Unit D Electron Configurations and Chemical Bonding Electron

Unit D: Electron Configurations and Chemical Bonding -Electron Configurations -Valence Electrons -Lewis Dot Structures -Ionic & Covalent Compounds -Naming Chemical Formulas

Quantum Mechanics • In the early 1900’s, a relatively unknown Jewish scientist named Albert Einstein published a paper on the behavior of light • He argued that light was both a particle (photon) and a wave, and that light was released in small packets, called quanta • This led to the emergence of a new branch of Chemistry/Physics, called Quantum Mechanics

Quantum Mechanics • Is concerned with the behavior of extremely small particles, such as electrons • Relies heavily on mathematics and probability to explain how atoms work

Quantum Uncertainty • A German scientist named Werner Heisenberg came up with a new, more accurate view of the atom that was based upon probabilities of where electrons could be rather than actual locations • He argued that, because electrons were so small, it was impossible to know with certainty both the location and the momentum of a particle, and that observing something inevitably changes it

Heisenberg Uncertainty Principle 1. It is impossible to know both the location AND the momentum of a subatomic particle 2. Observing something changes it

Schrödinger's Cat • An Austrian Scientist named Erwin Schrödinger devised a thought experiment to poke fun of the Heisenberg theorem in which a cat was placed in a giant box with a poison bomb with a 50/50 chance of detonating • According to the Uncertainty Principle, the cat was both alive and dead • This became known as Schrödinger's Cat


The Quantum Mechanical Model of the Atom • Rather than 2 d rings as in the Bohr model, electron orbitals are represented by 3 D probability clouds • Electrons move very quickly and behave like waves instead of particles

Electrons are everywhere…and nowhere

Arrangement of Electrons in Atoms Electrons in atoms are arranged as Energy LEVELS/SHELLS (n) SUBSHELLS (l) ORBITALS (ml)

Energy Levels (n) • Represent the distance of electrons from the nucleus • Require a specific amount of energy to be on (quantum number) • Lower energy levels require less energy; this is why they fill first
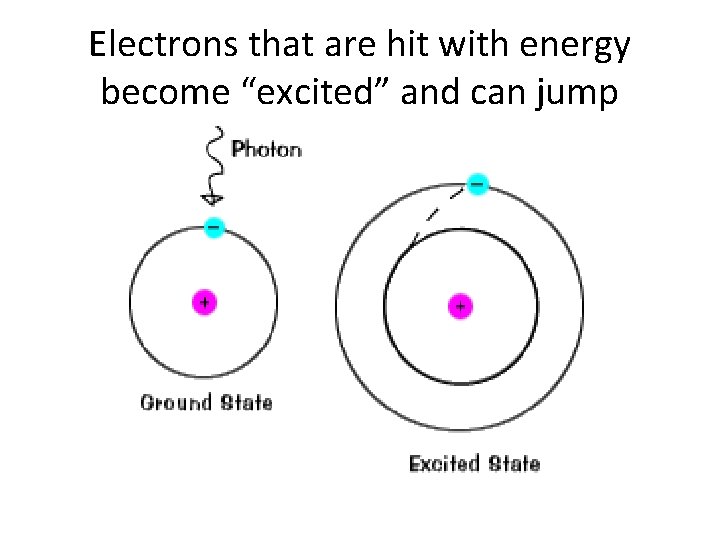
Electrons that are hit with energy become “excited” and can jump
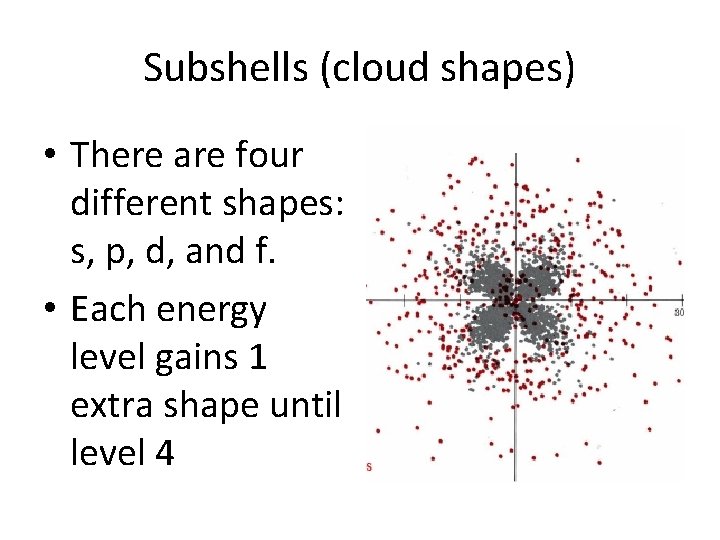
Subshells (cloud shapes) • There are four different shapes: s, p, d, and f. • Each energy level gains 1 extra shape until level 4
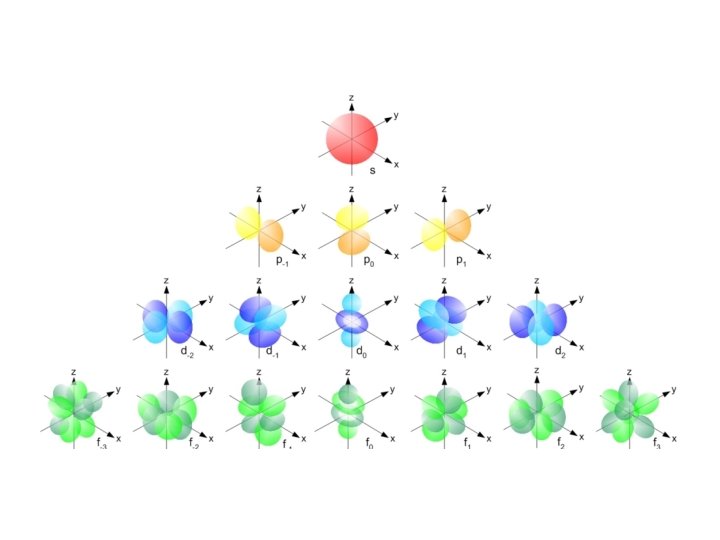
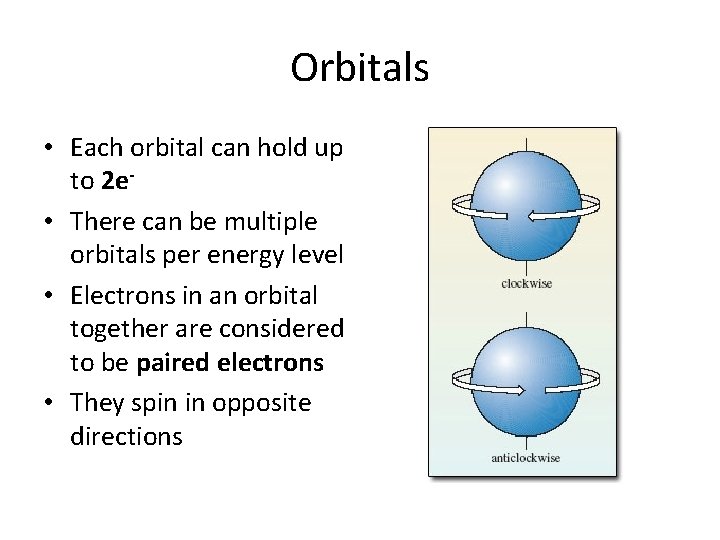
Orbitals • Each orbital can hold up to 2 e • There can be multiple orbitals per energy level • Electrons in an orbital together are considered to be paired electrons • They spin in opposite directions
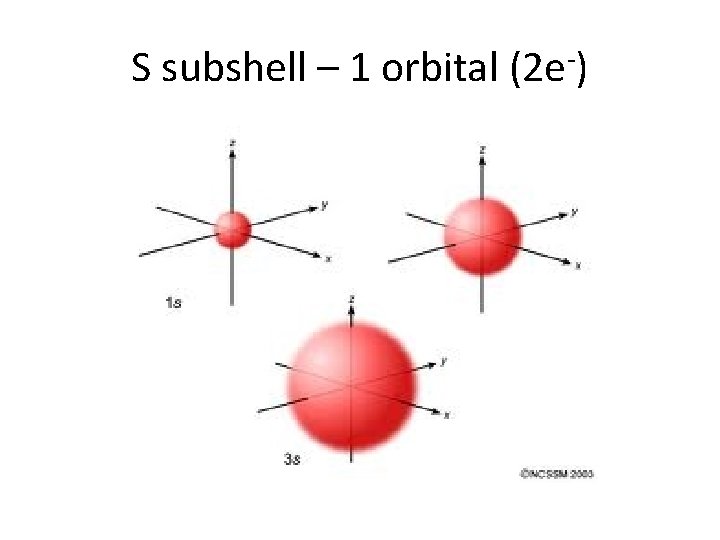
S subshell – 1 orbital (2 e-)
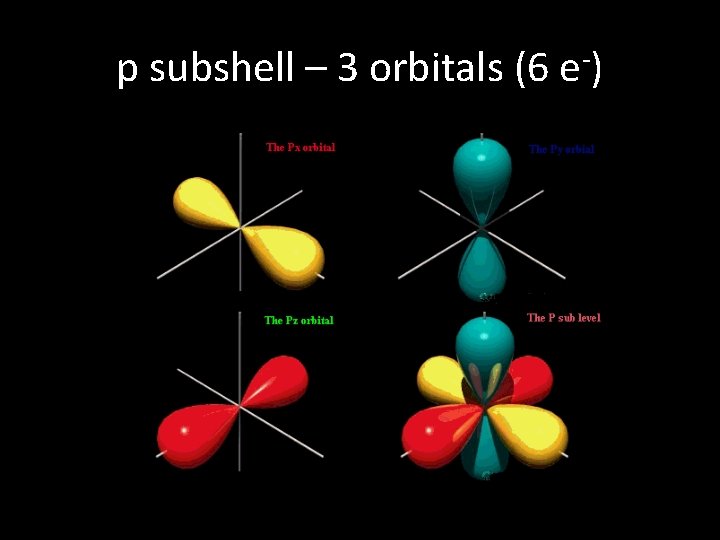
p subshell – 3 orbitals (6 e-)
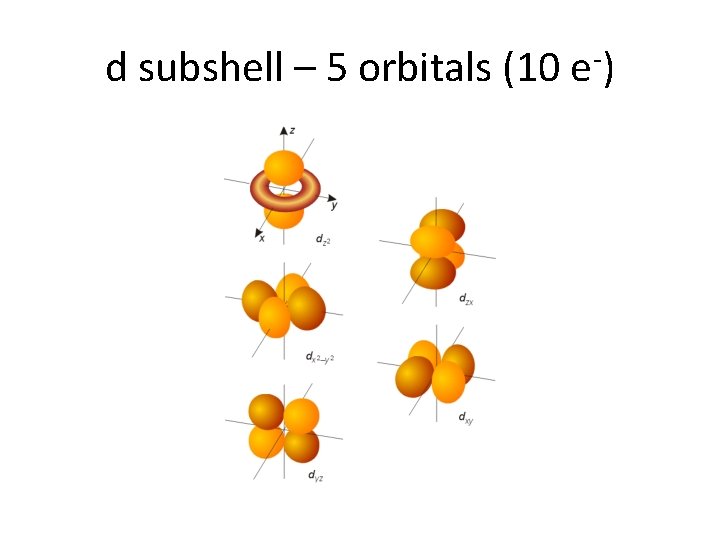
d subshell – 5 orbitals (10 e-)
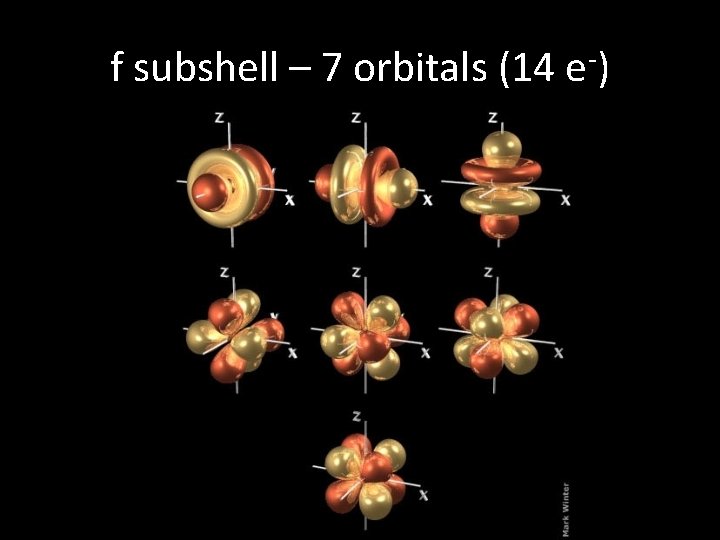
f subshell – 7 orbitals (14 e-)
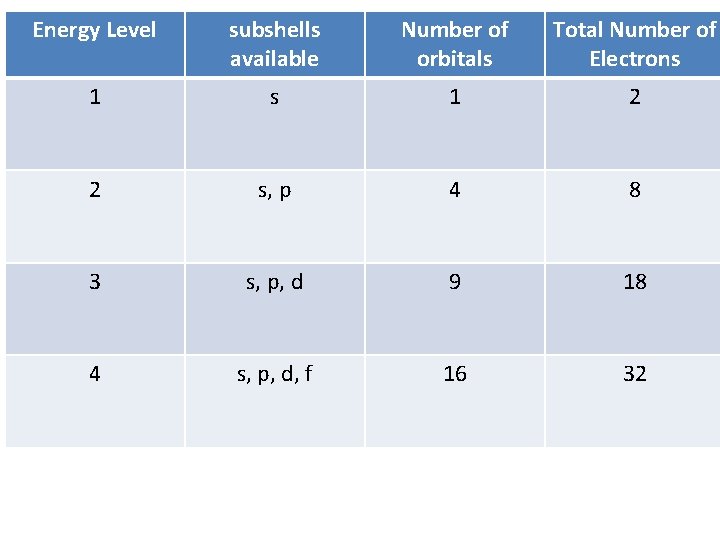
Energy Level subshells available Number of orbitals Total Number of Electrons 1 2 2 s, p 4 8 3 s, p, d 9 18 4 s, p, d, f 16 32

In Summary • Energy Levels n (shells) refer to WHERE the electrons are (distance from nucleus) • Subshells l (spdf) refer to the shape of their waves (clouds) • Orbitals m are where the electrons are located and refer to their spin (up, down) • All of these are quantum numbers
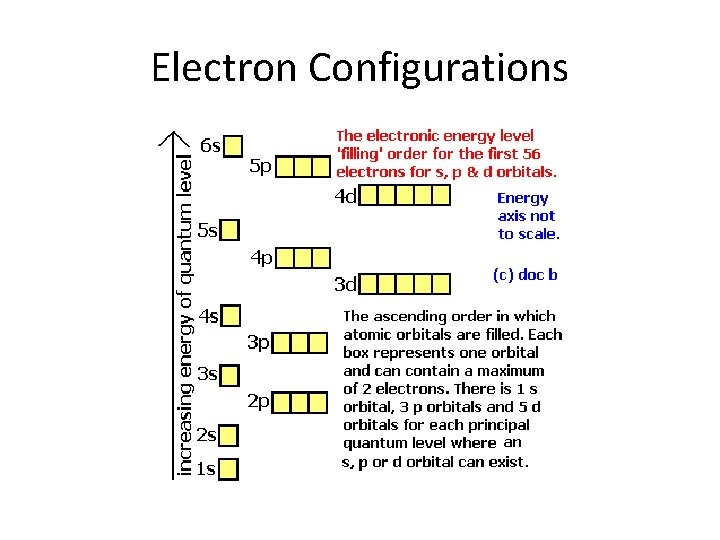
Electron Configurations
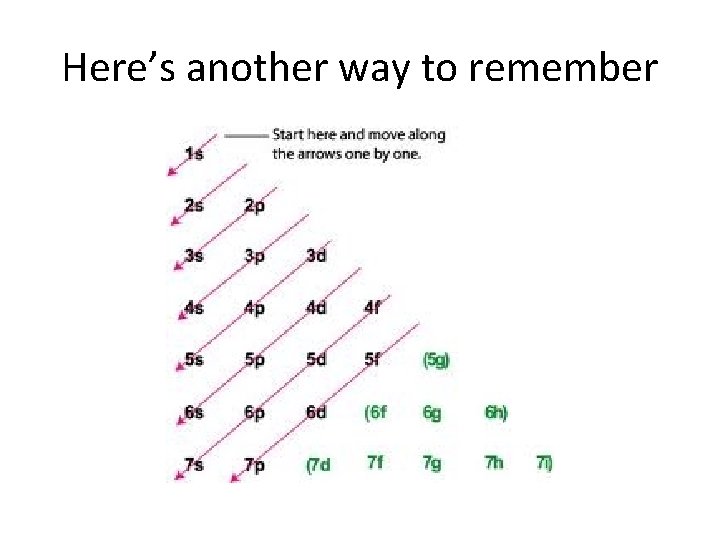
Here’s another way to remember

Rules for electron filling • Pauli’s Exclusion Principle • Aufbau Principle • Hund’s Rule

Pauli’s Exclusion Principle • A maximum of two electrons can fit on a single orbital, and they must have opposite spins

Aufbau Principle • Electrons fill orbitals from the bottom up (1 s before 2 s, 2 s before 2 p)

Hund’s Rule • Electrons occupy empty orbitals in a subshell before doubling up (strangers on a bus)
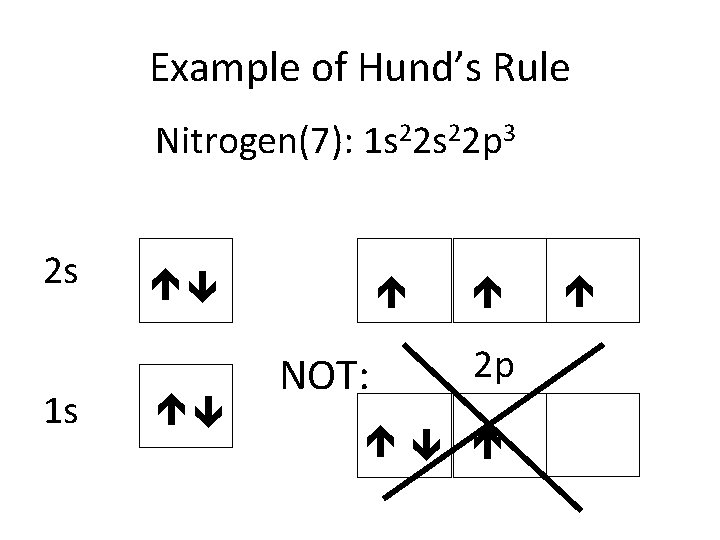
Example of Hund’s Rule Nitrogen(7): 1 s 22 p 3 2 s 1 s NOT: 2 p
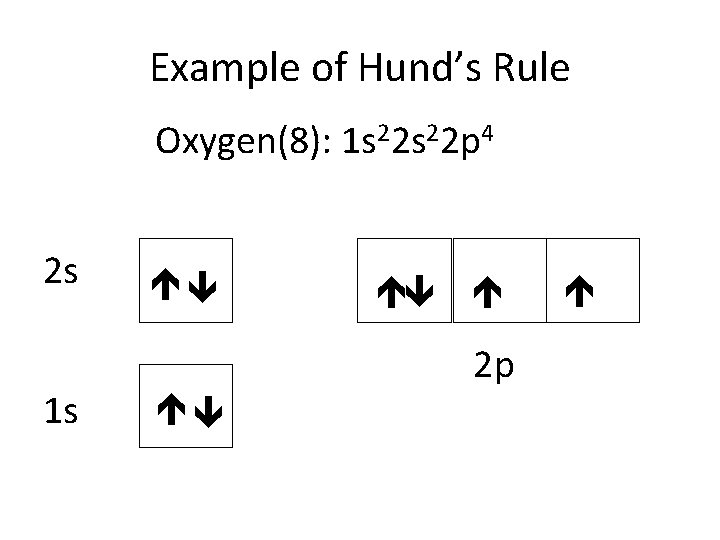
Example of Hund’s Rule Oxygen(8): 1 s 22 p 4 2 s 1 s 2 p
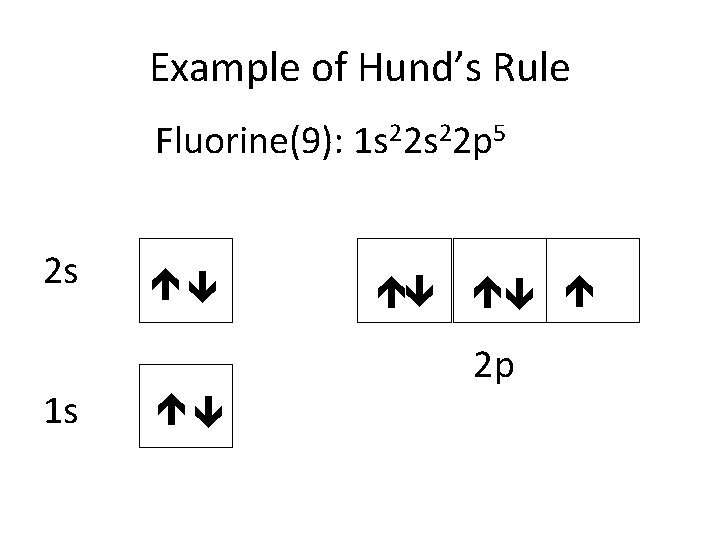
Example of Hund’s Rule Fluorine(9): 1 s 22 p 5 2 s 1 s 2 p
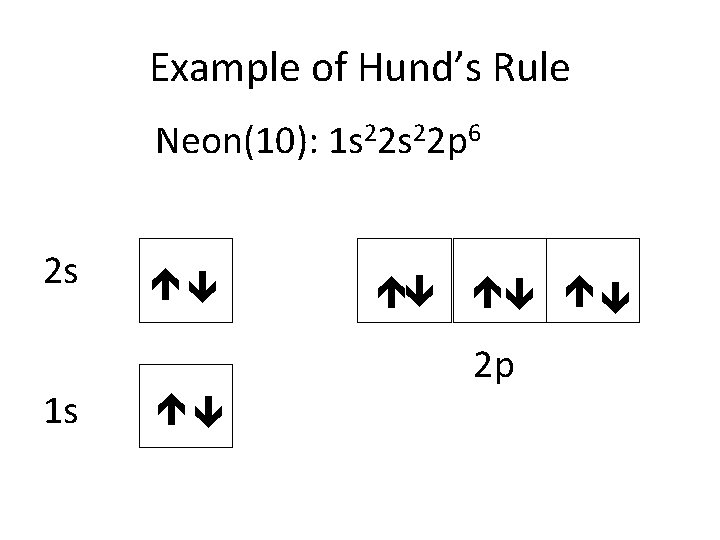
Example of Hund’s Rule Neon(10): 1 s 22 p 6 2 s 1 s 2 p

Examples • • Hydrogen (atomic # = 1), would be 1 s 1 Helium (atomic # = 2), would be 1 s 2 Lithium (atomic # = 3), would be 1 s 22 s 1 Oxygen (atomic # = 8), would be 1 s 22 p 4

Electron Configuration Practice 1. 2. 3. 4. 5. 6. Magnesium(12): Nitrogen(7): Boron(5): Fluorine(9): Chlorine(17): Argon(18): 22 s 22 p 63 s 2 1 s • ______ 1 s 22 p 3 • ______ 22 s 22 p 1 1 s • ______ 22 s 22 p 5 1 s • ____________ 22 s 22 p 63 s 23 p 6 1 s • ______

Now try these for homework 1. 2. 3. 4. 5. 6. Calcium(20): Sodium(11): Carbon(6): Sulfur(16): Neon(10): Silicon(14): 22 s 22 p 63 s 23 p 64 s 2 1 s • ______ 1 s 22 p 63 s 1 • ______ 22 s 22 p 2 1 s • ______ 22 s 22 p 63 s 23 p 4 1 s • ____________ 22 s 22 p 63 s 23 p 2 1 s • ______
Valence Electrons • Electrons in the outermost shell (highest level) of an atom are called valence electrons • Chemical bonding involves the exchange or sharing of valence electrons

Finding Valence e • Oxygen (8) has an electron configuration of 1 s 22 p 4 2 • Highest energy level: ___ • # of valence electrons: 2 + 4 = 6 • Oxygen has 6 valence electrons
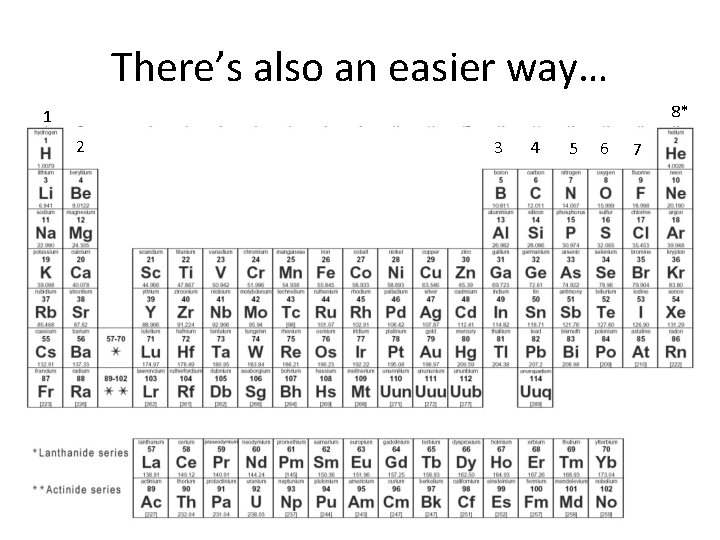
There’s also an easier way… 8* 1 2 3 4 5 6 7
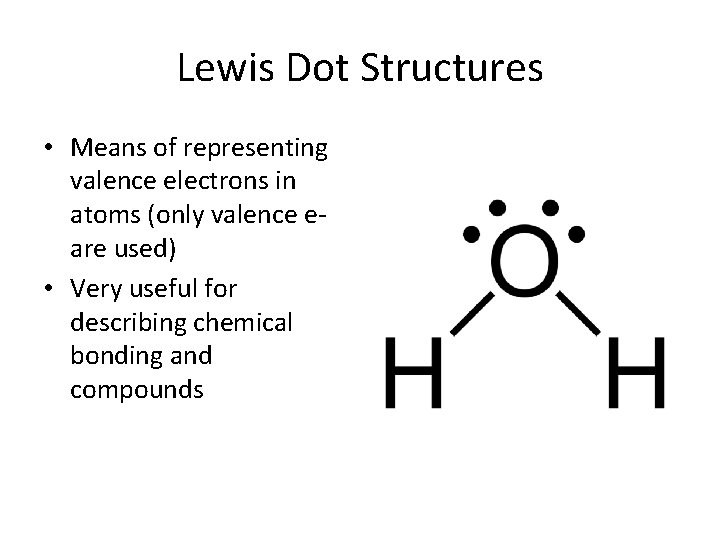
Lewis Dot Structures • Means of representing valence electrons in atoms (only valence eare used) • Very useful for describing chemical bonding and compounds

How to draw Lewis Structures • An element symbol represents each atom • Dots are drawn around the symbol to represent valence electrons (inner electrons are not drawn) • a maximum of 2 dots can occupy 1 side, and each element can fit a maximum of 8* electrons • In general, electrons fill empty spaces before pairing up

Examples Lithium has a configuration of 1 s 22 s 1 It has 1 electron in its highest energy level (2) Li The placement of the lone electron doesn’t matter

Examples Beryllium has a configuration of 1 s 22 s 2 It has 2 electrons in its highest energy level (2) Be

Examples Boron has a configuration of 1 s 22 p 1 It has 3 electrons in its highest energy level (2) B

Examples Carbon has a configuration of 1 s 22 p 2 It has 4 electrons in its highest energy level (2) C
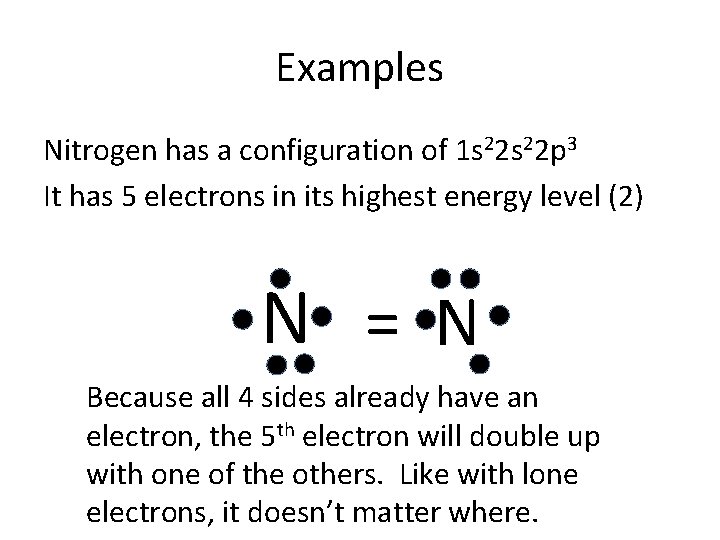
Examples Nitrogen has a configuration of 1 s 22 p 3 It has 5 electrons in its highest energy level (2) N = N Because all 4 sides already have an electron, the 5 th electron will double up with one of the others. Like with lone electrons, it doesn’t matter where.

Part 2 – Chemical Bonding Essential Question: Why does Chemical Bonding Occur?

Why does bonding occur? • All elements are more stable when their outer shells (valence electrons) are full • They can add extra electrons to fill an outer shell that is almost there, or • They can give away extra electrons, making the next level down (which is already full) their “outer shell. ”
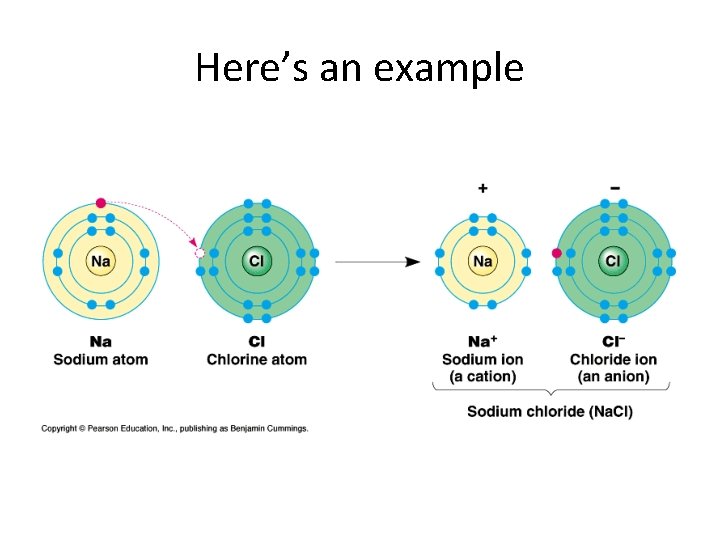
Here’s an example

Here’s what that actually looks like

Ions • An atom that has gained or lost an electron becomes an ion • Cations+ are atoms that have lost electrons and so have a positive charge because they have more protons than electrons • Anions- are atoms that have gained electrons and so have a negative charge because they have more electrons than protons • Charges are represented as superscripts above the element symbol

Examples Na+ = Sodium, +1 charge (11 p+, 10 e-) Cl- = Chlorine, -1 charge (17 p+, 18 e-) Fe 2+ = Iron(II), +2 charge (26 p+, 24 e-) S 2 - = Sulfur, -2 charge (16 p+, 18 e-)

Metals and non-metals • All metals give up their electrons and become cations (+) • All non-metals (can) steal electrons and become anions (noble gases don’t bother) (-)

Trends • All metals lose electrons and become cations • All non-metals (except for Hydrogen) can gain electrons and become anions • In general, atoms only exchange their valence electrons, but there are exceptions (transition metals cheat*)

Electronegativity • The tendency of an atom to attract electrons is known as electronegativity • The stronger the electronegativity of an atom, the more likely it is to take electrons from other atoms, and the less likely it is to lose them • Non-metals are, in general, more electronegative than metals

Higher Numbers = More attraction

Periodic Trends • As you move from left to right across the periodic table, electronegativity INCREASES • As you move from top to bottom, electronegativity DECREASES • Because more electronegative atoms tend to pull harder on their electrons, they tend to be smaller
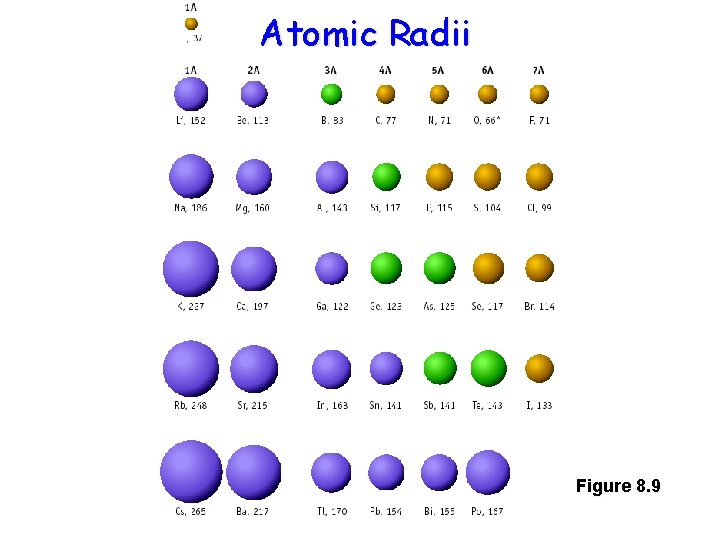
Atomic Radii Figure 8. 9

Bonding • Ionic • Covalent • Metallic

Ionic Bonding • occurs between metal cations and non-metal anions (+-) • Results in an ionic compound • The more electronegative non-metal “steals” one or more electrons from the metal • The two atoms are then attracted to each other because of their opposite charges
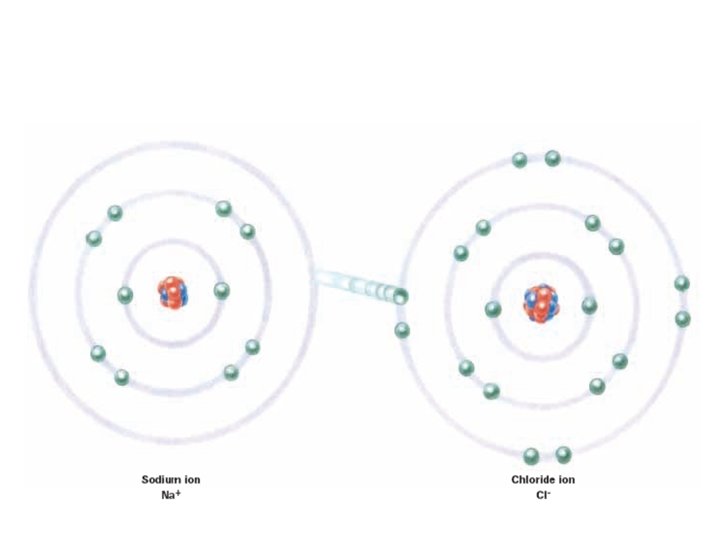

Properties of Ionic Compounds • Contain at least 1 metal and nonmetal • Usually solid crystals • Most can dissolve in water • Very high melting points

Covalent Bonding • Occurs between 2 or more non -metals • Covalently bonded atoms share electrons • Results in a molecule
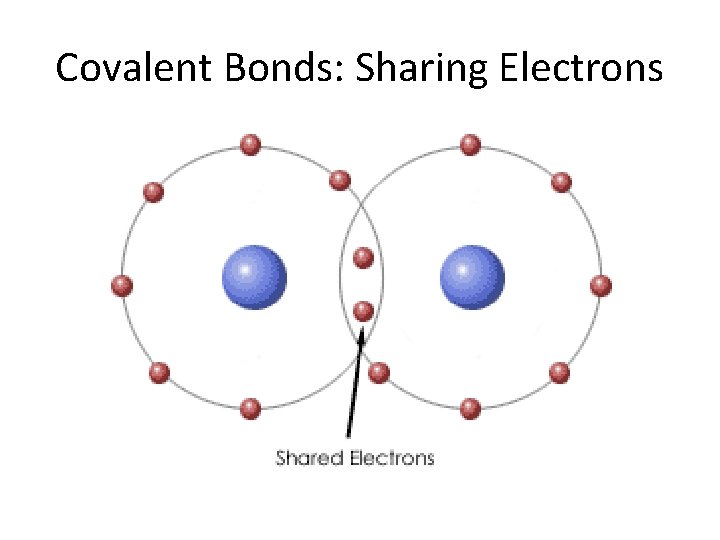
Covalent Bonds: Sharing Electrons
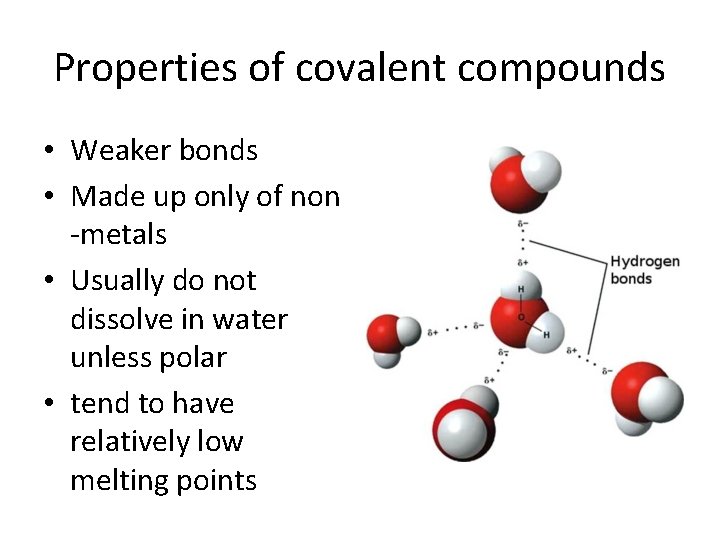
Properties of covalent compounds • Weaker bonds • Made up only of non -metals • Usually do not dissolve in water unless polar • tend to have relatively low melting points

Sugar vs. Salt Sugar – Melting point 186 o. C Salt – Melting Point 804 o C
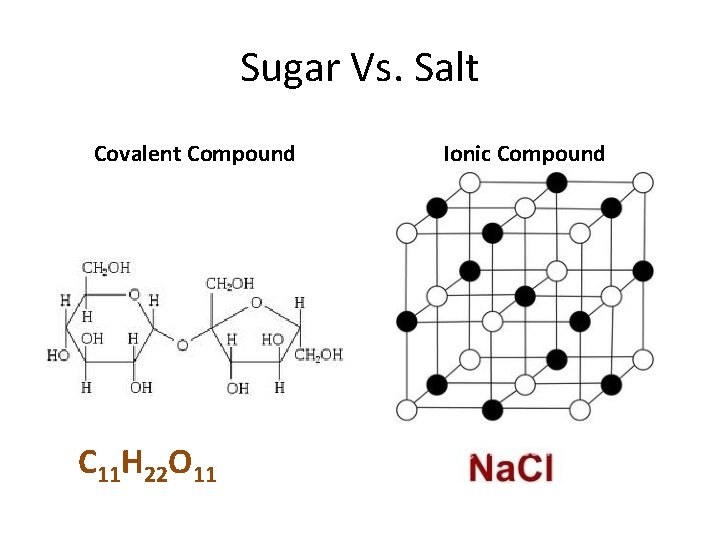
Sugar Vs. Salt Covalent Compound C 11 H 22 O 11 Ionic Compound

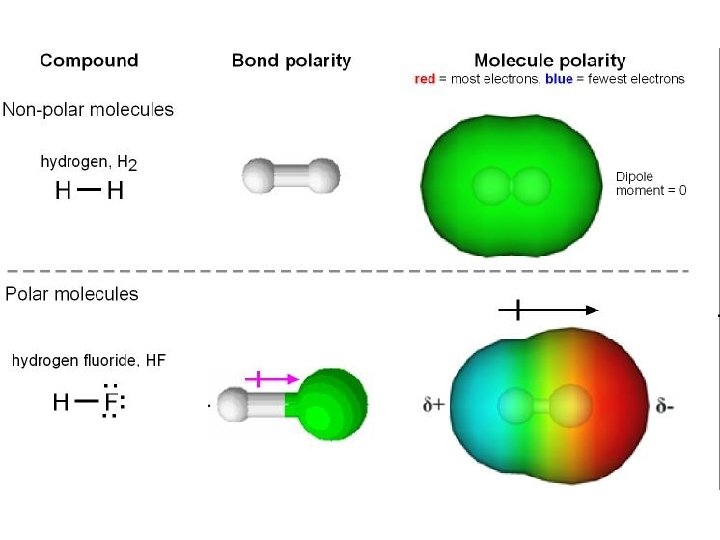
Polar Molecules • If one element is significantly more electronegative than the other, a polar covalent bond is formed • Atoms share electrons unevenly, resulting in one side which is slightly positive and one that is negative • Polar molecules can mix with water; non-polar (hydrophobic) molecules can not
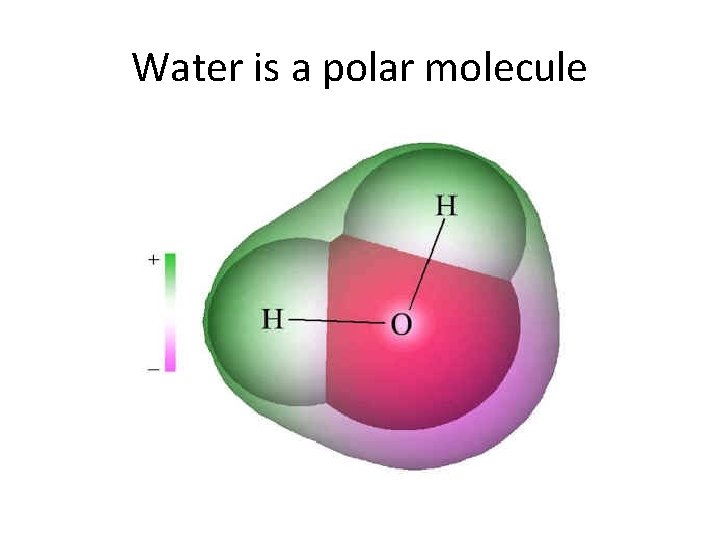
Water is a polar molecule

CO 2 (Carbon Dioxide) is not

Fats and oils are nonpolar and do not mix with water
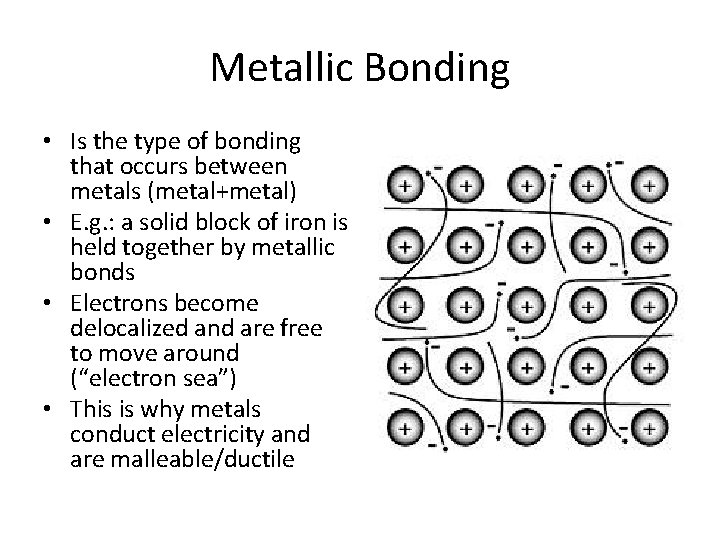
Metallic Bonding • Is the type of bonding that occurs between metals (metal+metal) • E. g. : a solid block of iron is held together by metallic bonds • Electrons become delocalized and are free to move around (“electron sea”) • This is why metals conduct electricity and are malleable/ductile

Lewis Structures for Covalent Bonding • Bonds between electrons are represented by lines between the atoms • Each line represents 2 electrons • Non-bonding electrons are represented as dots • Each atom (except H) needs exactly 8 electrons (octet rule)
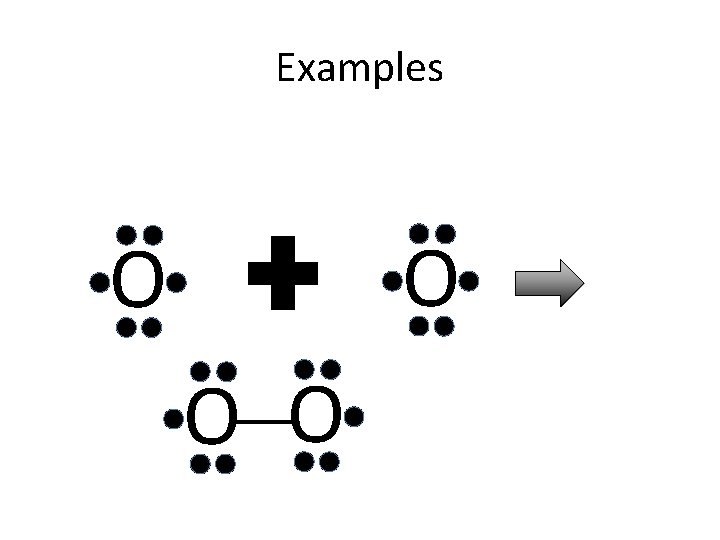
Examples O O

NAMING COMPOUNDS

Naming Ionic Compounds • • • The regular name of the cation comes first The suffix of the anion changes to –ide Na. Cl = Sodium Chloride Na. F = Sodium Fluoride Ca. Br 2 = Calcium Bromide

Try these. 1. 2. 3. 4. 5. Na. S Li 3 N Ag 2 O Ca. Br 2 Zn. O Sodium Sulfide ________ Lithium Nitride ________ Silver Oxide ________ Calcium Bromide ________ Zinc Oxide ________

Writing Formulas from Names • Use periodic trends to predict ionic charges of atoms • Overall charge of atom should be 0 • Use the “crisscross method” to assign number of atoms • “Sodium Fluoride” = Na+ + F- = Na. F • “Calcium Chloride” = Ca 2+ + Cl- = Ca. Cl 2

Ions with multiple charges • Transition metals can have different ionic states • Iron, for example, can sometimes have a +2 charge, while other times it will have a +3 • To avoid confusion, we notate Iron with a +2 charge as Iron(II), while Iron with a +3 charge would be Iron(III)
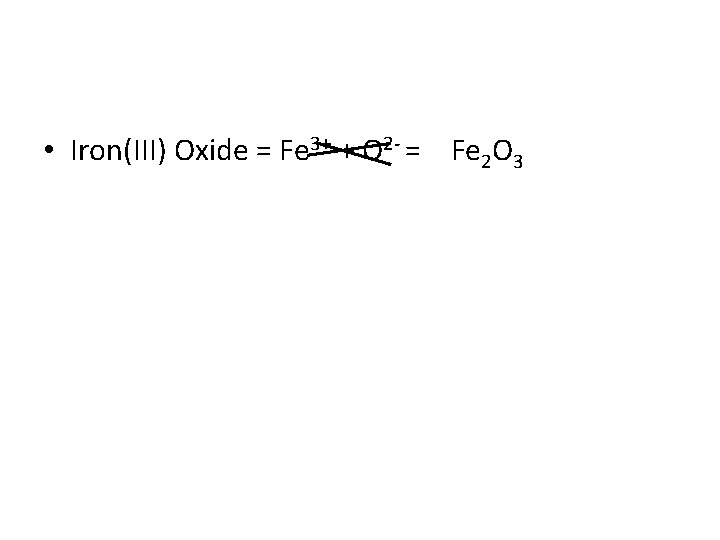
• Iron(III) Oxide = Fe 3+ + O 2 - = Fe 2 O 3

Practice

Polyatomic ions • Some molecules have an overall charge, like ions • These are called polyatomic ions • They are treated as a single unit and can NOT be separated!

Naming Covalent Compounds • Number of atoms is represented by prefix • All atoms in a molecule have their own separate prefixes and are listed in order of appearance • First element only has a prefix if it is greater than 1 • Last element ends in -ide
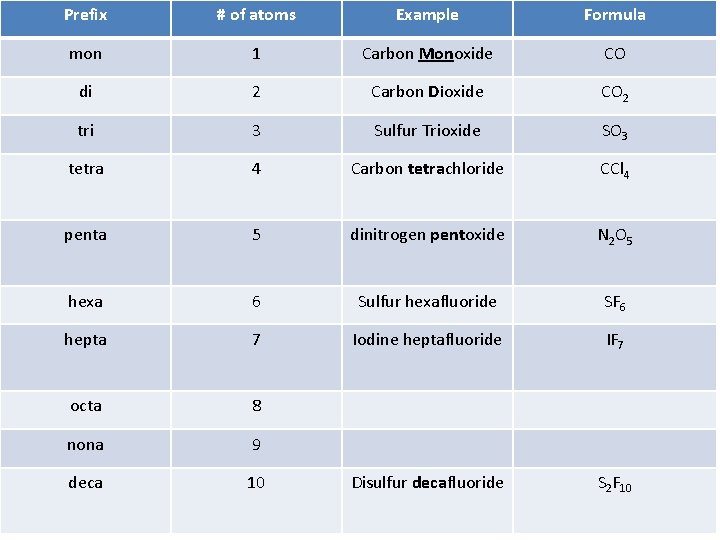
Prefix # of atoms Example Formula mon 1 Carbon Monoxide CO di 2 Carbon Dioxide CO 2 tri 3 Sulfur Trioxide SO 3 tetra 4 Carbon tetrachloride CCl 4 penta 5 dinitrogen pentoxide N 2 O 5 hexa 6 Sulfur hexafluoride SF 6 hepta 7 Iodine heptafluoride IF 7 octa 8 nona 9 deca 10 Disulfur decafluoride S 2 F 10

Practice






- Slides: 91