Unit B The Chemistry of Life Affixes and

Unit B: The Chemistry of Life

Affixes and Roots • • • iso macro poly mer ose co = equal = large = many = parts = sugar = with, together

Chemistry • is the study of the structure, properties, and composition of substances, and the changes that substances undergo

Elements • a substance that cannot be broken down into simpler substances: • gold • silver • carbon

Symbols • Hydrogen is the first element. It can be abbreviated using a single letter or symbol.

Trace Elements • Elements make up all things, including living things. • Some are present in very small amounts in living things. These are known as trace elements.

Atoms: Building Blocks of Elements • An atom is the smallest particle of an element that has the characteristics of that element.
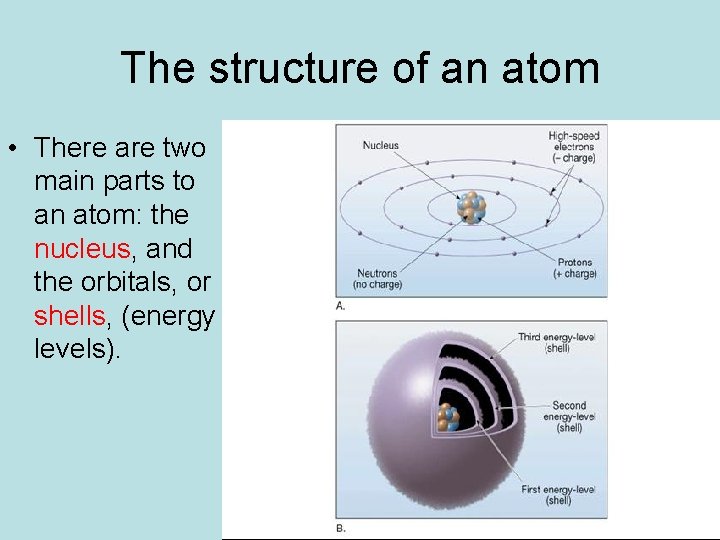
The structure of an atom • There are two main parts to an atom: the nucleus, and the orbitals, or shells, (energy levels).
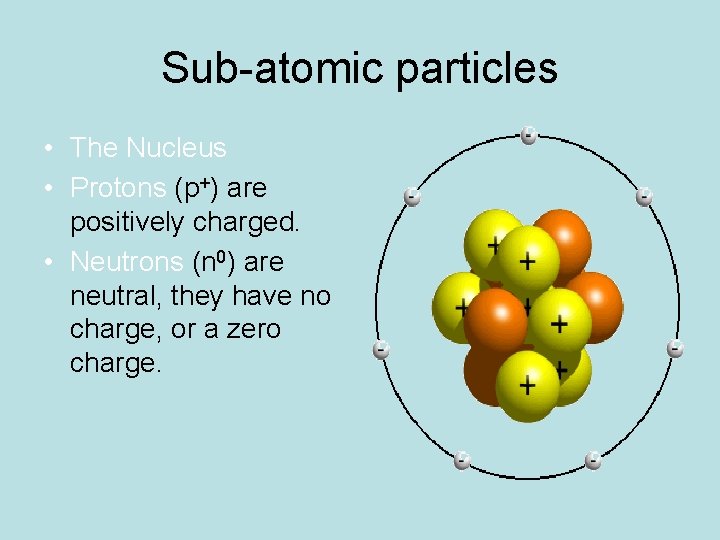
Sub-atomic particles • The Nucleus • Protons (p+) are positively charged. • Neutrons (n 0) are neutral, they have no charge, or a zero charge.
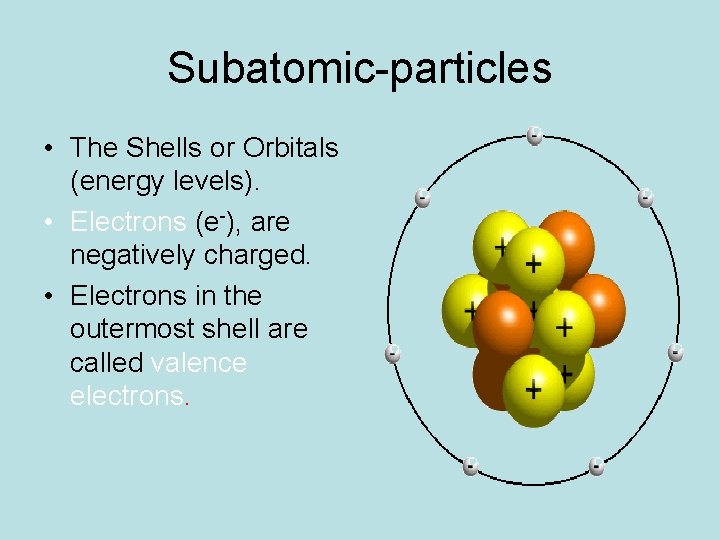
Subatomic-particles • The Shells or Orbitals (energy levels). • Electrons (e-), are negatively charged. • Electrons in the outermost shell are called valence electrons.

Electron Energy Levels • The 1 st shell can hold up to 2 valence e-. • The 2 nd shell can hold up to 8 valence e-. • The 3 rd shell can hold up to 8 valence e-. • (Although there can be as many as 18 e- in the 3 rd energy level, for the purposes of understanding bonding we only concern ourselves with the 8 valence e-. )

The numbers • The large number one in the upper left hand corner is the atomic number. It is the number of protons.

The numbers • The number on the bottom is the atomic mass number. • It is also known as the molar mass, or atomic weight. • It is the number of protons + the number of neutrons. * * (when you round it to the nearest whole number).
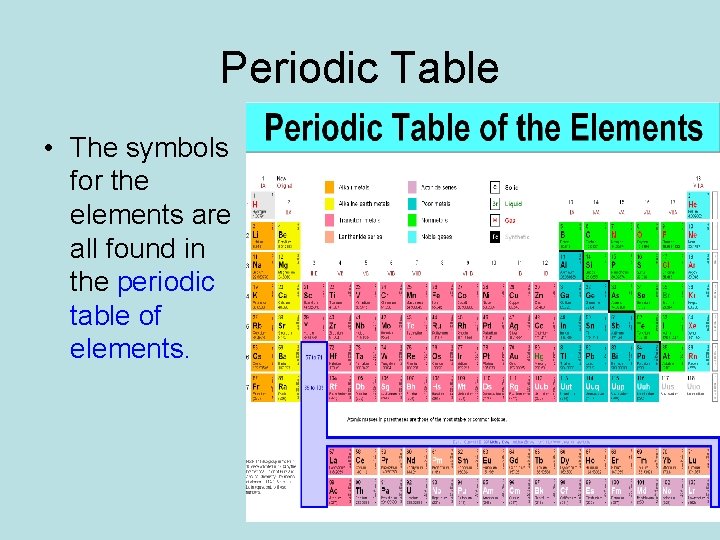
Periodic Table • The symbols for the elements are all found in the periodic table of elements.

Variations • Different scientists use different versions of the periodic table but they all have the same information, even if it is all switched around.

Here’s one version…

This one was in X-men 3 and in Room 148.
This one is from a Chicago man.
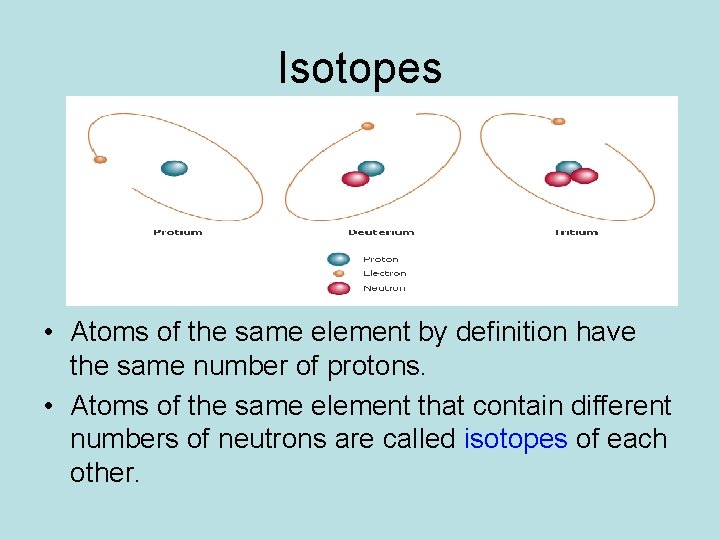
Isotopes • Atoms of the same element by definition have the same number of protons. • Atoms of the same element that contain different numbers of neutrons are called isotopes of each other.

Compounds • a substance that is composed of two or more atoms of different elements

Bonds • There two types of bonds: • a. covalent • b. ionic
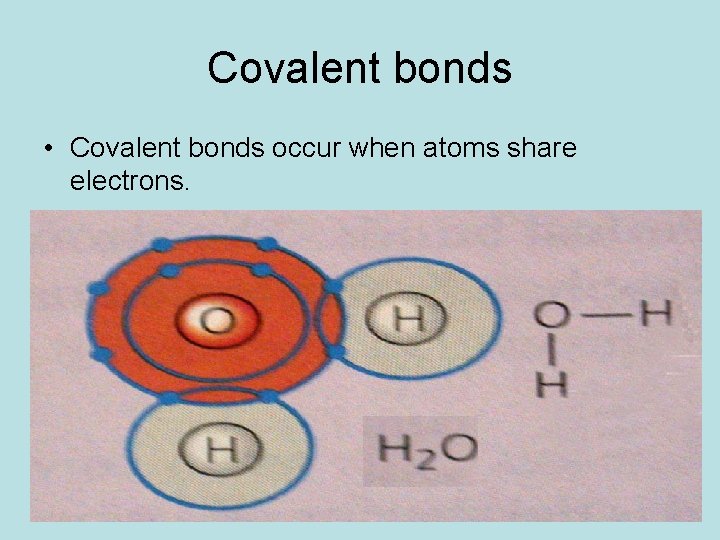
Covalent bonds • Covalent bonds occur when atoms share electrons.

Ionic Bonds • Ions are atoms with a charge. They have a charge due to an imbalance in the number of protons and electrons. • The attractive force between two ions of opposite charges is known as an ionic bond.

Quick terms • metabolism = all of the chemical reactions that take place within an organism

• mixture = a combination of substances in which the individual components retain their properties

• solution = a mixture in which one or more substances (solutes) are distributed evenly in another substance (solvent)
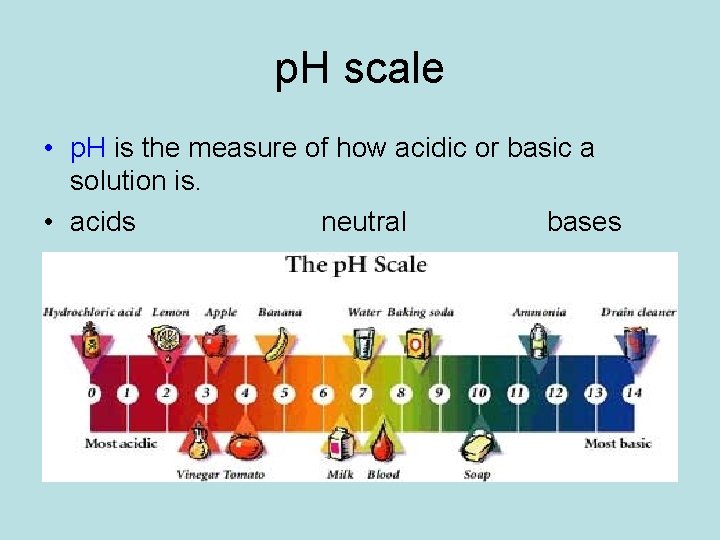
p. H scale • p. H is the measure of how acidic or basic a solution is. • acids neutral bases

Buffers • Buffers are substances that resist changes in p. H. • They absorb H and OH ions.
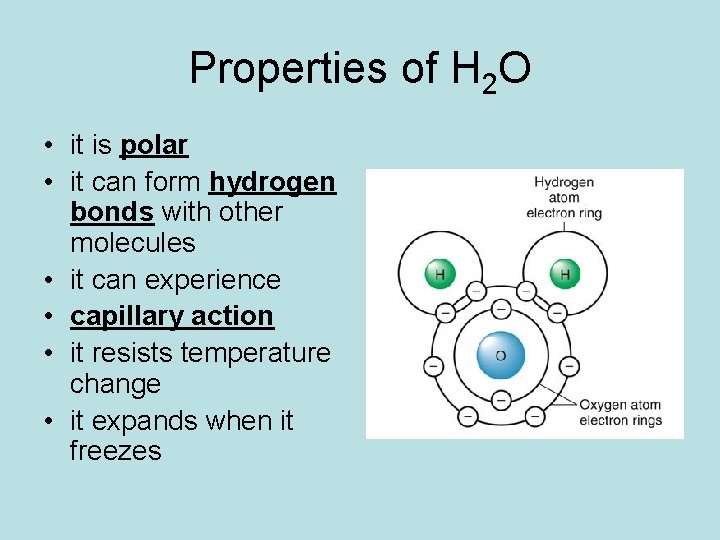
Properties of H 2 O • it is polar • it can form hydrogen bonds with other molecules • it can experience • capillary action • it resists temperature change • it expands when it freezes

Polar Molecules • Water is polar because the electrons spend more time around the oxygen atom. • This gives oxygen a partial negative charge and, consequently, gives the hydrogen atoms partially positive charges.
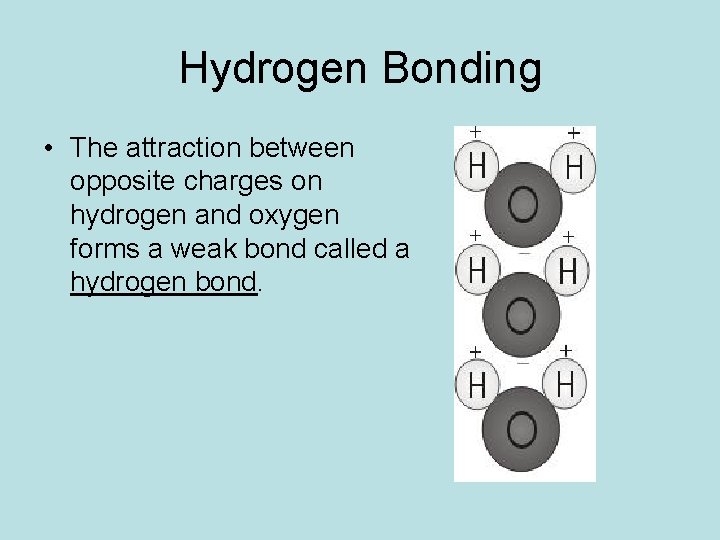
Hydrogen Bonding • The attraction between opposite charges on hydrogen and oxygen forms a weak bond called a hydrogen bond.

Capillary Action… • is the ability of water to crawl up thin tubes. • This property makes it possible for water to get from roots in the ground to the leaves of the tallest trees.
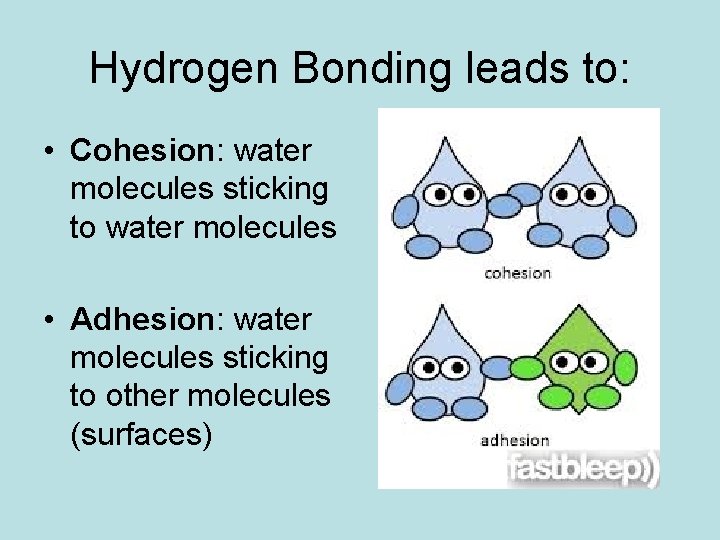
Hydrogen Bonding leads to: • Cohesion: water molecules sticking to water molecules • Adhesion: water molecules sticking to other molecules (surfaces)

Buffers • Buffers are substances that help prevent changes in p. H

Diffusion • is the net movement of particles from an area of higher concentration to an area of lower concentration.

Life Substances • Carbon is the most important element to life on Earth. • This is due to the fact that it can form up to four bonds.

Why is that important? • Because of this fact carbon can form many different shaped molecules, even using the same atoms.
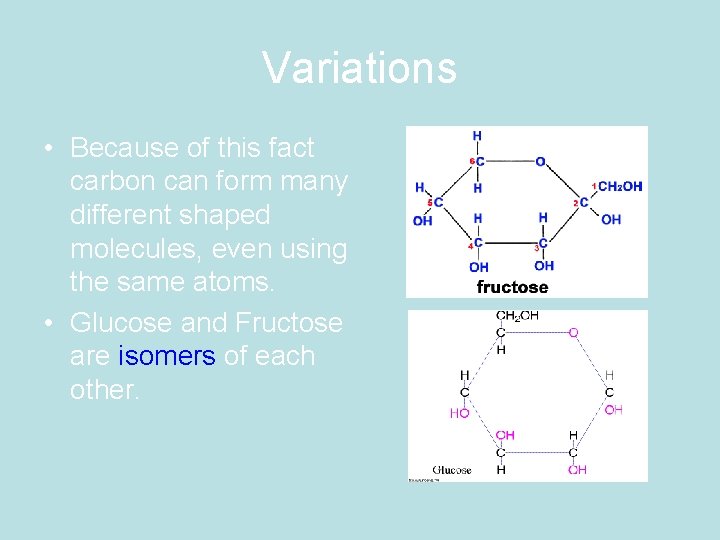
Variations • Because of this fact carbon can form many different shaped molecules, even using the same atoms. • Glucose and Fructose are isomers of each other.
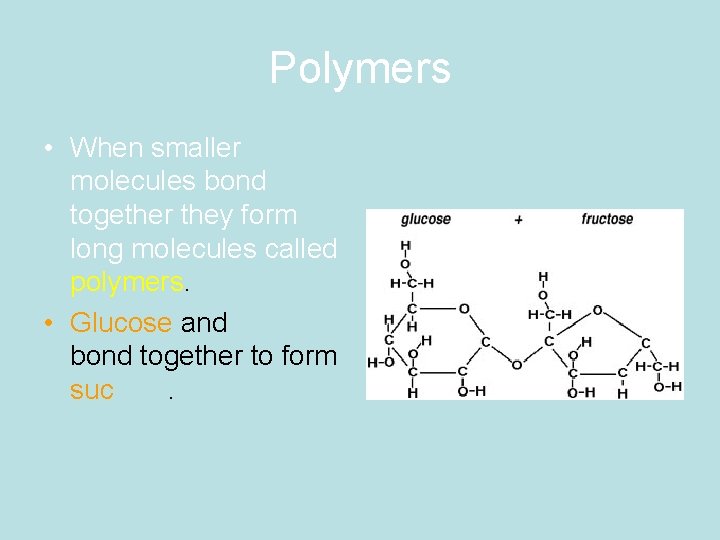
Polymers • When smaller molecules bond together they form long molecules called polymers. • Glucose and fructose bond together to form sucrose.

4 major macromolecules • • carbohydrates lipids proteins nucleic acids

Carbohydrates • are composed of carbon, hydrogen and oxygen. • 1 C: 2 H : 1 O • Cx. H 2 x. Ox • These are basically sugars: mono-, diand poly- saccharides

Lipids • are composed mostly of carbon and hydrogen with a small amount of oxygen • Fats, oils, waxes and steroids are all lipids. • They are non-polar and so don’t mix with water. •
Proteins • A protein is a large complex polymer composed of carbon, hydrogen, oxygen, nitrogen and sometimes sulfur. • Two or more amino acids held together by a peptide bonds are considered a protein.
Special Proteins • Enzymes are proteins that change the rates of chemical reactions. They are necessary for life as we know it.

Nucleic acids… • are complex biomolecules that store cellular information in the form of a code. • They are polymers made up of subunits called nucleotides.
- Slides: 45