Unit 9 Gases 1 Pressure Gas pressure the




































- Slides: 45

Unit 9: Gases

1. Pressure • • • Gas pressure: the result of collisions of billions of rapidly moving gas particles with other objects • Atmospheric pressure results from the collisions of atoms and molecules in air with objects SI unit of pressure: Pascal (Pa) Other units include • millimeters of mercury: mm. Hg • atmospheres: atm • torr

Pressure conversions 1 atm = 760 mm. Hg =760 torr = 101. 3 k. Pa ** Remember- STP is defined as a temp of 0°C (273 K) and 101. 3 k. Pa (1 atm)

Practice A pressure gauge records a pressure of 89. 0 k. Pa. What is this measurement in a) atmospheres? b) mm. Hg? 1 atm = 760 mm. Hg = 101. 3 k. Pa

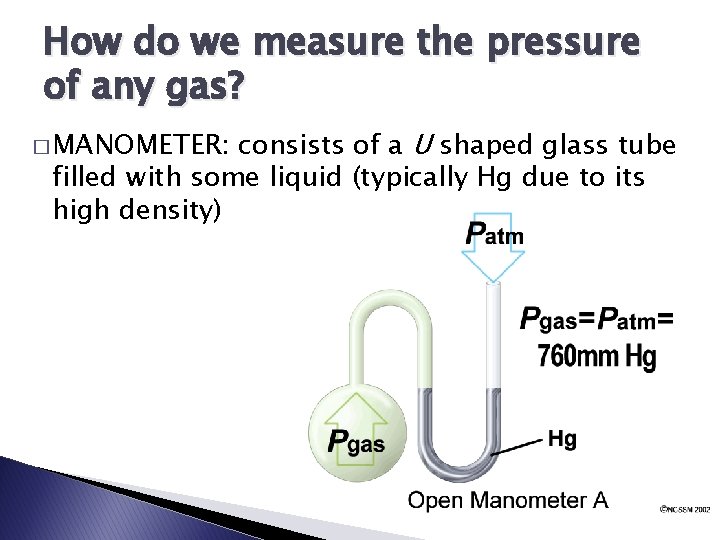
How do we measure the pressure of any gas? consists of a U shaped glass tube filled with some liquid (typically Hg due to its high density) � MANOMETER:

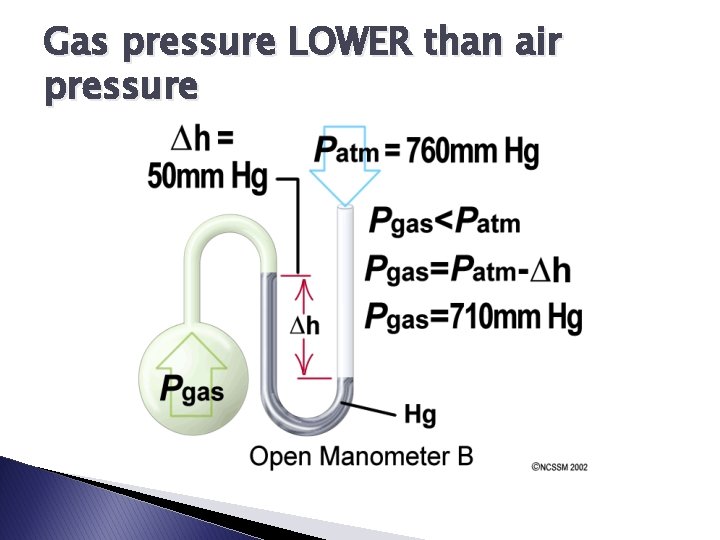
Gas pressure LOWER than air pressure

Gas pressure HIGHER than air pressure

2. Volume • Volume is a measure of how much space an object takes up • SI unit for volume = Liter • Remember: 1 L = 1000 m. L

3. Temperature • Temperature is a measure of the average kinetic energy of the particles in a substance • SI unit for temp: Kelvin • Remember: K = °C + 273

The Gas Laws � Show relationships between pressure, volume and temperature for an ideal gas � 3 major gas laws **All temperatures MUST be in Kelvins.

1. Boyle’s Law: Pressure and Volume � Under constant temperature, as the pressure of a gas increases, the volume decreases � As the pressure decreases, the volume increases

Boyle’s Law: Pressure and Volume P 1 V 1 =P 2 V 2

2. Charles’ Law: Temperature and Volume � Under constant pressure, as the temperature of a gas increases, the volume increases � As temperature decreases, the volume decreases

Charles’ Law: Equation V 1 = V 2 T 1 T 2 **You must convert all temperatures to Kelvin!

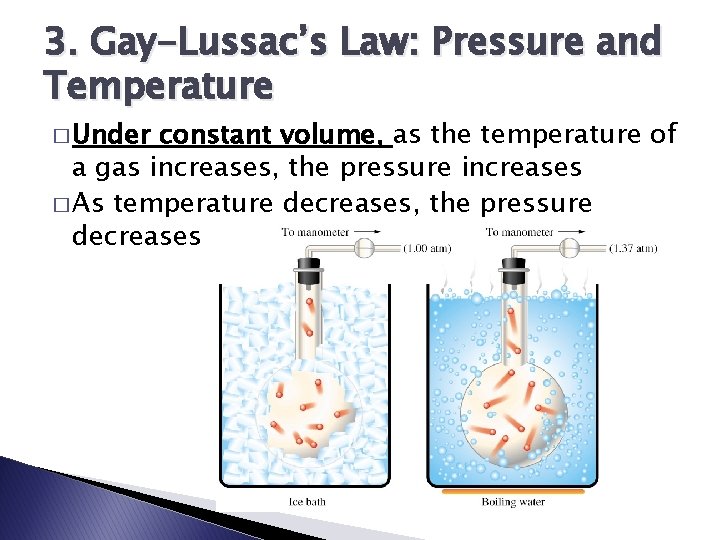
3. Gay-Lussac’s Law: Pressure and Temperature � Under constant volume, as the temperature of a gas increases, the pressure increases � As temperature decreases, the pressure decreases

Gay-Lussac’s Law: Equation P 1 = P 2 T 1 T 2

The Combined Gas Law

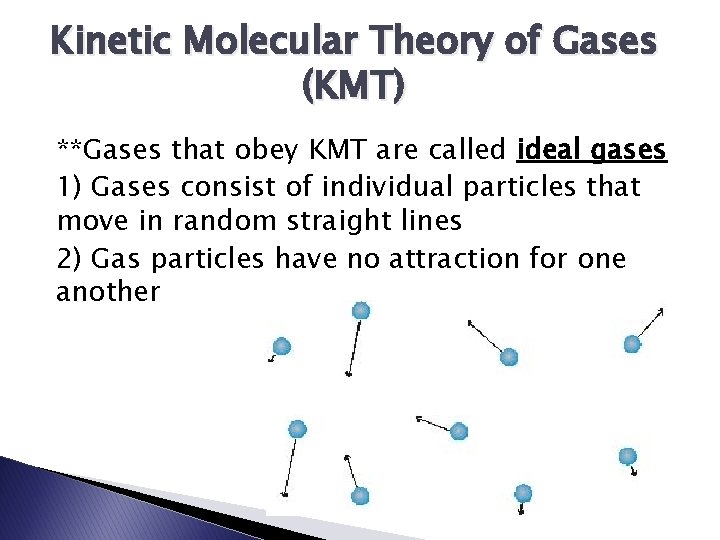
Kinetic Molecular Theory of Gases (KMT) **Gases that obey KMT are called ideal gases 1) Gases consist of individual particles that move in random straight lines 2) Gas particles have no attraction for one another

Kinetic Molecular Theory of Gases (KMT) 3) Gas particles are separated by such large distances that the particles themselves have negligible volume 4) When gas particles collide, the collisions are perfectly elastic (particles may transfer energy, but there is no net loss of energy)

When do gases behave most like ideal gases? � Under conditions of low pressure and high temperature � Lighter (smaller) gases behave more like ideal gases than heavier ones

These ideal “laws” are OK for molecules that are: �Small �Nonpolar ***2 real gases that behave most ideal: �H 2 and He

Ideal Gas Law � Relates ◦ ◦ ◦ 4 measureable quantities: P = Pressure (atm) V= Volume (L) n= number of moles R = ideal gas constant (0. 0821 L • atm/mol • K) T = Temp (K) Refer to text for derivation (p. 351)

Ideal Gas Law- Examples �What is the pressure, in atm, of a. 500 mol sample of N 2(g) in a 10. 0 L container at 298 K? �How many grams of O 2 are in a 125 m. L container at 45°C and 340. mm. Hg?

Finding Molar Mass or Density from Ideal Gas Law � Moles = mass (g) Molar Mass (g/mol) PV = n. RT

Finding Molar Mass or Density from Ideal Gas Law �PV = m. RT M Rearranged: M= m. RT PV

Since �d = m v M= m. RT = PV Rearranged: d. RT P d = MP RT

Warm-Up Find the molar mass of a 1. 05 g gas sample at 37° C and 0. 840 atm that occupies 2. 35 L.

Avogadro’s Principle � One mole of any gas at STP occupies a volume of 22. 4 L

Avogadro’s Principle � Equal volumes of gases at the same temperature and pressure contain an equal number of particles

mol volume calculations 1 mol = 22. 4 L at STP Assuming STP: � Convert 0. 42 mol H 2 (g) to liters � Convert 64. 3 L O 2 (g) to moles

volume calcs �Since moles = volume for gases at same T and P, treat vol as mol calcs using mole ratios �Ex. 3 H 2(g) + N 2(g) 2 NH 3(g) ◦ How many L of H 2(g) are required to produce 4 L of NH 3(g) at STP? ◦ How many L of H 2(g) are required to produce 8. 19 L of NH 3(g) at STP?
Mass volume calculations �Ex. What is the mass of 1. 33 x 104 m. L of O 2 @STP?

Gas Density �Usually given in g/L @ STP �Density (@STP) = Molar Mass (g/mol) Molar Volume (22. 4 L/mol)

Example �What is the density of CO 2(g), in g/L @STP?

Warm-Up �A 0. 519 g gas sample is found to have a V= 200. m. L @STP. What is the molar mass of this gas? ◦ Molar Mass = Density * Molar Volume

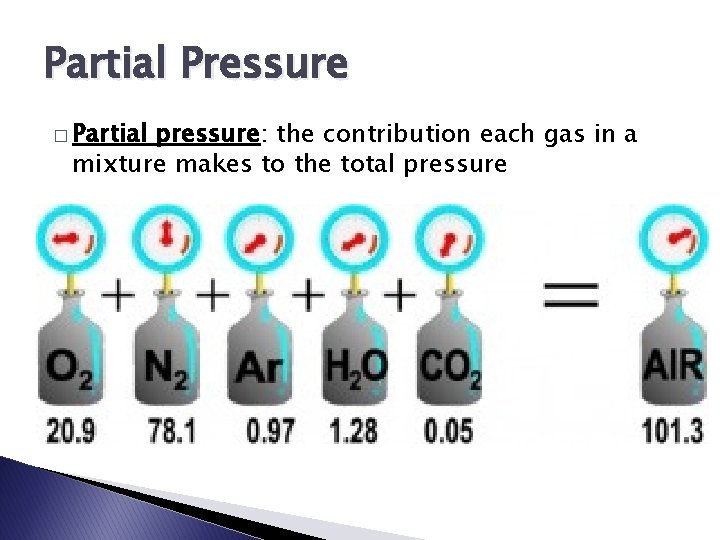
Partial Pressure � Partial pressure: the contribution each gas in a mixture makes to the total pressure

Dalton’s Law of Partial Pressure Ptotal = P 1 + P 2 + P 3 + … - - At constant volume and temperature, the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures of the component gases The partial pressure of an individual gas is equal to the total pressure multiplied by the mole fraction of that gas

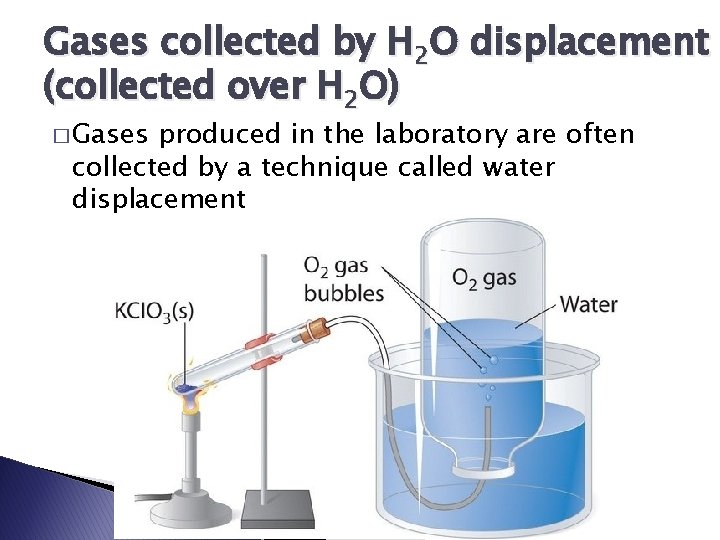
Gases collected by H 2 O displacement (collected over H 2 O) � Gases produced in the laboratory are often collected by a technique called water displacement

Gases collected by H 2 O displacement � “wet” gas = dry gas + H 2 O(g) � PT = Pdry gas + PH 2 O(g) � PH 2 O (g) can be found using this table

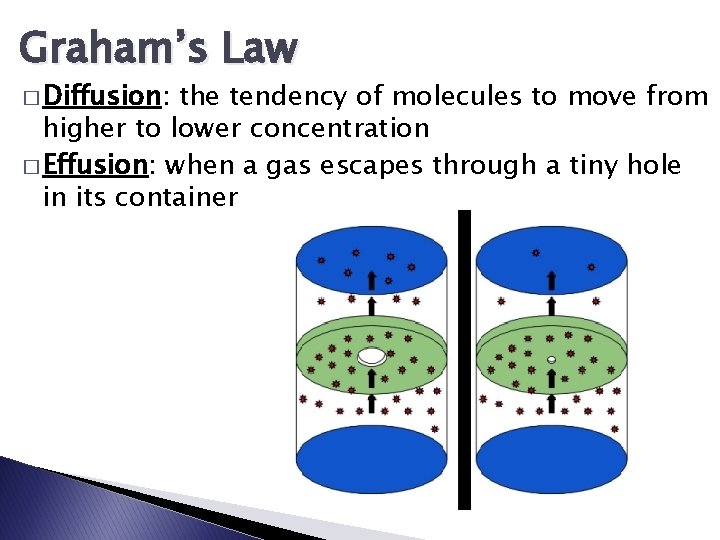
Graham’s Law � Diffusion: the tendency of molecules to move from higher to lower concentration � Effusion: when a gas escapes through a tiny hole in its container

Graham’s Law Qualitatively… �Smaller, lighter gas molecules (lower molar mass) will diffuse faster than bigger, heavier gas molecules.

Graham’s Law Quantitatively. . . �The rates of diffusion (effusion) of gases @ the same T and P are inversely proportional to the square roots of their molar masses.

Example �Compare H 2(g) rates of diffusion of and O 2(g).

Because density varies directly with Molar Mass… � rate of diffusion of A = √density B rate of diffusion of B √density A

Real vs. Ideal gases � Particles of a real gas ARE attracted to one another (and can condense) � Real gas particles DO have volume � When gas particles collide, the collisions are NOT perfectly elastic (there is some loss of energy)