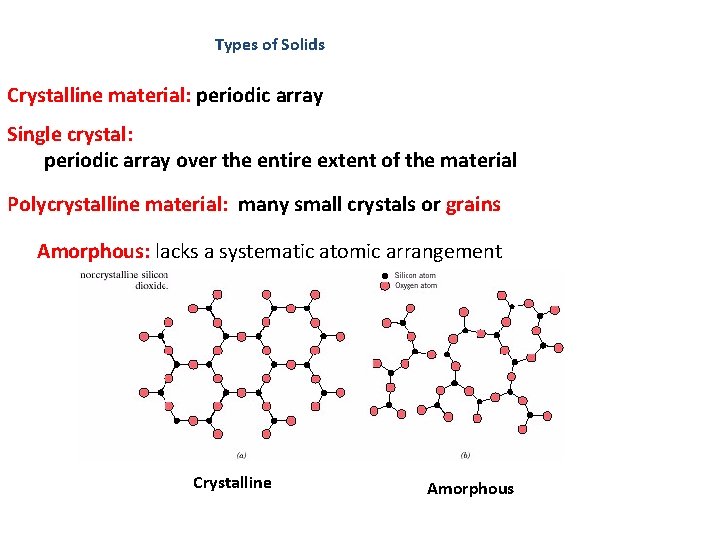
Types of Solids Crystalline material: periodic array Single crystal: periodic array over the entire extent of the material Polycrystalline material: many small crystals or grains Amorphous: lacks a systematic atomic arrangement Crystalline Amorphous

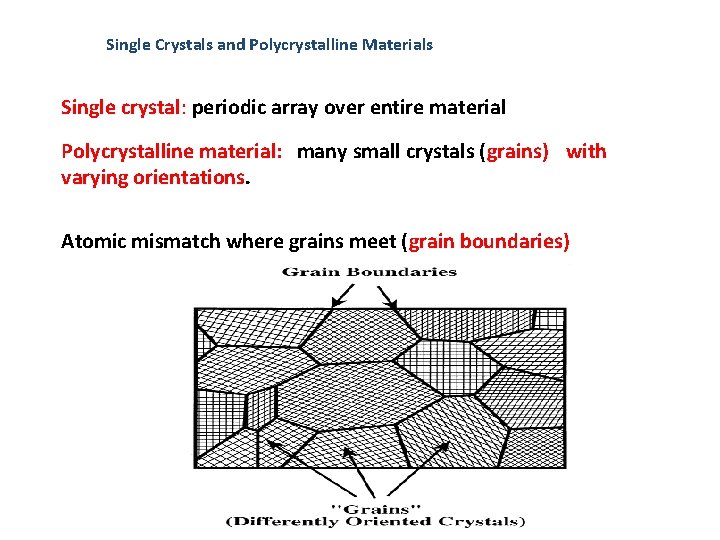
Single Crystals and Polycrystalline Materials Single crystal: periodic array over entire material Polycrystalline material: many small crystals (grains) with varying orientations. Atomic mismatch where grains meet (grain boundaries)

Polycrystalline Materials

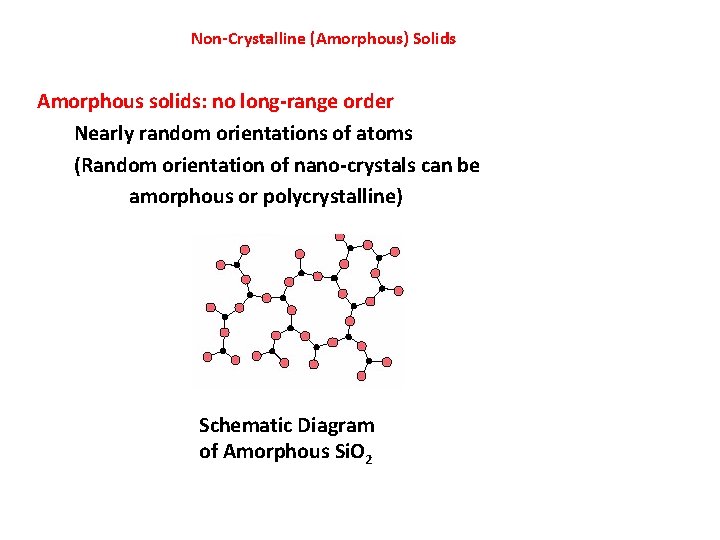
Non-Crystalline (Amorphous) Solids Amorphous solids: no long-range order Nearly random orientations of atoms (Random orientation of nano-crystals can be amorphous or polycrystalline) Schematic Diagram of Amorphous Si. O 2

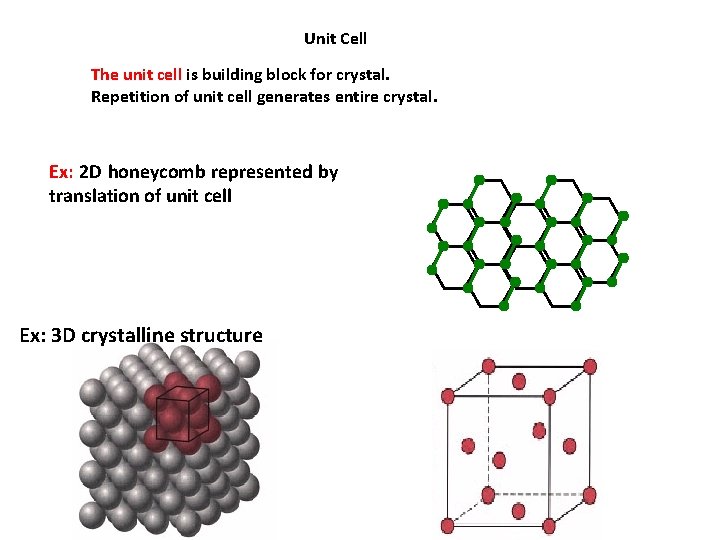
Unit Cell The unit cell is building block for crystal. Repetition of unit cell generates entire crystal. Ex: 2 D honeycomb represented by translation of unit cell Ex: 3 D crystalline structure

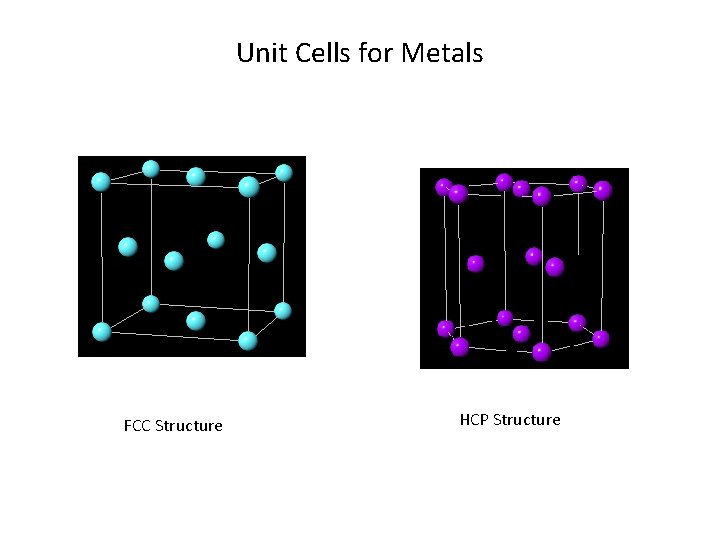
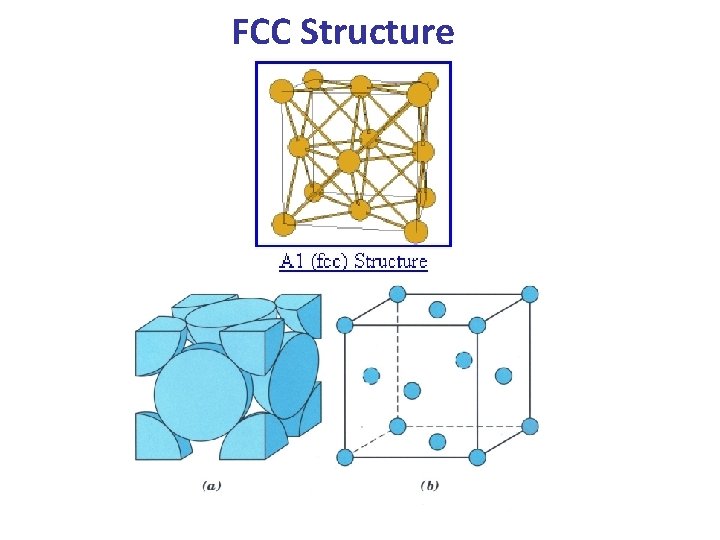
Unit Cells for Metals FCC Structure HCP Structure

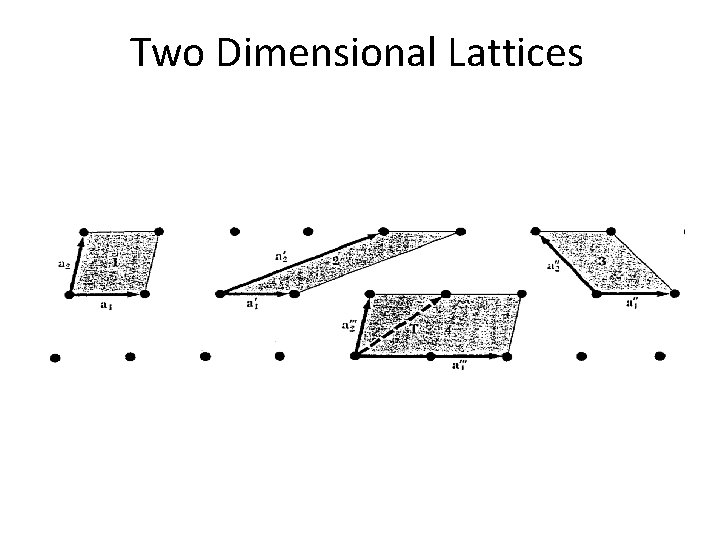
Two Dimensional Lattices

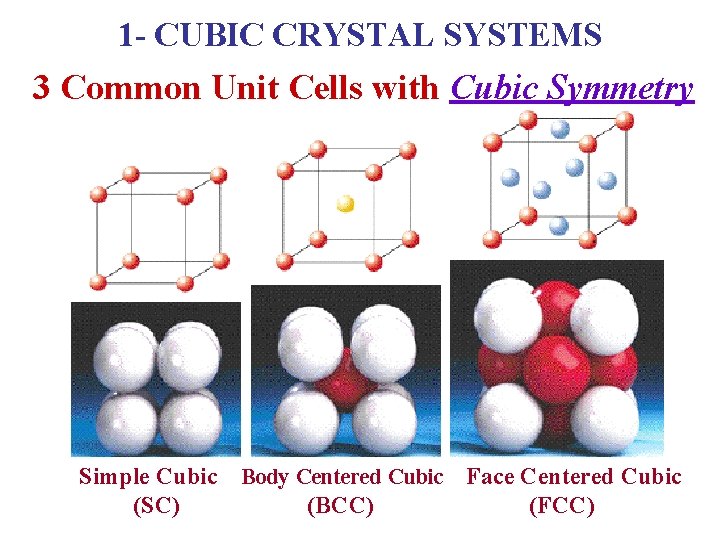
1 - CUBIC CRYSTAL SYSTEMS 3 Common Unit Cells with Cubic Symmetry Simple Cubic (SC) Body Centered Cubic (BCC) Face Centered Cubic (FCC)

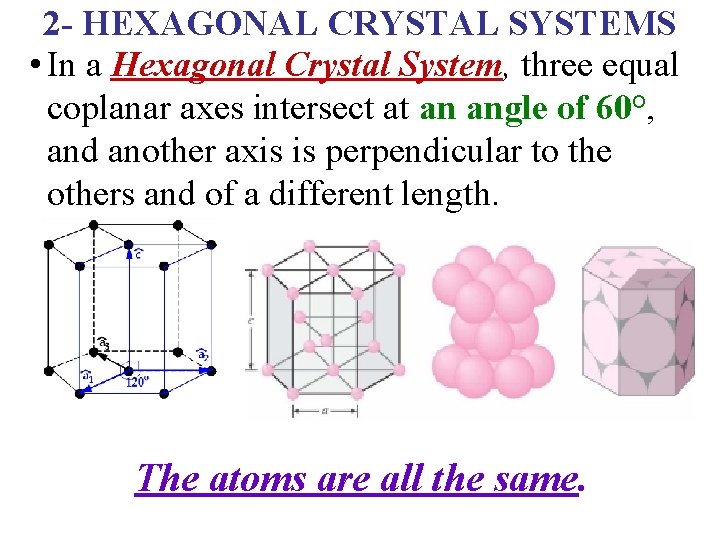
2 - HEXAGONAL CRYSTAL SYSTEMS • In a Hexagonal Crystal System, three equal coplanar axes intersect at an angle of 60°, and another axis is perpendicular to the others and of a different length. The atoms are all the same.

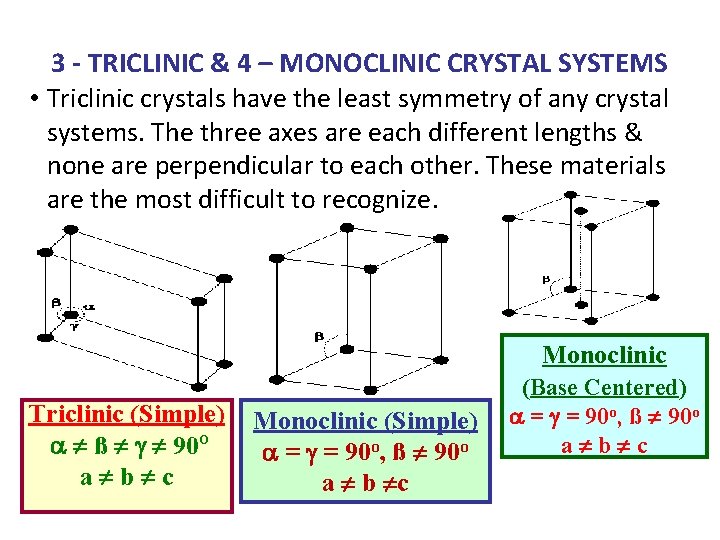
3 - TRICLINIC & 4 – MONOCLINIC CRYSTAL SYSTEMS • Triclinic crystals have the least symmetry of any crystal systems. The three axes are each different lengths & none are perpendicular to each other. These materials are the most difficult to recognize. Monoclinic Triclinic (Simple) a ¹ ß ¹ g ¹ 90 O a¹b¹c Monoclinic (Simple) a = g = 90 o, ß ¹ 90 o a ¹ b ¹c (Base Centered) a = g = 90 o, ß ¹ 90 o a¹b¹c

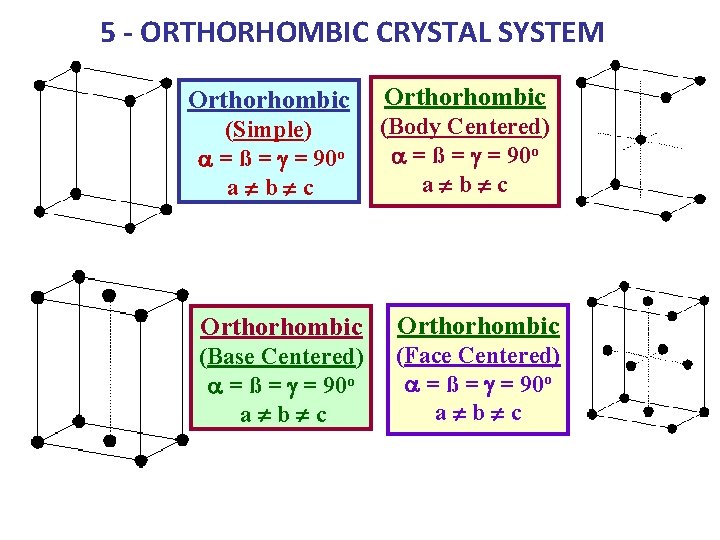
5 - ORTHORHOMBIC CRYSTAL SYSTEM Orthorhombic (Simple) a = ß = g = 90 o a¹b¹c Orthorhombic (Body Centered) a = ß = g = 90 o a¹b¹c Orthorhombic (Base Centered) a = ß = g = 90 o a¹b¹c (Face Centered) a = ß = g = 90 o a¹b¹c

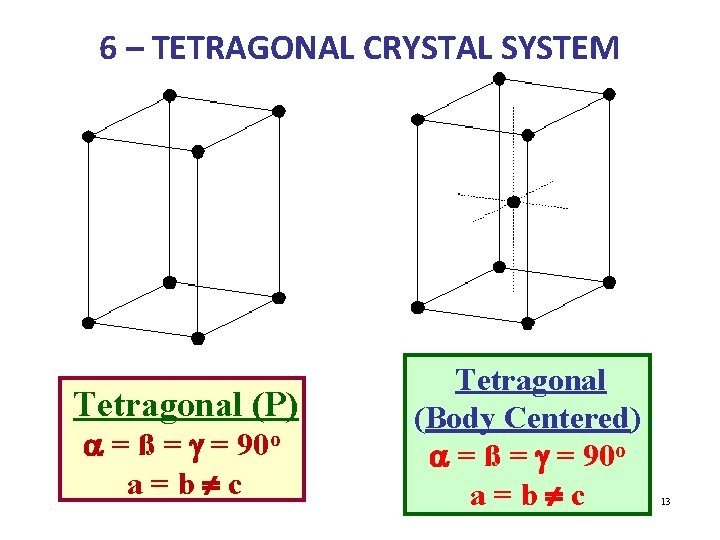
6 – TETRAGONAL CRYSTAL SYSTEM Tetragonal (P) a = ß = g = 90 o a=b¹c Tetragonal (Body Centered) a = ß = g = 90 o a=b¹c 13

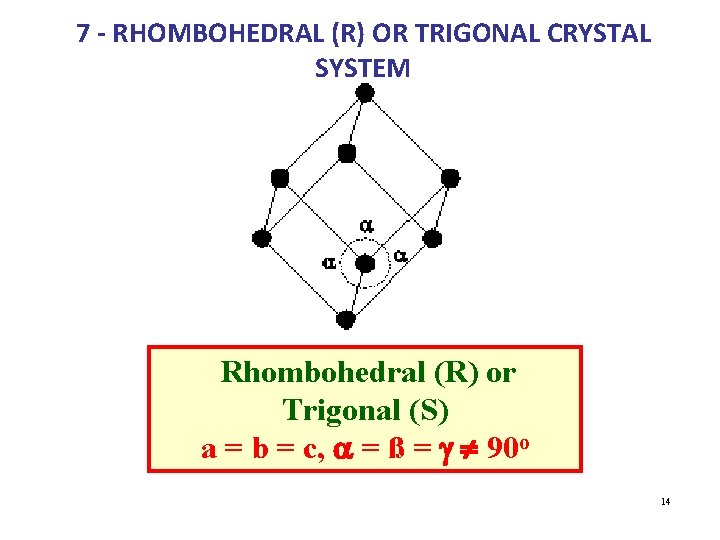
7 - RHOMBOHEDRAL (R) OR TRIGONAL CRYSTAL SYSTEM Rhombohedral (R) or Trigonal (S) a = b = c, a = ß = g ¹ 90 o 14

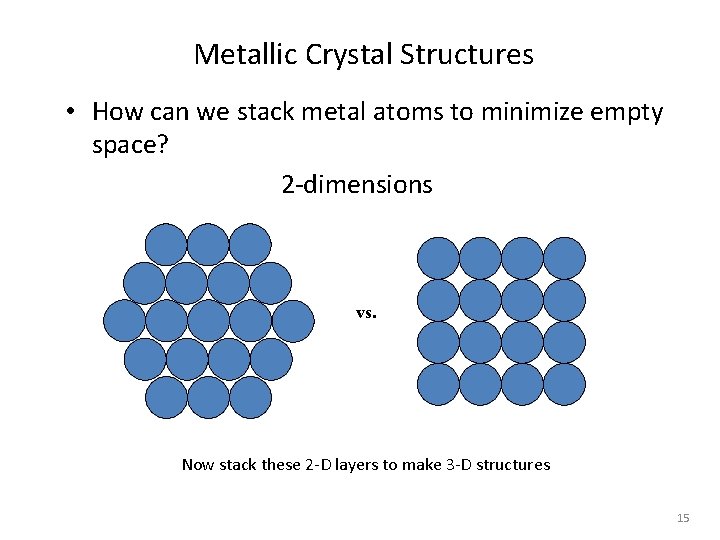
Metallic Crystal Structures • How can we stack metal atoms to minimize empty space? 2 -dimensions vs. Now stack these 2 -D layers to make 3 -D structures 15

Simple Cubic and BCC Structure • Rare due to low packing density (only Po has this structure) • Close-packed directions are cube edges. 16

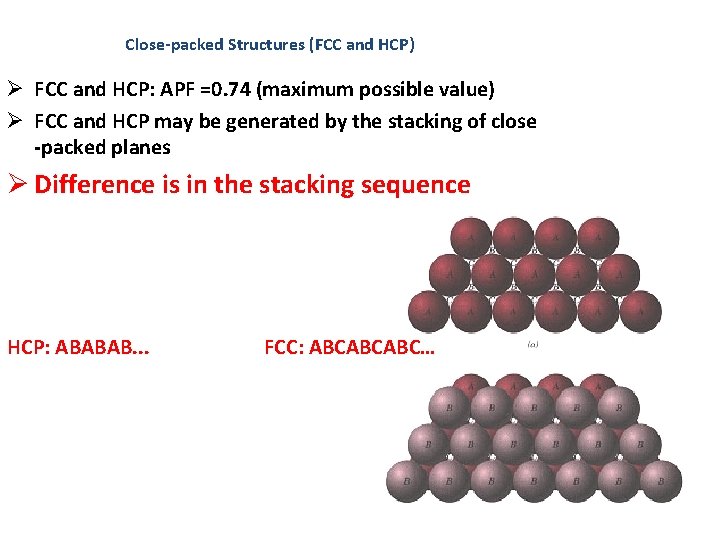
Close-packed Structures (FCC and HCP) Ø FCC and HCP: APF =0. 74 (maximum possible value) Ø FCC and HCP may be generated by the stacking of close -packed planes Ø Difference is in the stacking sequence HCP: ABABAB. . . FCC: ABCABCABC…

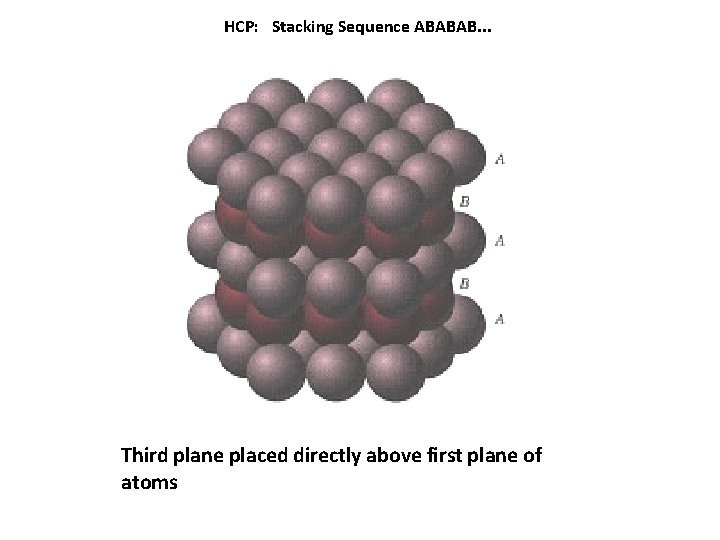
HCP: Stacking Sequence ABABAB. . . Third plane placed directly above first plane of atoms

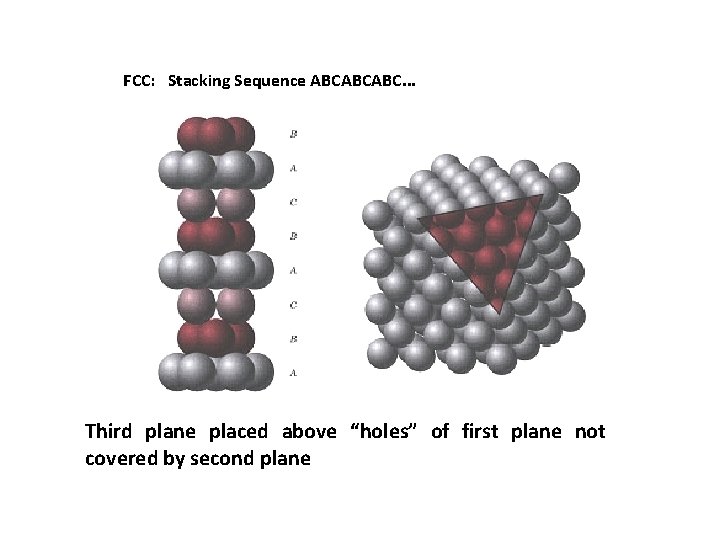
FCC: Stacking Sequence ABCABCABC. . . Third plane placed above “holes” of first plane not covered by second plane

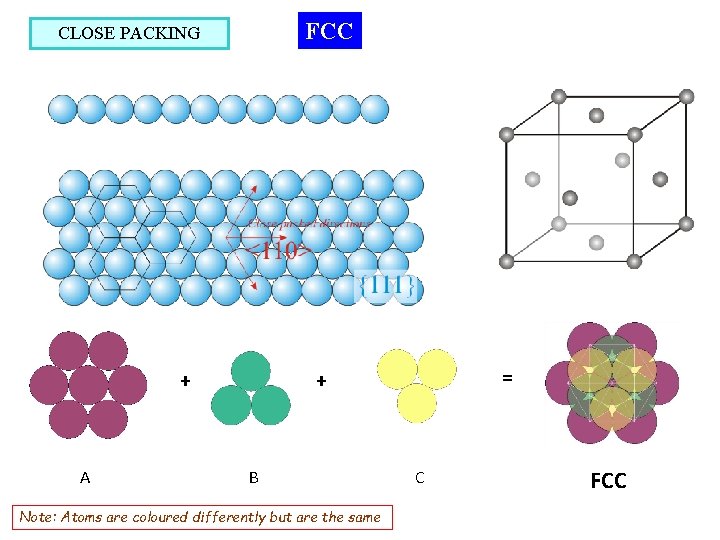
FCC CLOSE PACKING A = + + B Note: Atoms are coloured differently but are the same C FCC

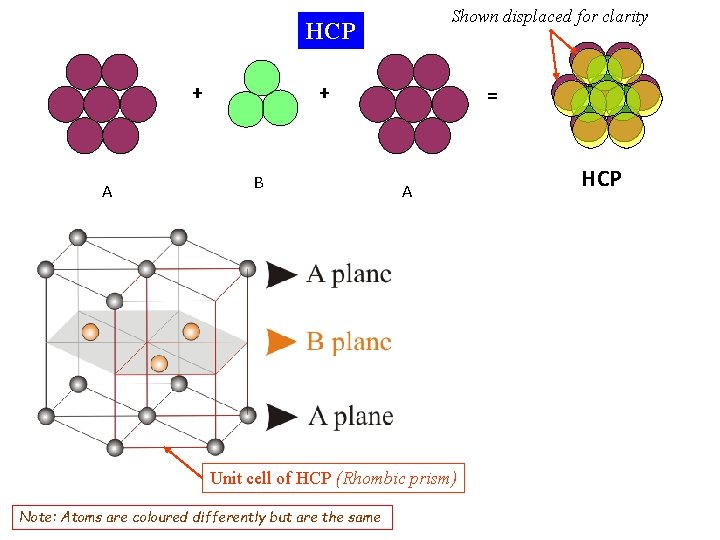
Shown displaced for clarity HCP + A + B = A Unit cell of HCP (Rhombic prism) Note: Atoms are coloured differently but are the same HCP

Coordination Number(CN) for SC:

FACE CENTERED CUBIC STRUCTURE COORDINATION NUMBER In its own plane it touches four face centered atoms. The face centered atoms are its nearest neighbors. In a plane, which lies just above this corner atom, it has four more face centered atoms as nearest neighbors. Similarly, in a plane, which lies just below this corner atom it has yet four more face centered atoms as its nearest neighbors. PH 0101, UNIT 4, LECTURE 5 23

Atomic Packing Factor (or Atomic Packing Fraction) • For a Bravais Lattice, The Atomic Packing Factor (APF) the volume of the atoms within the unit cell divided by the volume of the unit cell. • When calculating the APF, the volume of the atoms in the unit cell is calculated AS IF each atom was a hard sphere, centered on the lattice point & large enough to just touch the nearest-neighbor sphere. • Of course, from Quantum Mechanics, we know that this is very unrealistic for any atom!!

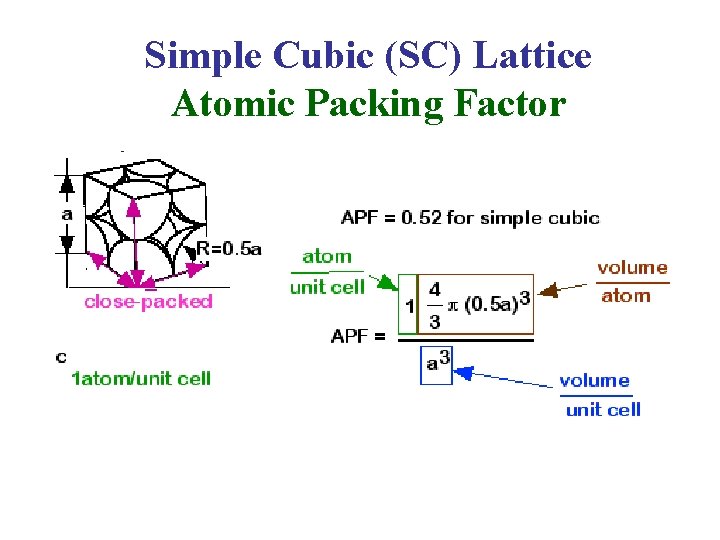
a- The Simple Cubic (SC) Lattice • The SC Lattice has one lattice point in its unit cell, so it’s unit cell is a primitive cell. • In the unit cell shown on the left, the atoms at the corners are cut because only a portion (in this case 1/8) “belongs” to that cell. The rest of the atom “belongs” to neighboring cells. • The Coordination Number of the SC Lattice = 6. b c a

Number of atoms in SC Figure shows how a single atom in corner of Unit Cell of SC is being shared by other Unit cells

Total Number of atom in SC • • Number of atoms in each corner = 1/8 Total number of corners = 8 Total number of atoms in corner = 1/8*8=1 No other atoms in any part of unit cell

Simple Cubic (SC) Lattice Atomic Packing Factor

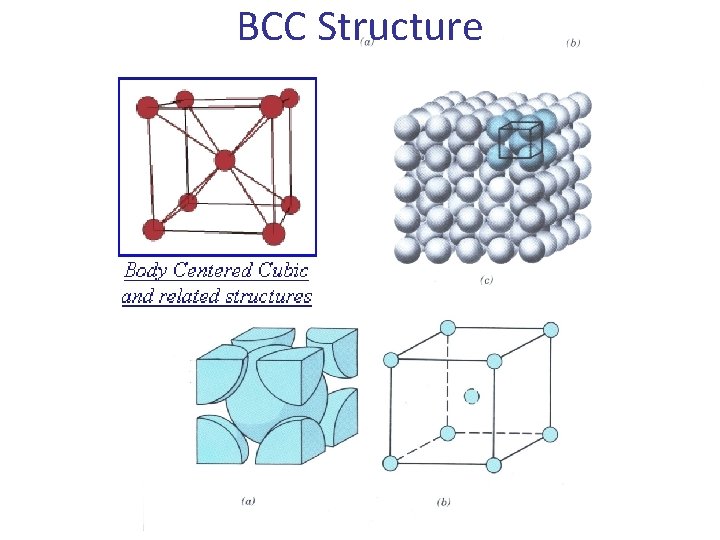
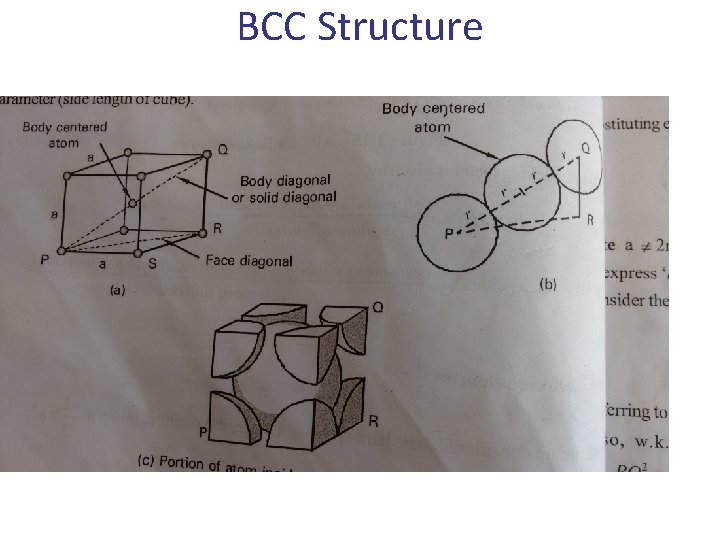
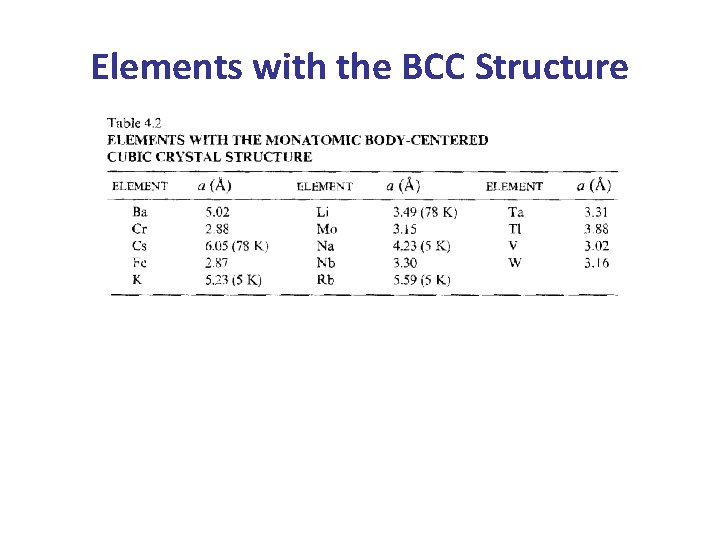
b- The Body Centered Cubic (BCC) Lattice • The BCC Lattice has two lattice points per unit cell so the BCC unit cell is a non-primitive cell. • Every BCC lattice point has 8 nearest- neighbors. So (in the hard sphere model) each atom is in contact with its neighbors only along the body-diagonal directions. • Many metals (Fe, Li, Na. . etc), including the alkalis and several transition elements have the BCC structure. b c a 29

BCC Structure

BCC Structure

Total Number of atoms in BCC • • • Number of atoms in each corner = 1/8 Total number of corners = 8 Total number of atoms in corner = 1/8*8=1 Atoms Inside the Unit cell = 1 Total of atoms = 1+1 = 2

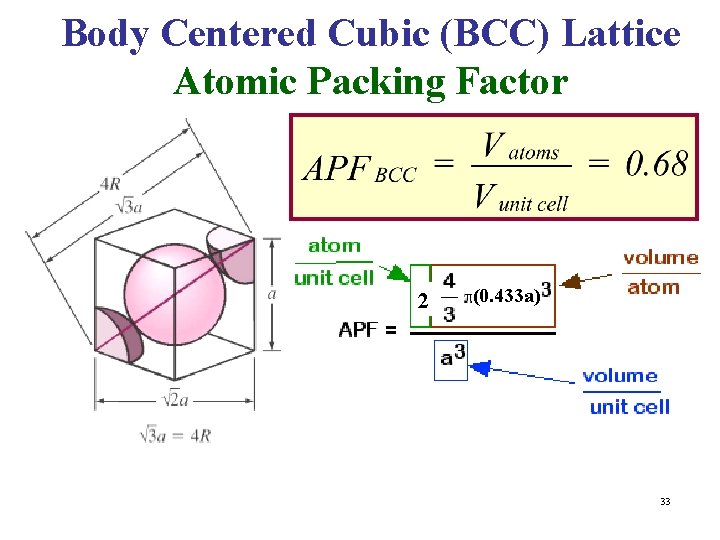
Body Centered Cubic (BCC) Lattice Atomic Packing Factor 2 (0. 433 a) 33

Elements with the BCC Structure

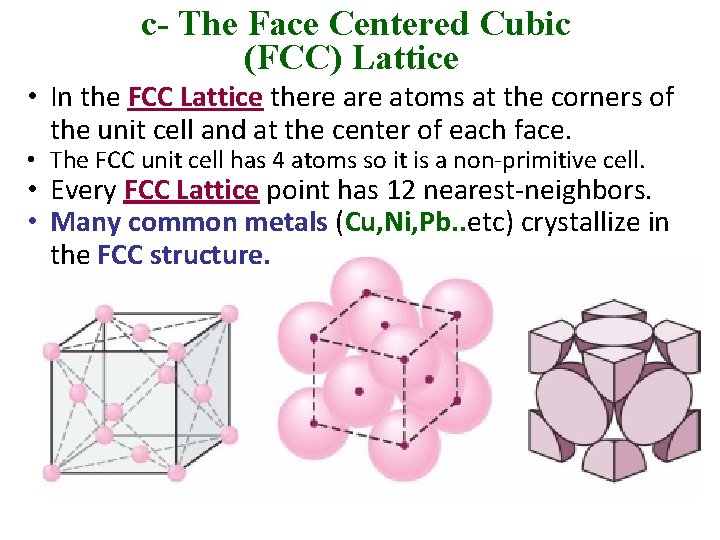
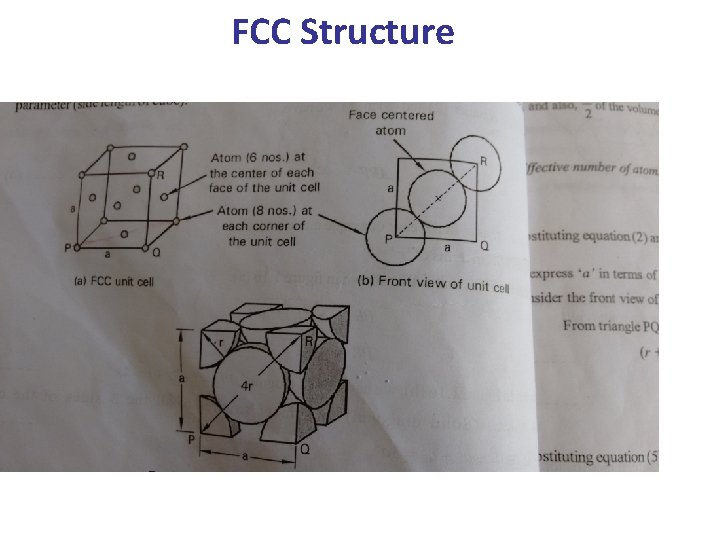
c- The Face Centered Cubic (FCC) Lattice • In the FCC Lattice there atoms at the corners of the unit cell and at the center of each face. • The FCC unit cell has 4 atoms so it is a non-primitive cell. • Every FCC Lattice point has 12 nearest-neighbors. • Many common metals (Cu, Ni, Pb. . etc) crystallize in the FCC structure.

FCC Structure

FCC Structure

Total Number of Atoms in FCC • • In Coners, Each corner is having 1/8 atom Total number of corners = 8 Total number of atoms in corner = 1/8*8=1 The face of hexagonal is having 1/2 of atom Total number of face = 6 Total number of atoms in face = 1/2*6= 3 Total = 1+3= 4

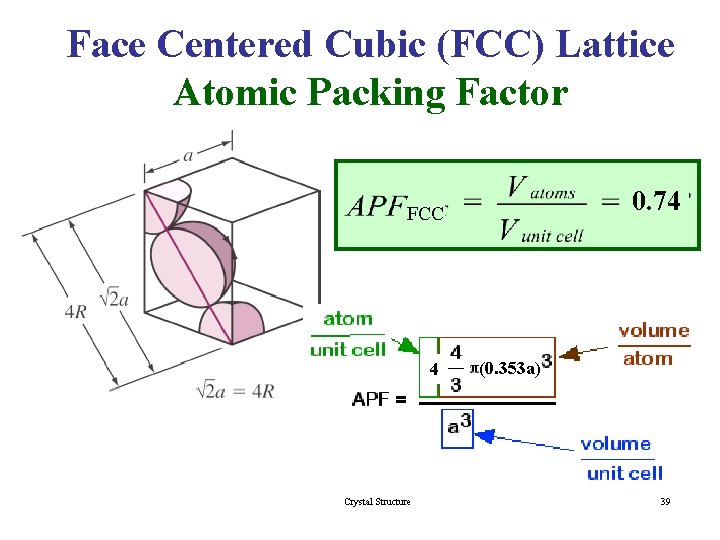
Face Centered Cubic (FCC) Lattice Atomic Packing Factor 0. 74 FCC 4 Crystal Structure (0. 353 a) 39

Elements That Have the FCC Structure

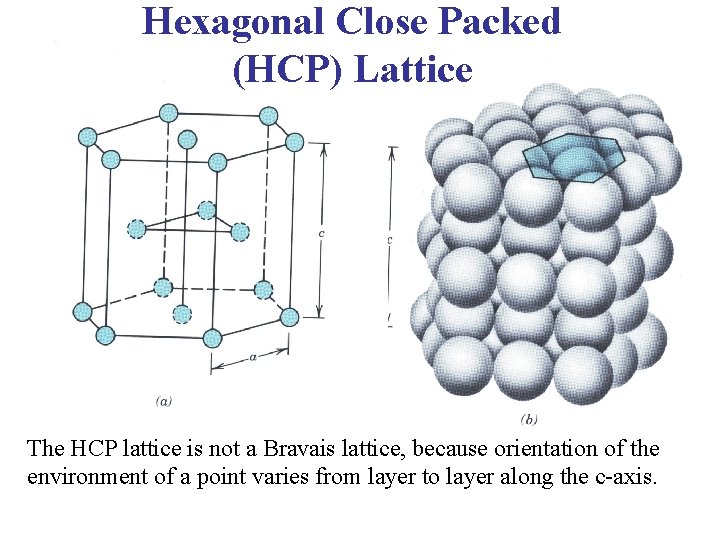
Hexagonal Close Packed (HCP) Lattice • This is another structure that is common, particularly in metals. In addition to the two layers of atoms which form the base and the upper face of the hexagon, there is also an intervening layer of atoms arranged such that each of these atoms rest over a depression between three atoms in the base.

Hexagonal Close Packed (HCP) Lattice The HCP lattice is not a Bravais lattice, because orientation of the environment of a point varies from layer to layer along the c-axis.

Hexagonal Close Packed (HCP) Lattice

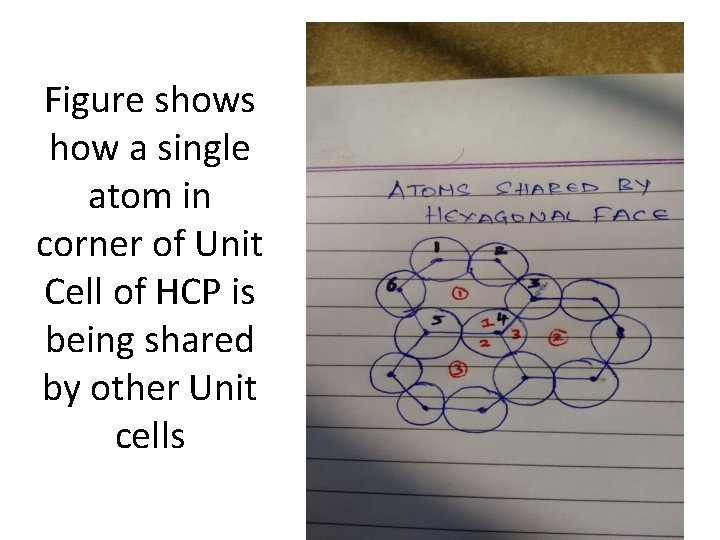
Figure shows how a single atom in corner of Unit Cell of HCP is being shared by other Unit cells

Total Number of Atoms in HCP • • In Coners, Each corner is having 1/6 atom Total number of corners = 12 Total number of atoms in corner = 1/6*12=2 The face of hexagonal is having 1/2 of atom Total number of face = 2 Total number of atoms in face = 1/2*2= 1 Atoms Inside the Unit cell = 3 Total = 2+1+3 = 6

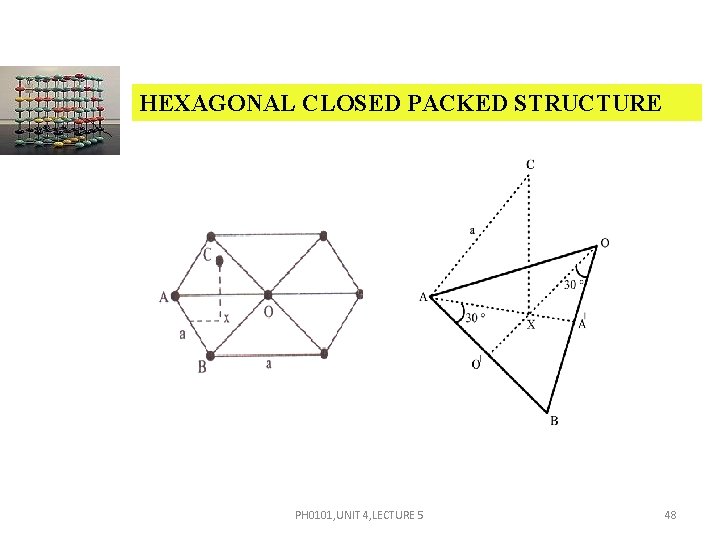
HEXAGONAL CLOSED PACKED STRUCTURE a a PH 0101, UNIT 4, LECTURE 5 46

HEXAGONAL CLOSED PACKED STRUCTURE ATOMIC PACKING FACTOR (APF) APF = v = 6 4/3 r 3 Substitute r = , v = 6 4/3 v = a 3 PH 0101, UNIT 4, LECTURE 5 47

HEXAGONAL CLOSED PACKED STRUCTURE PH 0101, UNIT 4, LECTURE 5 48

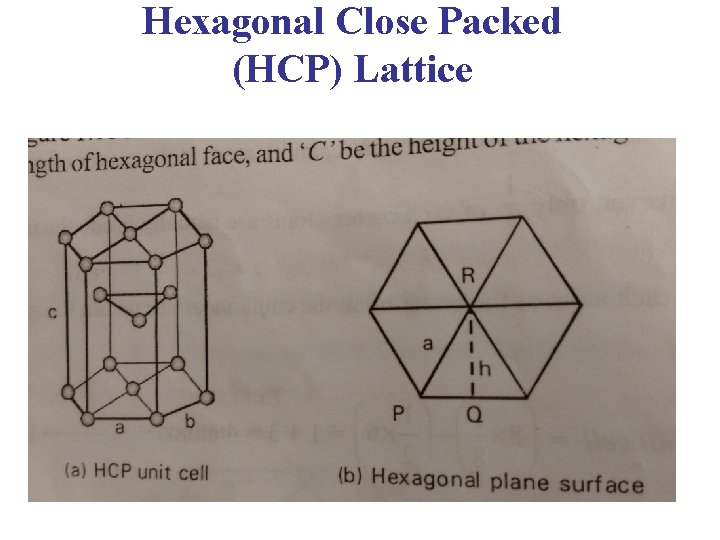
HEXAGONAL CLOSED PACKED STRUCTURE AB = AC = BO = ‘a’. CX = where c height of the hcp unit cell. Area of the base = 6 area of the triangle – ABO = 6 1/2 AB OO Area of the base = 6 1/2 a OO In triangle OBO PH 0101, UNIT 4, LECTURE 5 49

HEXAGONAL CLOSED PACKED STRUCTURE cos 30º = OO = a cos 30º = a Now, substituting the value of OO , Area of the base = 6 a a = V = Area of the base × height PH 0101, UNIT 4, LECTURE 5 50

HEXAGONAL CLOSED PACKED STRUCTURE V= c APF = = APF = PH 0101, UNIT 4, LECTURE 5 51

CALCULATION OF c/a RATIO In the triangle ABA , Cos 30º = AA = AB cos 30º = a But AX = AA = i. e. AX = PH 0101, UNIT 4, LECTURE 5 52

CALCULATION OF c/a RATIO In the triangle AXC, AC 2 = AX 2 + CX 2 Substituting the values of AC, AX and CX, a 2 = PH 0101, UNIT 4, LECTURE 5 53

CALCULATION OF c/a RATIO Now substituting the value of to calculate APF of an hcp unit cell, PH 0101, UNIT 4, LECTURE 5 54

HEXAGONAL CLOSED PACKED STRUCTURE APF = = APF = Packing Fraction =74% PH 0101, UNIT 4, LECTURE 5 55

Imperfections in Solids • The properties of some materials are profoundly influenced by the presence of imperfections. • It is important to have knowledge about the types of imperfections that exist and the roles they play in affecting the behavior of materials. • If volume imperfection is 0. 01%, while calculating properties like density, imperfection wont affect. • But impurity of Al is only few parts per million in silicon semiconductor destroy the rectifying and transisting action. 56

Atom Purity and Crystal Perfection • If we assume a perfect crystal structure containing pure elements, then anything that deviated from this concept or intruded in this uniform homogeneity would be an imperfection. 1. There are no perfect crystals. 2. Many material properties are improved by the presence of imperfections and deliberately modified (alloying and doping). 57

Types of Imperfections • Vacancy atoms • Interstitial atoms • Substitutional atoms Point defects • Dislocations Line defects • Grain Boundaries Area defects 1 -2 atoms Zero dimensional 1 -dimensional 2 -dimensional 58

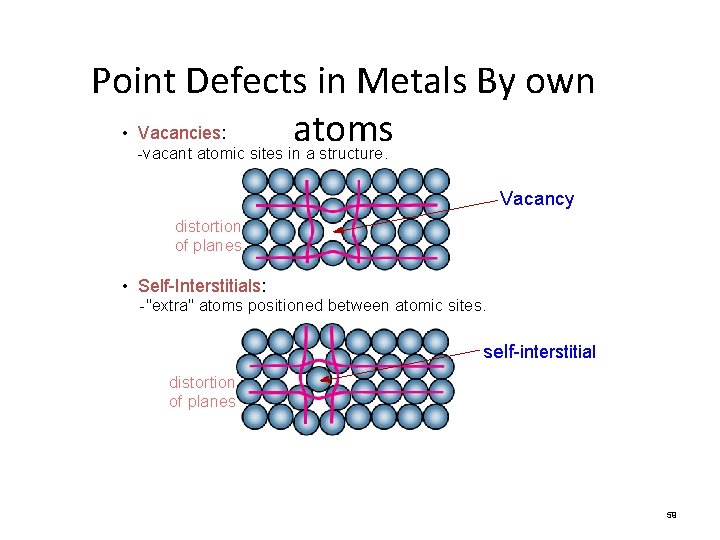
Point Defects in Metals By own • Vacancies: atoms -vacant atomic sites in a structure. Vacancy distortion of planes • Self-Interstitials: -"extra" atoms positioned between atomic sites. self-interstitial distortion of planes 59

Point Defects in Metals By different atoms Solid Solution • The addition of impurity atoms to a metal results in the formation of a solid solution. • The solvent represents the element that is present in the greatest amount (the host atoms). For example, in Lab 8 (MSE 227) Precipitation Hardening of Aluminum , aluminum is the solvent and copper is the solute (present in minor concentration ). • Solid solutions form when the solute atoms (Cu) are added to the solvent (Al), assuming the crystal structure is maintained and no new structures are 60

Solid Solution - continued • A solid solution is a homogenous composition throughout. • The impurity atoms (Cu) are randomly and uniformly dispersed within the solid. • The impurity defects in the solid solution are either substitutional or 61

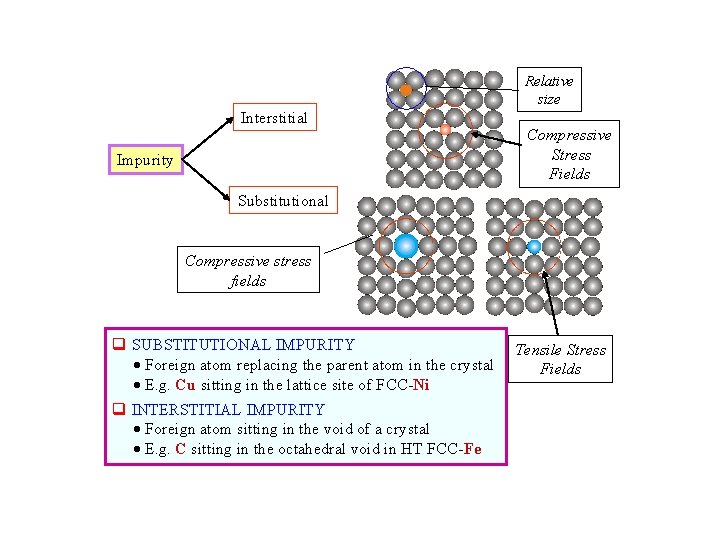
Relative size Interstitial Impurity Compressive Stress Fields Substitutional Compressive stress fields q SUBSTITUTIONAL IMPURITY Foreign atom replacing the parent atom in the crystal E. g. Cu sitting in the lattice site of FCC-Ni q INTERSTITIAL IMPURITY Foreign atom sitting in the void of a crystal E. g. C sitting in the octahedral void in HT FCC-Fe Tensile Stress Fields

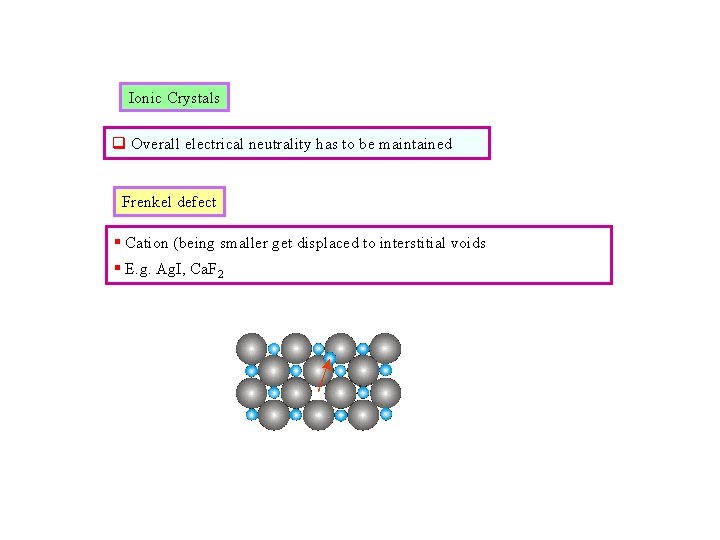
Ionic Crystals q Overall electrical neutrality has to be maintained Frenkel defect § Cation (being smaller get displaced to interstitial voids § E. g. Ag. I, Ca. F 2

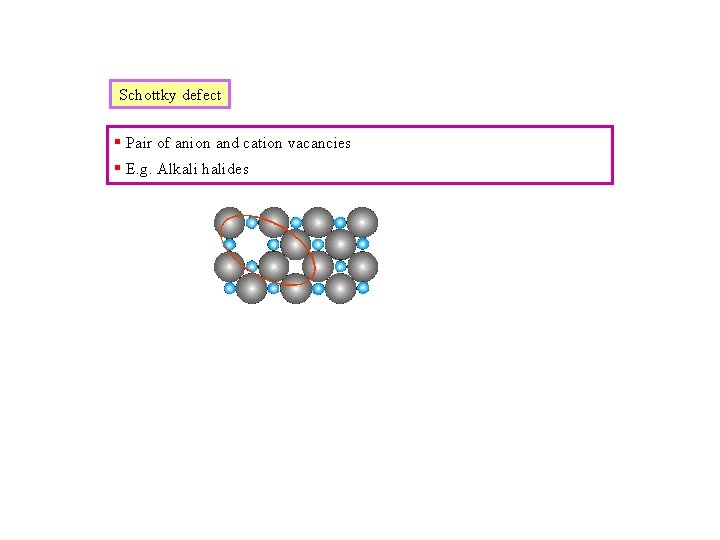
Schottky defect § Pair of anion and cation vacancies § E. g. Alkali halides

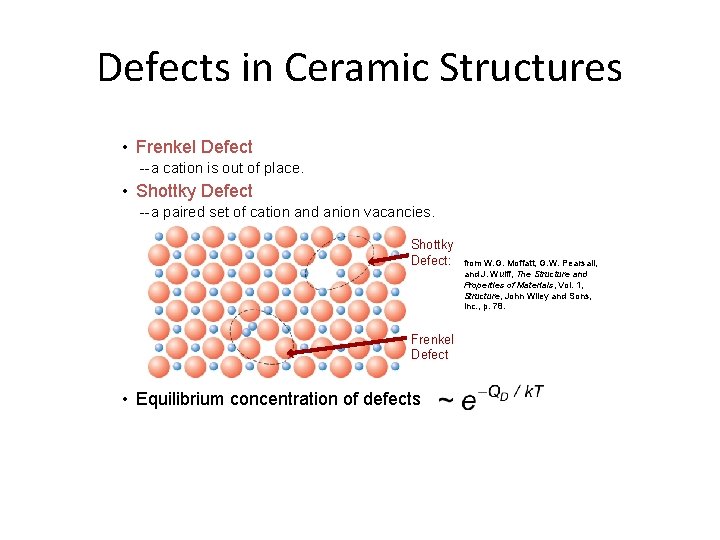
Defects in Ceramic Structures • Frenkel Defect --a cation is out of place. • Shottky Defect --a paired set of cation and anion vacancies. Shottky Defect: Frenkel Defect • Equilibrium concentration of defects from W. G. Moffatt, G. W. Pearsall, and J. Wulff, The Structure and Properties of Materials, Vol. 1, Structure, John Wiley and Sons, Inc. , p. 78.

Line Defects Are called Dislocations: And: • slip between crystal planes result when dislocations move, • this motion produces permanent (plastic) deformation. Schematic of Zinc (HCP): • before deformation • after tensile elongation Adapted from Fig. 7. 8, Callister 7 e. slip steps which are the physical evidence of large numbers of dislocations slipping along the close packed plane {0001}

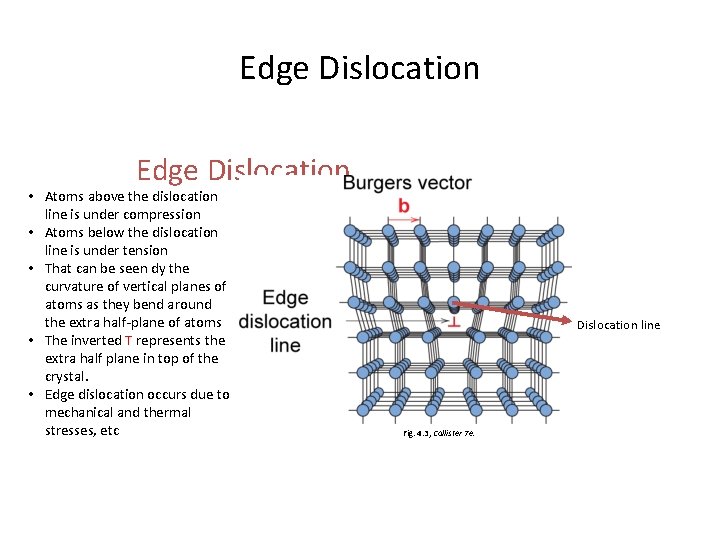
Linear Defects (Dislocations) – Are one-dimensional defects around which atoms are misaligned • Edge dislocation: – extra half-plane of atoms inserted in a crystal structure – b (the berger’s vector) is (perpendicular) to dislocation line • Screw dislocation: – spiral planar ramp resulting from shear deformation – b is (parallel) to dislocation line Burger’s vector, b: is a measure of lattice distortion and is measured as a distance along the close packed directions in the lattice

Edge Dislocation • Atoms above the dislocation line is under compression • Atoms below the dislocation line is under tension • That can be seen dy the curvature of vertical planes of atoms as they bend around the extra half-plane of atoms • The inverted T represents the extra half plane in top of the crystal. • Edge dislocation occurs due to mechanical and thermal stresses, etc Dislocation line Fig. 4. 3, Callister 7 e.

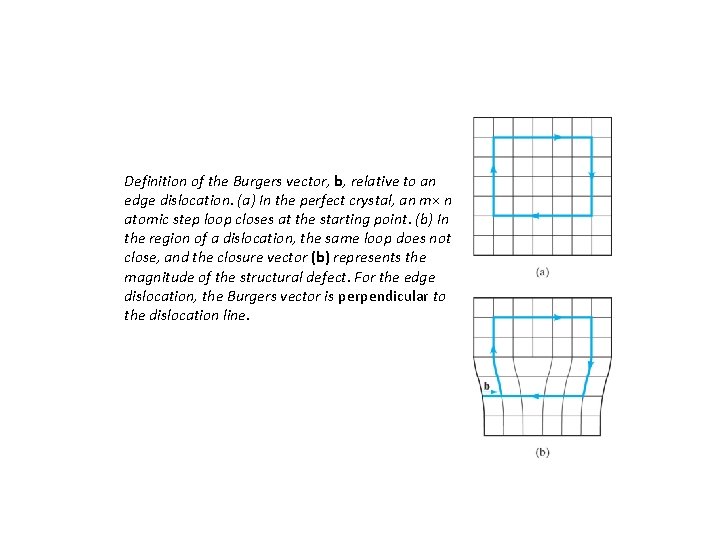
Definition of the Burgers vector, b, relative to an edge dislocation. (a) In the perfect crystal, an m× n atomic step loop closes at the starting point. (b) In the region of a dislocation, the same loop does not close, and the closure vector (b) represents the magnitude of the structural defect. For the edge dislocation, the Burgers vector is perpendicular to the dislocation line.

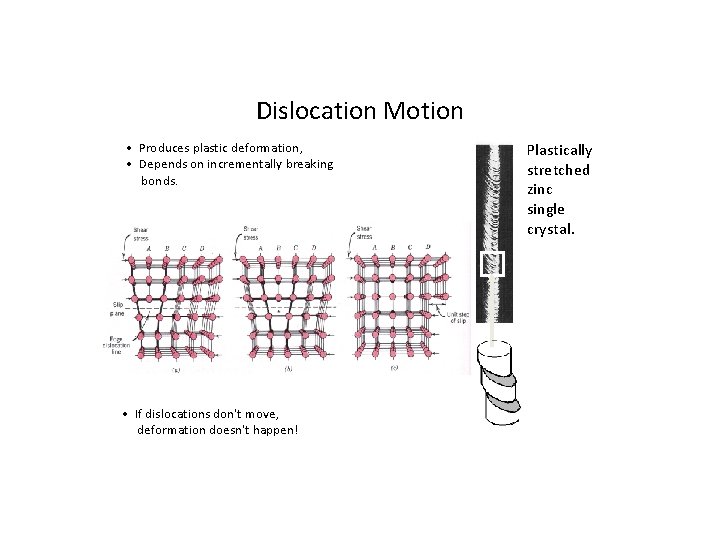
Dislocation Motion • Produces plastic deformation, • Depends on incrementally breaking bonds. • If dislocations don't move, deformation doesn't happen! Plastically stretched zinc single crystal.

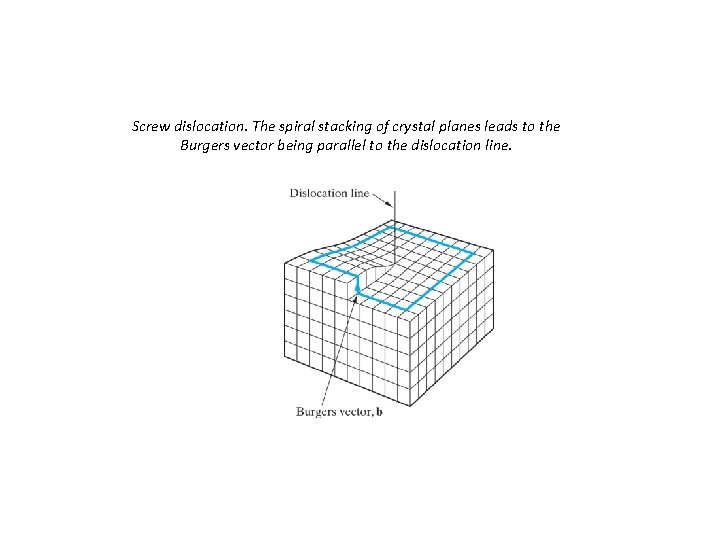
Screw dislocation: – Named for the spiral stacking of crystal planes around the dislocation line; results from shear deformation – b parallel ( ) to dislocation line 71

Screw dislocation. The spiral stacking of crystal planes leads to the Burgers vector being parallel to the dislocation line.

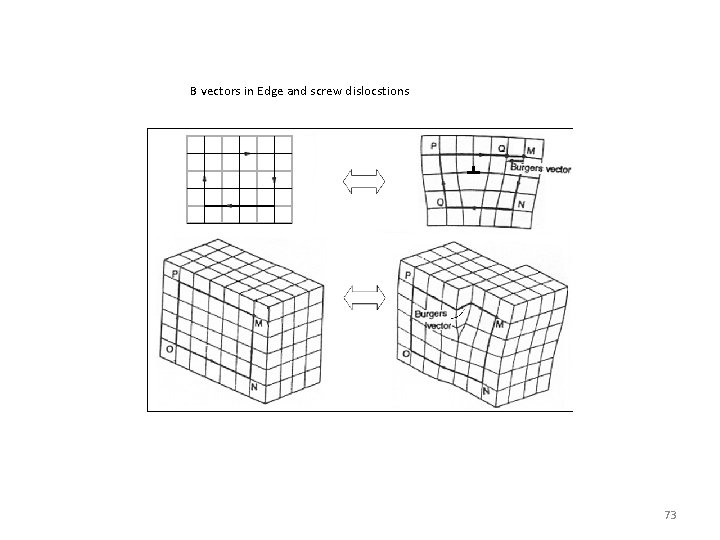
B vectors in Edge and screw dislocstions 73

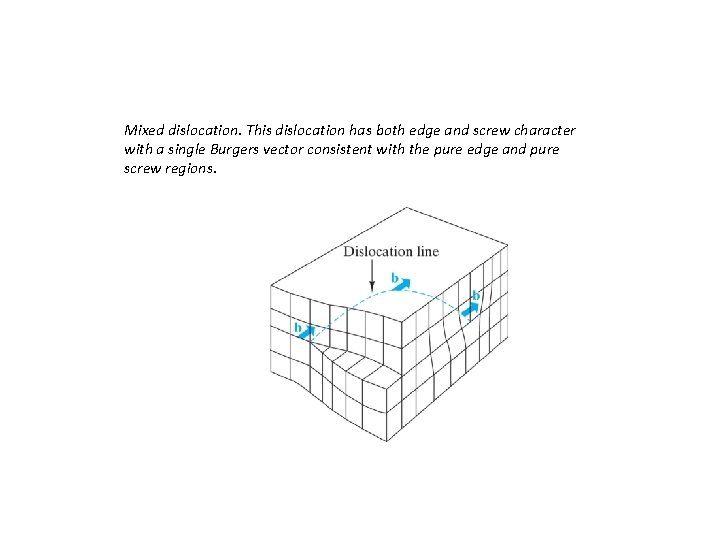
Mixed dislocation. This dislocation has both edge and screw character with a single Burgers vector consistent with the pure edge and pure screw regions.

Edge, Screw, and Mixed Dislocations Mixed Edge Adapted from Fig. 5. 10, Callister & Rethwisch 3 e. Screw 75

Imperfections in Solids Dislocations are visible in electron micrographs; TEM Titanium alloy, dark lines are dislocations 51, 450 x Fig. 5. 11, Callister & Rethwisch 3 e. 76

Polycrystalline Materials Grain Boundaries § § regions between crystals transition from lattice of one region to another (a) The atoms near the boundaries of the 3 grains do not have an equilibrium spacing or arrangement; slightly disordered. (b) Grains and grain boundaries in a stainless steel sample. low density in grain boundaries 77

Typical optical micrograph of a grain structure, 100×. The material is a low-carbon steel. The grain boundaries have been lightly etched with a chemical solution so that they reflect light differently from the polished grains, thereby giving a distinctive contrast.

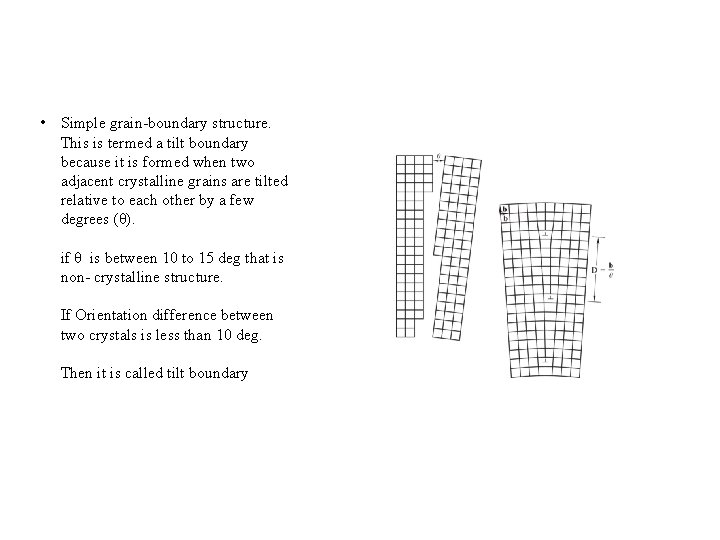
• Simple grain-boundary structure. This is termed a tilt boundary because it is formed when two adjacent crystalline grains are tilted relative to each other by a few degrees (θ). if θ is between 10 to 15 deg that is non- crystalline structure. If Orientation difference between two crystals is less than 10 deg. Then it is called tilt boundary

Planar Defects in Solids - Twinning • • • A shear force that causes atomic displacements such that the atoms on one side of a plane (twin boundary) mirror the atoms on the other side. A reflection of atom positions across the twin plane. Displacement magnitude in the twin region is proportional to the atom’s distance from the twin plane. Takes place along defined planes and directions depending upon the system. • Ex: BCC twinning occurs on the (112)[111] system

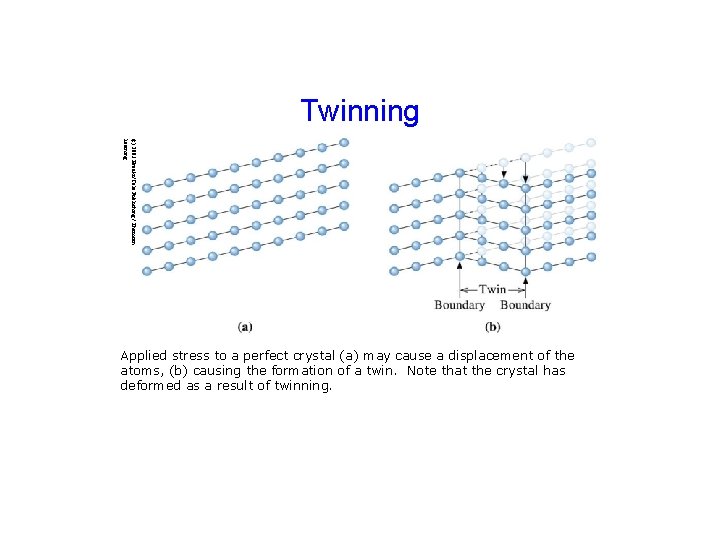
Twinning (c) 2003 Brooks/Cole Publishing / Thomson Learning Applied stress to a perfect crystal (a) may cause a displacement of the atoms, (b) causing the formation of a twin. Note that the crystal has deformed as a result of twinning.

Properties of Twinning § Of the three common crystal structures BCC, FCC and HCP, the HCP structure is the most likely to twin. § FCC structures will not usually twin because slip is more energetically favorable. § Twinning occurs at low temperatures and high rates of shear loading (shock loading) conditions where there are few present slip systems (restricting the possibility of slip) §Small amount of deformation when compared with slip.

• Stacking faults – For FCC metals an error in ABCABC packing sequence – Ex: ABCAB—ABC – C is missing in the stacking order – For HCP metals an error in ABAB packing sequence – Ex: ABCABAB – Extra layer called C is present in the stacking order.

(c) 2003 Brooks/Cole Publishing / Thomson Learning A micrograph of twins within a grain of brass (x 250).