Unit 9 Day 4 Colligative Properties Learning Objectives

Unit 9 Day 4 Colligative Properties

Learning Objectives • I can describe what happens to solutes when dissolved in solvents • I can define and explain how colligative properties work • I can calculate freezing point depression and melting point elevation

Instruction -complete all notes and example problems -complete summary -silently -raise your hand to ask or answer a question

KP 1: What happens to a solute when it is dissolved in water (solvent) • Depends on if the solute is ionic or covalent – Covalent compounds do not dissociate in water • The particles become isolated and surrounded by water particles – Ionic Compounds do dissociate in water • The compound breaks apart into individually charged ions • Each ion is surrounded by water particles
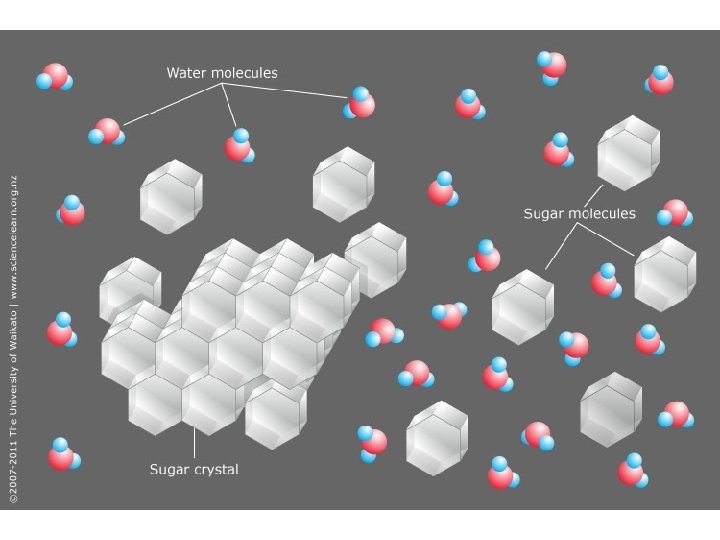
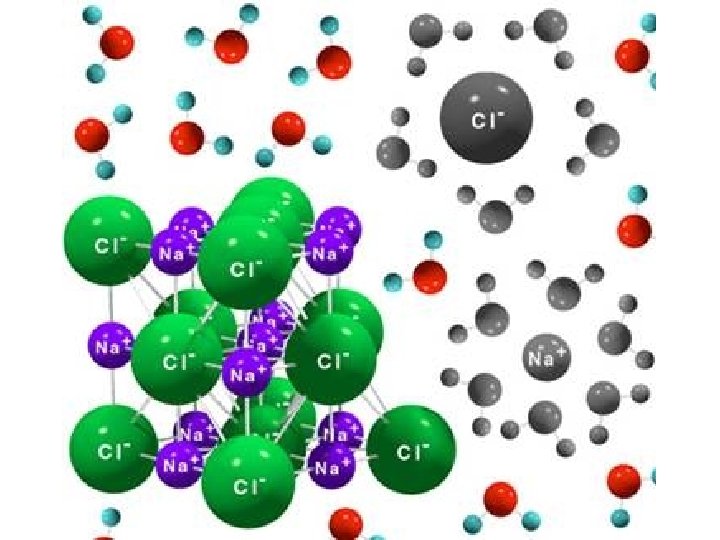

How many parts does a compound separate into? • Is it ionic or covalent? – Ionic = metal + nonmetal – Covalent = nonmetal + nonmetal • If covalent it stays as one particle • If ionic , add subscripts together
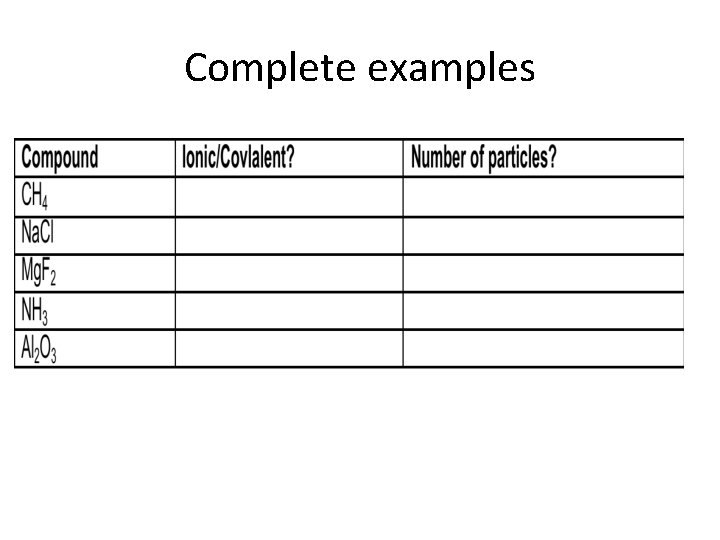
Complete examples

What is a colligative properties? • Properties of solutions that depend on the number of dissolved particles – Freezing point elevation – Boiling point depression

How do colligative properties work? • The compounds form bonds with the water • You need energy to break these bonds, which changes freezing and boiling points

The boiling point will increase with more solute

The freezing point will decrease with more solute

How can you calculate boiling point elevation? • Boiling Point Elevation: ΔTb= i. Kb m – ΔTb = Freezing point of pure solvent – Freezing point of solution – i = Van’t Hoff factor • associated with the amount of dissociation -> how many particles does the solute dissolve into – Kb = Freezing point depression constant • Depends on the substance – m = molality

How can you calculate freezing point depression? • Freezing Point Depression: ΔTf = i. Kf m – ΔTf = Freezing point of pure solvent – Freezing point of solution – i = Van’t Hoff factor • associated with the amount of dissociation -> how many particles does the solute dissolve into – Kf = Freezing point depression constant • Depends on the substance – m = molality

Example 1: A 5 molal solution of sodium chloride (Na. Cl) is dissolved in water. How will this affect the boiling point of water, assuming original bp=100? Kb water = 0. 52°C kg/mol • a. Is Na. Cl ionic or covalent? • b. How many parts will it break into? • c. What is the change in boiling point?

Example 2: 2. 5 moles of Mg. I 2 is dissolved in 30 kg of water. What will the new boiling point be if its original boiling point is 100°C and Kb is 0. 52 • a. Is Na. Cl ionic or covalent? • b. How many parts will it break into? • c. What is the new boiling point?

Example 3: A 5 molal solution of Mg. I 2 and water is created. How will this affect the freezing point of water, assuming freezing point of water is 0°C? Kf water = 1. 86°C kg/mol • a. Is Mg. I 2 ionic or covalent? • b. How many parts will it break into? • c. What is the new freezing point?
- Slides: 17