Unit c Solubility A The Concept of Solubility

Unit c Solubility

A. The Concept of Solubility Solutions can be either ionic or molecular.

1. • • • Ionic compounds are made of a metal ad a non-metal Are composed of one negative ion and one positive ion The ions will dissociate in solution Examples: Na. Cl(s) → Na+(aq) +Cl-(aq) Mg(NO 3)2(s) →
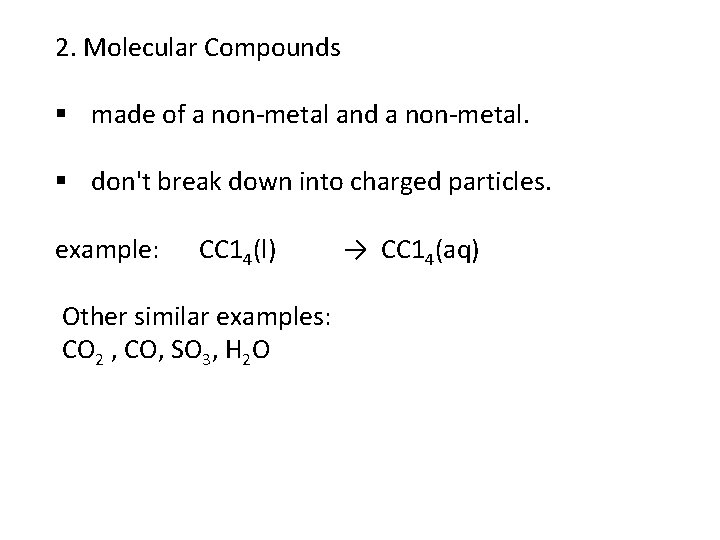
2. Molecular Compounds § made of a non-metal and a non-metal. § don't break down into charged particles. example: CC 14(l) Other similar examples: CO 2 , CO, SO 3, H 2 O → CC 14(aq)

Definitions § Saturated solution (the Chem 12 definition): When the dissolved substance is in equilibrium with some of the undissolved substance. § Solubility: The equilibrium concentration of the substance (at a given temperature). • Molar solubility: The solubility expressed in "moles per liter"

Saturation exists when … a) Some undissolved material is present AND b) Equilibrium exists between the dissolved and undissolved material A saturated solution is at equilibrium! dissolving Ag. Br. O 3(s) ⇌ Ag+(aq) +Br. O 3 -(aq) crystallizing

Assignment Hebden #1, 2 acd, 3, 4, 5, 7

Solutions and Solubility Calculations Example 1: Find the concentration of a solution of. Na. CH 3 COO if 1. 5 mol is dissolved in 200. 0 m. L of solution.

Solutions and Solubility Calculations Example 2: 15 g of Na. Cl is dissolved in 65 m. L of solution. Find [Na. Cl]

Solutions and Solubility Calculations Example 3: How many moles of H 2 SO 4 are there in 325 m. L of a 0. 512 M solution?

Solutions and Solubility Calculations Example 4: 55 m. L of 1. 6 M Ca. C 12 is boiled dry. What mass of solid is recovered?

Solutions and Solubility Calculations Example 5: Find the molar solubility of KNO 3 at 45°C if 85 g of it can be dissolved to make 135 m. L of solution.

Solutions and Solubility Calculations Example 6: The molar solubility of KI is 0. 850 M. Find the solubility in g/L

Solutions and Solubility Calculations Example 7: The molar solubility of Ag. Br. O 3 is 8. 30 X 10 -3 M. Find the mass of silver bromate that can be dissolved in 55. 0 m. L of water.

Solutions and Solubility Calculations Example 8: 35 m. L of 6. 0 M HCl is added to 52 m. L of water. Find the new [HCl].

Assignment Hebden #8 -10 and 12 -14.

Calculating ion concentrations Example 1: Calculation the concentration of each ion in a 0. 50 M Cu 3(PO 4)2 solution.

Calculating ion concentrations Example 2: A saturated solution of Fe. I 3 contains 6. 4 g of Fe. I 3 per L of solution. Find the [ ] of each ion.

Calculating ion concentrations Example 3: 2. 0 L of 0. 50 M Cu. Cl 2 is mixed with 3. 0 L of 0. 80 Mn. Cl 4. Calculate the concentration of each ion in the final mixture. Assume no reactions occur.

Assignment Hebden #17 a-d, 18 a, 19 abc, 20 abgh. Workbook

predicting the solubility of salts § Most ionic compounds are soluble in water because of water’s polar nature. § Others may dissolve to a slight, but appreciable extent. § Definition of low solubility: a saturated solution of the substance is less than 0. 1 M. § A table of solubility is provided to help you determine which compounds are soluble and which have low solubility.

Assignment Hebden #21

Molecular, Ionic, and Net Ionic Equations Ø Is it possible to predict when a precipitate will form when two solutions are mixed? Ø Example: A solution of lead nitrate is mixed with a solution of potassium iodide. A precipitate forms. Which product is the precipitate (ppt)?

The Molecular Equation: (or Formula equation): Pb(NO 3)2 (aq) + KI (aq) → Pb. I 2 +2 KNO 3

The Reactants: Since the reactants are ionic compounds, they will actually dissociate in water and be present as ions And Pb 2+ + NO 3 And 2 K+ + 2 I-

The products: Ø the products are Pb. I 2 and KNO 3 Ø Which of these is the precipitate? (refer to your data booklets or to p 459 of Heath) Ø Pb. I 2 : not soluble, so it remains a solid. Ø KNO 3 : soluble, so it remains dissociated as 2 K+ + NO 3 -

The Ionic Equation: - Pb 2+ + 2 NO 3 - + 2 K+ + 2 I- → Pb. I + 2 K+ +2 NO 3 - § Note the spectator ions. § Cancel them out.

Th Net Ionic Equation: Pb 2+ (aq) + 2 I- (aq) →Pbl 2 (s)

Assignment Hebden #22, 25 abcghi workbook

Qualitative Analysis Separating Mixtures of ions by Precipitation Ø The composition of an ionic substance can be determined by selecting a reagent that gives a precipitate with some ions but not with others Ø Generally, we try to precipitate out only one ion at a time.

Example 1: Devise a method for precipitating out Ag+ and Sr 2+ separately from a solution. Step 1 2 Add… Reasoning equation

Example 2: Separate by precipitation Br- and OH- from a solution of Na. OH and NH 4 Br Step 1 2 Add… Reasoning equation

Example 3: Separate a mixture of Ca 2+ , Mg 2+and Cu 2+ Step 1 2 3 Add… Reasoning equation

Assignment Hebden #26 -32, 35, 36 workbook

B. Quantitative Aspects of Solubility Ø We have said that if solubility is <0. 1 mol/L the substance is insoluble. Ø But even 'insoluble' substances dissolve somewhat, but quickly become saturated. Ø They are best represented by equilibrium reactions.


Ksp Ø A special kind of Keq is defined for ionic solutions. Ø It is called Ksp, where sp stands for solubility product.

Example 1: The solubility of Ca. CO 3 is 6. 9 x 10 -5 mol∕L at 25°C. Calculate the Ksp.

Example 2: The solubility of Mg. F 2 is 7. 3 x 10 -2 g∕L at 25°C. Calculate the Ksp.

Assignment

Example 3: Calculate the solubility of Cu. CO 3 given Ksp = 4. 8 X 10 -4. Ø Let ion concentration at equilibrium = x. Ø Solve for x in Ksp expression

Example 4: Calculate the solubility of Fe. I 2 given Ksp = 1. 5 X 10 -14 at 25°C Ø Let ion concentration at equilibrium = x. Ø Solve for x in Ksp expression

Assignment

The significance of Ksp Ø at 25°C : Ksp of Cu. CO 3 =4. 8 X 10 -4 Ksp of Ag. Cl =1. 8 X 10 -10 Ø Which one is more soluble? why? Cu. CO 3 is more soluble because its Ksp is larger, which means [ion] is larger, so more has dissolved. . Ø Generally: A larger Ksp means greater solubility but there are exceptions:

Ag. Cl Ksp =1. 8 X 10 -10 Ca. F 2 Ksp =4. 9 X 10 -11 We expect _______to be more soluble because it has a larger Ksp. Lets check it out by finding the solubility of each. Ø Solubility of Ag. CI: Solubility of Ca. F 2 :

so… Ø Ca. F 2 is more soluble than Ag. Cl even though it has a smaller Ksp Ø Moral: you can reliably compare Ksp only when the compounds produce the same number of ions.

Assignment Hebden # 40 b, 41, 43, 47, 52

Predicting Precipitation Using Ksp Ø Consider a situation where 20. 0 m. L of 0. 10 M Ag. NO 3 is mixed with 30. 0 m. L of 0. 010 M Na. CI. Ø Does a ppt of Ag. Cl form? (Ksp Ag. Cl= 1. 8 x 10 -10) Ø Find the concentrations of each of the ions that form Ag. CI. Ø Calculate the trial Ksp( Ktrial or TIP) Ø Compare the TIP to the real Ksp if TIP ≥Ksp then a ppt will form

Example 1: Will Ag. CI precipitate if 1. 0 m. L of 0. 10 M Ag. NO 3 is mixed into l. 0 L of water with [ CI-] = 1. 0 x l 0 -6 M? (Ksp Ag. Cl = 1. 8 x 10 -10)
![Example 2: What is the highest [I-] that can exist without precipitating Agl in Example 2: What is the highest [I-] that can exist without precipitating Agl in](http://slidetodoc.com/presentation_image_h2/4998cc68665e6d5816b00938933e97e9/image-50.jpg)
Example 2: What is the highest [I-] that can exist without precipitating Agl in a 0. 010 M Ag. NO 3 solution if Ksp for Agl =8. 3 X 10 -17? Ø To get ppt: [Ag+][I-] ≥Ksp Ø At minimum [I-], [Ag+][I-] = Ksp. Ø Solve for the only unknown, the [I-]!

Example 3: 0. 10 M Ag. NO 3 is added to a solution containing 0. 020 M Na. Cl and 0. 020 M K 2 Cr. O 4. Identify the first precipitate that forms Ø Identify the possible precipitate: Ø Find [Ag+] needed to ppt with each ion.
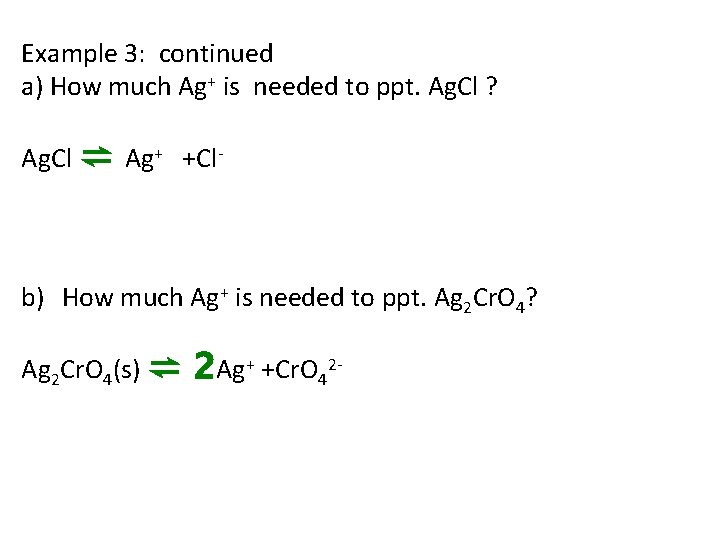
Example 3: continued a) How much Ag+ is needed to ppt. Ag. Cl ? Ag. Cl ⇌ Ag+ +Cl- b) How much Ag+ is needed to ppt. Ag 2 Cr. O 4? Ag 2 Cr. O 4(s) ⇌ 2 Ag+ +Cr. O 42 -
![Since Ag. Cl requires the least [Ag+], it will ppt. first. Since Ag. Cl requires the least [Ag+], it will ppt. first.](http://slidetodoc.com/presentation_image_h2/4998cc68665e6d5816b00938933e97e9/image-53.jpg)
Since Ag. Cl requires the least [Ag+], it will ppt. first.

Assignment Hebden #57, 59, 62, 65, 69 workbook
 Ø Example 1: Ag. NO 3 is titrated against Using precipitation to measure [ion](titration) Ø Example 1: Ag. NO 3 is titrated against](http://slidetodoc.com/presentation_image_h2/4998cc68665e6d5816b00938933e97e9/image-55.jpg)
Using precipitation to measure [ion](titration) Ø Example 1: Ag. NO 3 is titrated against swimming pool water to measure the [Cl-]. If 14. 00 m. L of 0. 100 M Ag. NO 3 is needed to ppt. all the Cl- in a 100. 0 m. L sample, find the [Cl-] in the sample. (Na. Cr. O 4 is used as an indicator to show when all the Cl- is gone). Step 1: Find moles Ag. NO 3 used. Step 2: Find moles Cl- present Step 3: Find [Cl-]

Assignment Hebden #70, 71, 73 workbook

Common lon Effect Ø Example 1. Ag. I(s) ⇌ Ag+ (aq) + Cl- (aq) Ø Adding a common ion (eg Ag+) from another source will shift the equilibrium to the left, meaning less Ag. I will dissolve (it becomes less soluble). Example 2. Agi(s) !: ; Agaq) + r-(aq) : > Ifyou add some S 2 - ions they will precipitate out the silver ions as Ag 2 S. : > This causes the [Ag+] to drop and the

Common lon Effect Ø Example 2. Ag. I(s) ⇌ Ag+ (aq) + Cl- (aq) Ø If you add some S 2 - ions they will precipitate out the silver ions as Ag 2 S. Ø This causes the [Ag+] to drop and the equilibrium will shift right, meaning more Ag. I will dissolve (it becomes more soluble). Ø A common ion decreases the solubility. Removing a common increases solubility.


Assignment Hebden #81 -86 Workbook Common ion practice ws

Applications of Solubility Ø Hard water usually contains Ca 2+ or Mg 2+, often from limestone, Ca. CO 3. Ø Effects of hard water: causes precipitates in hot water systems, including coffee makers and kettles. funny taste. stops soap from working properly by forming a soap scum (precipitate) which stops the soap from lathering. Why? Ca 2+ precipitates with part of the soap molecule.

How do we soften permanently hard water? Ø Permanently hard water contains mostly sulfates and chlorides of Mg 2+ and Ca 2+, such as Ca. SO 4, Ca. Cl 2, Mg. SO 4, Mg. Cl 2. Ø Solution: Add 'washing soda' (Na 2 CO 3). The CO 32 - forms a ppt with the Mg 2+ and Ca 2+ , removing them so that they can' t precipitate the soap.

How do we soften temporarily hard water? Ø temporarily hard water contains Ca(HCO 3)2 OR Mg(HCO 3)2 Ø These come from limestone as in this reaction Ca. CO 3(s) + H 2 O(l) +CO 2(g) ⇌ Ca(HCO 3)2 + heat Ø Solution: just heat the water! This shifts the equilibrium left, cause the Ca. CO 3 to precipitate out, and leave the water soft.

Example calculate the maximum mass of Na 2 CO 3 that can be added to 2. 5 L of water containing a Ca 2+ concentration of 0. 0040 M, without causing a precipitate.

Assignment Hebden #77, 78, 79. Practice Test Unit C-2 Test
















































- Slides: 113